CHEM100 LEC EXAM 2
1/77
Earn XP
Description and Tags
CHEM 100 LEC
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
What are Ionic Compounds?
Metal + Nonmetal
Metal + Nonmetal is what Compound?
Ionic Compound
What are Molecular/Covalent Compounds?
Nonmetal + Nonmetal
Nonmetal + Nonmetal is what Compound?
Molecular/Covalent Compound
What is an Acid?
Formula begins with hydrogen (EXCEPT H2O). Examples: HCI, HNO3, H3PO4, H2SO4
What is a Covalent?
Bond between 2 nonmetals. Examples: CO2, H2O, I4O9, BrF2, N2O, CCL4
What is a Salt?
Contains a Metal/Ammonium (NH4+) and an anion other than Hydroxide (OH-). Examples: NaCl, FeS, NH4I, MgSO4, AgNO3, K2O
What is a Base?
Contains a metal and hydroxide (OH-). Examples: KOH, Al(OH)3, Ba(OH)2, Ca(OH)2, Fe(OH)2, NaOH
What is a Polar Covalent Bond?
Bond between 2 different nonmetals. Examples: H2O, HCI
What is London?
A bond between two nonpolar molecules. Examples: Cl2, CH4, Ar
What is a Dipole-Dipole Bond?
A bond between the partially positive end of one polar molecule and the partially negative end of another polar molecule
What is a Nonpolar Covalent Bond?
Bond between the same 2 nonmetals. Examples: O2, Cl2, N2
When does Hydrogen Bonding Happen?
When hydrogen is directly bonded to: N, O, F. Look For: H-N, H-O, H-F
In a given atom, the Maximum Number of Electrons that can occupy the Fifth Shell is:
50 electrons
In a given atom, the Maximum Number of Electrons that can occupy the Fourth Shell is:
32 electrons
In a given atom, the Maximum Number of Electrons that can occupy the Third Shell is:
18 electrons
In a given atom, the Maximum Number of Electrons that can occupy the Second Shell is:
8 electrons
In a given atom, the Maximum Number of Electrons that can occupy the First Shell is:
2 electrons
In a given atom, the Maximum Number of Electrons that can occupy the Sixth Shell is:
72 electrons
In a given atom, the Maximum Number of Electrons that can occupy the Seventh Shell is:
98 electrons
A bond in which an Electron Pair is equally Shared by Two Atoms is
Nonpolar covalent
What is a Mole in Chemistry?
6.02 X 10^23
An Cation is an atom that has blank in a Chemical Reaction
Lost an electron
An anion is an atom that has blank in a Chemical Reaction
Gained an electron
Which Subatomic Particles contribute substantially to the Mass of the Atom?
Protons and Neutrons
In a Single Covalent Bond, there are blank Electrons shared between Two Atoms
2
In a Double Covalent Bond, there are blank Electrons shared between Two Atoms
4
In a Triple Covalent Bond, there are blank Electrons shared between Two Atoms
6
What Type of Elements tend to Lose their Outer Electrons easily?
Metals
What Type of Compound may have a Partially Negative and a Partially Positive Charge?
Covalent/Molecular
The Expected Charge for the Iron ion is
2+ and 3+
The Expected Charge for the Cobalt ion is
2+ and 3+
The Expected Charge for the Nickel ion is
2+ and 3+
The Expected Charge for the Copper ion is
+ and 2+
The Expected Charge for the Zinc ion is
2+
The Expected Charge for the Aluminum ion is
3+
The Expected Charge for the Silver ion is
+
The Expected Charge for the Gold ion is
+ and 3+
The Expected Charge for the Mercury ion is
+ and 2+
The Expected Charge for the Tin ion is
2+ and 4+
The Expected Charge for the Lead ion is
2+ and 4+
The Expected Charge for the Antimony ion is
3+ and 5+
How can you estimate Electronegativity?
By where the elements are located on the periodic table (How far apart)
Metals/Positive Charges Blank Electrons
Lose Electrons
Nonmetals/Negative Charges Blank Electrons
Gain Electrons
What is the Formula of Mass?
Mass=Moles X Molar Mass
What is the Formula of Moles?
