IR Spectroscopy
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Alkanes
C-H
H-C-H
C-C
C-H: <3000
H-C-H: ~1465
C-C: can’t interpret
Alkenes
=C-H
C=C
C=C-C=C
Weaker signal
=C-H: sp2 >3000
C=C: 1600-1660
C=C-C=C: conjugation shifts carbon-carbon streches to lower frequencies
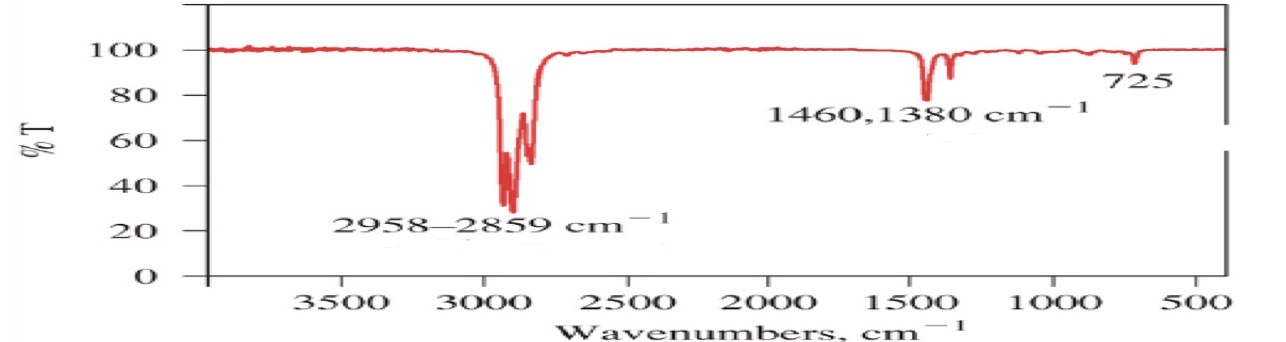
Alkanes
Alkynes
=-C-H
C-=C
=-C-H: sp ~3250-3350
C-=C: 2150
Nitriles
-C=-N
C=C-C=-N
-C=-N: ~2250
C=C-C=-N: decreases frequency of C=-N stretch (red shift)
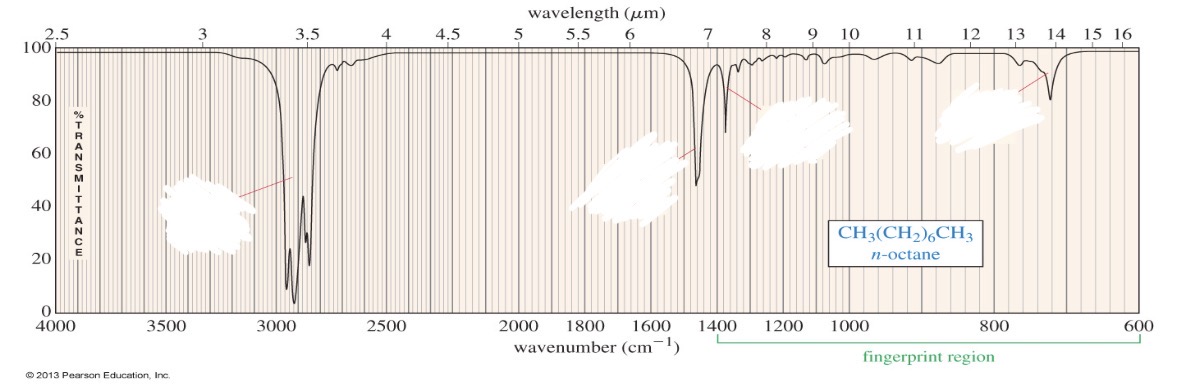
Alkanes
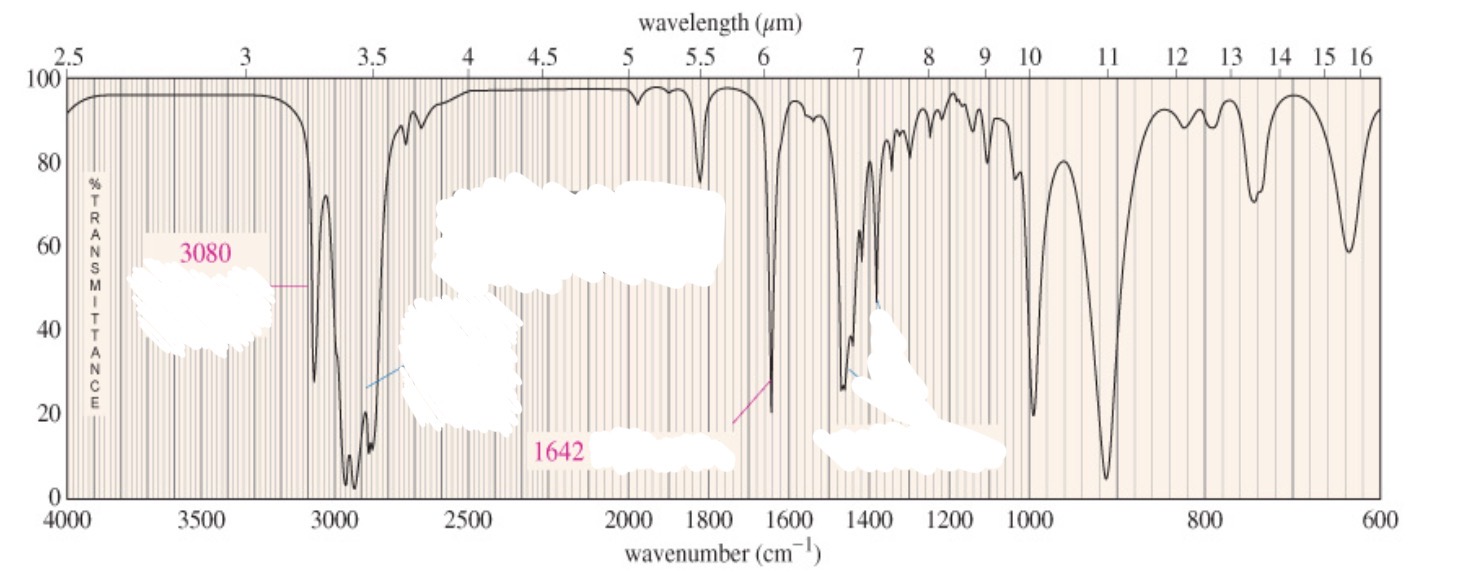
Alkene
