Mass Spectrophotometers: Ionization Methods
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
What type of molecules does Mass Spectrometry detect?
mass spec detects a variety of ionized molecules including large biomolecules (proteins, peptides, DNA, carbohydrates), small organic compounds, drug metabolites, and inorganic elements
measures their mass-to-charge ratio (m/z) to determine molecular weight, quantify compounds, and identify chemical structures
Goldilocks Molecules
molecules that fall within the detectable m/z range of a mass spec
“just right” = not too small and not too large for the instrument
lower limit: an e- or smth smaller than an e- (small ions); typically 1-10 Da and above)
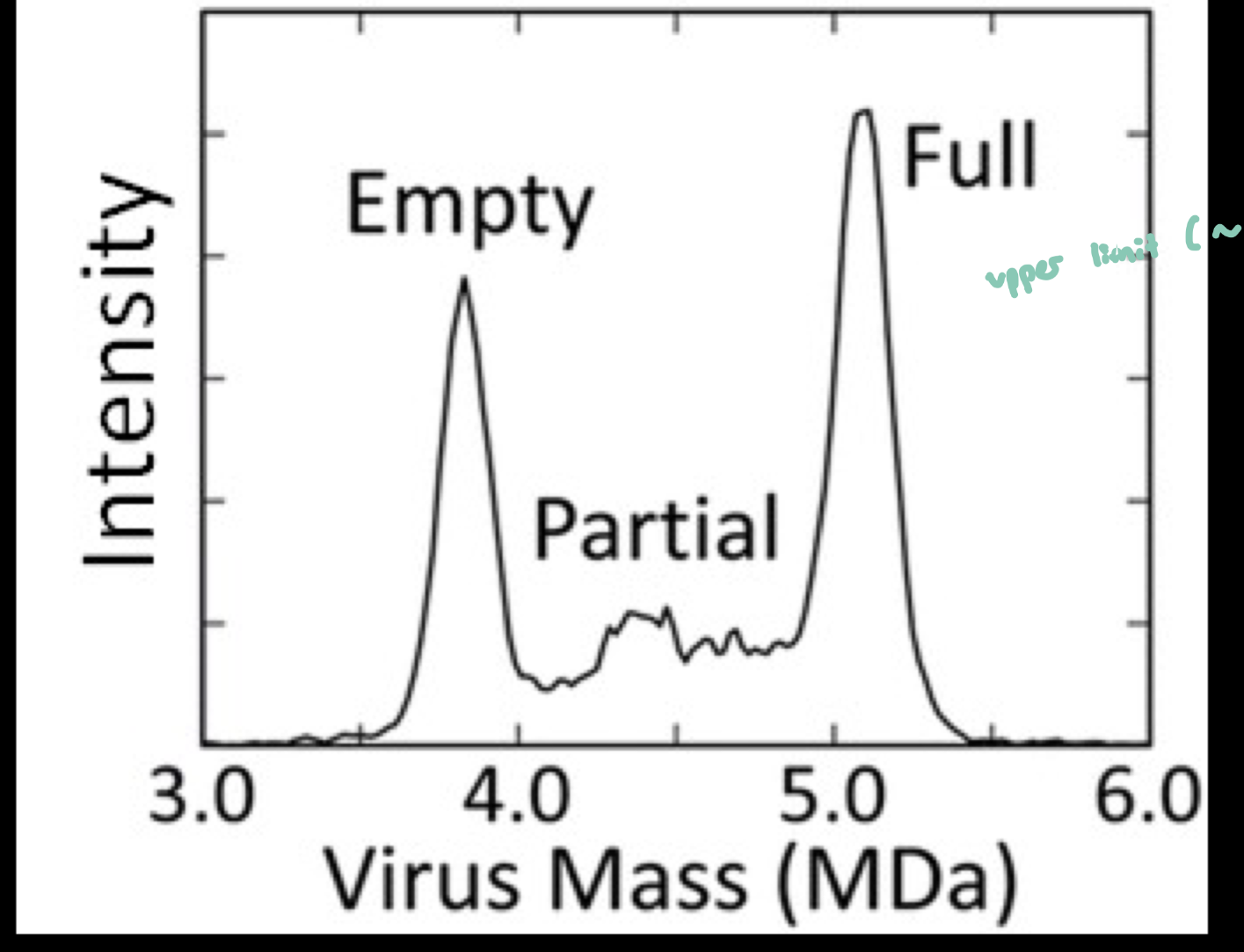
upper limit: ~5 mil Daltons (depends on the instrument)
the limits refer to the instrument’s detection limits; the smallest or largest m/z that the instrument can measure)
Different mass spectrometers have different detectable m/z ranges, and therefore different “Goldilocks zones.”
