Chemistry 11 - Atoms and Molecules
1/72
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
What is Matter?
What are the three states of Matter?
Anything that has mass and takes up space
Solid, liquid gas
What is an Atom?
The smallest possible unit of an element which contains it’s fundamental components
What is a Molecule?
How is it held together?
2 or more atoms
Held together through STRONG electrical forces
Is a pure substance a homogenous or heterogenous mixture?
Does its composition change? Do they have predictable or unpredictable properties? Why or why not?
What forms of matter can it be?
Can it be an element, compound, or both?
Provide 1 example of a pure substance.
Homogenous mixture;
No, it’s composition does not change. Pure substances have predictable properties because of it’s uniform composition
The forms of matter of pure substances are solid, liquid, and gas
Pure substances can be elements and compounds
Examples: Water, salt, gold, copper, diamonds
What is an element?
Can it be broken down into smaller pieces?
Does it fall under heterogeneous or homogenous substances? If so, does it fall under mechanical mixtures, pure substances or homogenous mixtures?
What makes it qualified to be an element?
Provide 1 example of an element.
A pure substance that cannot be broken down into smaller pieces.
Elements fall under homogenous mixtures, as a pure substance.
It becomes qualified as an element when there’s only one type of atom present in a pure substance (all atoms must be the same).
Examples: Hydrogen, Helium, Lithium, Beryllium
What is a Compound?
Does it fall under heterogeneous or homogenous substances? If so, does it fall under mechanical mixtures, pure substances or homogenous mixtures?
Is there only one type of molecule in a compound? Why or why not.
A compound is a pure substance that contains two or more types of atoms
Compounds fall under homogenous mixtures, as a pure substance
There is only one type of molecule in a compound because a pure substance must have constant composition; therefore, having 1 type of molecule keeps a pure substance’s composition consistent.
What is a System?
Anything under observation
What is a substance?
Is it a physical or chemical property?
What are the two categories of substances?
Physical properties that are used to classify the substances into various categories:
Heterogeneous Substance
Homogenous Substance
What is a phase? (guiding questions below)
What is it within?
Do phases stick together by choice? Why or why not?
Are phases distinctly and visibly separated within a system? What separates them?
Anything within a system that remains the same in both it’s composition and properties
Phases stick together by choice because they have similar characteristics.
Phases are distinctly and visibly separated within a system. They are separated by boundaries.
ex. Coin in water. This is a TWO PHASE system, as there is a distinct boundary of where the coin stops and water exists.
What is a mixture?
What are the categories of mixtures?
Do the properties depend on the composition?
What is the difference between a mixture and a pure substance?
A mixture is a multitude of different types of atoms and/or molecules
Mixtures have more categories than solid, liquid, and gas. The other categories are heterogeneous and homogeneous mixtures.
Yes, the properties depend on the composition. If it’s a homogeneous or a heterogeneous mixture, then it’s properties can change
The difference between a mixture and a pure substance is that mixtures have different types of atoms, and pure substances have the same molecules throughout it’s composition.
What is a Solution?
Is it a homogenous mixture?
What are the 5 types of solutions?
A solution is a homogenous mixture that contains 2 or more substances
Yes, it is a homogenous mixture;
5 types of solutions:
gas in gas
gas in liquid
liquid in liquid
solid in liquid
solid in solid
(just remember that there’s no gas in solid)
What are Mechanical Mixtures?
Is it a homogeneous or a heterogeneous mixture?
Mechanical Mixtures are heterogeneous mixtures with 2 or more substances.
It is a heterogeneous mixture; (it’s the only subcategory of heterogeneous mixtures)
What is a Particle?
A particle is a general term to describe a small bit of matter
What are Homogeneous Substances?
Provide an example of a homogeneous substance.
Homogenous substances are one phase systems, meaning their composition stays the same throughout.
Water, gold, diamond, vodka
What is an proton, electron, and neutron?
Where can you locate each of these?
How much does each weigh?
A proton is a positively charged subatomic particle. A neutron is a neutrally charged subatomic particle. An electron is a negatively charged subatomic particle.