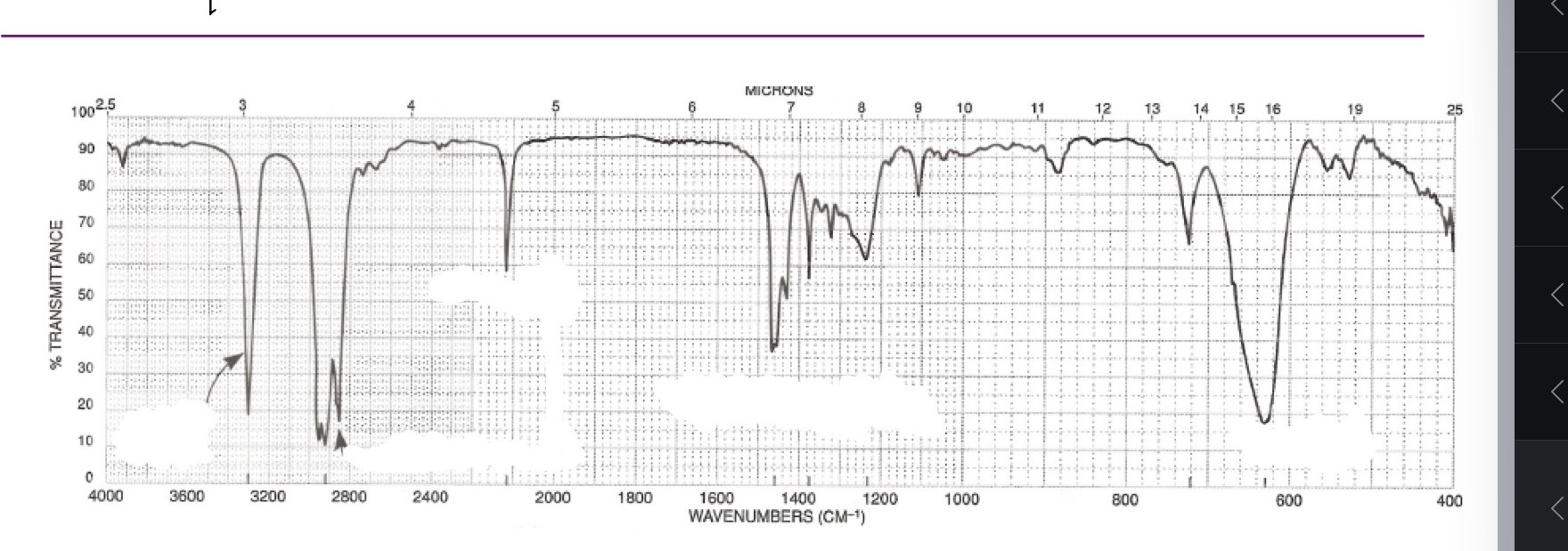
Alkynes
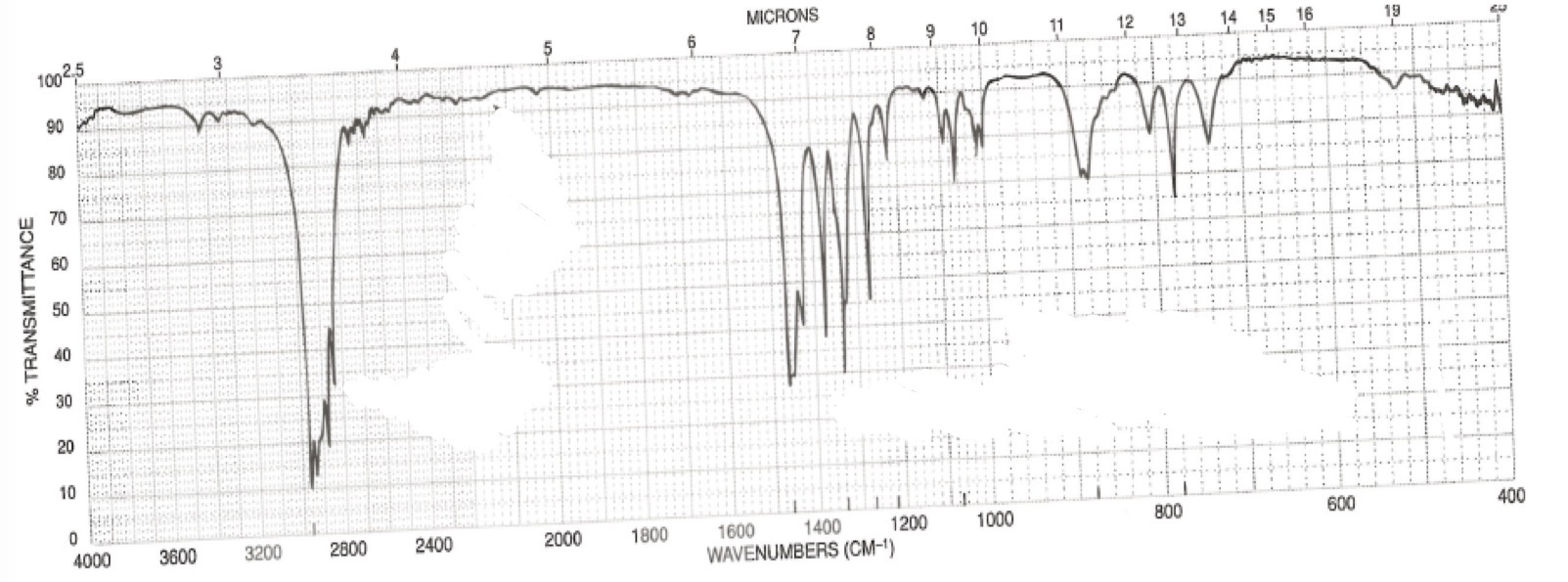
No C=-C stretch bc the alkyne is symmetrical
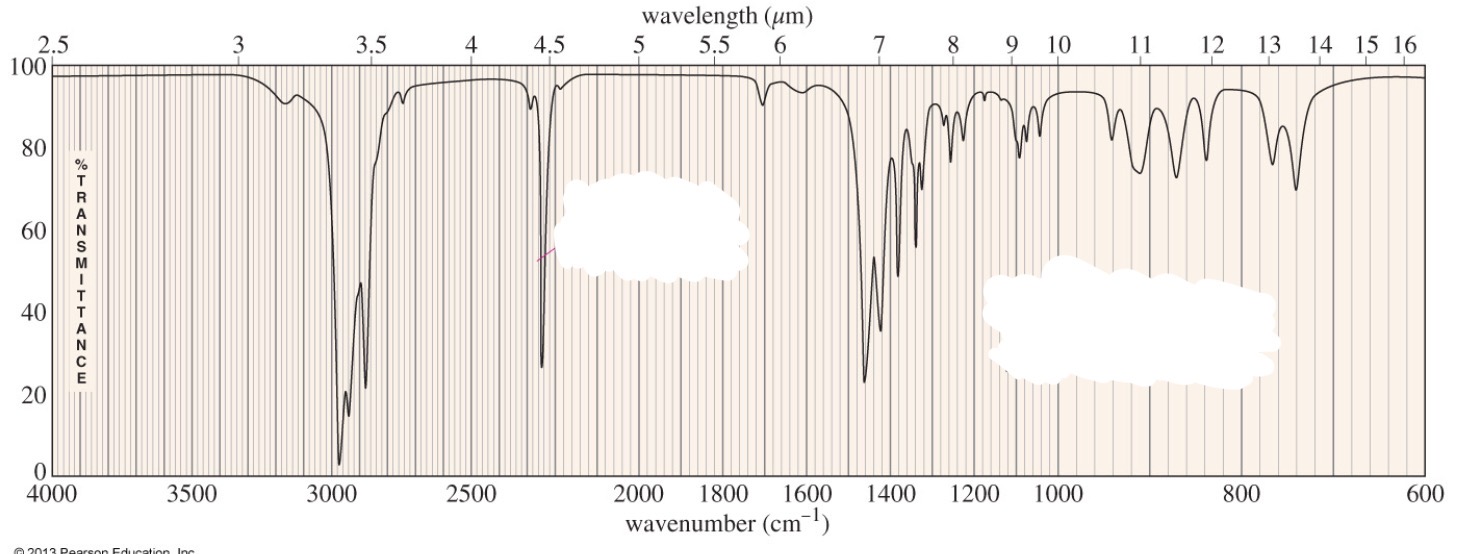
Nitrile
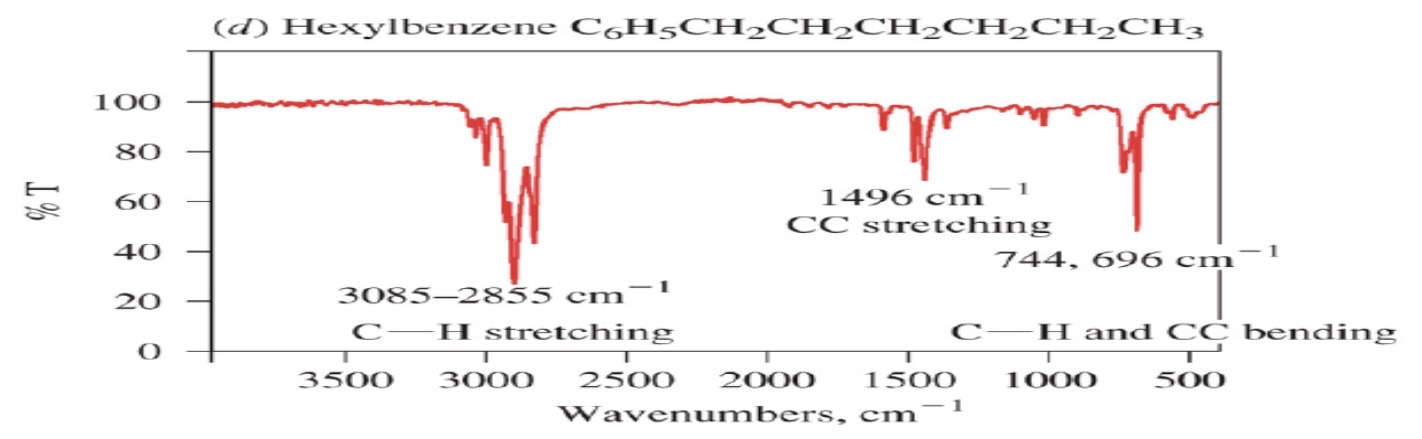
Aromatic compounds
=C-H: sp2
C=C:
isolated C=C:
conjugated C=C:
aromatic C=C:
