Orgo Chapter 18 Reactions at the α Carbon of Carbonyl Compounds
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
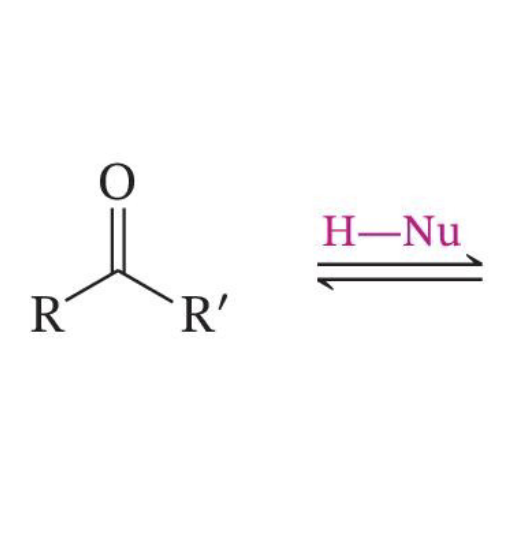
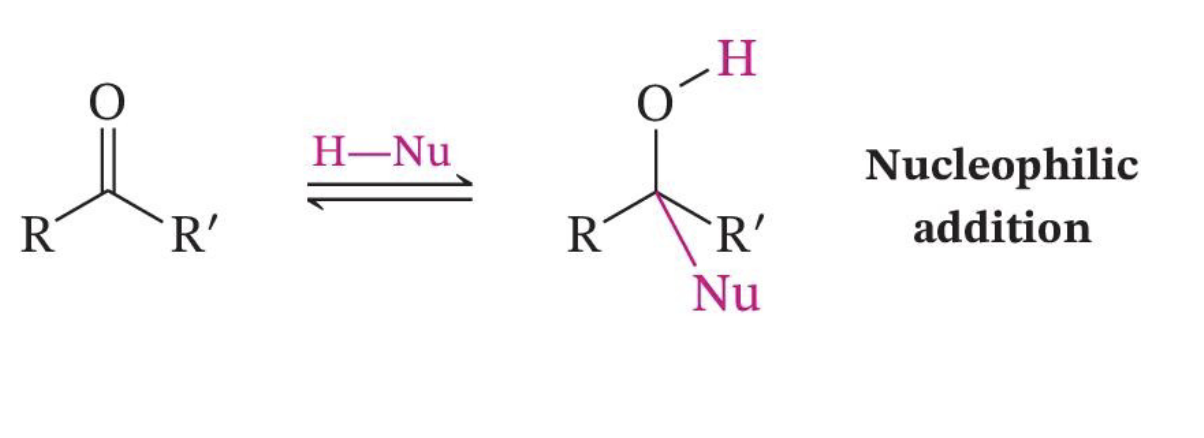
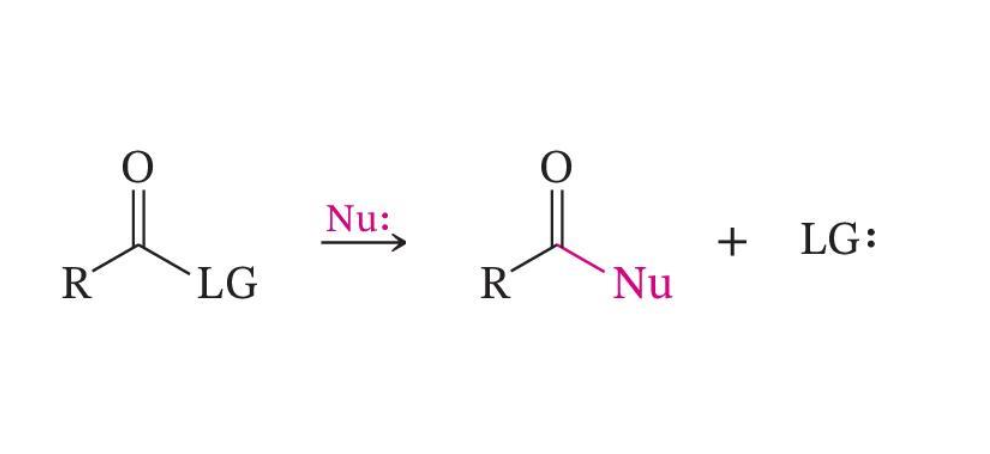
Direct Nucleophilic Attack
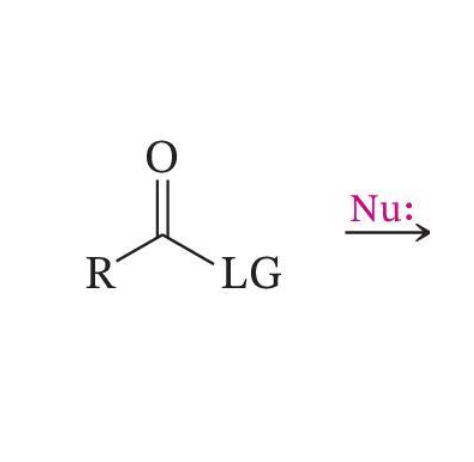