They are all found within an atom. Protons and neutrons are found in the nucleus. Meanwhile, electrons can be found in orbitals on the energy shells surrounding the nucleus.
Proton weighs one.
Neutron weighs one.
Electron weighs 0.
What is an ion?
How does an atom become an ion? how is it for Ionic Bonds? How does it work for Covalent bonds?
Why do atoms want to become an ion/why do they want a full valence shell?
An ion is a charged atom.
For ionic bonds, atoms become an ion by losing or gaining electrons. For covalent bonds, atoms share electrons.
Atoms want to become an ion because their current state is unstable, due to the equal number of protons and electrons. When they lose, gain, or share electrons, they gain full valence shells, which makes them stable.
What is an isotope?
Does it affect an atom’s chemical activity? Why or why not?
An isotope is an atomic species that have the same atomic number/number of protons but has a different atomic mass. (it has different amounts of neutrons)
It does NOT affect chemical activity because only the protons and electrons can. It only affects atomic mass.
What does a mass number/atomic mass refer to?
Where can you find it on the periodic table?
Why does it have decimal points?
The mass number refers to the weight of the neutrons and protons in an atom’s nucleus
It can be found on every single element box.
Atomic mass has decimal points because of isotopes (same number of protons, but different electrons). Atomic mass of an atom is the average mass of all of that element’s atoms in the world. Because isotopes vary the weight, it causes decimal points.
What does an atomic number refer to?
How is it useful? What is it’s relation to electronegativity and ionization energy?
An atomic number refers to the amount of protons there are in an element’s nucleus.
It is useful to find number of neutrons, and relative electronegativity and ionization energy.
What is Avogrado’s number?
Why is it useful?
Avogrado’s Number is 6.022 × 10²³
It is useful to see if your atomic mass of your sample has the same mass as the periodic table’s mass for that element. Because atomic mass on the periodic table is found by finding the average mass of each of that element’s atoms, the mass for a single sample may differ. If it has 6.022 × 10²³ atoms, then it will have the same atomic mass as stated on the periodic table.
What is a mole?
How can you calculate molar mass?
Why do you calculate molar mass?
A mole is a unit of measurement in chemistry. The mole of something is equal to as many as there are atoms.
