Medical Case Module 6: Quantum Model of Light
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
Are the next two scenarios true or false?
False, False
What is the peak wavelength for a blackbody at 4800 K? For a blackbody at 1000 K?
604 nm, 2900 nm
What is the single photon energy for light of wavelength 400 nm? For light of wavelength 650 nm?
3.1 eV, 1.9 eV
Calculate the emission spectra for a material with the following energy levels: -7.50 eV, -5.0 eV, and -3 eV. Tip: there will be 3 wavelengths.
275 nm, 496 nm, and 620 nm
The emission spectra of a low pressure source includes wavelengths at 510 nm and 610 nm. If the ground state energy level is -15.00 eV calculate two different possibilities for the other energy levels.
(-12.97 eV, -12.57 eV) and (-12.57 eV, -10.54 eV)
A substance has energy levels of: Eo = -15.00 eV, E1 = -13.00 eV, and E2 = -10.50 eV. Can this material fluoresce? If so, calculate the energy of the incident wavelength needed to excite the material and calculate the wavelengths of emission of the fluorescence.
Yes, it can. Incident energy: +4.5 eV. Wavelengths of emission: 496 nm and 620 nm.
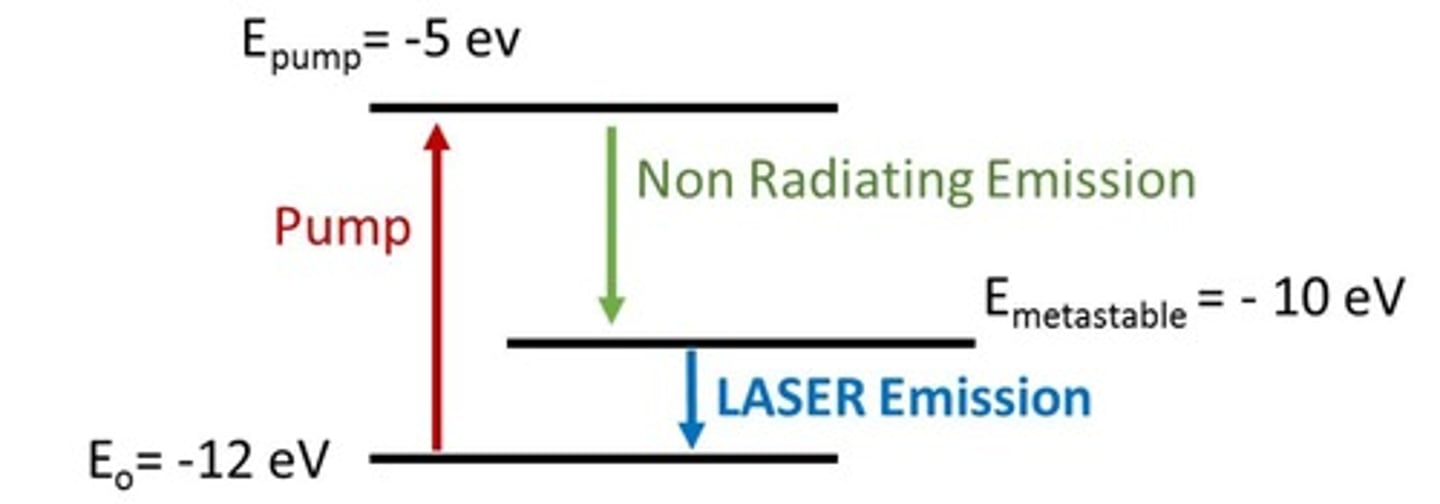
The energy levels of a laser are described on the graph below. Eo = -12.00 eV, Epump = -5.00 eV, and Emetastable = -10 eV. Calculate the energy of the incident wavelength needed to pump electrons and the wavelength of emission of the LASER.
Energy of the incident wavelength: 177 nm. LASER emission on 619.5 nm.
Why the laser used for LASIK surgery has a wavelength of emission on the range of the 200 nm.
Because the cornea absorbs 100% of the radiation with that wavelength.