Nucleophilic Acyl Substitution
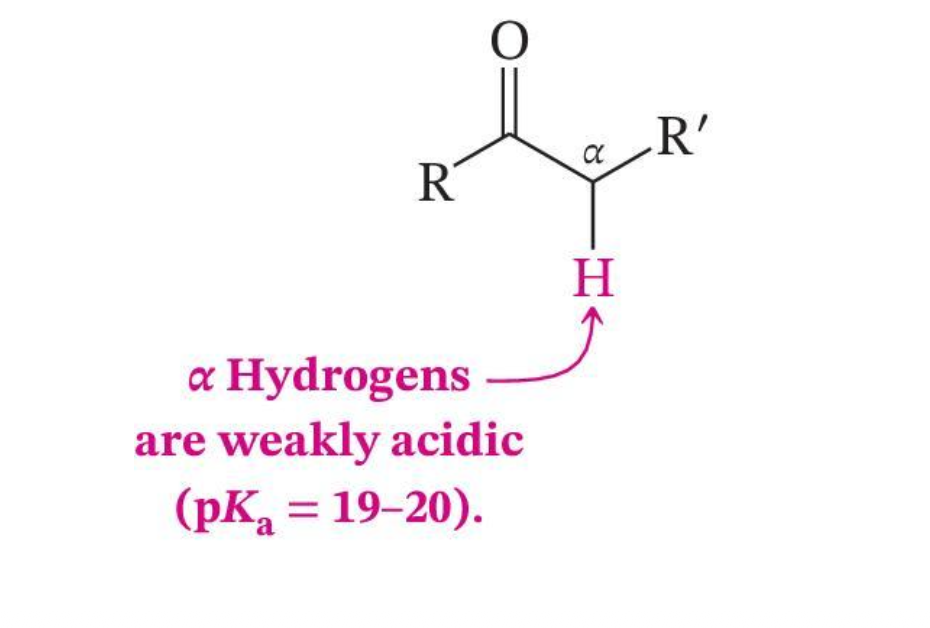
α-Hydrogens Are Acidic
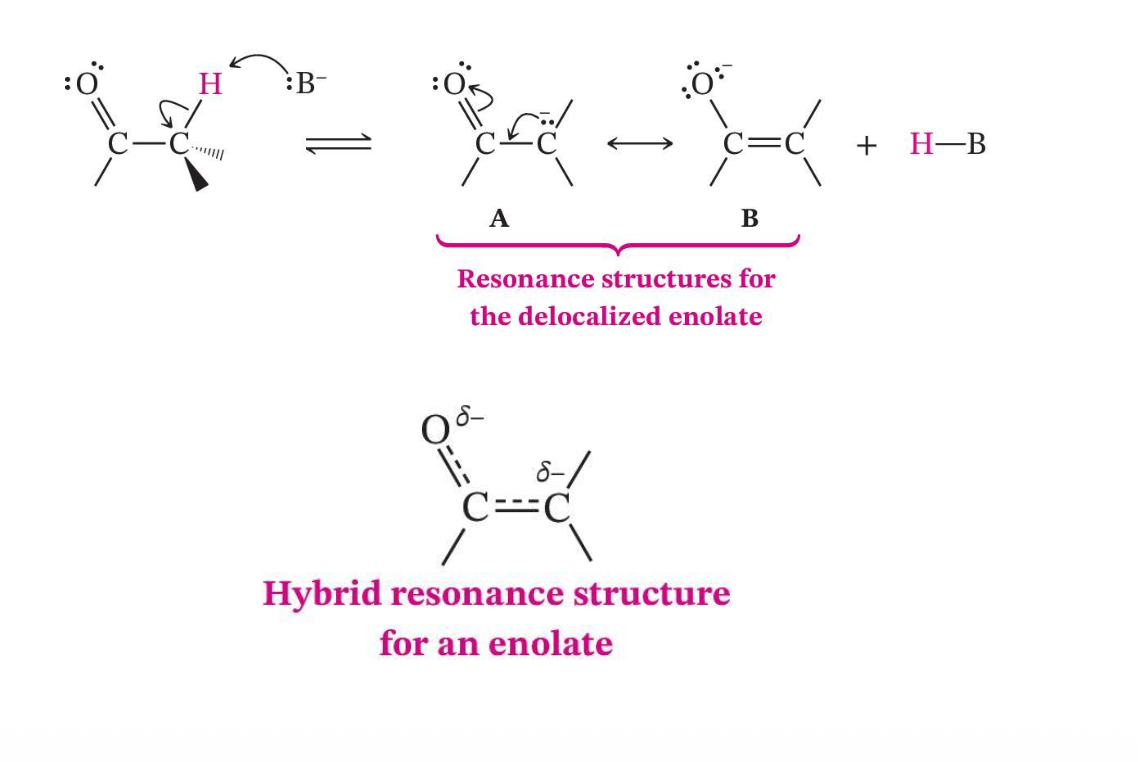
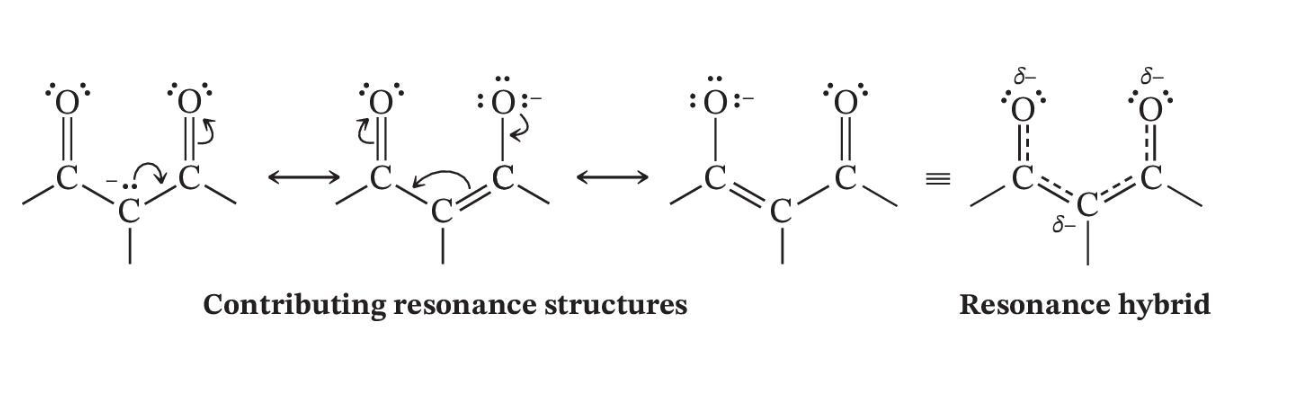
Enolate Ions Are Resonance Stabilized
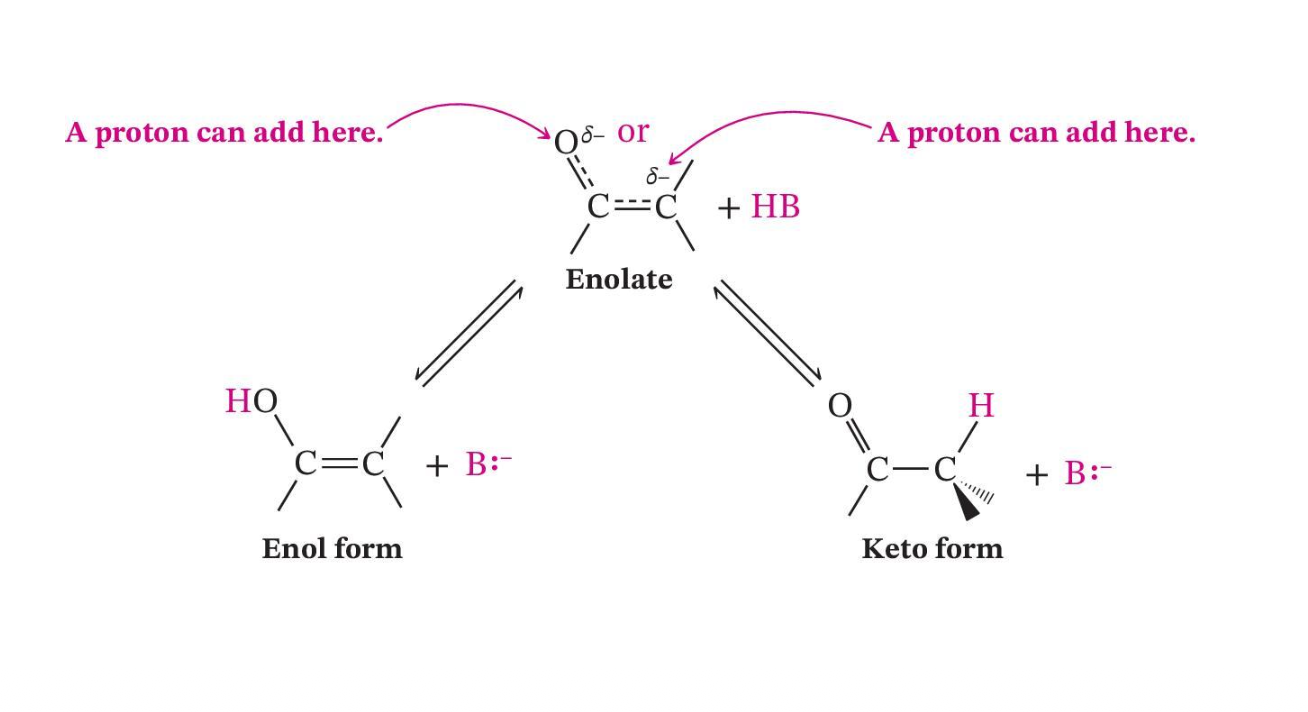
Conjugate Base of Both Forms