you can calculate molar mass by multiplying a percentage (ex. 75% → 0.75) by the number of atoms there are (usually the superscript in this scenario [ex. ²³C])
You calculate molar mass to see the exact mass of an atom within an experiment.
What is an aqueous solution?
What is a solute solution?
What is a solvent solution?
Is water a solute, solvent, or aqueous solution?
An aqueous solution is refers to a solvent in water (ex. saltwater)
A solute solution is a component in a solution that exists in a small quantity (ex. a child in a mother’s belly)
A solvent solution is a component in a solution that exists in a greater quantity (ex. the lemon in Chinese icetea)
Water is ALWAYS THE SOLVENT
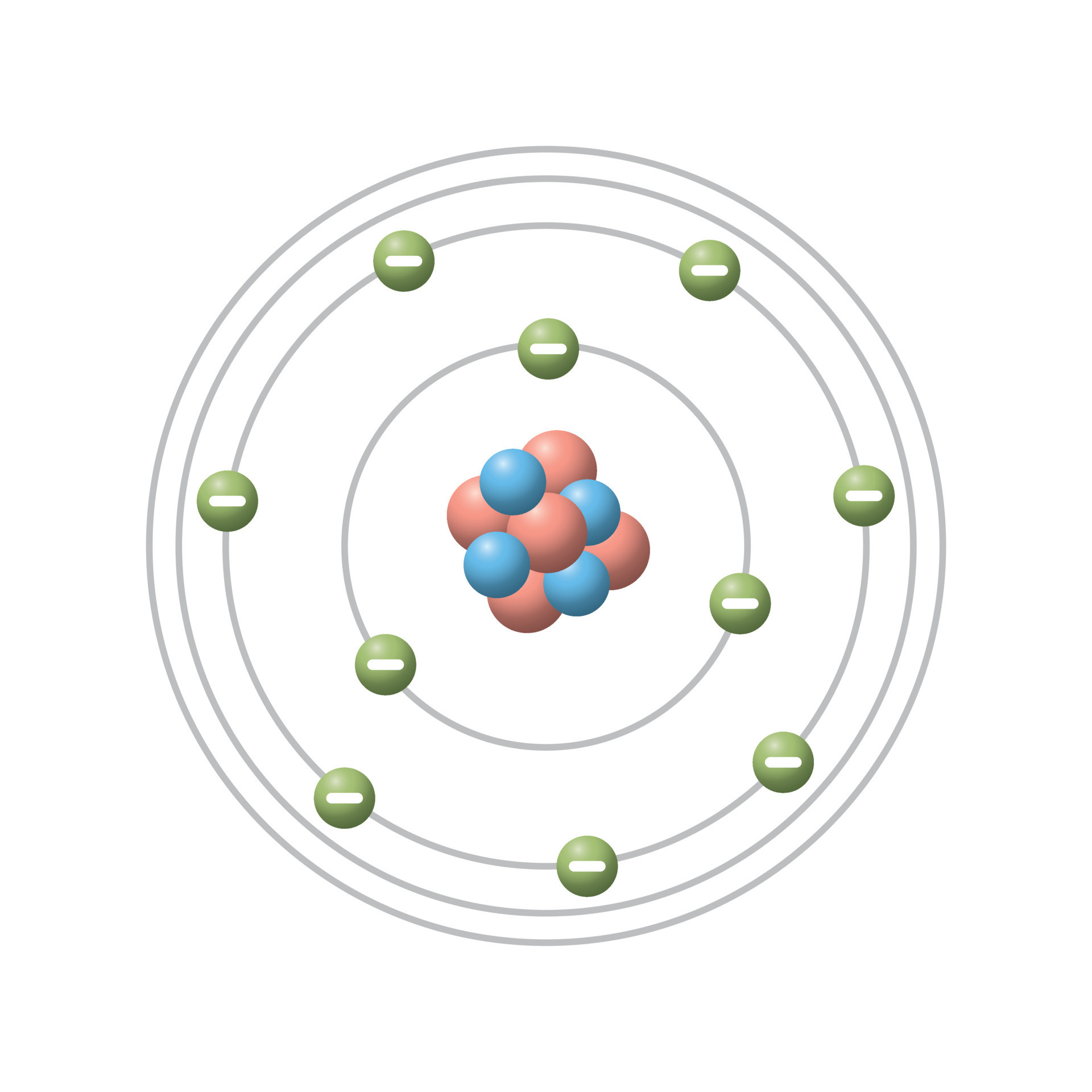
Assign the following terms to the diagram
Proton
Neutron
Electron
Valence Shell
Energy Shell/Orbital
Proton is red
Neutron is Blue
Electron is Green
Valence Shell is outermost shell
Energy Shell/Orbital are the circles surrounding the nucleus
Periodic Table Info:
What direction is a period?
What direction is a group/family?
What direction do protons and electrons increase on?
What direction does atomic mass increase? Why does it increase?
What direction do valence shells increase?
A period is right to left
A group/family is up and down
Protons and electrons increase across periods from left to right, and along groups/families from up to down.
Atomic mass increases the same way as protons and electrons increase ^ This is because atomic mass depends on the number of protons and neutrons in a nucleus, so it increases when their quantity increases.
A valence shell is added each element down a group
Atomic Notation:
What are superscripts? What do they represent?
What are subscripts? What do they represent?
Superscripts are like exponents – they are on top. They represent the charge an ion has
Subscripts are beside and below. They represent the amount of atoms there are of that particular element (this is scenario based)
What is an ionic charge?
What are the three ways an atom gain a charge?
How does this concept relate to electronegativity and Ionization Energy?
An ionic charge is when an atom takes, gains, or shares an outer electron, they get a charge.
Three ways an atom gains a charge is through taking, gaining, or sharing electrons.
This relates to electronegativity and ionization energy because the more electronegativity an atom has, they better ability it has to take electron. The more ionization energy it has, the more it can keep their outer electrons in place.