Moles=Grams divided by Molar Mass
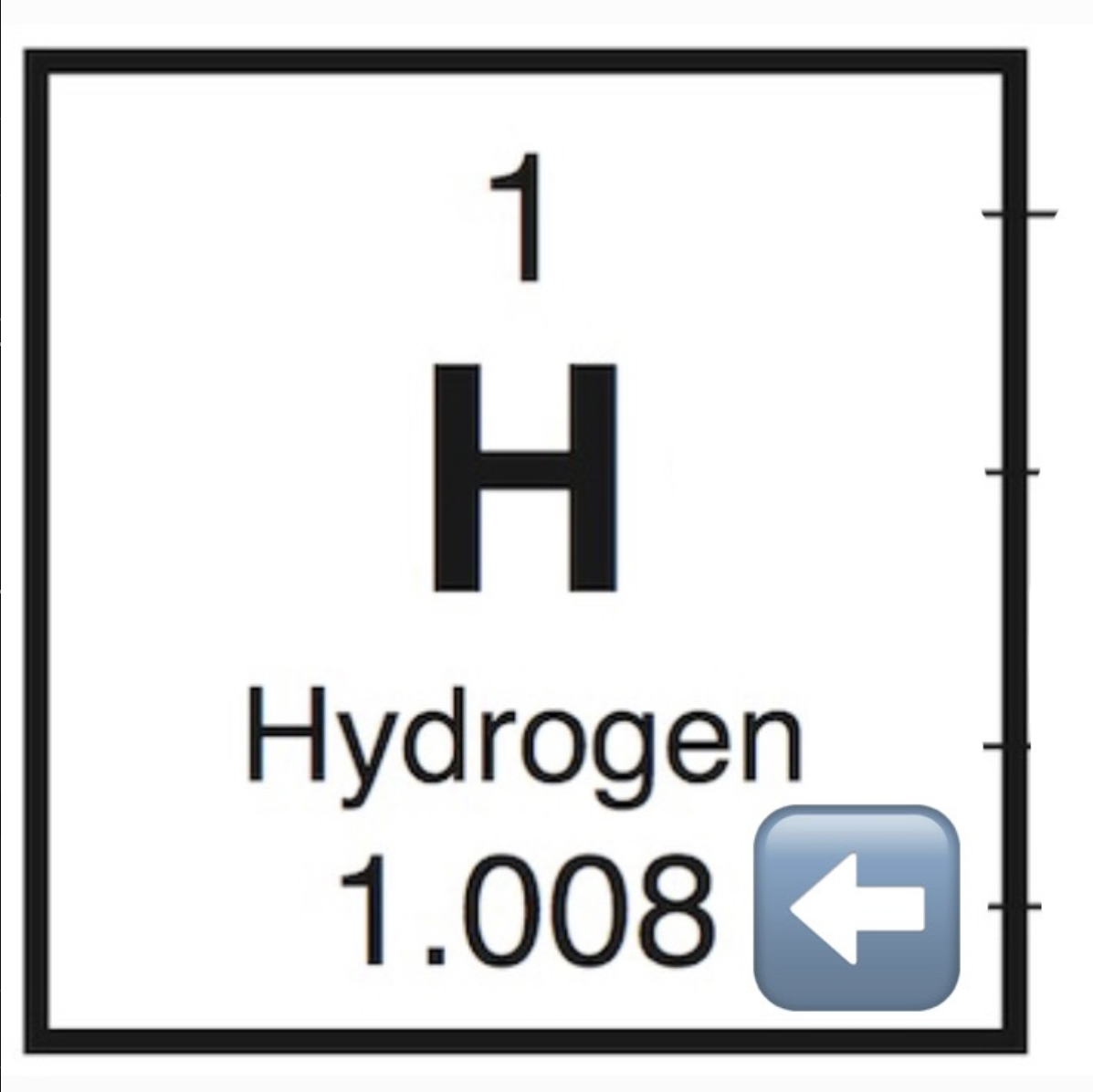
Atomic Mass
OH-
Hydroxide
C2H3O2-
Acetate
NO2-
Nitrite
NH4+
Ammonium
NO3-
Nitrate
HSO3-
Bisulfite
HSO4-
Bisulfate
HCO3-
Bicarbonate
ClO3-
Chlorate
ClO4-
Perchlorate
SO3²-
Sulfite
SO4²-
Sulfate
Co3²-
Carbonate
C2O4²-
Oxalate
PO4³-
Phosphate
Hydroxide
OH-
Acetate
C2H3O2-
Nitrite
NO2-
Nitrate
NO3-
Ammonium
NH4+
Bisulfite
HSO3-
Bisulfate
HSO4-
Bicarbonate
HCO3-
Chlorate
ClO3-
Perchlorate
ClO4-
Sulfite
SO3²-
Sulfate
SO4²-
Carbonate
CO3²-
Oxalate
C2O4²-
Phosphate
PO4³-