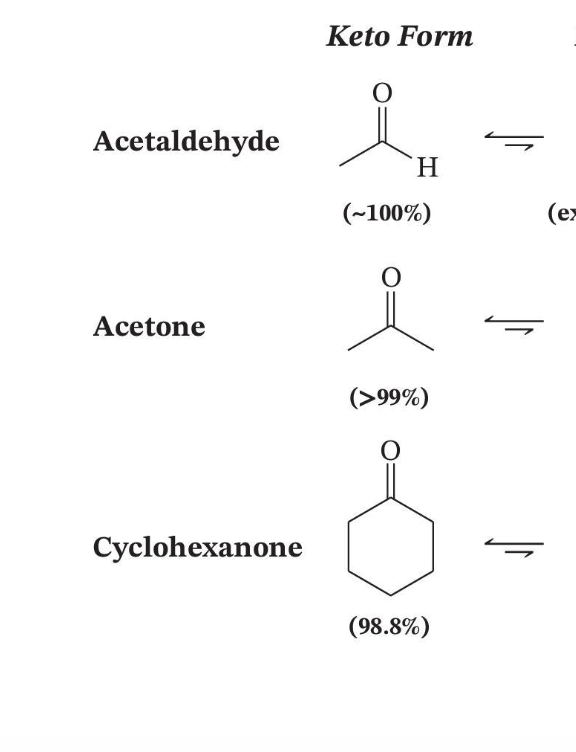
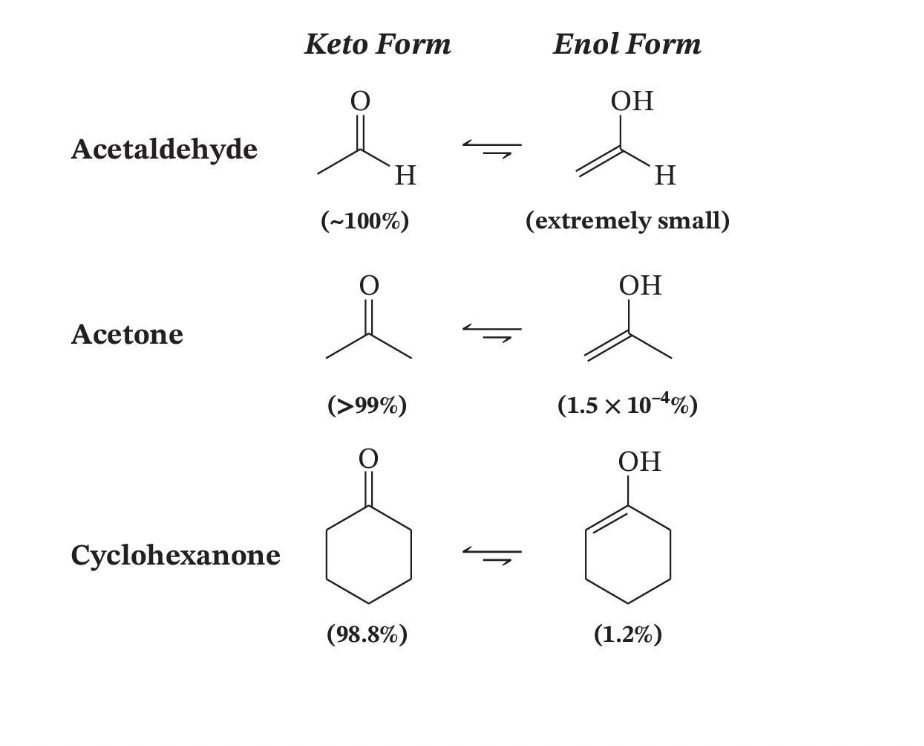
Tautomer Ratios in Solution
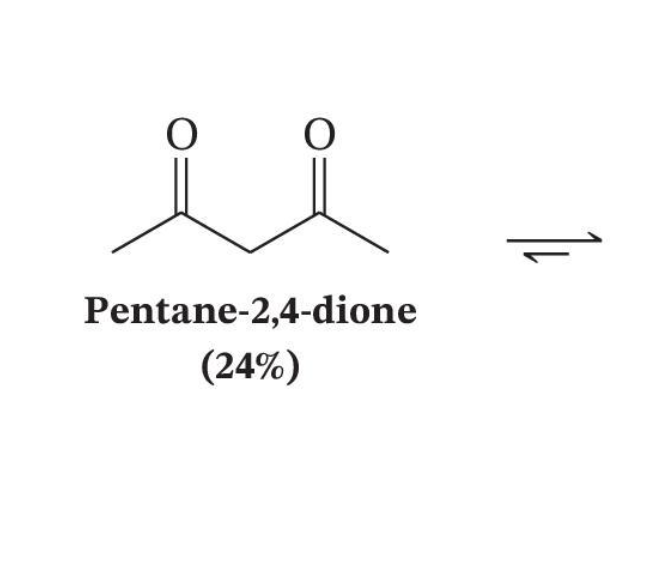
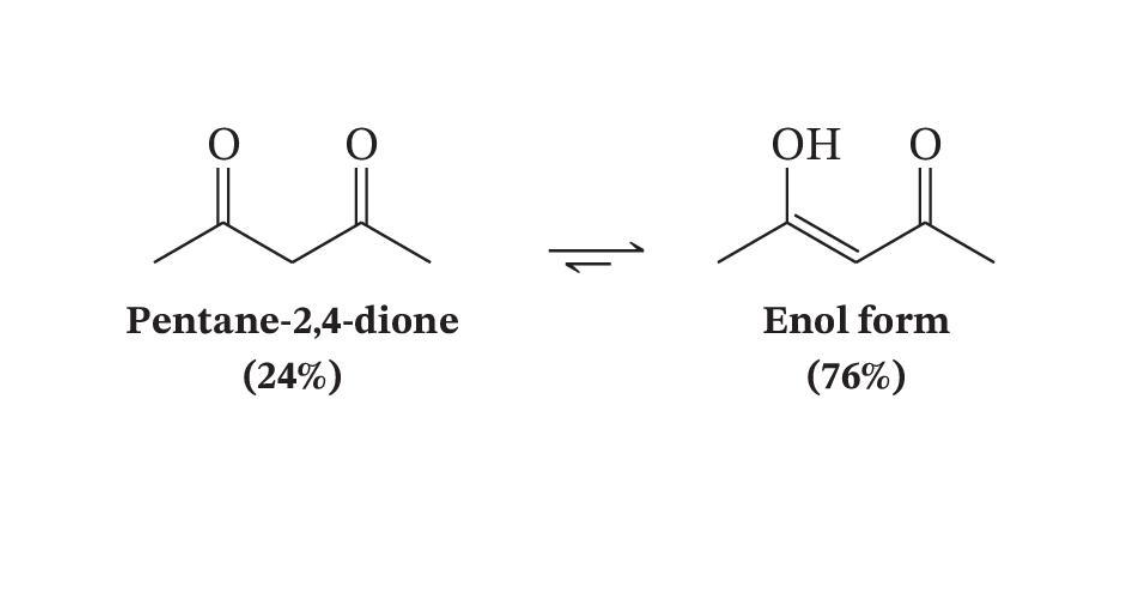
Conjugation Favors the Enol
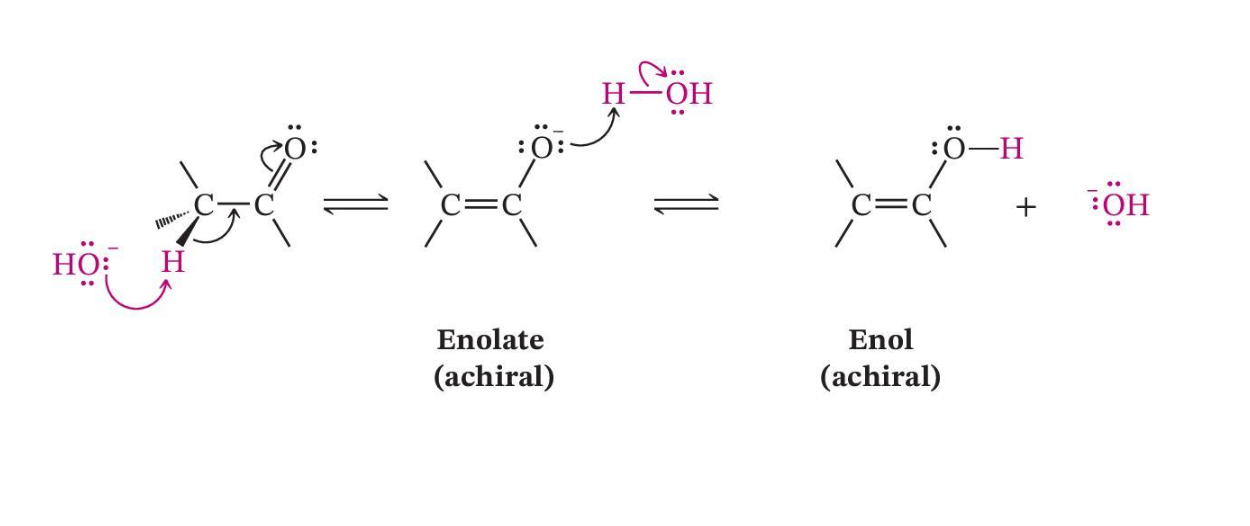
Base-Catalyzed Enolization