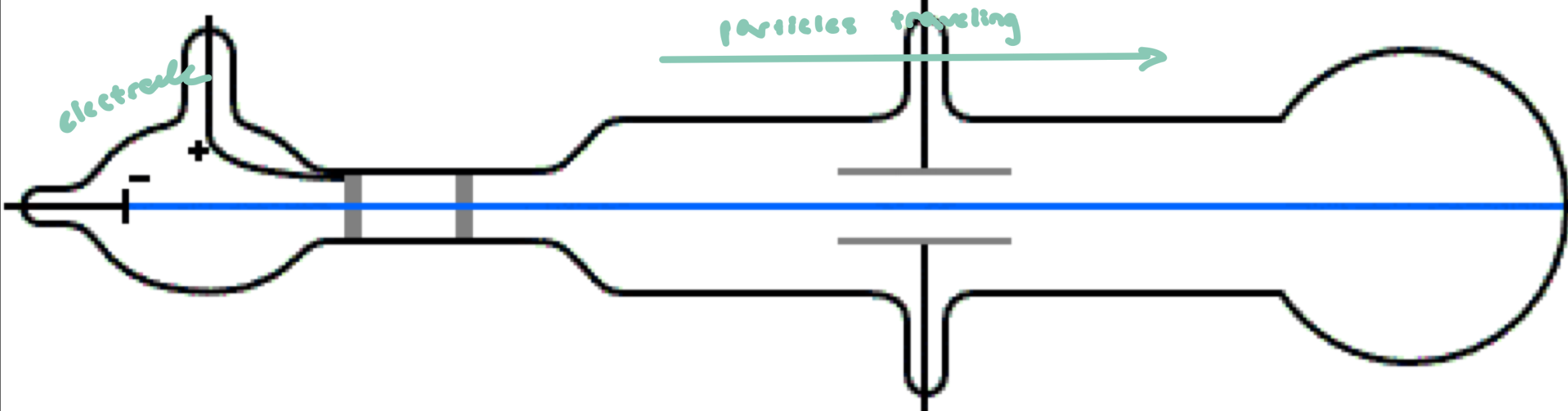
JJ Thomson
sealed glass container (vacuum sealed) to prevent collisions w/ air
electrode on 1 end to create charged particles (ions)
looking at how charged atoms move in an electric field
measure deflection → determine m/z
Charged particles are deflected by electric fields
if (-) charged, deflected from (-) plate and toward (+) plate
if (+) charged, deflected from (+) and toward (-) plate
ionization method: used high energy to remove electrons to create ions
often caused fragmentation of molecules
works well for small atoms/molecules
John Fenn
Developed Electrospray Ionization (ESI) for mass spectrometry; Solved the problem of analyzing large biomolecules (DNA, RNA, proteins)
converts biomolecules from solution → gas phase gently, allowing intact mass spec analysis of large molecules
Key challenge:
Molecules must be in the gas phase for mass spectrometry
Traditional high-energy ionization would fragment large biomolecules