=C-H: sp2: >3000
C=C:~1600 and ~1475
isolated C=C: 1640-1680
conjugated C=C: 1620-1640
aromatic C=C: ~1600
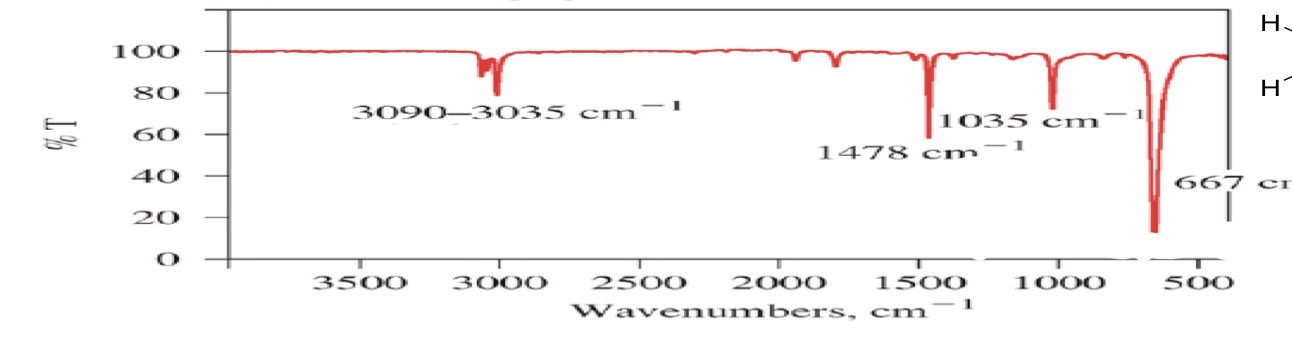
Aromatic
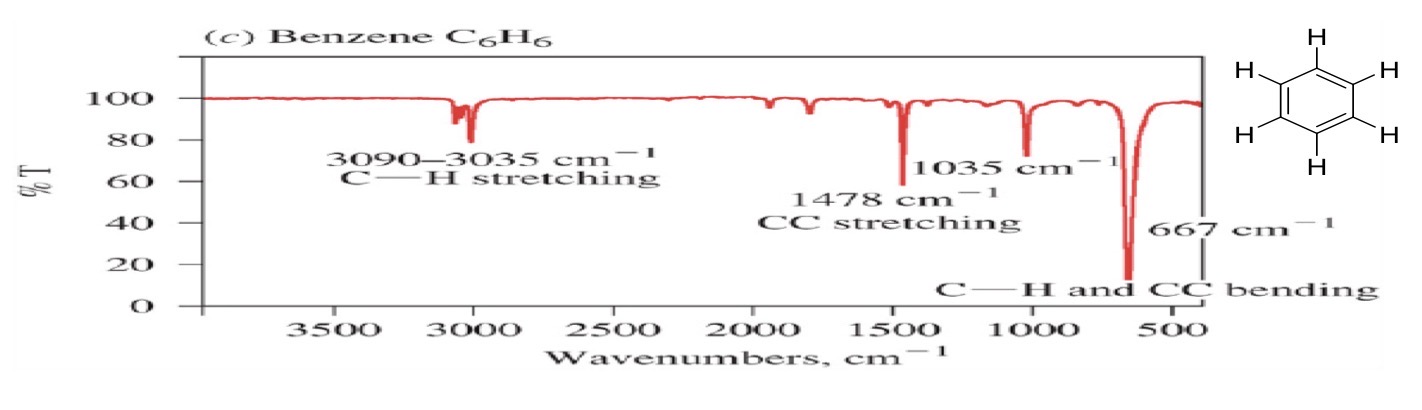
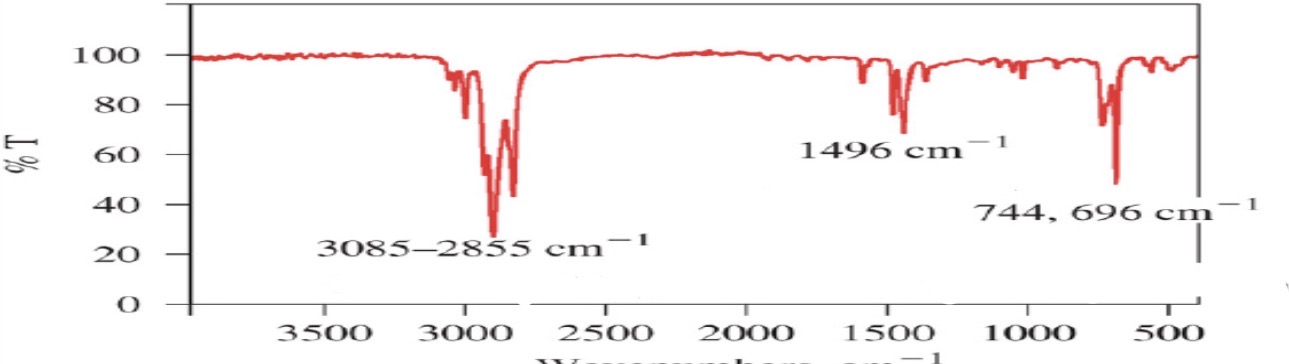
Aromatic
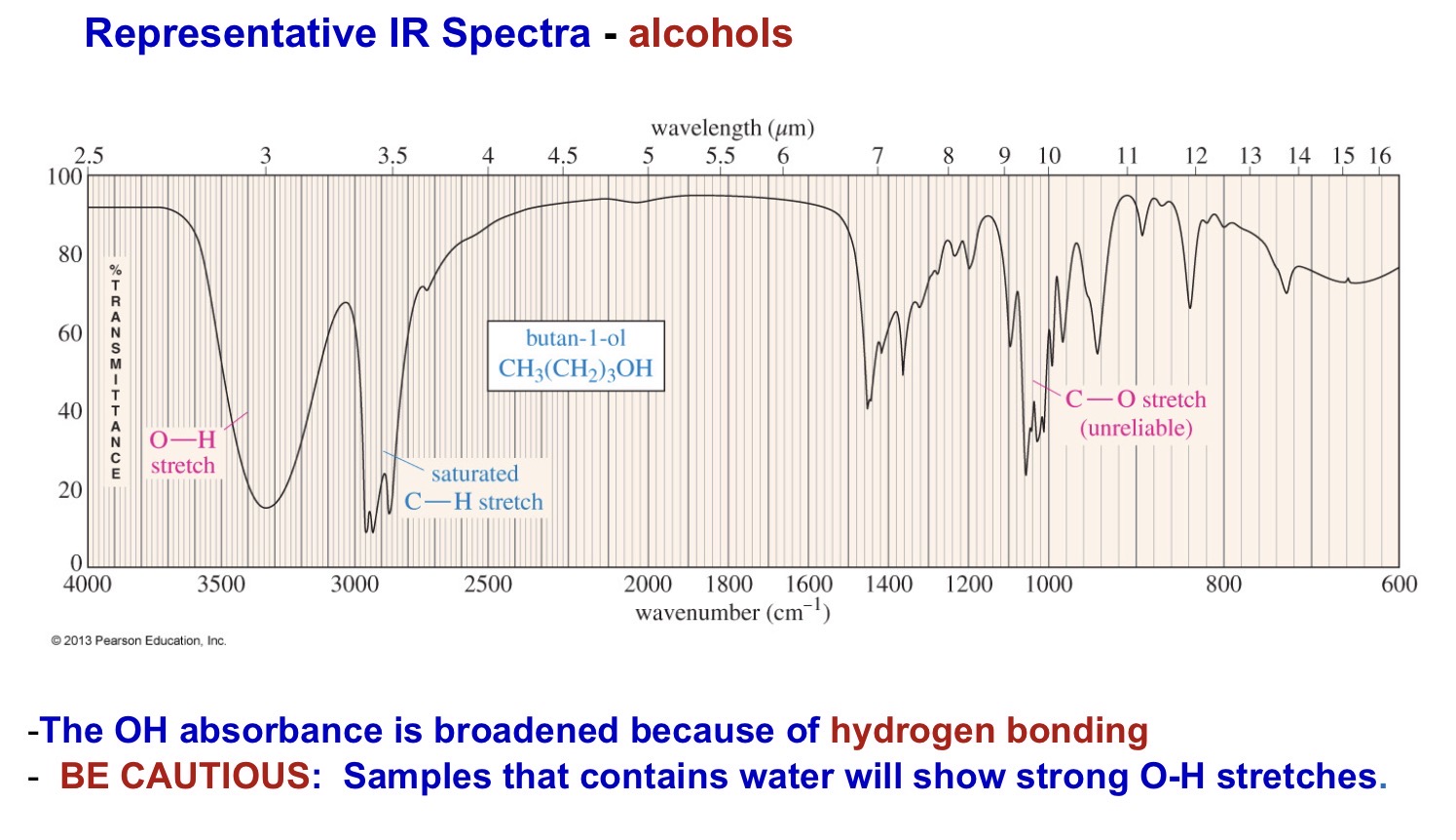
