Ionic bonding
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Formula for carbonate
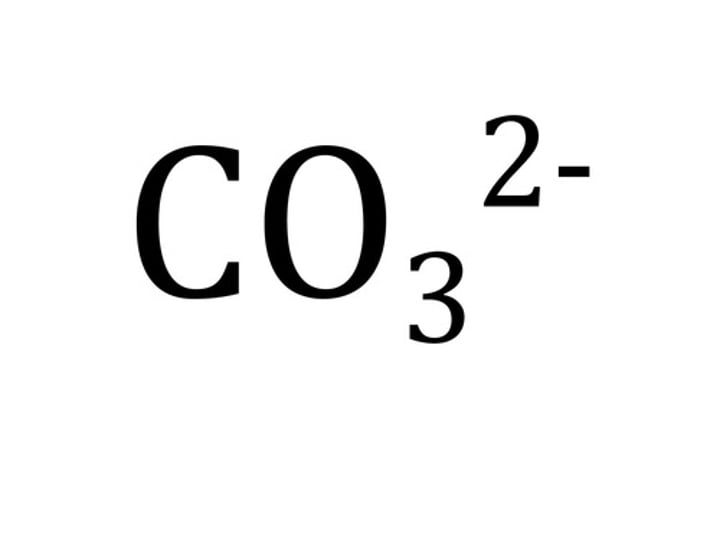
What is ionic bonding?
The electrostatic attraction between ions of opposite charges
When does ionic bonding take place?
When a metal reacts with a non-metal
Which is stronger, ionic bonds or covalent bonds?
Covalent bonds
An atom will lose or gain electrons until it has the same electron configuration as what?
A noble gas
What is a compound ion?
An ion which is made of multiple atoms
Formula for nitrate
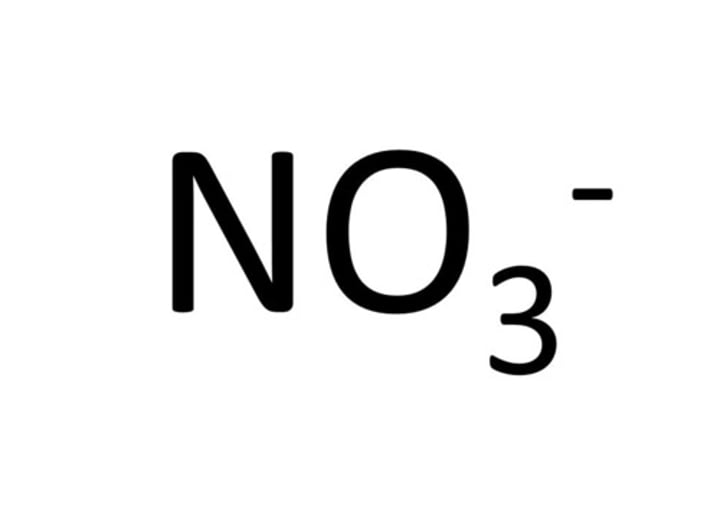
Formula for hydroxide
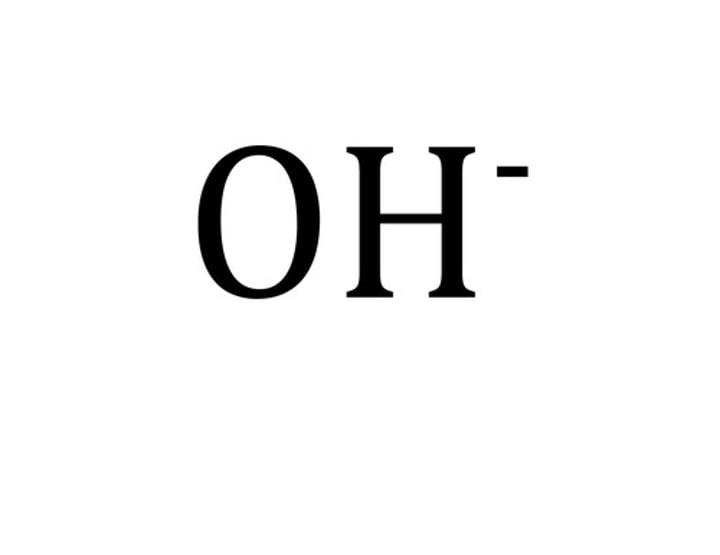
Formula for sulfate
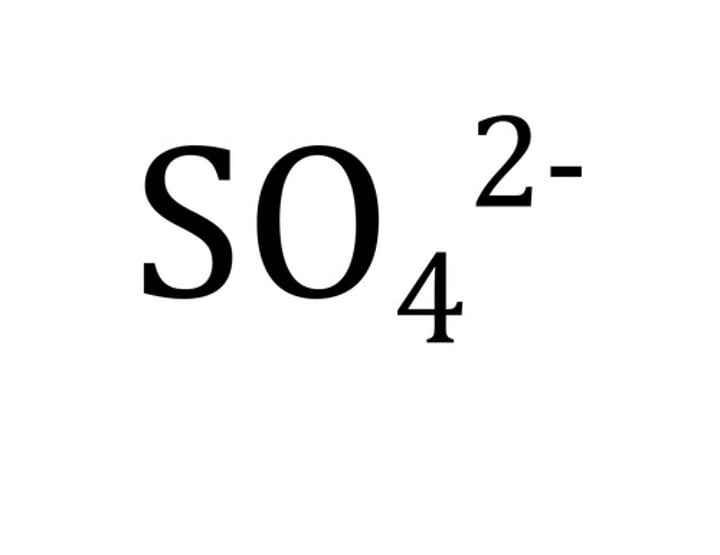
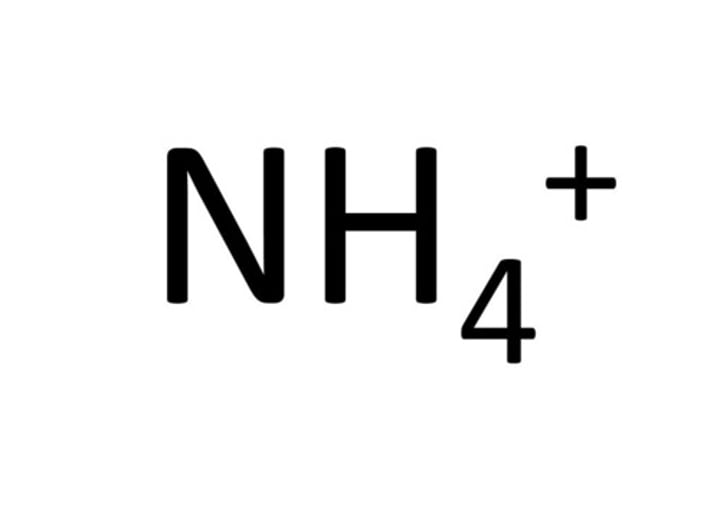
Formula for ammonium
What is the principle of electroneutrality?
The overall charge of a compound ion must be zero
Ionic compounds have a giant ionic _________ structure
lattice
What are the three properties of ionic compounds?
• They dissolve in water.
• They conduct electricity when molten or dissolved.
• They have high melting points
Why do ionic compounds dissolve in water?
Water is a polar solvent. This allows them to attract the ions away from their structure, breaking regular crystal lattice.
What does polar mean?
When charges in a compound are unevenly distributed
Why can ionic compounds conduct electricity when molten or dissolved in water?
The ions that carry the current are free to move in the liquid state but not the solid state
Why do ionic compounds have high melting points?
They have strong attractions between oppositely charged ions, which take a lot of energy to break