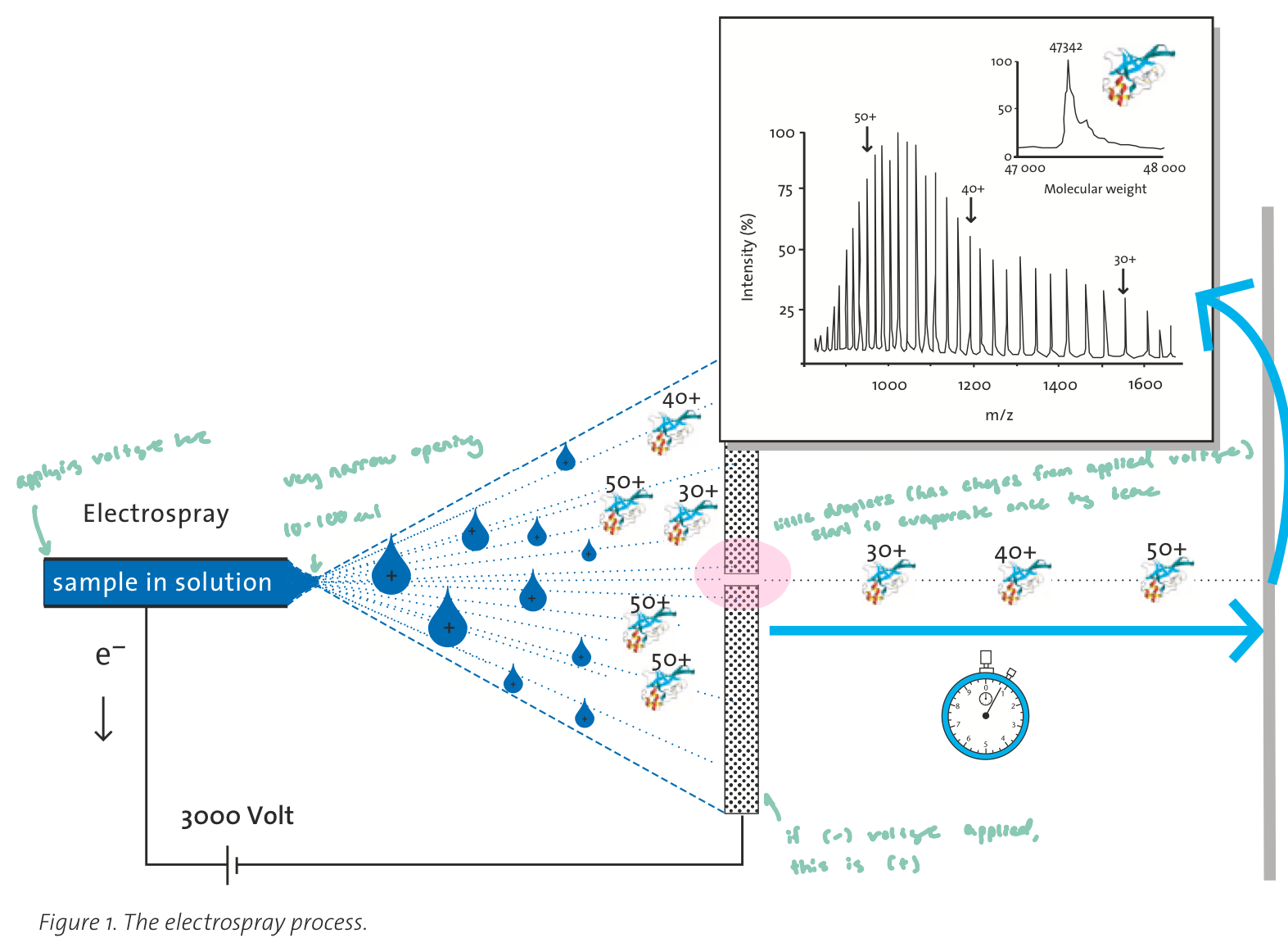
John Fenn: How ESI Works
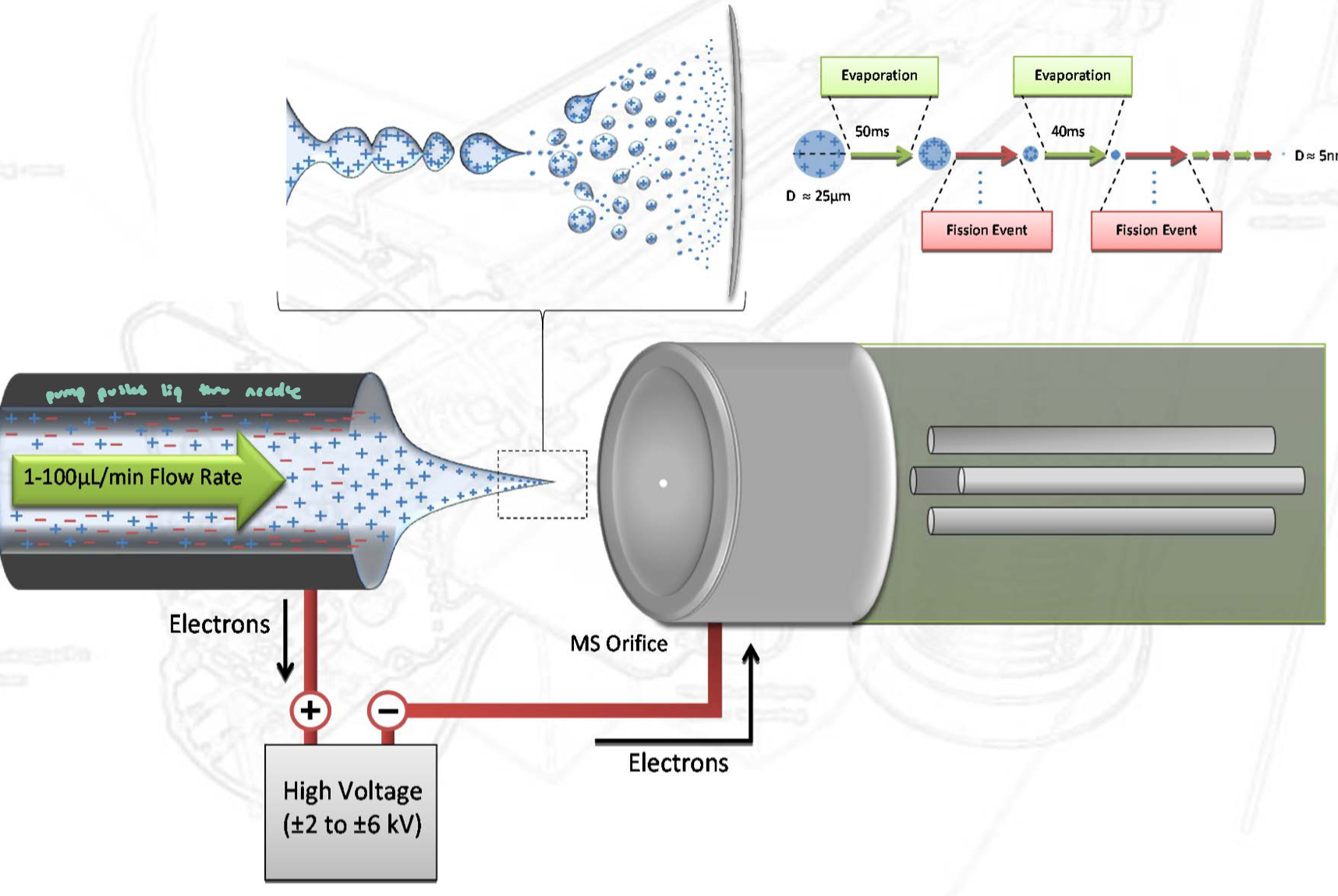
Sample is in liquid solution, creating an electrical connection between the sol. and mass spec
ions in sol. allow current to flow, effectively “closing the circuit”
A high voltage is applied to a narrow capillary
This produces a fine spray of charged droplets
only very small charged droplets are emitted from the capillary
What happens next:
Droplets evaporate solvent as they travel toward an oppositely charged plate (electrode)
Droplets become smaller → charges become more concentrated
Eventually, ions are released into the gas phase
(large biomolecules remain intact)
ESI: Coulombic Repulsion & Fission
like charges in a droplet repel each other (Coulombic repulsion
when repulsion becomes too strong, the droplets becomes unstable and undergoes Coulomb fission → “explodes into smaller droplets”
repeated evaporation + fission leads to formation of gas-phase ions that can enter the mass spec for detection