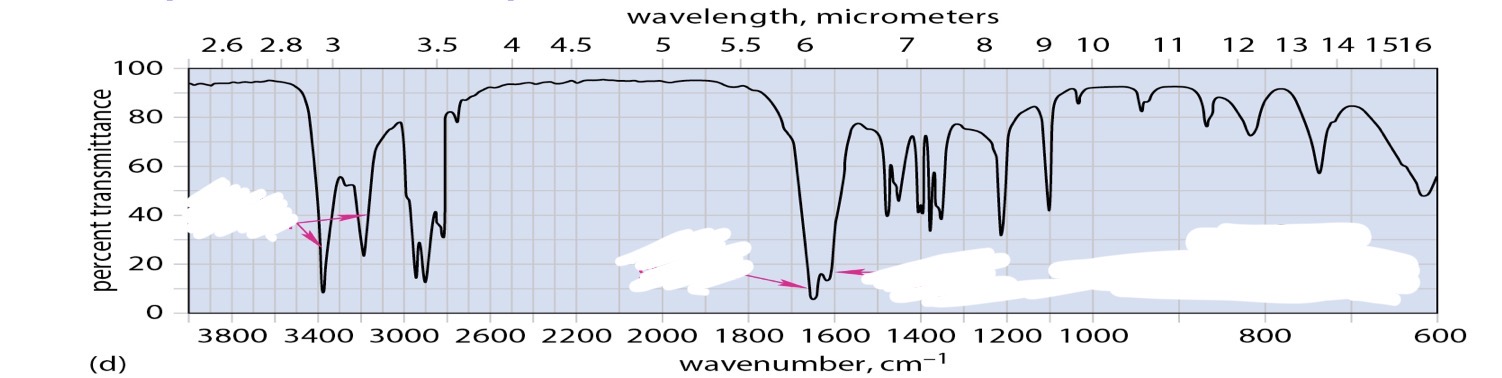
Alcohol
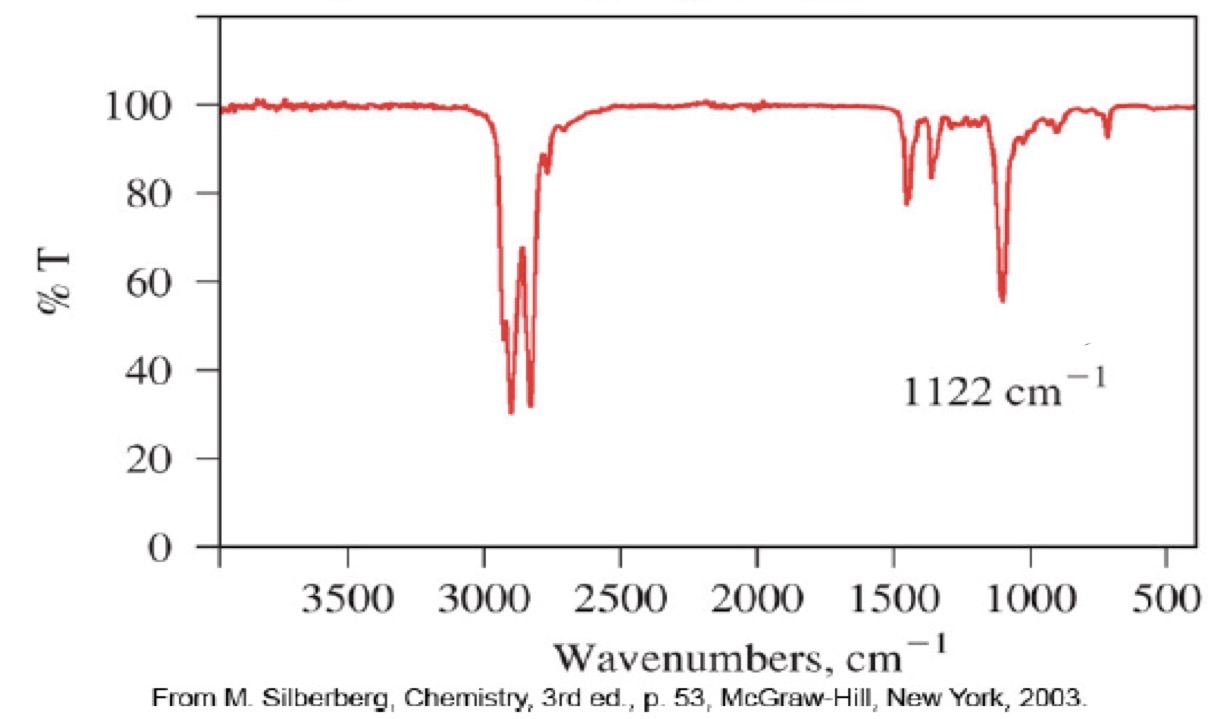
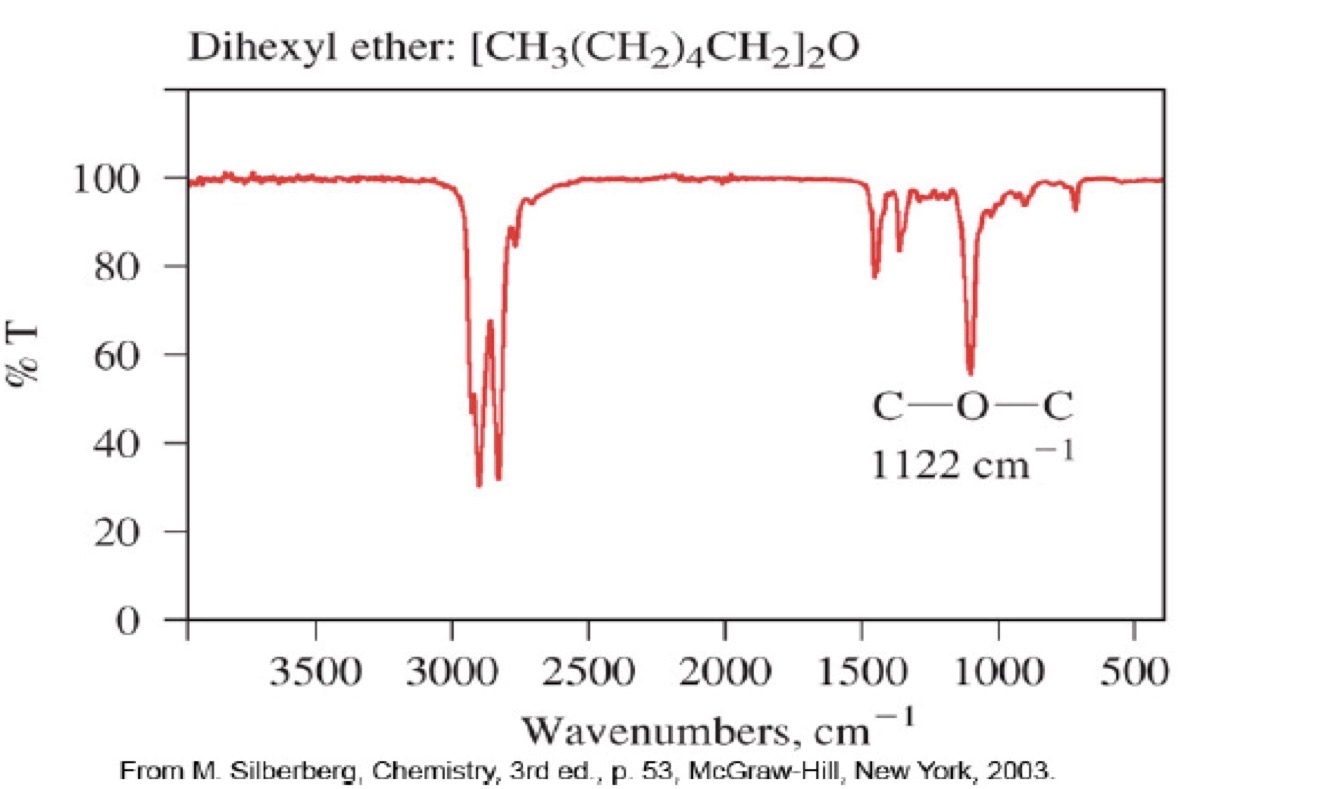
Ether
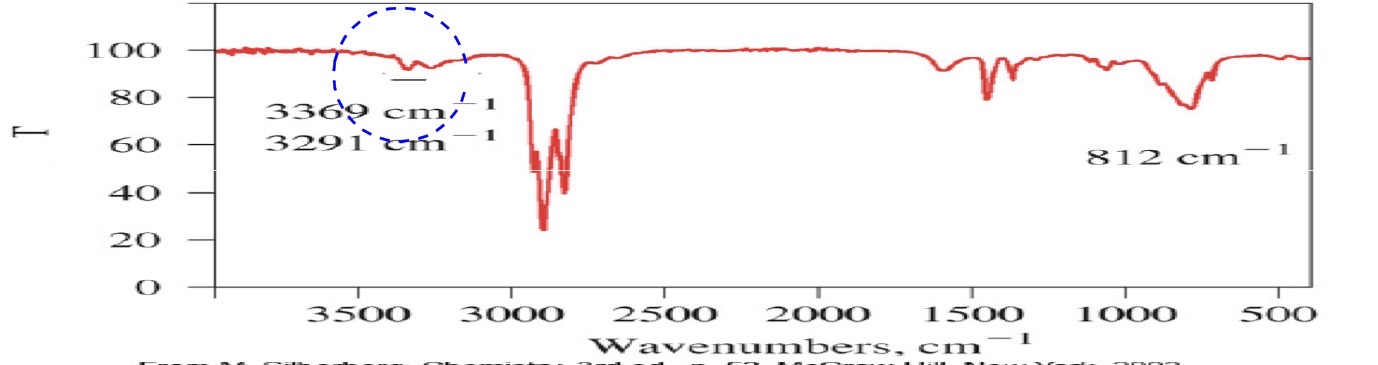
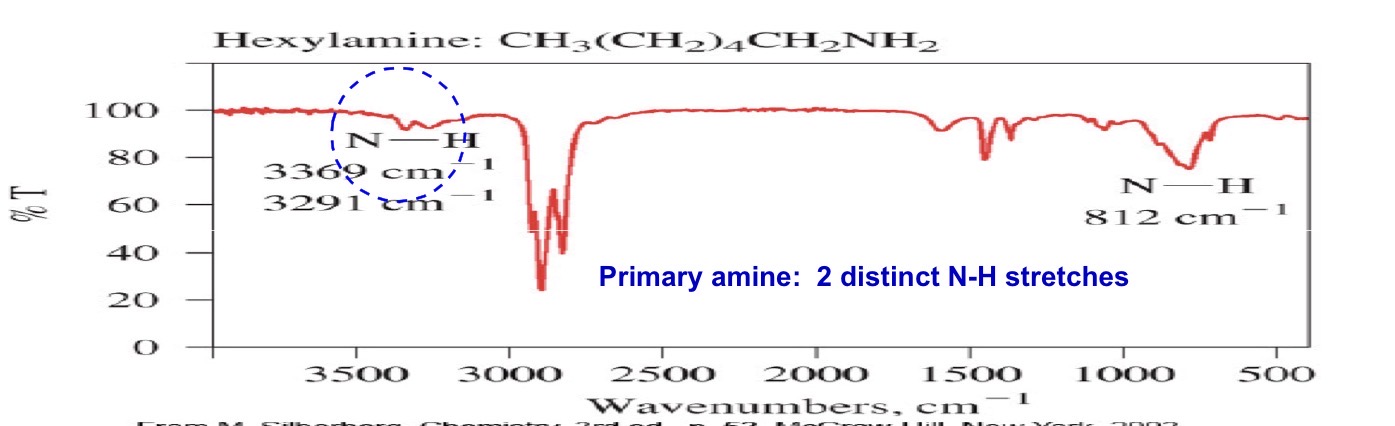
Primary amine
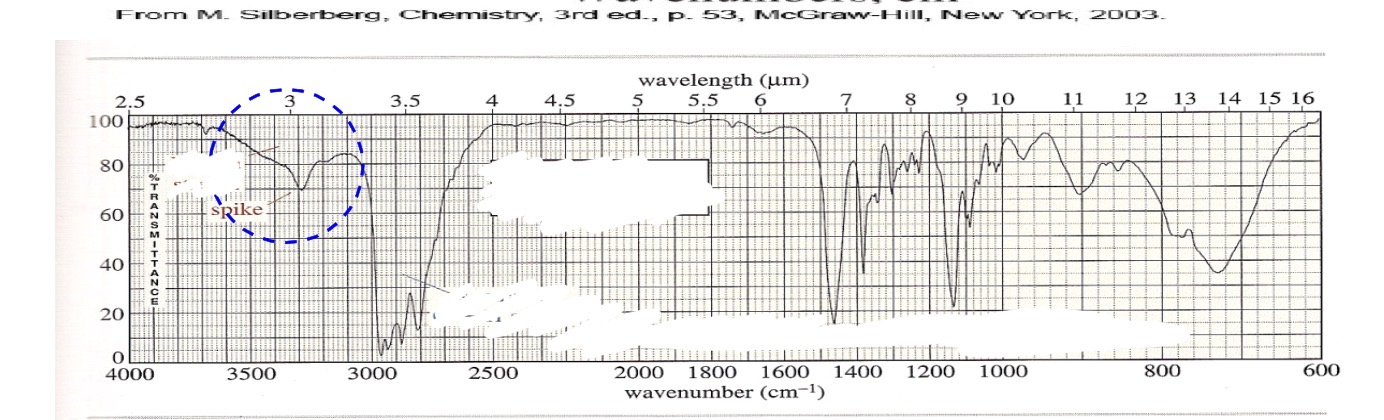
