unit 4: systems at equilibrium
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
reversible reaction
a chem reaction that proceeds in the forward and reverse directions
chemical equilibrium
a state in a chemical system in which the forward and reverse reaction are occuring at the same rate
dynamic equilibrium
A chem system at equilibrium that is changing at the molecular level, but macroscopic properties are constant
characteristics of dynamic eq.
rate of fwd rxn = rate of rev rxn
Macroscopic properties (ex. color, pressure) are constant
The system is closed
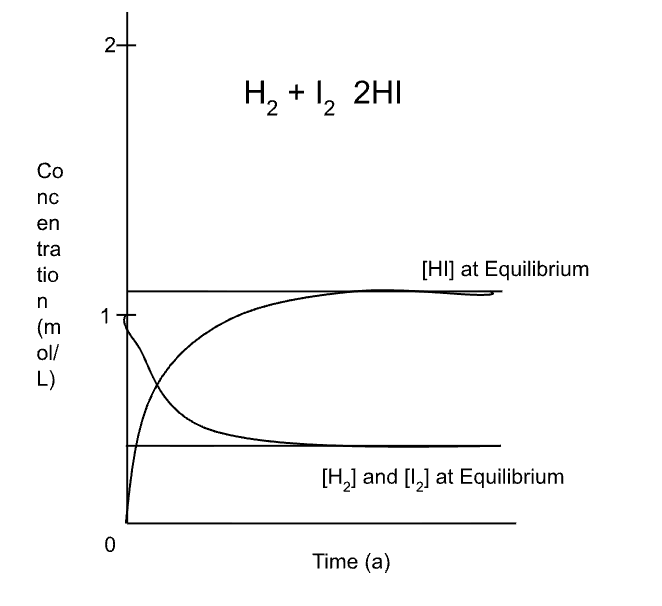
equilibrum graph - fwd rxn (Ex: H2 + I2 → 2HI)
* Remember, chem equilibrium is where rate fwd rxn = rate bwd rxn, so the rate of reactants consumed equals the rate of products formed.
H2 and I2 have a conc of 1, so they start at one and get consumed
2HI starts at 0, since none created, and more gets formed.
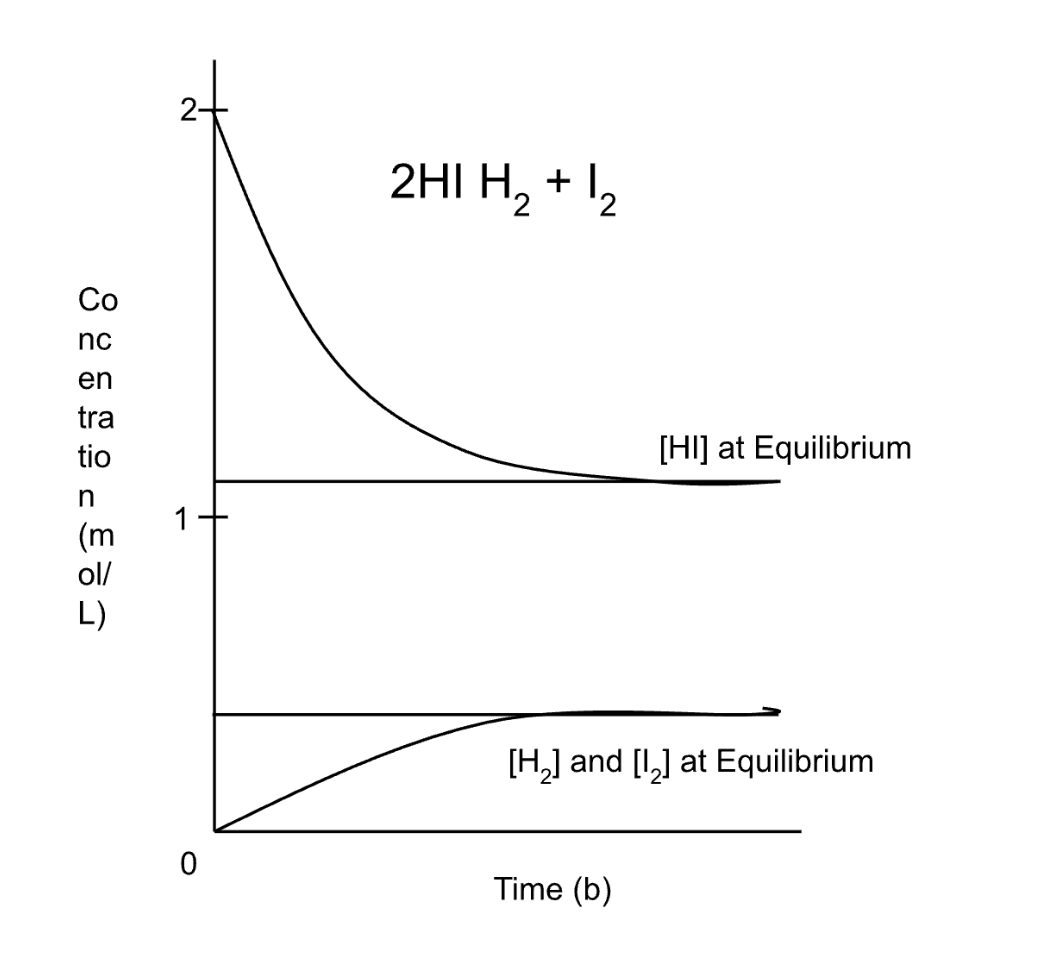
equilibrium graph - rev rxn (2HI —> H2 + I2
2HI starts at conc of 2, and gets consumed, while the prod start at zero and get formed.
equilibrium constant, Keq
The ratio of equilibrium concentrations for a particular chemical system at a particular temperature
what is Keq equal to
aA + bB ↔ cC + dD K = ([C]c[D]d)/