More ESI Figures
Tanaka & MALDI
developed laser-based ionization (MALDI approach) for analyzing large biomolecules like proteins
MALDI allows gentle ionization: proteins and other large biomolecules must be converted into the gas phase, where high-energy methods would normally fragment them
How MALDI works
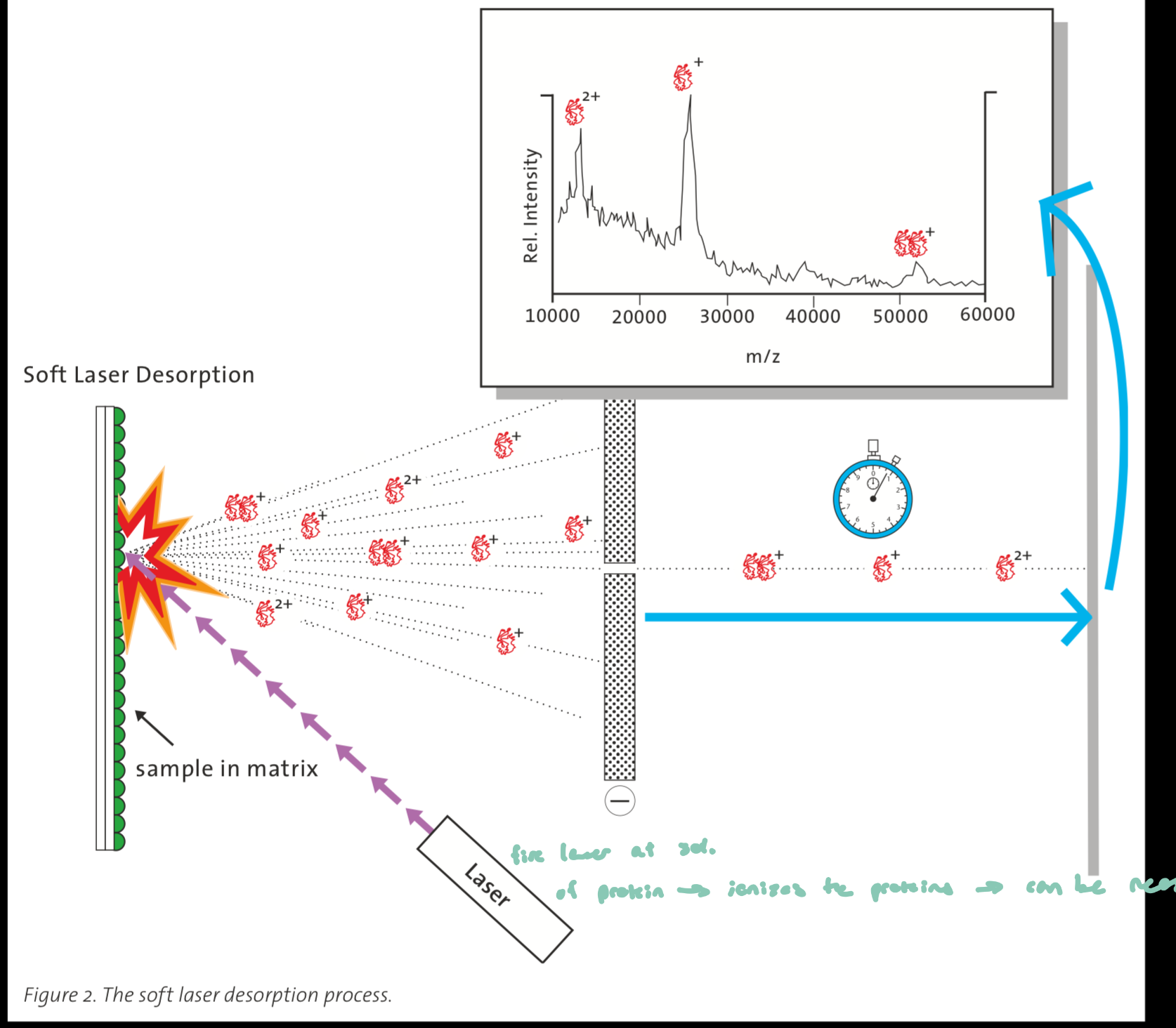
sample (protein) is mixed with a matrix (light absorbing chemical) and crystallized
a laser pulse is fired at the sample surface, hitting the matrix which absorbs the laser energy and rapidly vaporizes
it carries embedded protein molecules into the gas phase, and in this process, proteins become ionized
MALDI vs ESI
matrix-assisted laser desorption ionization
electrospray ionization (more popular)
MALDI: 1 charge, solid, smaller (better for smaller molecular weights)
ESI: many charges, liquid (good for DNA/proteins, which are happier in solution)
Types of Mass Spectrometers
Why mass spectrometry?