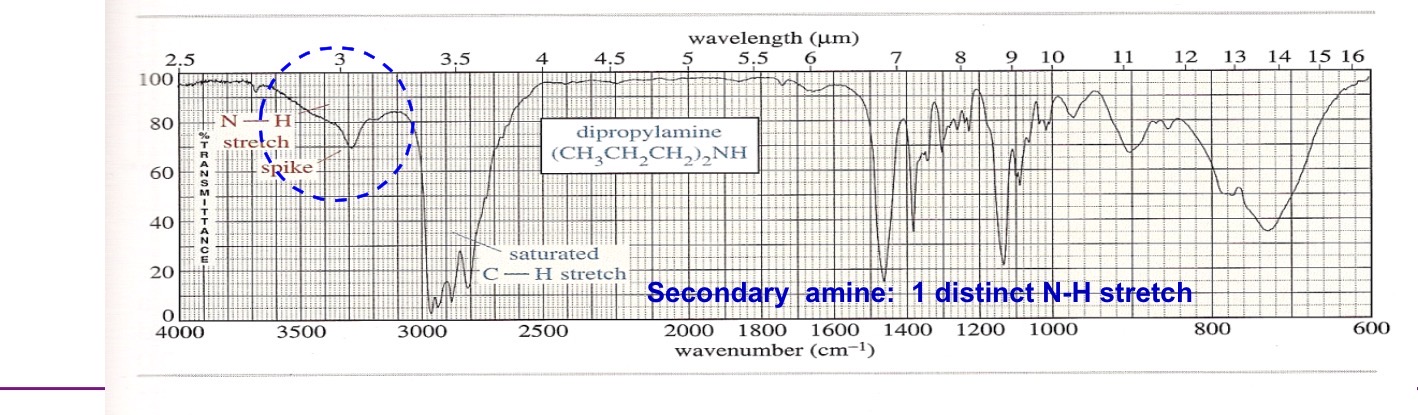
Secondary amine
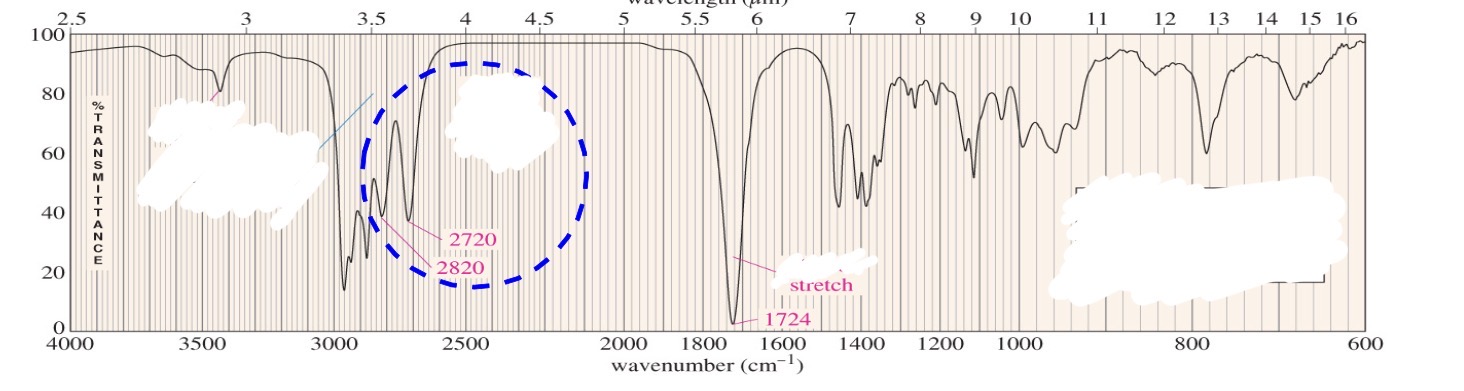
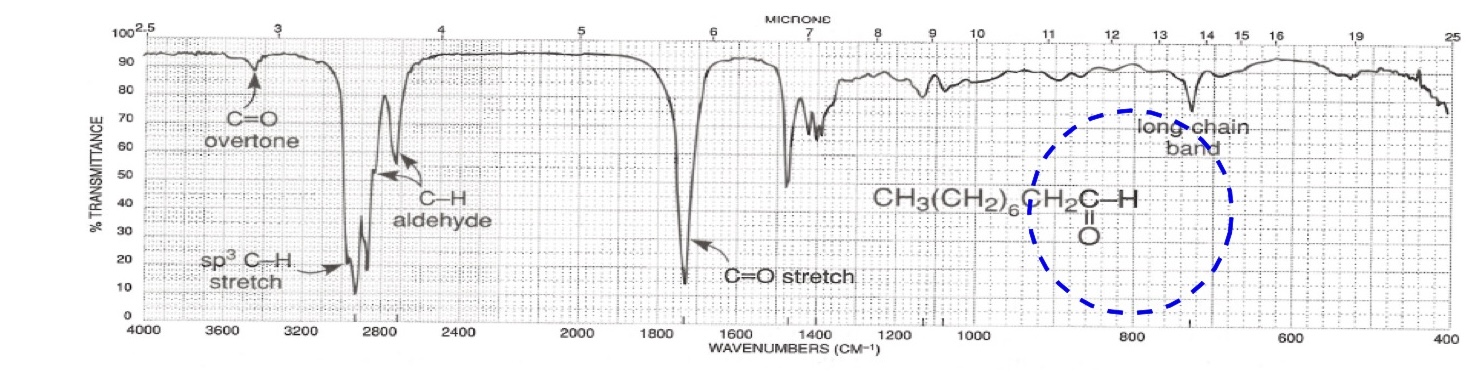
Aldehyde
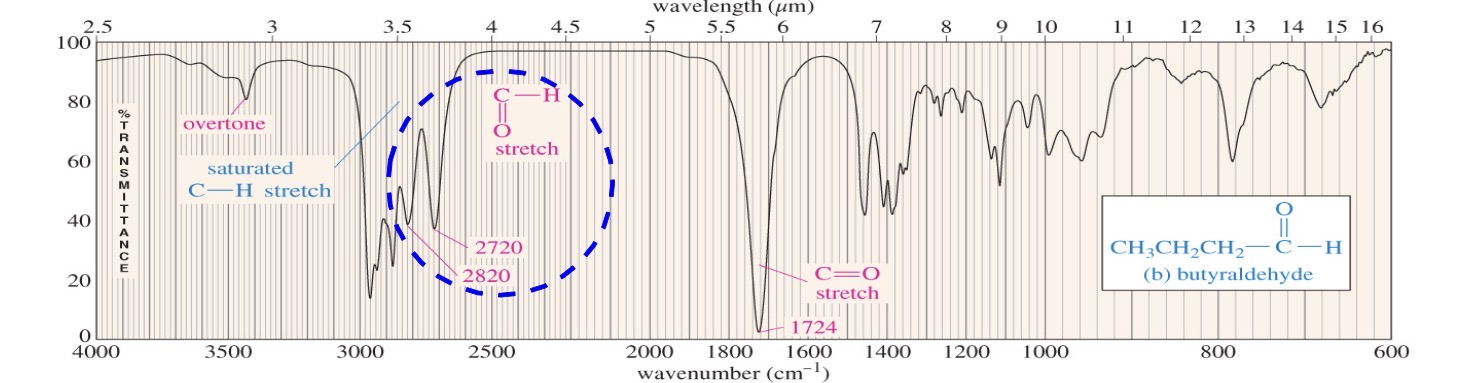

Aldehyde
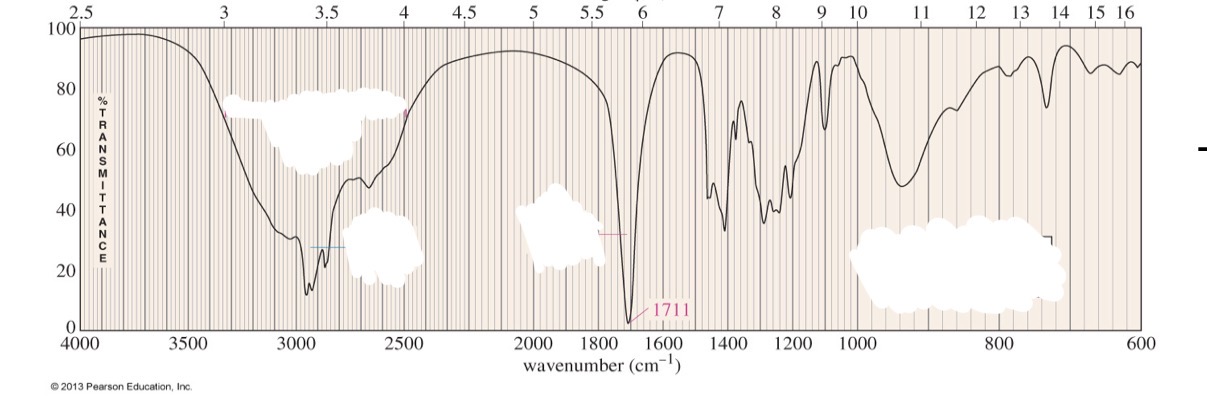
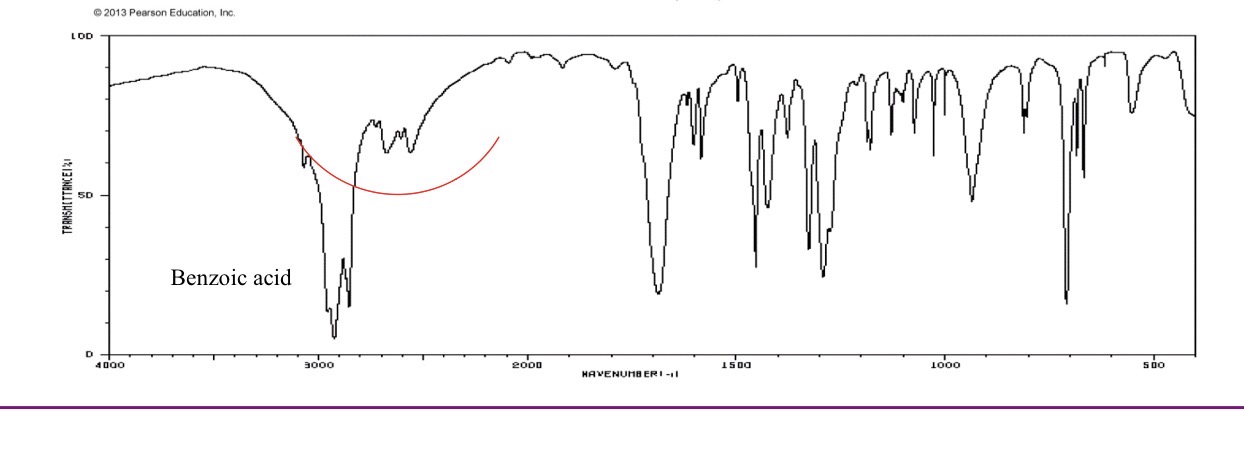
Carboxlyic acid