What did Dalton believe?
What does Dalton’s model look like? What is it called?
What does his model explain about an atom?
Dalton believed that elements are made of atoms, and atoms are identical.
(image attached to see model – it is a circle). It is called the billiard ball model.
His model explains that atoms are identical.

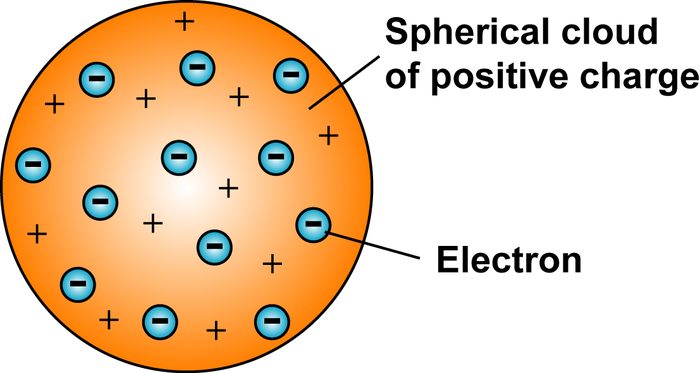
What did Thompson discover?
What does Thompson’s model look like? What is it called?
What does his model explain about an atom?
Thompson discovered that atoms have positive and negative charges (he called electrons corpuscles).
Thompson’s model looks like a circle with dots in it. The circle is the positive charge, and the electrons are dots inside it. It is called the plum pudding model.
His model explained that atoms have positive and negative charges.
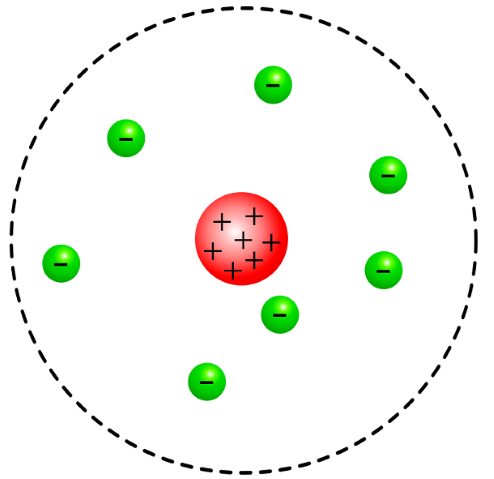
What are the three things that Rutherford believed?
What does Rutherford’s model look like? What is it called?
What does his model explain about an atom (besides his beliefs)
Rutherford believed that
atoms have a positively charged nucleus surrounded by negatively charged electrons,
atoms are innately neutrally charged,
atomic mass is from the nucleus
Rutherford’s model looks like a circle with positive charges in the middle, and negative charges surrounding it. It is called the Rutherford Model.
It explains his beliefs, and that particles can through, but bounce off the positively charged nucleus.
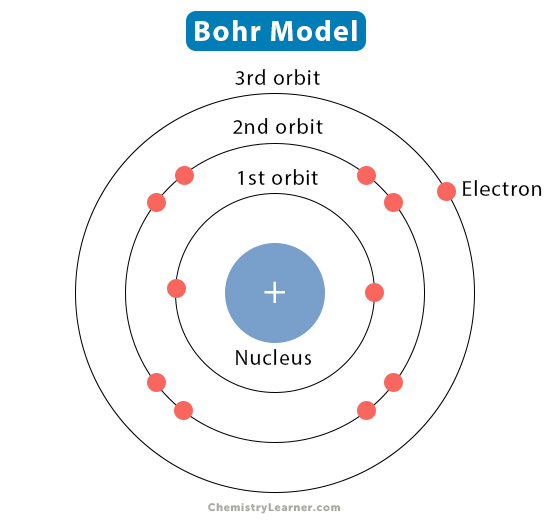
What are the 3 things Bohr believed?
What does Bohr’s model look like? What is it called?
What does his model explain about an atom?
Bohr believed that
electrons have specific energy,
electrons are confined to moving within orbitals
electrons only omit or absorb energy while moving
Bohr’s model looks like a nucleus surrounded by energy shells with electrons. It is called the bohr model.