the most powerful of MS is its selectivity
very little ambiguity, however may struggle with isomers
measures mass very specifically; can fragment the molecules to figure out the original molecule
Versus UV/visible spectrophotometry of a complex mixture, we may be unsure of what we’re looking at
World Records of Mass Spec
Can distinguish very small mass differences (~0.01 Da or even smaller in high-resolution instruments)
allows detection of isotopes, small chemical modification, and tiny differences
applying (-) or (+) voltage allows us to see diff things
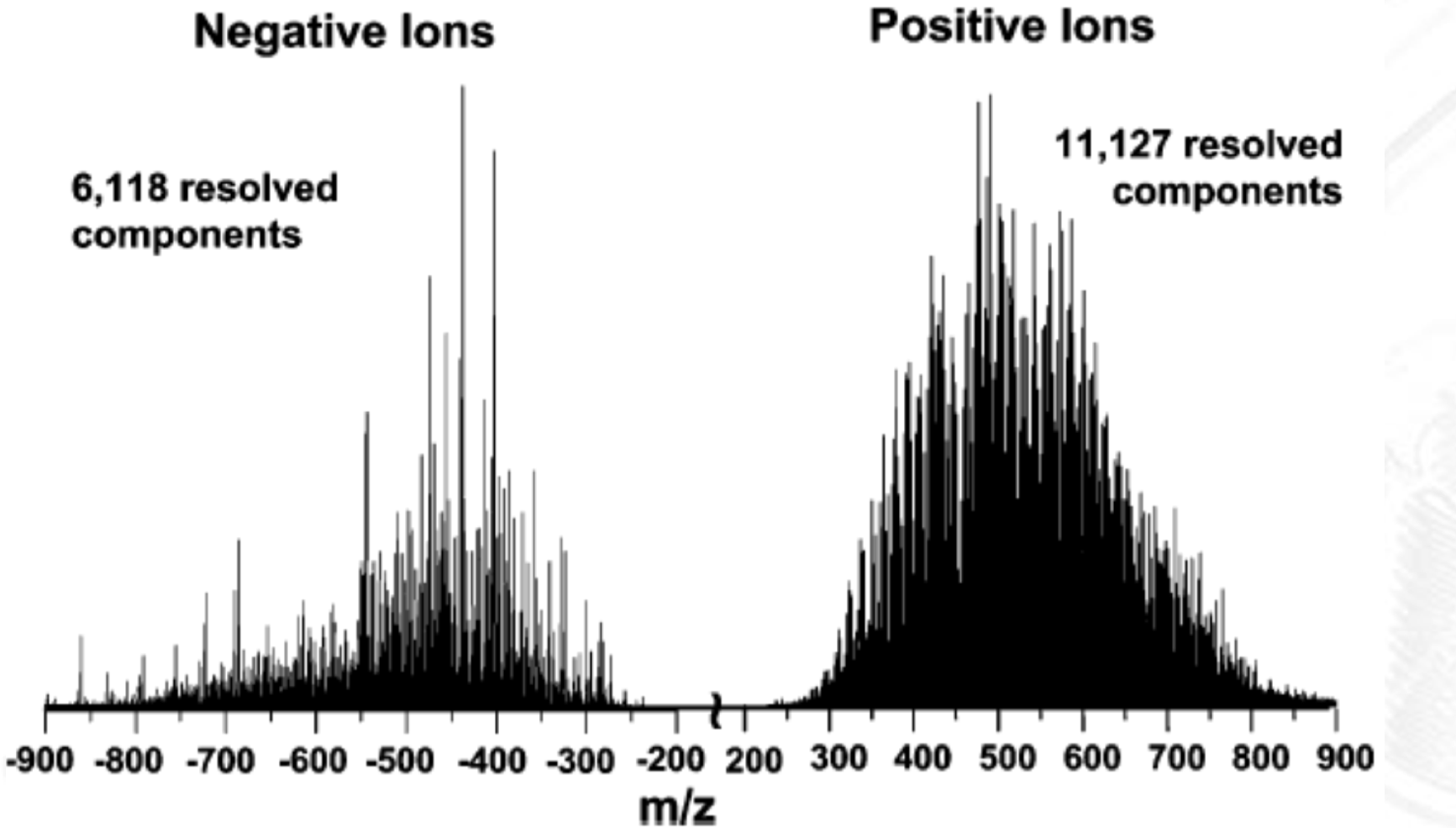
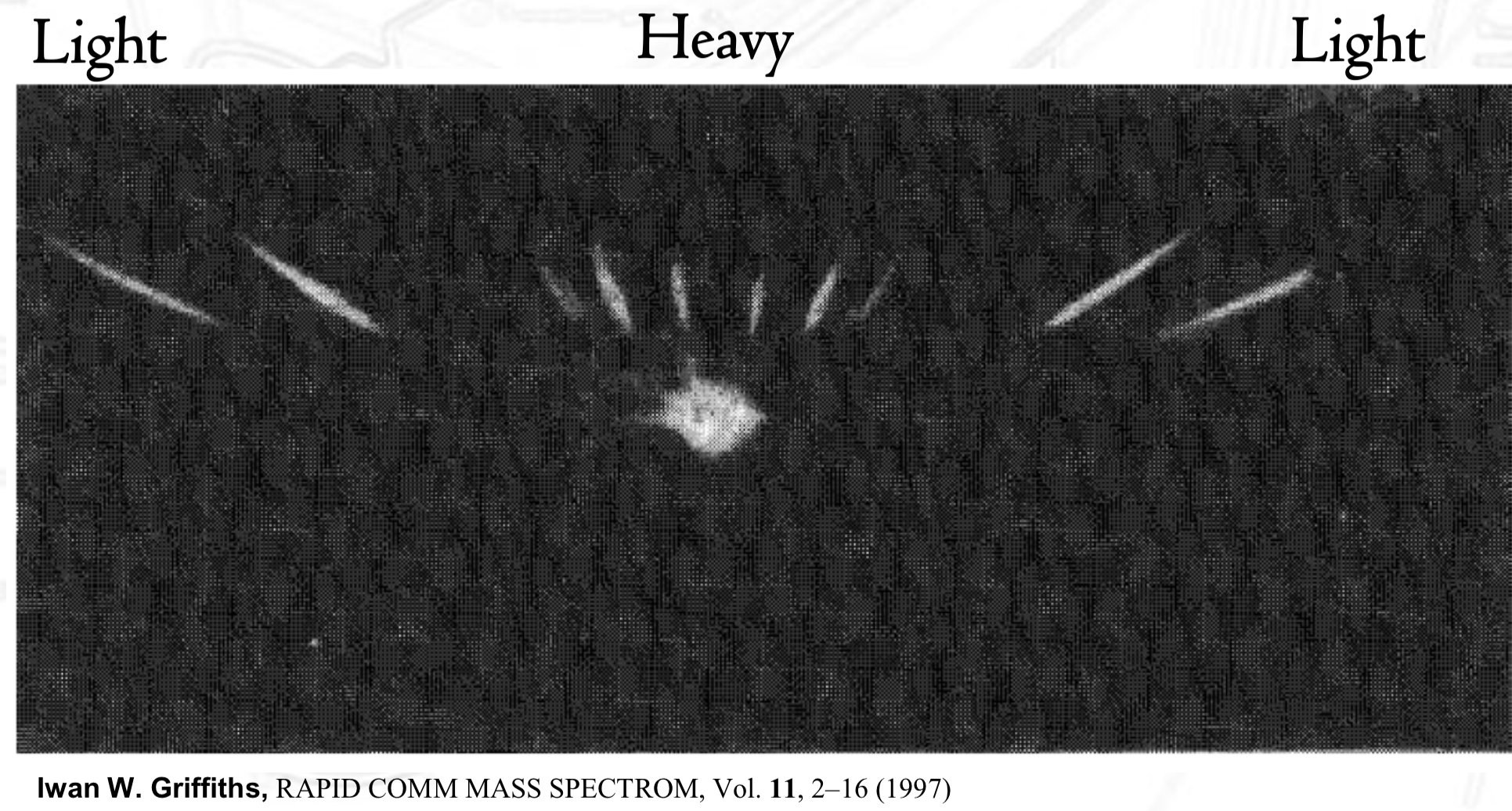
The 1st Mass Spectrum by Iwan. W. Griffiths
showed that mass spec can separate ions by m/z ratio
demonstrated that ions form a pattern of peaks based on m/z
helped establish that mass spec can be used to analyze and distinguish ions based on mass differences (an analytical method)
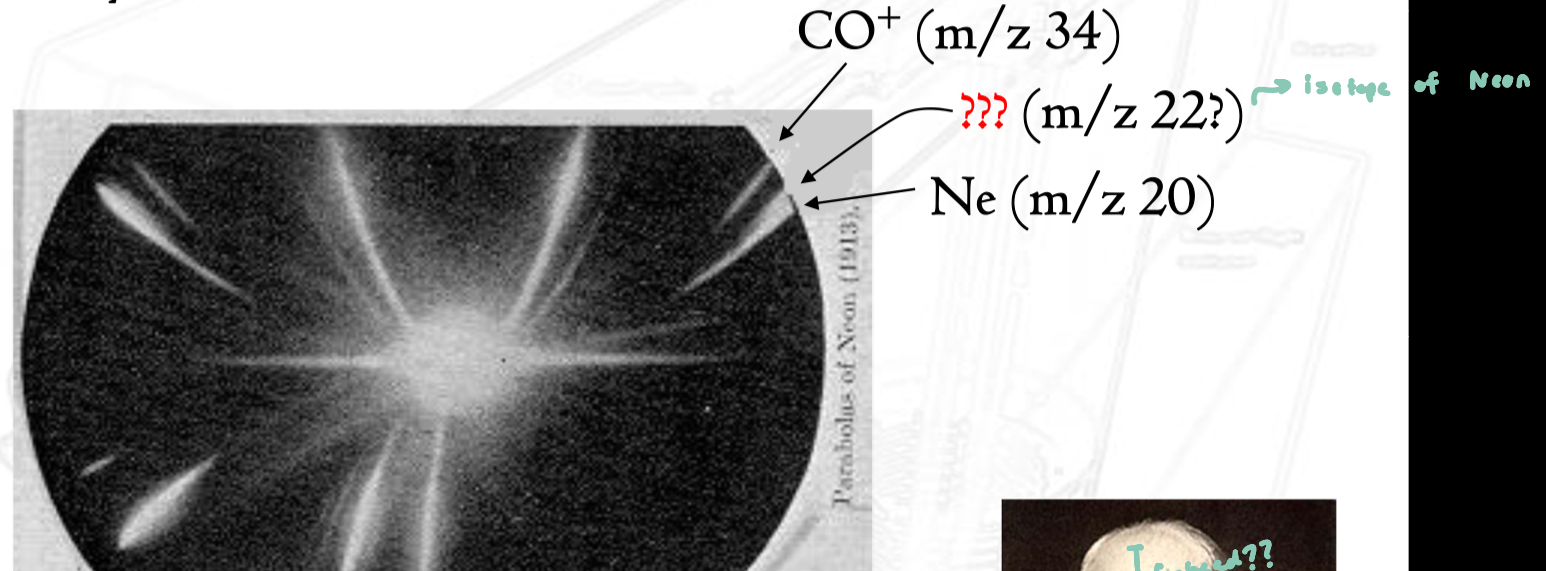
Mystery of Neon