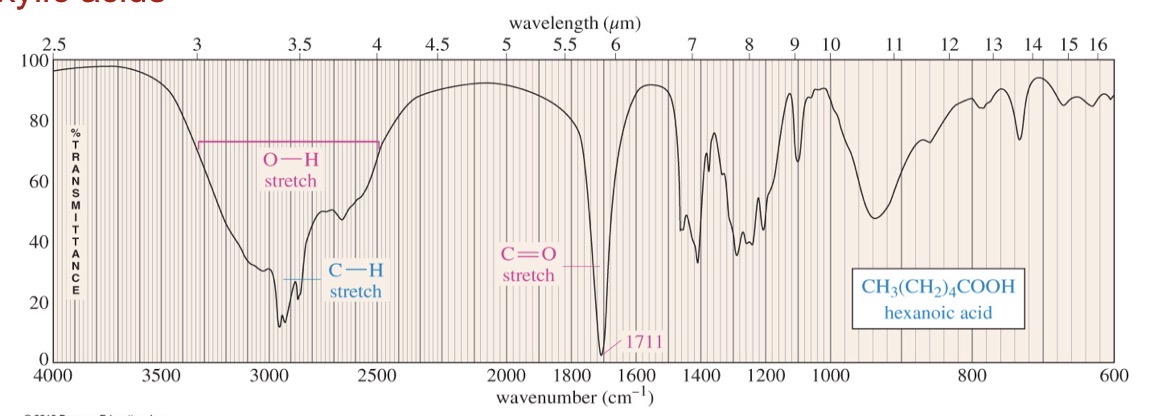
Carboxlyic acid
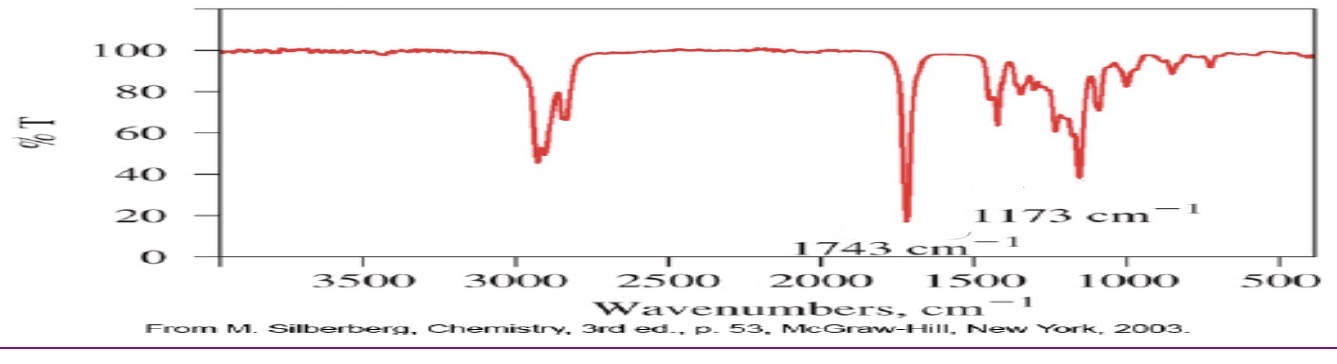
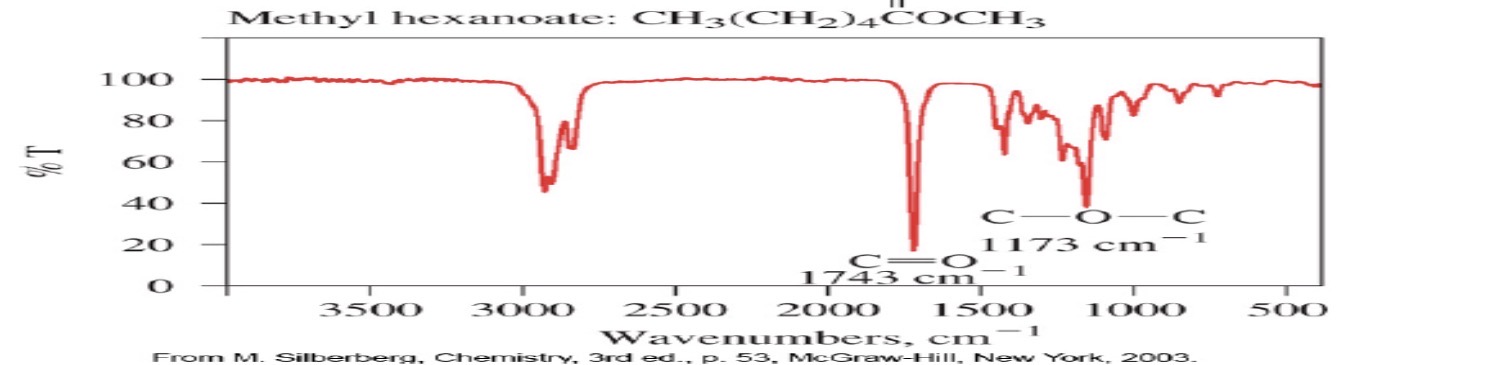
ester
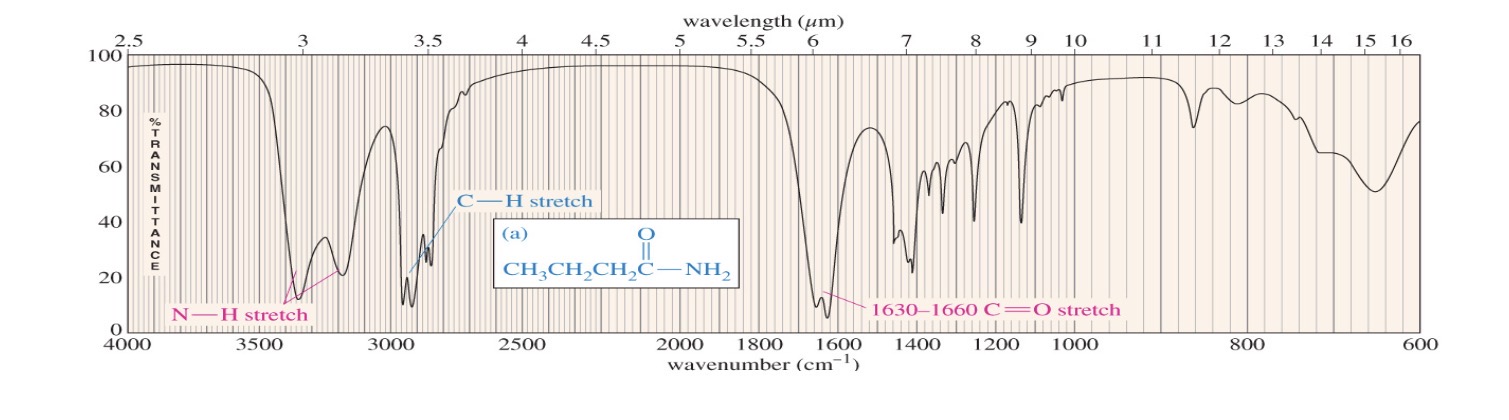
Amide primary 2 stretches
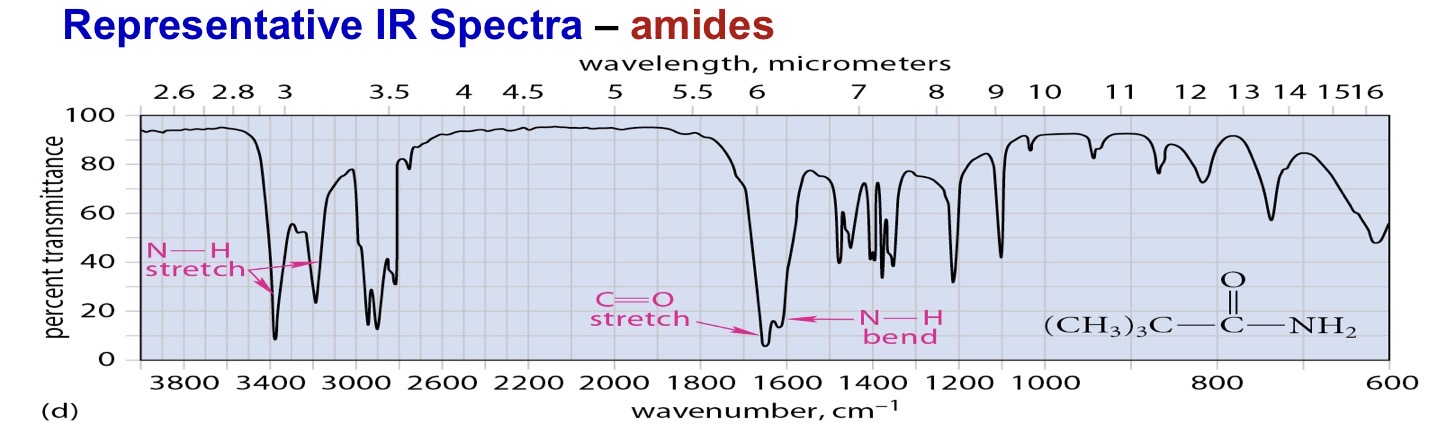
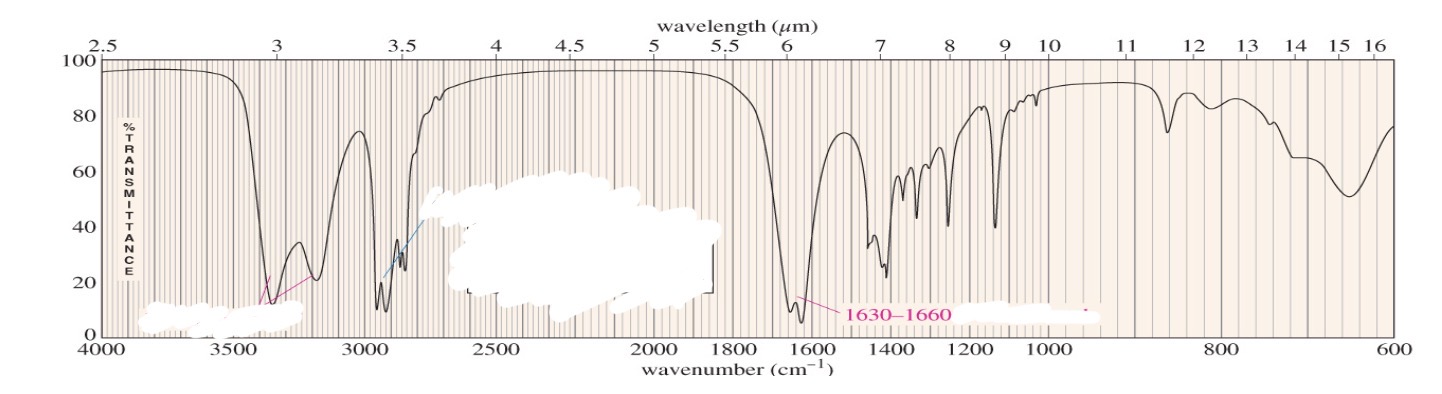