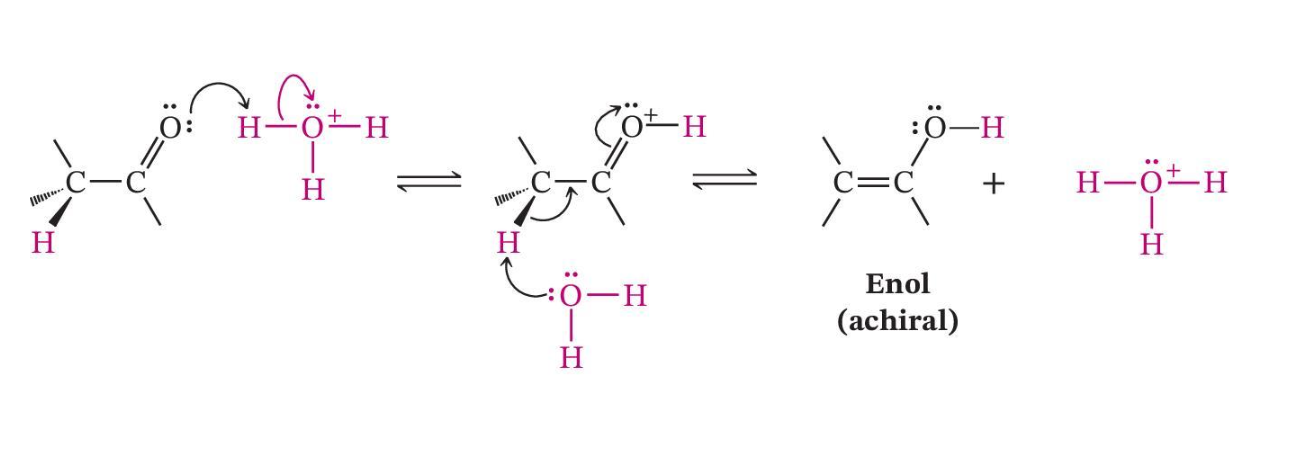
Acid-Catalyzed Enolization
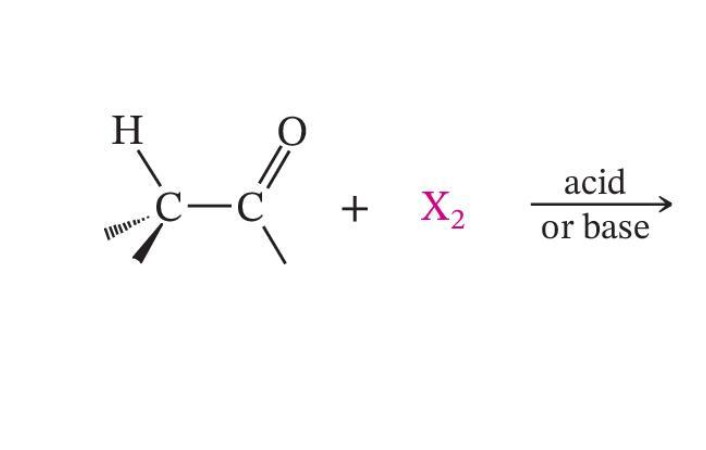
α-Halogenation
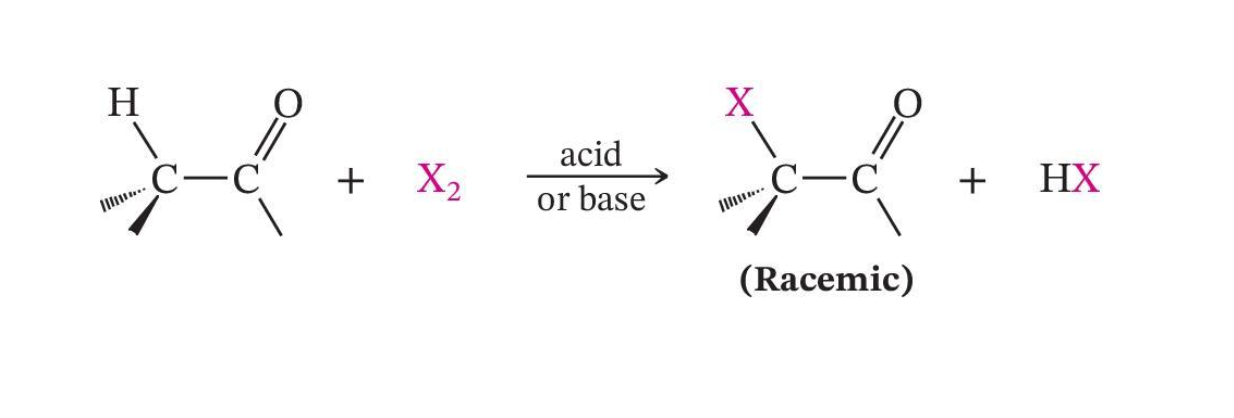
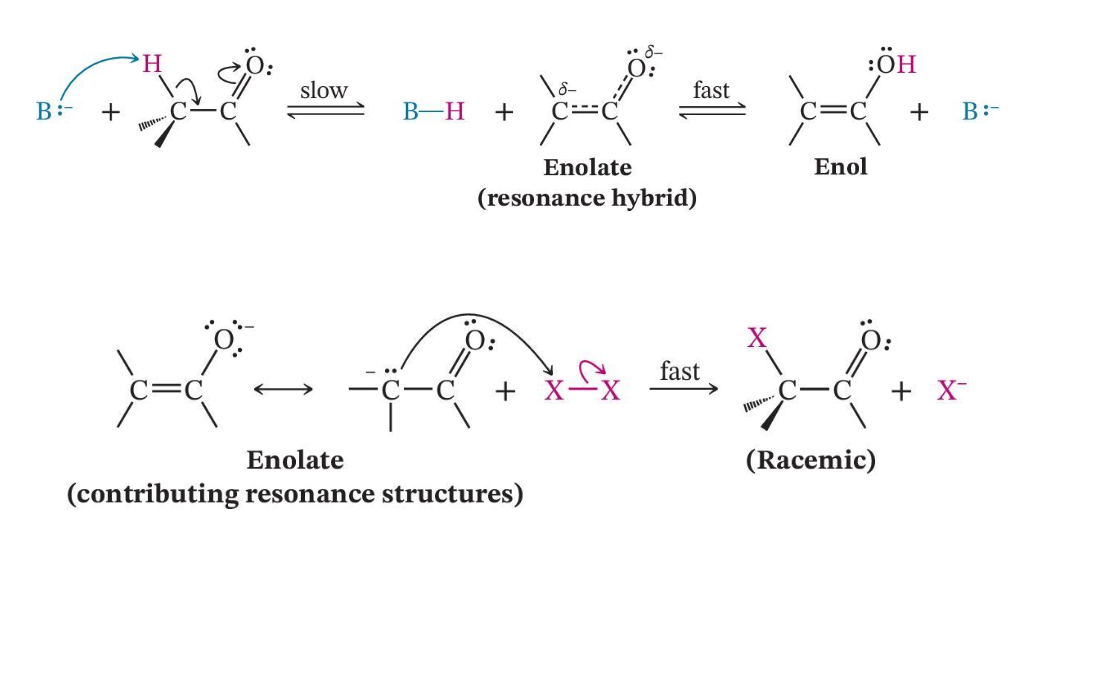
Base-Promoted Halogenation
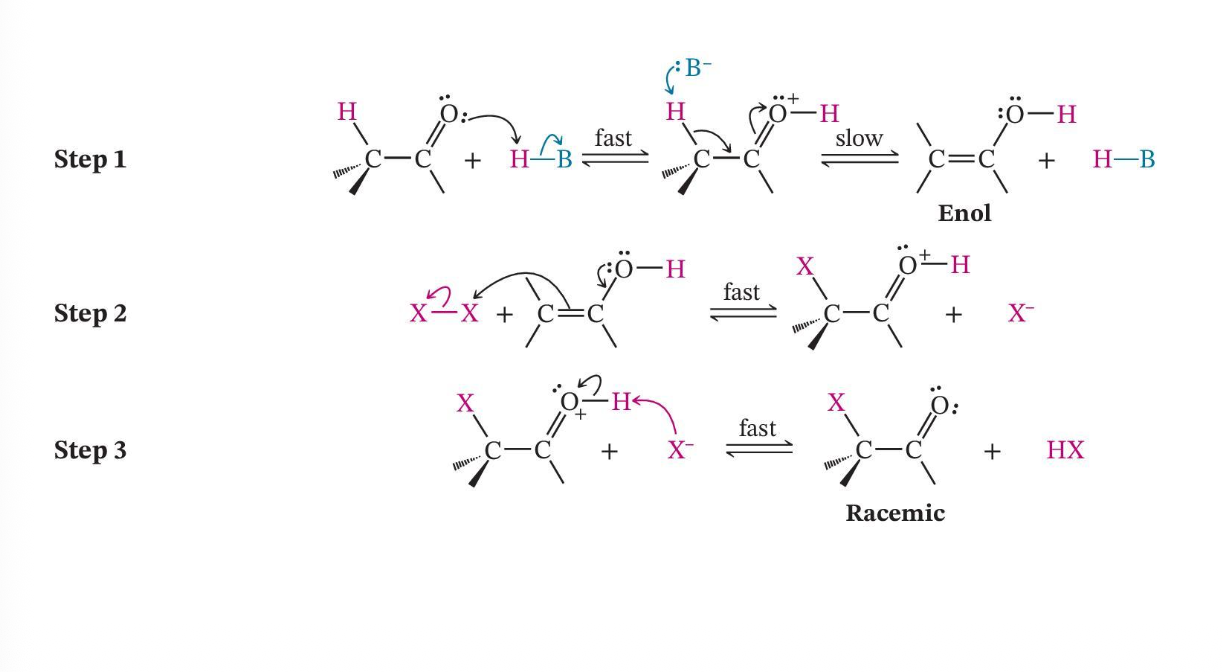
Acid-Promoted Halogenation