the mass spectrum of Ne showed two distinct peaks instead of one
at the time, this could not be explained by existing atomic theory
Thomson thought he had discovered a new element
actual explanation: the two peaks were isotopes of neon
Preparative Mass Spec: Calutron
Calutron: large scale mass spec used for isotope separation
purpose: Separate U235 and U238 isotopes (purifying uranium)
mass spec is very sensitive
U atoms are ionized into charged particles and accelerated thru an electric field
ions are separated based on m/z, where light ions (U235) deflect slightly differently than heavier (U238)
Ionization
removal or addition of an electron
Energy required depends on how tightly a nucleus binds its highest energy electrons
outer orbit e-’s are easier to pull off
biomolecules (larger molecules) tend to break apart
you risk breaking apart the molecule when trying to peel off just one e-
noble gases are most reluctant to give up e-
Soft Ionization - MALDI
usually N2 laser (337 nm, 3-10 ns pulses)
very short, high energy pulses that rapidly excite the matrix
causes desorption and ionization in a very short burst
soft method: minimizes fragmentation of biomolecules
how ionization by MALDI occurs is poorly understood (what’s going on is occurring at the atomic/molecular level isn’t understood)
MALDI Matrices
biological samples coated by a matrix
must absorb light at wavelengths emitted by laser
efficient at converting light energy to heat
charge-transfer reactions
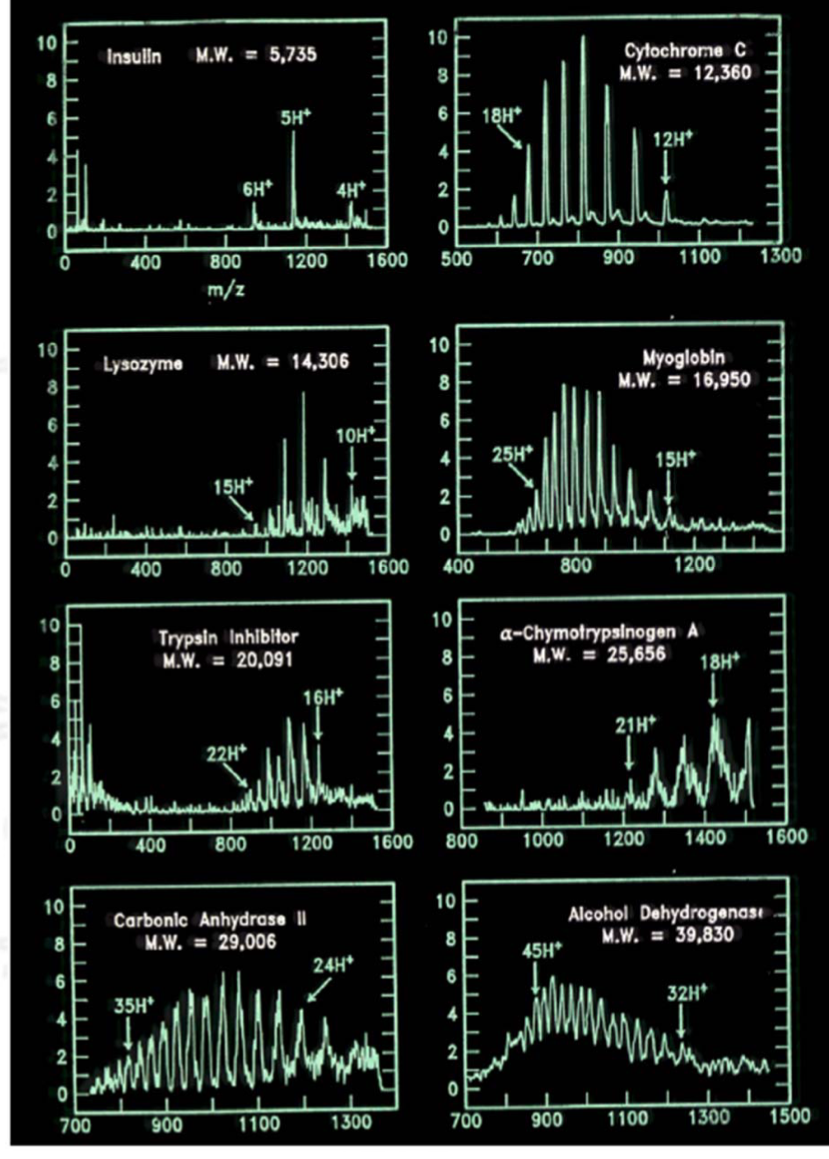
First ESI-MS of Proteins
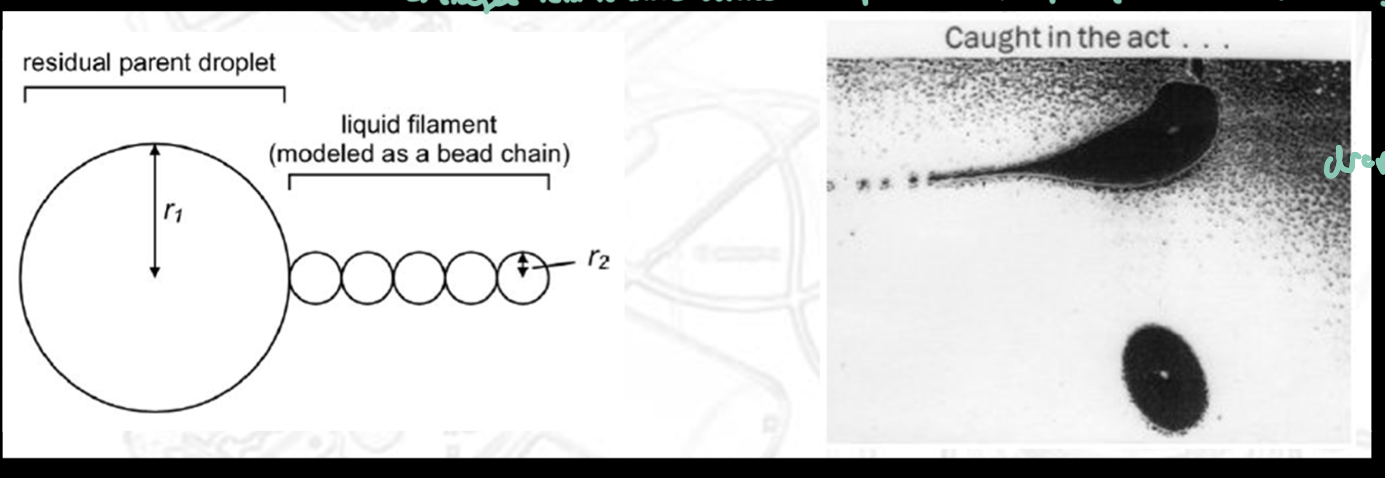
ESI: Taylor Cone
When high voltage is applied to a liquid at the capillary tip, the liquid does not spray straight out
Instead, it forms a cone-shaped meniscus called the Taylor cone
From the tip of this cone, a fine charged jet of liquid is emitted
The cone forms because…
Strong electric field pulls liquid outward; Surface tension pulls inward
The balance of forces creates a stable cone shape rather than a flat droplet surface
Charge distribution in droplets