Primary amide 2 N-H stretches

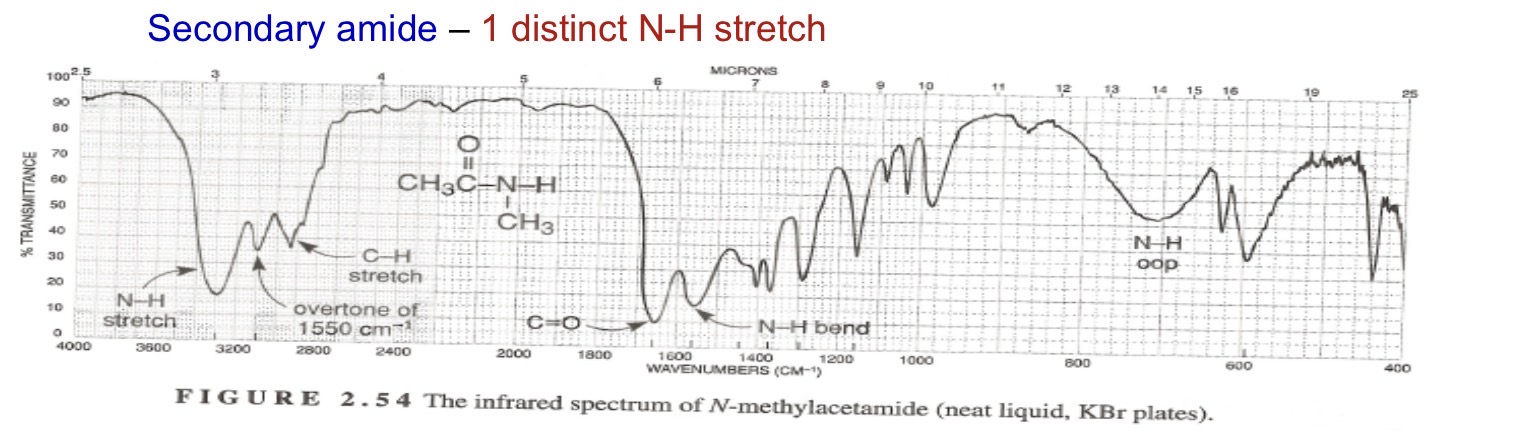
Secondary amide 1 stretch
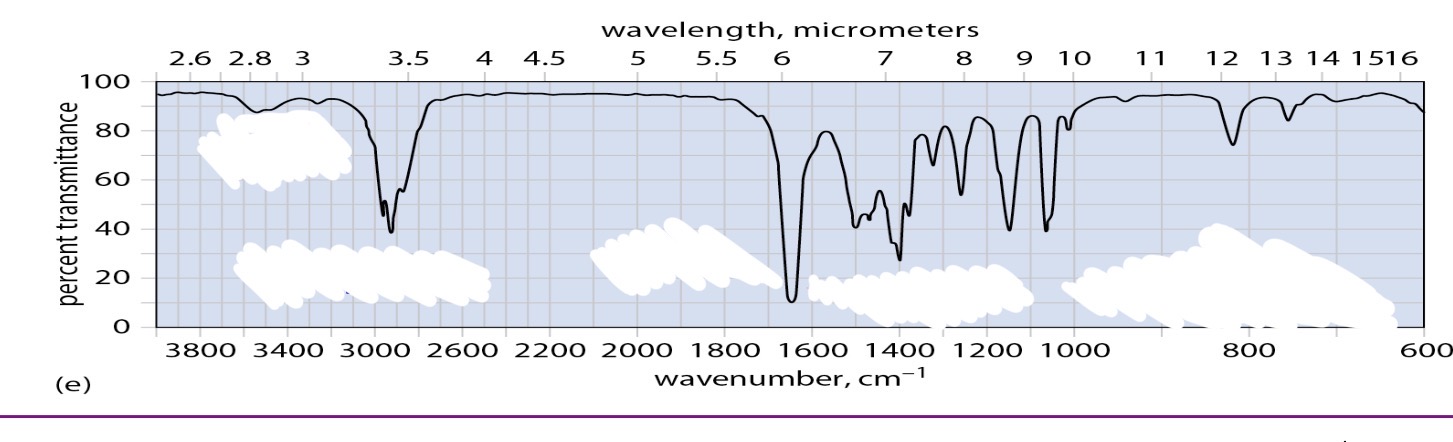
Teritary amide no stretches
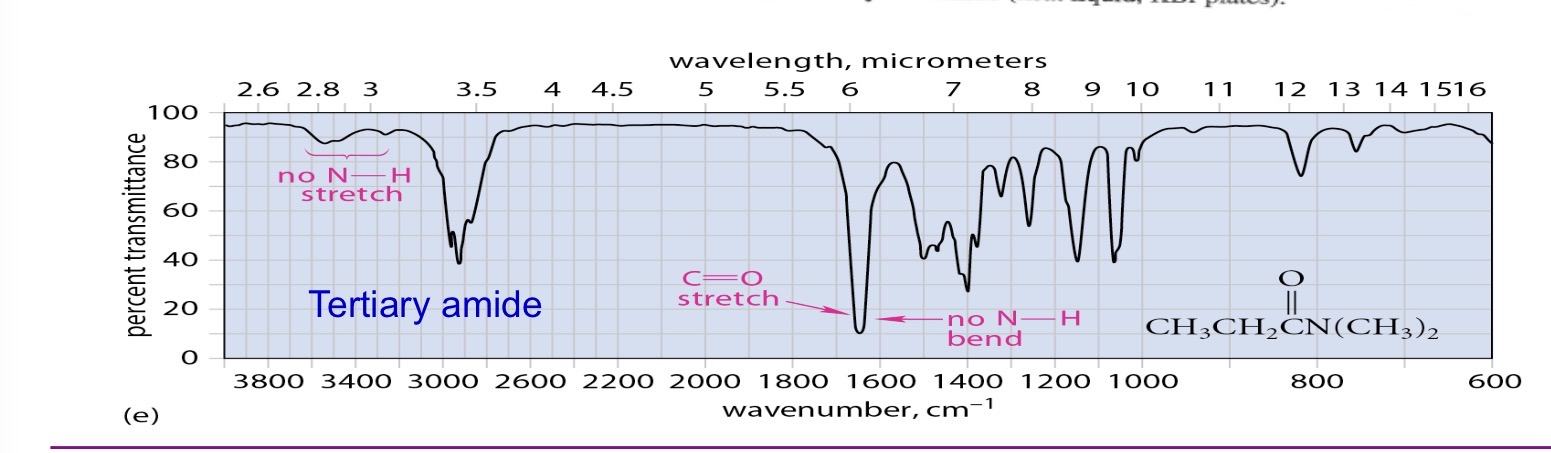
Red shift
more single bond character so vibrates at lower frequences
due to resonance