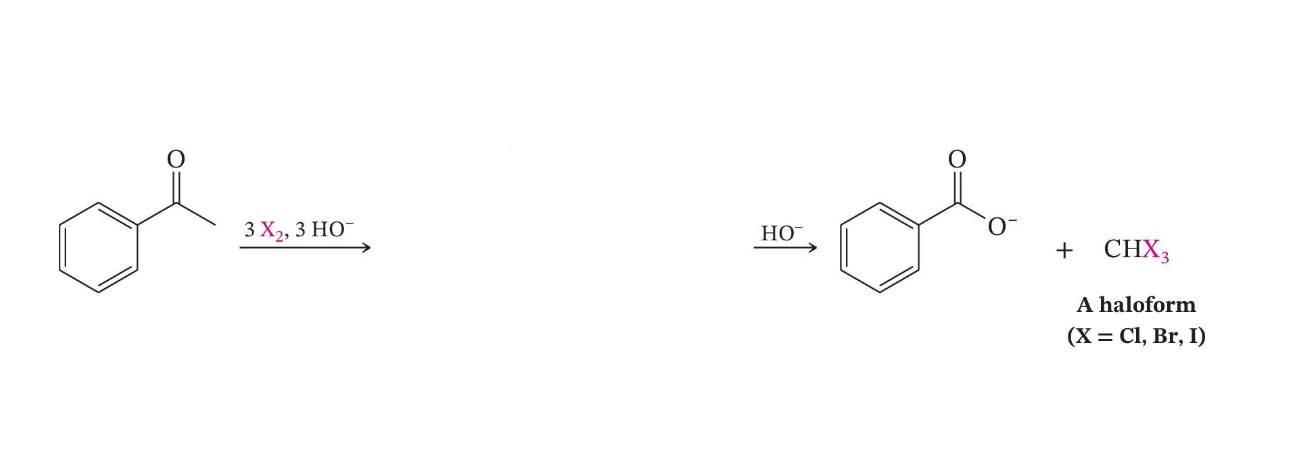
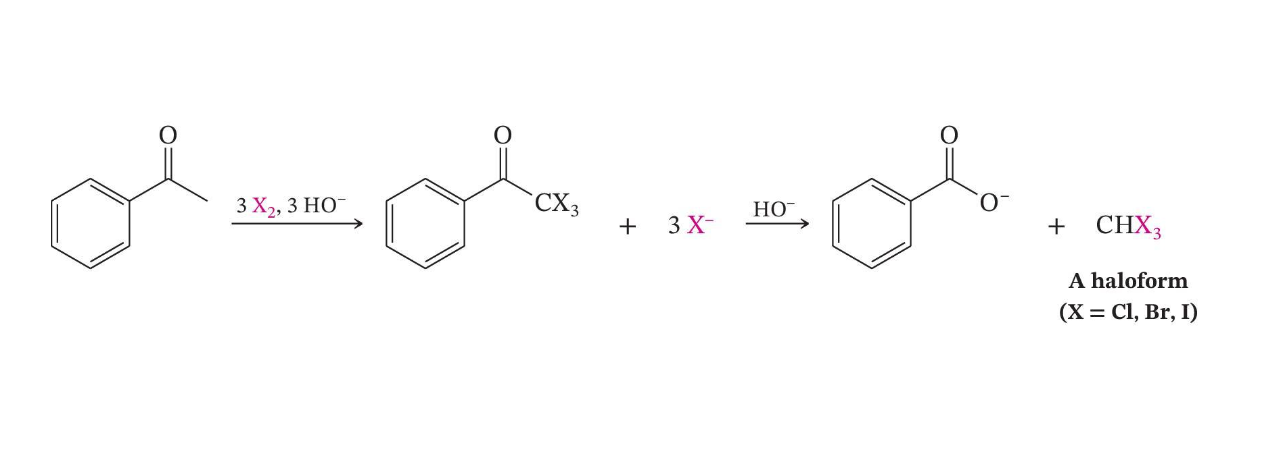
Haloform Reaction
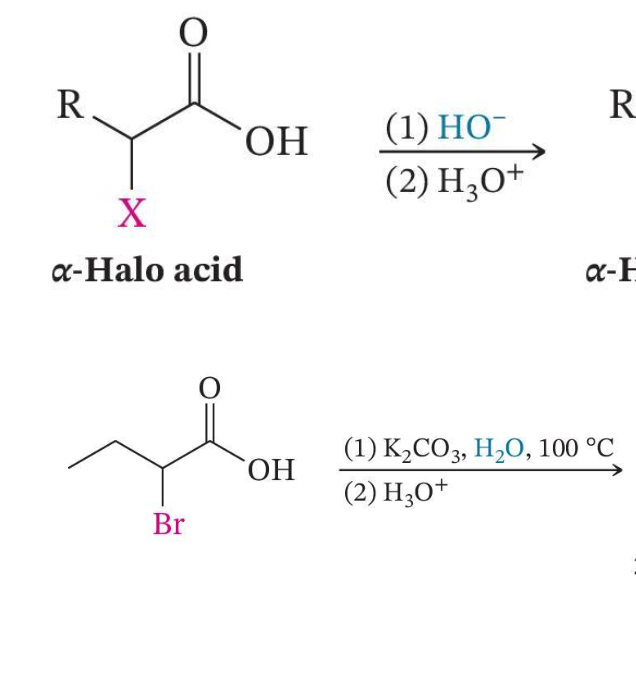
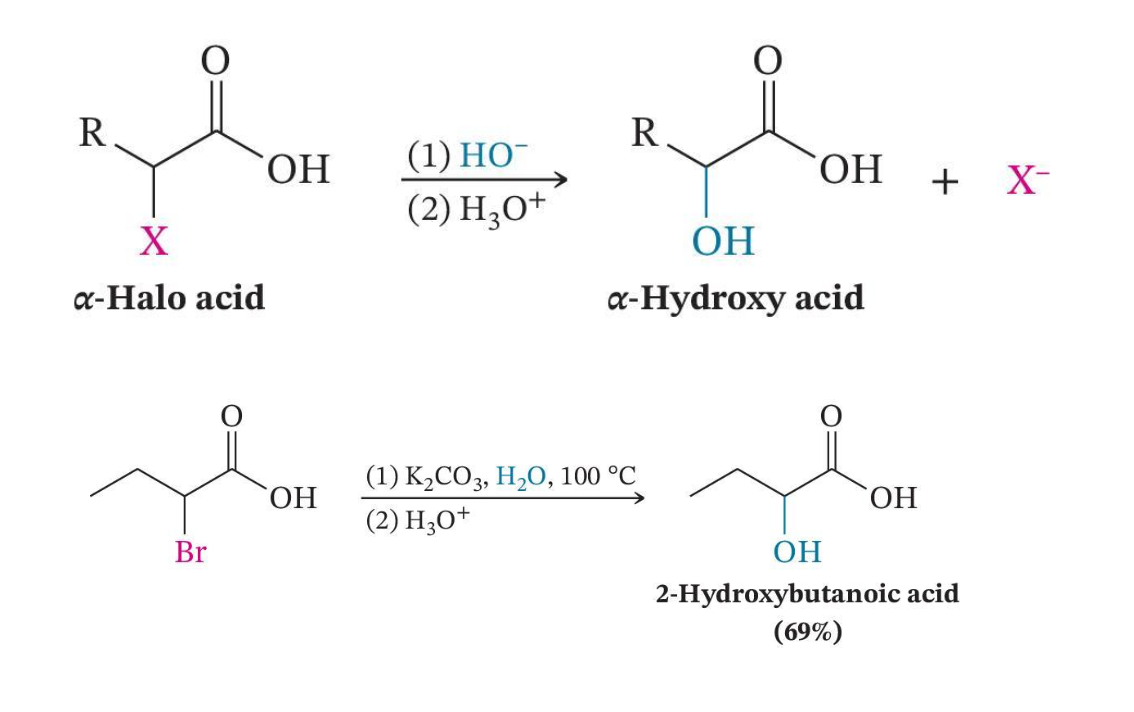
Conversion to α-Hydroxy Acids
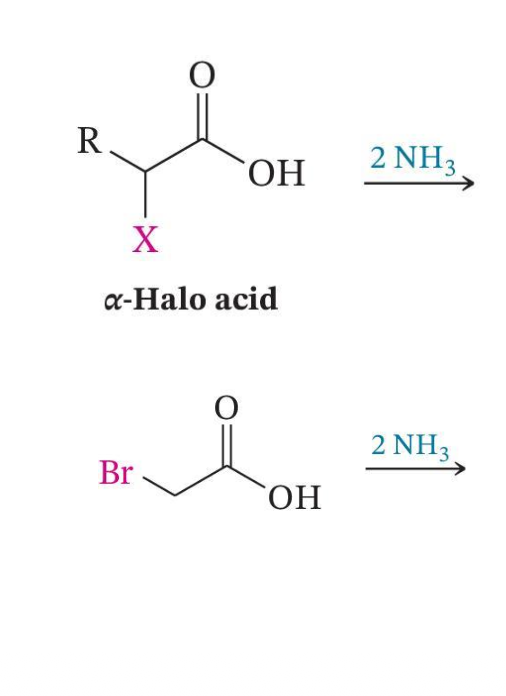
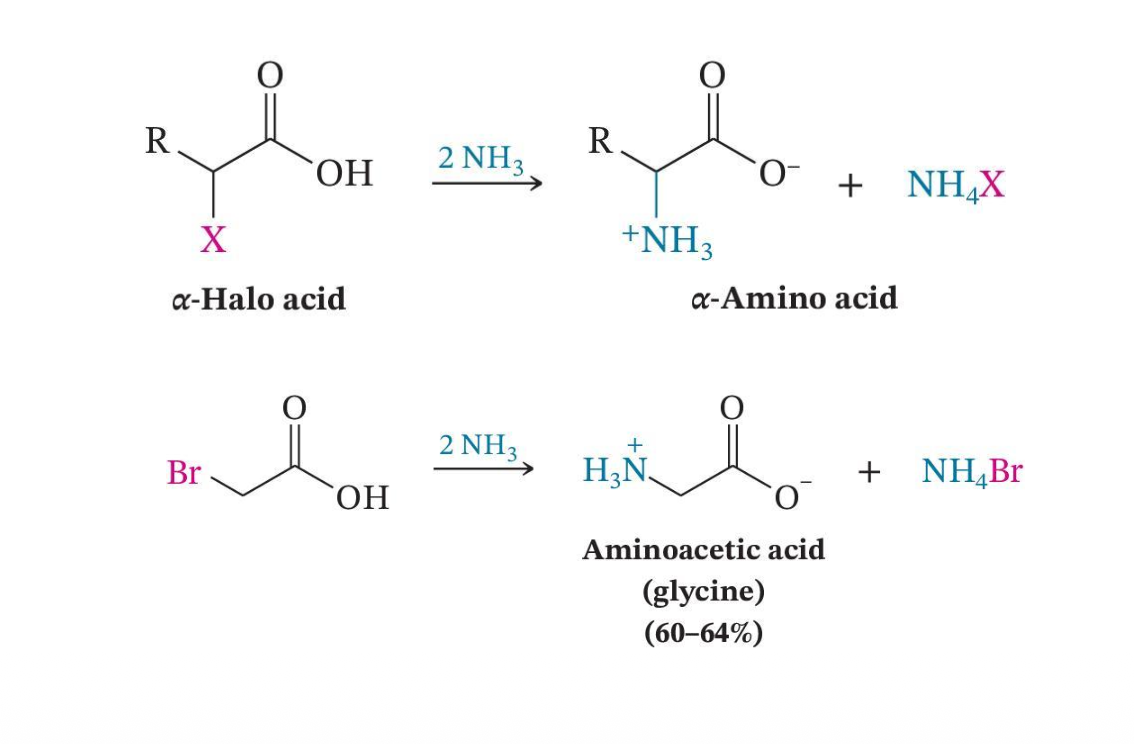