Bohr’s model explains everything about an atom – that’s why it’s used today.
What did Greek Philosophers discover?
they discovered atoms;
Greek Philosophers believed that differences in substances were because of differences of size and shape of tiny, uniform, and uncuttable particles.
What is Quantum Mechanics?
Why is it important?
Quantum Mechanics is a branch of physics that studies atomic and subatomic systems
It is important because it helps us theoretically understand the structures of atoms and it’s affect on other things.
What are the three characteristics of Electron Placement?
Does this fall under quantum mechanics?
Electrons are on specific energy shells
Electrons are found based off of probability: where it is now and where it is going
Electrons do not move in predictable paths
yes, electron placement falls under quantum mechanics, as quantum mechanics studies atomic and subatomic structure
What are energy shells? (guiding questions below)
Where can they be found?
What are on energy shells?
Energy shells are fixed distances from an atom’s nucleus where electrons can be found.
note: they are also called orbitals
What are orbitals? (guiding questions below)
Where can they be found?
What are inside orbitals?
How much ___ can be found within orbitals
Do they move in the same direction or different directions?
How do electrons fill orbitals
Orbitals is an actual region where electrons can exist in a particular energy level
They can be found within an energy shell, and within a subshell
Two electrons can be found within orbitals
Electrons move in different directions
Electrons fill orbitals by going to the orbital that requires the least amount of energy (the lowest one cause electrons are lazy). If that’s full, they will occupy empty orbitals. When all orbitals are half occupied, they will fill in the half full orbitals, starting from left to right. When all orbitals of that shell are full, they will begin on a new shell.
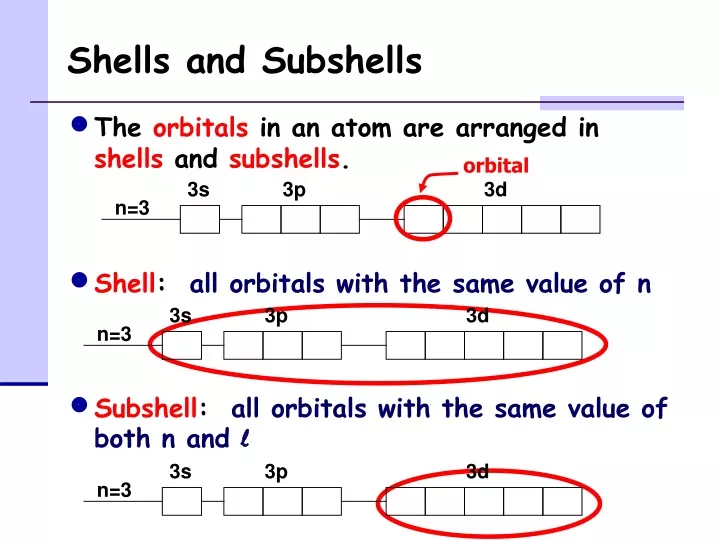
What is a shell? Are they the same as energy shells?
What is a subshell?
Where can it be found?
What is the difference between shells and subshells?
What is it’s relation to Orbitals?
A shell is a set of orbitals having the same n-value (ex. the third shell has 3s, 3p, and 3d orbitals). They are the same as energy shells.
A subshell is set of orbitals of the same type (ex. a set of 3 2p orbitals in the 2nd subshell
Shells can be found at a fixated distance away from an atom’s nucleus. Subshells can be found within a shell.
Shells are labels for the full set of orbitals, and subshells are specific to one set of orbitals within a shell
It’s relation to orbitals is that orbitals are within subshells, which are within shells.