Charges move toward the surface of the droplet due to Coulombic repulsion
This makes the droplet surface highly charged and unstable
ESI: Jet Fission (Droplet Breakup)
As solvent evaporates, droplets shrink and become unstable
They undergo Coulombic fission (explosive breakup)
During this process:
Small fragments of the droplet are ejected
Mass loss is relatively small (~2–5%)
Charge loss is proportionally larger (~20%) because charge is concentrated at the surface
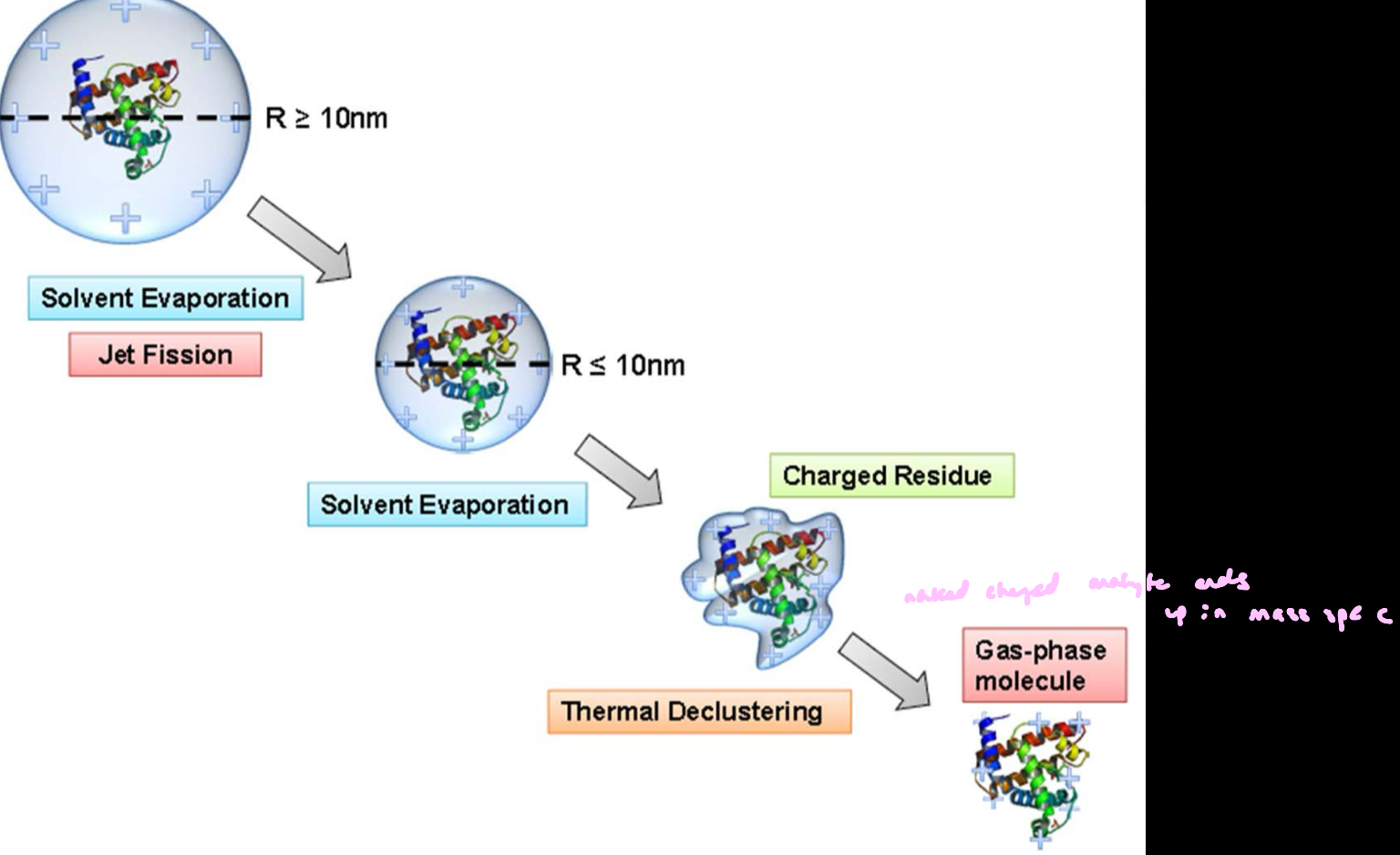
ESI: Charged Residue Model
Each droplet contains a single analyte molecule (e.g., a protein) as it gets smaller.
A charged droplet is formed from the Taylor cone jet
Solvent evaporates → droplet continuously shrinks
Repeated Coulomb fission reduces droplet size
Eventually, a droplet contains one analyte molecule + remaining charge
Final solvent evaporates completely
final outcome: The analyte is left as a charged, gas-phase ion; the charge originally on the droplet is transferred to the molecule