Conversion to α-Amino Acids
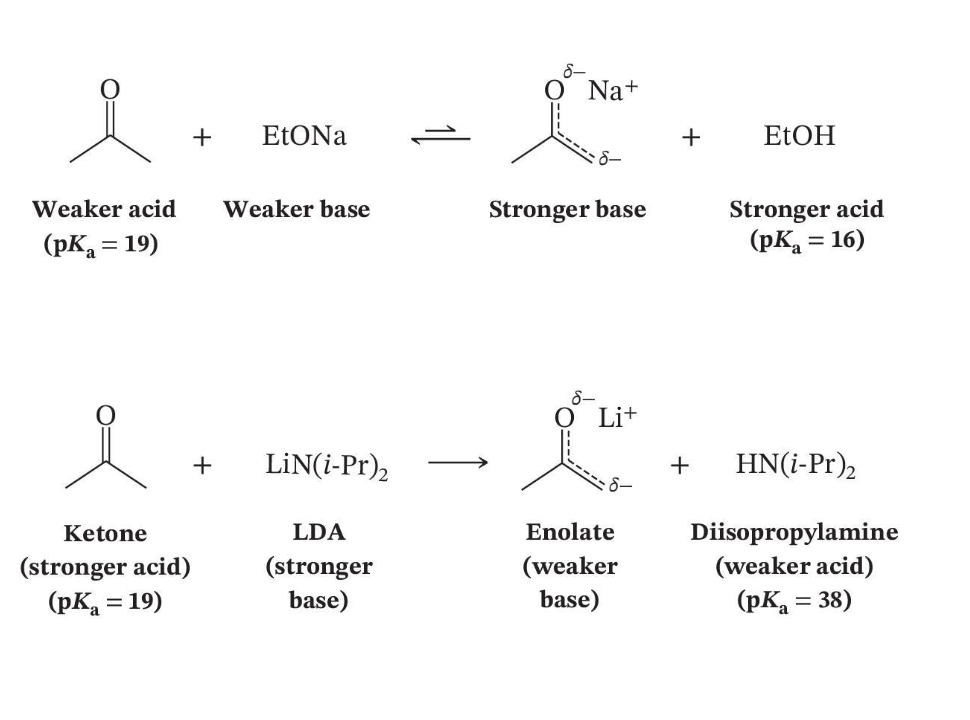
Equilibrium Direction Depends on Base
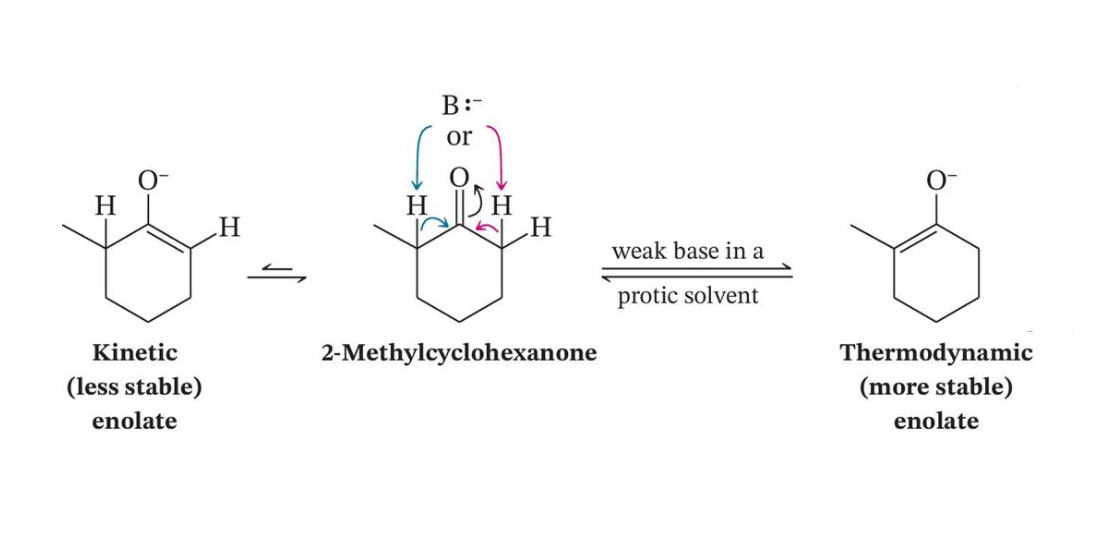
Kinetic Versus Thermodynamic Enolates
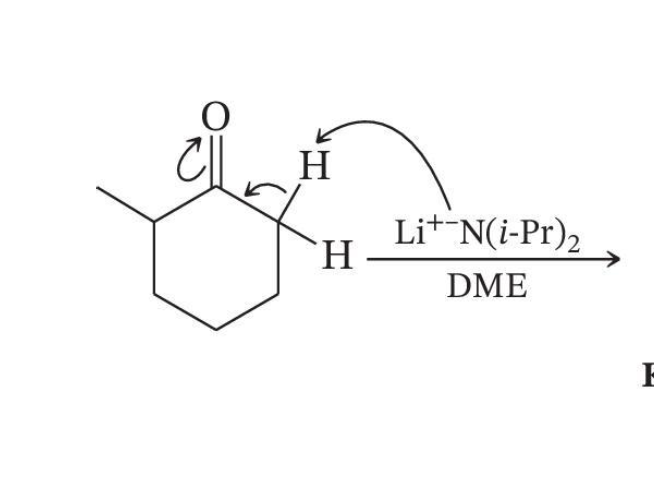
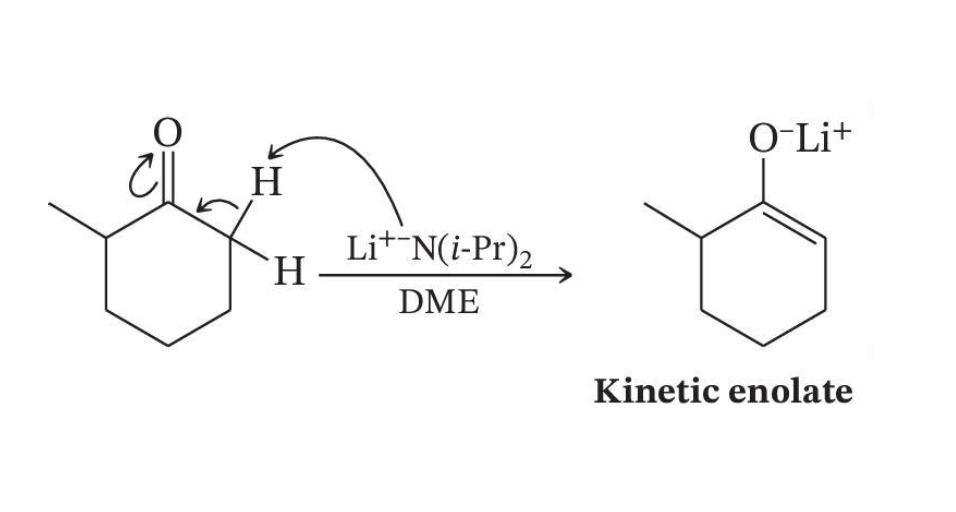
Kinetic Enolate Formation