Valence Electrons
Electrons situated on the outermost energy shell that can react
If they can’t react, they’re not valence
Atomic Radius
The distance from an atom’s nucleus to the outermost energy shell
Ionization Energy
The extent of how much an atom’s nucleus has a hold on it’s outermost electrons;
The energy required to remove the outermost electron;
Laws of Attraction
Opposite charges attract
Like charges repel
The more charges there are, the greater the attraction
The closer charges are to each other, the greater the attraction
Periodic table organization: state the trends of electronegativity, ionization energy, and atomic radius
Left to right across a period
Moving down a group
Atomic radius decreases and ionization energy increases due to laws of attraction (more subatomic particles → greater attraction → smaller it becomes → more ionization energy)
Atomic radius increases due to more energy shells being added per element. The ionization energy decreases because the outermost electrons are farther away (law of attraction)
Electronegativity
Ability of a nucleus to pull away outermost electrons from neighbouring atoms
What atom families are involved in an Ionic Bond?
Why do they bond?
What is its electronegativity?
A bond between a non-metal and metal;
Give or take electrons to achieve a full outermost shell and stability
The electronegativity difference between atoms is >1.7
Covalent bond
A bond between 2 or more non-metals because they have same or similar electronegativity values
Shares their outermost electrons in order to have a full valence shell
What is a Nonpolar Covalent Bond?
What is it’s electronegativity?
When electrons are shared equally in a covalent bond (a bond between non-metal(s));
Same electronegativity (the difference between the atoms are 0)
What is a Polar Covalent Bond?
What charges do the involved atoms get?
What is the range of it’s electronegativity?
When atom X has a larger electronegativity than atom Y, but atom X’s electronegativity is not strong enough to pull an electron away from the atom Y.
Therefore, they share the electron; however atom X gains a partial negative charge (δ−) because it’s gaining the electron more than losing it. Atom Y gets a partial positive charge (δ+) because its losing the electron more than gaining it.
0 < x < 1.7
Is an atom’s electronegativity greater if it’s ionization energy is greater? Why or why not?
Yes;
The nucleus has a stronger pull on it’s outermost electrons. Because the atomic radius is small, it’s positive energy radiates to other atoms, then having the ability to take away their outermost electrons as well
Why is the noble gases an exception in regards to the positive correlation between an atom’s electronegativity and ionization energy?
High ionization energy because it’s valence shells are innately full ← more charges = greater attraction
Low electronegativity because it doesn’t want other atom’s electrons
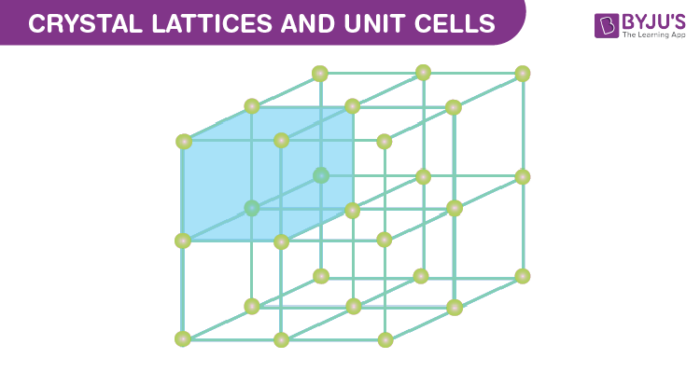
How do crystal lattices form? How are laws of attraction related?
When an ionic bond occurs, the atoms become ions with opposite charges. Because of the laws of attraction, they are drawn to each other
What does a higher electronegativity do?
What does a lower electronegativity do?
Why does this happen?
Pulls away outermost electrons from other atoms
Give their outermost electrons to other atoms
This occurs so atoms can leave their neutral state (neutral because of the equal number of protons and electrons) and become stable through full valence shells
What happens when an atom’s electronegativity is exactly 1.7?
It is classified as a polar covalent bond with ionic characteristics!
How does forming bonds allow atoms to achieve a more stable electron configuration?
Forming bonds allows atoms to achieve a full valence shell by taking, giving away, or sharing electrons, dependent on whether it’s a metal or non-metal. Achieving a full valence shell helps an atom become stable.
Lewis Dot Structure
How are electrons represented?
How are shared electrons represented? What are they actually called?
A 2D diagram outlining the type of bond and valence electron transfer that occurs between atoms.
Dots are used to represent an individual electron
Shared electrons are represented by a line. They care called bonding pairs!
How can you tell how many valence electrons are in the atoms of each group?