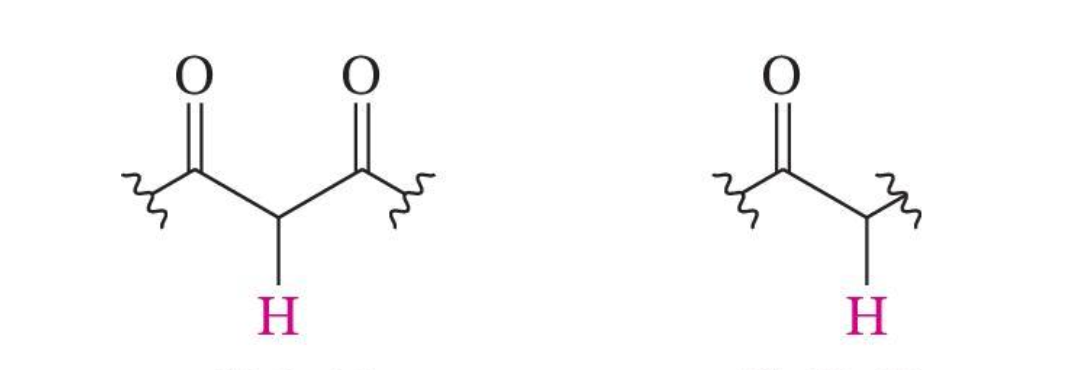
β-Carbonyl Group Increases Acidity
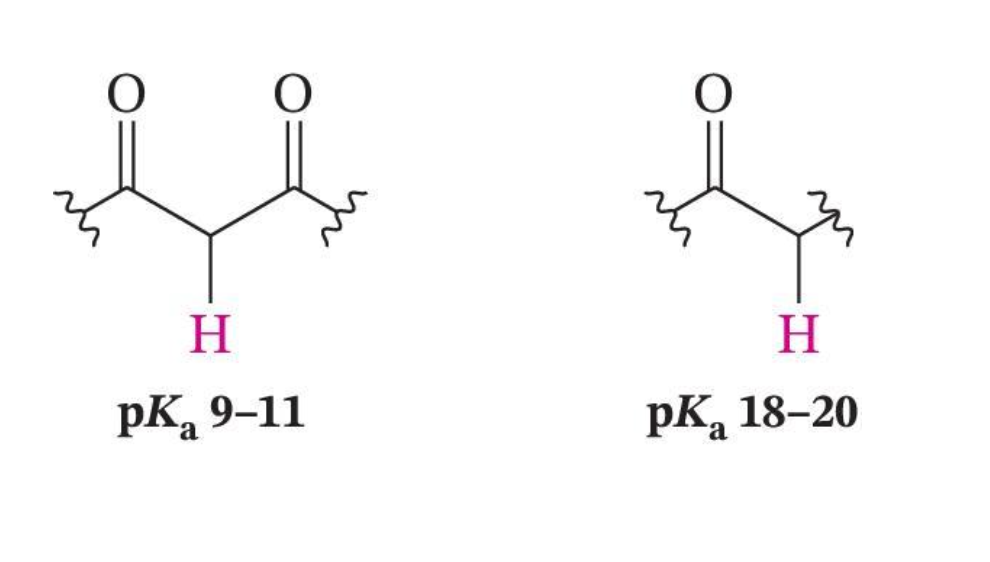
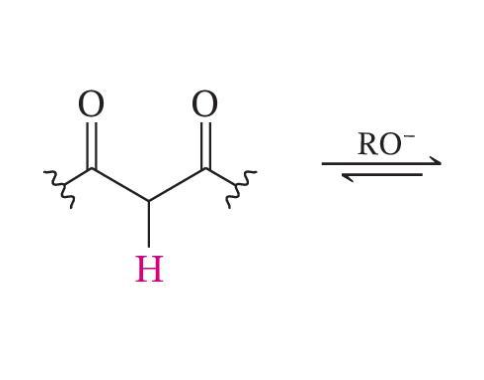
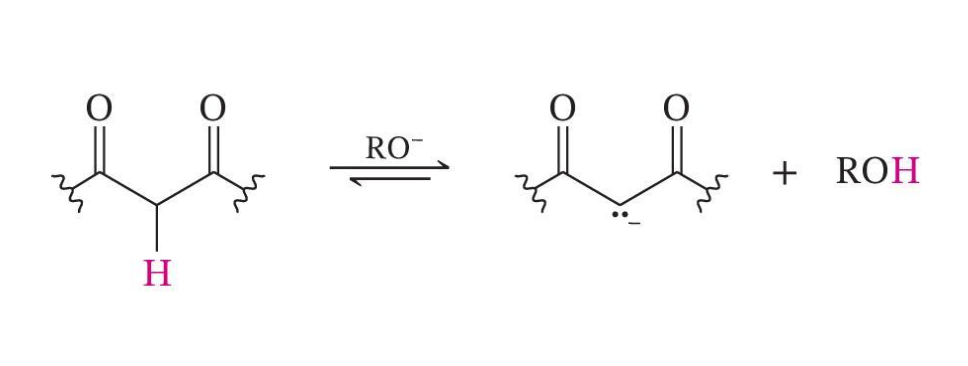
Weaker Bases Can Be Used
Enolate Resonates Through Both
Carbonyls
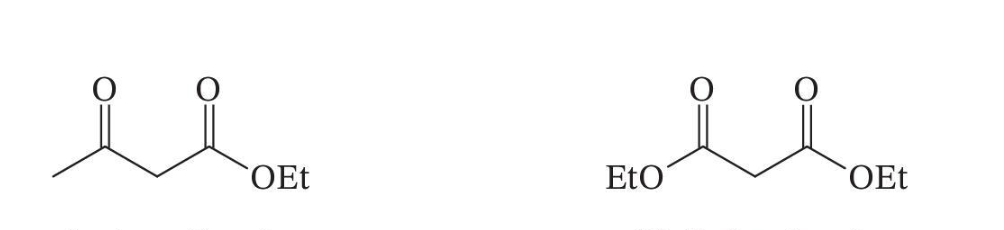