The amount of valence electrons corresponds with the final digit of the group number!
Open shell
A not-full valence shell
Close shell
An outer shell with all it’s electrons
What is the Octet Rule?
Is it used for covalent or ionic bonds/compounds?
Atoms achieve closed shells by covalently bonding
Only applicable to covalent compounds
What are Polyatomic Ions? Are they exclusive to ionic bonds, covalent bonds, or both?
Is it already an ion?
A covalent bond with more than 2 atoms associated with it
Yes it is an ion.
What does VSEPR stand for?
What is the VSEPR theory?
Valence Shell Electron Pair Repulsion Theory
Predicts the shape of molecules in 3D Space
How does the VSEPR theory predict the shape of molecules in 3D space?
According to the laws of attraction, like charges repel.
Because they repel, they adopt a specific formation to set themselves as far apart from each other as possible around the central atom.
What is a lone pair?
Electron pairs not in a bond
What is a bonding pair?
What is a single pair?
What is a double pair? Are they in one group, or two?
Electrons in a bond
Two electrons paired up to make 1 bond
4 electrons paired up to make 2 bonds. They are in 1 group, because they are going in the same direction and real life.
What is a group in accordance to the VSEPR theory?
A group is a pair (bonding or lone) that goes in 1 direction of the diagram and real life.
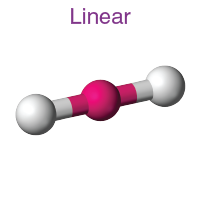
Linear Molecular Shape (VSEPR theory)
How many surrounding atoms are there?
How many lone pairs are there?
What is the bond angle?
How many groups are there?
How many bonding pairs are there?
two surrounding atoms
0 lone pairs
180°
two
two single bond pairs
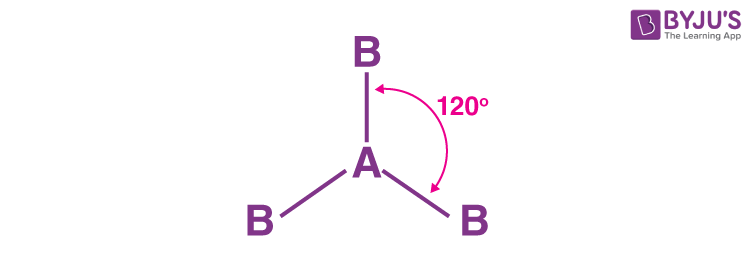
Trigonal Planar Molecular Shape (VSEPR theory)
How many surrounding atoms are there?
How many lone pairs are there?
What is the bond angle?
How many groups are there?
How many bonding pairs are there?
Three surrounding atoms
0 lone pairs
120° bond angle
three groups
three bonding pairs
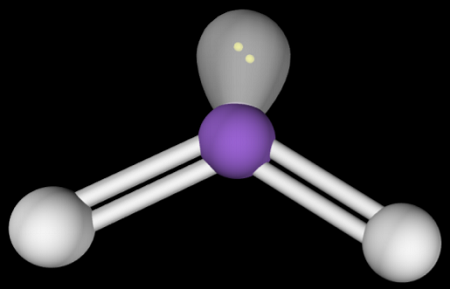
Bent 120º Molecular Shape (VSEPR theory)
How many surrounding atoms are there?
How many lone pairs are there?
What is the bond angle?
How many groups are there?
How many bonding pairs are there?
Two surrounding atoms
one lone pair
120° bond angle
three groups
two bonding pairs
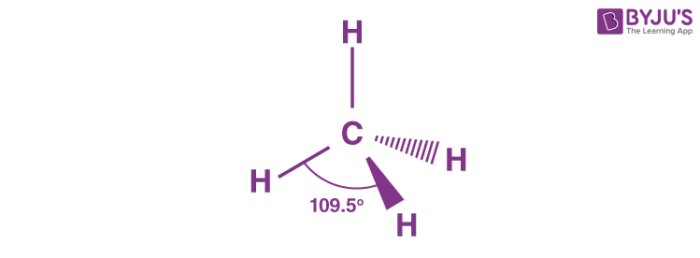
Tetrahedral/Tetrahedron Molecular Shape (VSEPR theory)
How many surrounding atoms are there?
How many lone pairs are there?
What is the bond angle?
How many groups are there?
How many bonding pairs are there?
Four surrounding atoms
zero lone pairs
108.5° bond angles
Four groups
1 points towards you
1 Points behind you
Four bonding pairs
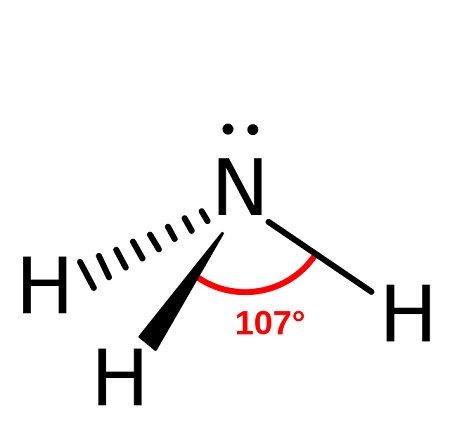
Trigonal Pyramidal Molecular Shape (VSEPR theory)
How many surrounding atoms are there?
How many lone pairs are there?
What is the bond angle?
How many groups are there?
How many bonding pairs are there?
three surrounding atoms
One lone pair
109.5°
Four Groups
Three bonding pairs
1 points towards you
1 points behind
Bent 109.5° Molecular Shape (VSEPR theory)
How many surrounding atoms are there?
How many lone pairs are there?
What is the bond angle?
How many groups are there?
How many bonding pairs are there?
two surrounding atoms
two lone pairs
109.5°
four groups
two bonding pairs
1 in front
1 behind
Trigonal Bipyramidal Molecular Shape (VSEPR theory)
How many surrounding atoms are there?
How many lone pairs are there?
What is the bond angle?
How many groups are there?
How many bonding pairs are there?
Five surrounding atoms
zero lone pairs
two different bond angles: 90° and 120°
Five groups
1 points towards you
1 points behind you
Five bonding pairs
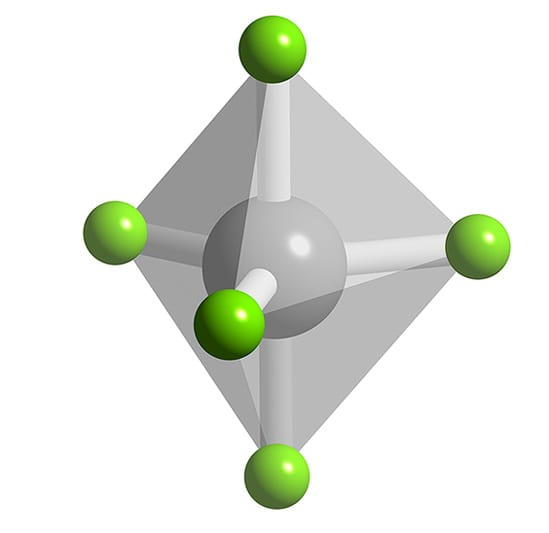
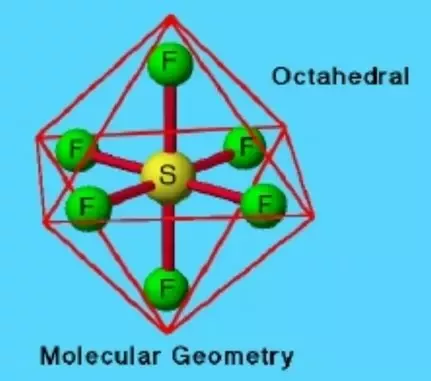
Octahedral Molecular Shape (VSEPR theory)
How many surrounding atoms are there?
How many lone pairs are there?
What is the bond angle?
How many groups are there?
How many bonding pairs are there?
Six surrounding atoms
Zero lone pairs
90° bond angles
Six groups
2 point behind
2 point forward
Six bonding pairs
Surrounding Atom
The atoms surrounding the central atom within the VSEPR theory
Why do some atomic masses have decimal points?