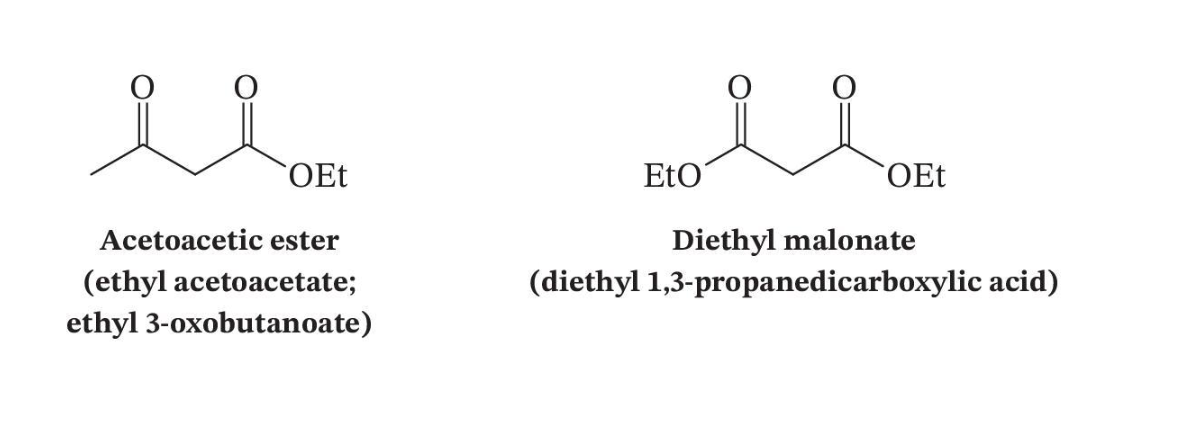
Useful β-Dicarbonyl Compounds
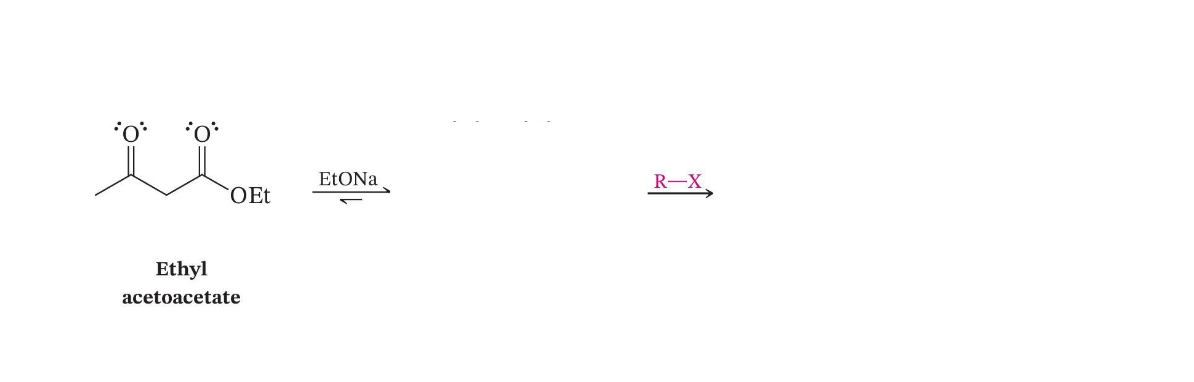
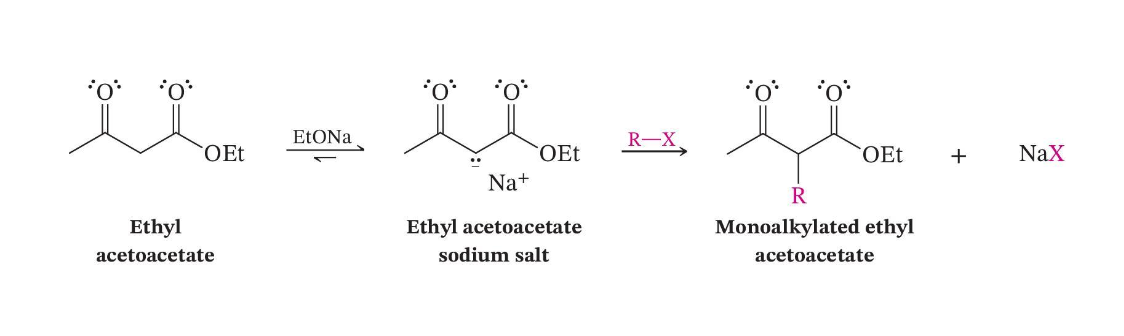
β-Keto Ester Alkylation

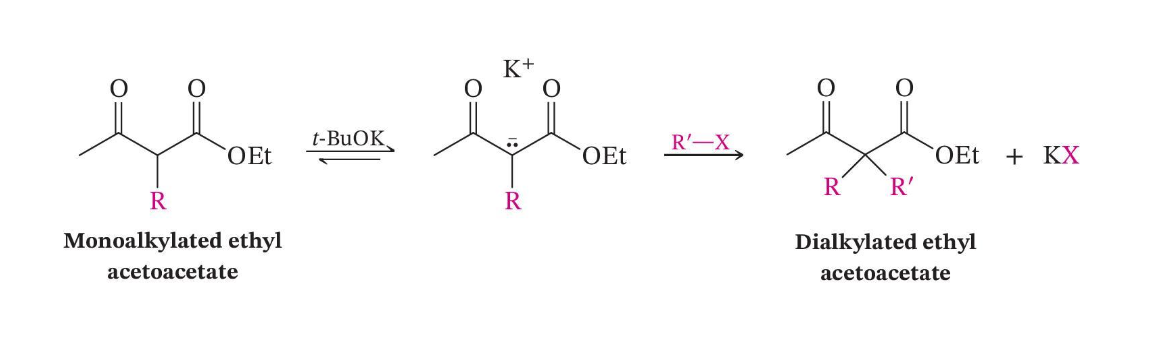
Dialkylation
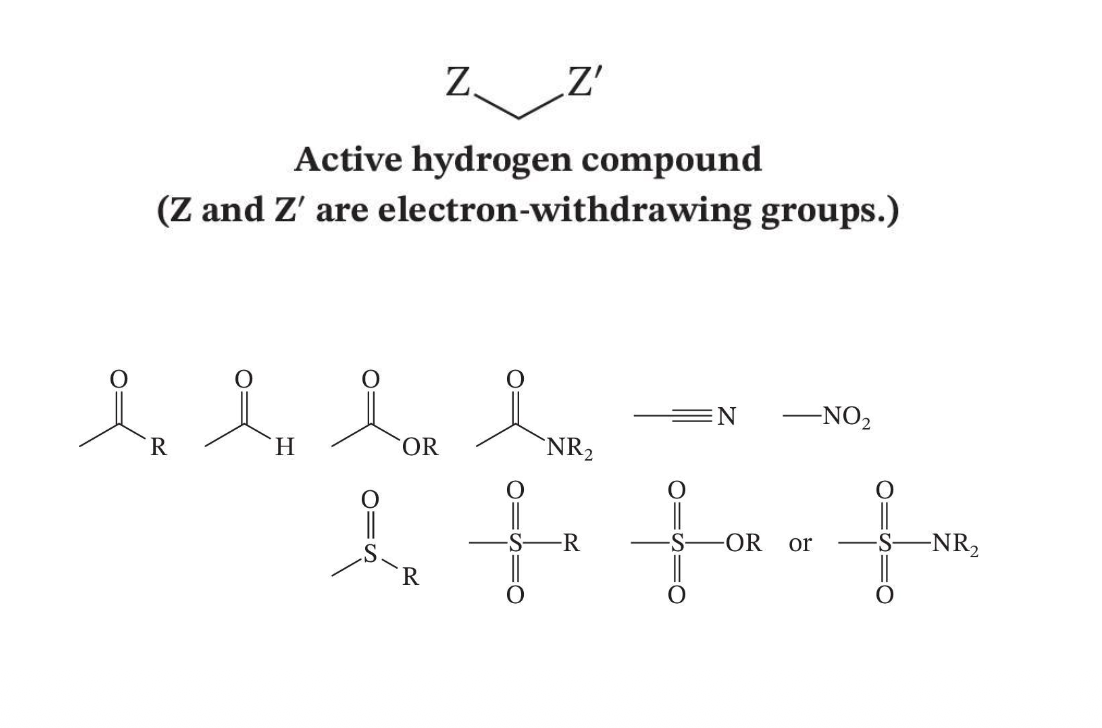
Active Hydrogen Compounds