Antigen Recognition by T Lymphocytes
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
How is the T cell (and its receptor) similar to/different from the B cell (and its receptor)?
Like B cells, they recognize and bind antigen through highly variable antigen-specific receptors
Originate in the bone marrow & mature in the thymus
Most of them are destroyed, and IF they make it through these checks, they move into circulation and wait for an antigen that binds their receptor. They do not get activated unless this happens!
A B cell receptor (surface Ig) can be secreted if the cell is activated and becomes an antibody
The T cell receptor is never secreted. Upon activation, they do different things depending on the surface glycoprotein co-receptor (CD4 or CD8)
What are some of the things that T cells do?
CD8 T cells are cytotoxic cells, which kill cells infected with a virus or bacterium (like the NK cells of innate immunity)
CD4 cells can be Helper T cells of several flavors:
They are called HELPER cells because they secrete cytokines that fully activate other effector cells
TH1 & TH17 cells activate macrophages and neutrophils to increase phagocytosis and kill phagocytosed pathogens
TH2 cells respond to parasites by secreting cytokines that activate mast cells, basophils, and eosinophils
TFH cells induce the differentiation of B cells to Plasma cells
Treg (regulatory) cells terminate the CD4 and CD8 T cell responses
What is the structure of the T cell receptor?
Similar structure to the BCR
Produced by somatic gene rearrangement
Highly diverse antigen specificities
Each T cell expresses just one form of an antigen receptor, but each clone has a different specificity for antigen
The mechanisms that produce diversity are similar to those that generate Ig diversity
Whereas Igs bind epitopes of intact macromolecules, TCRs recognize short peptide antigens that are derived by degradation of a pathogen’s proteins
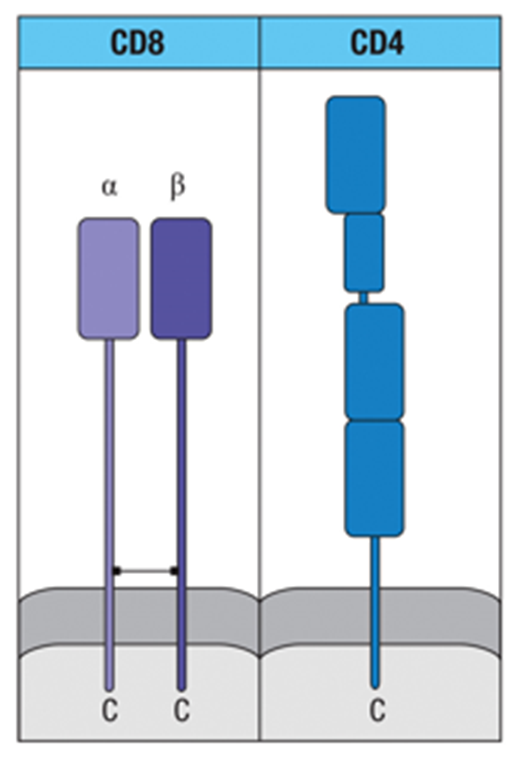
•Has an α and β chain, an antigen binding site, a variable and constant region, a transmembrane region, and a tiny cytoplasmic tail
Who was responsible for elucidating T cell receptors?
•Rolf Zinckernagel & Peter Doherty (1973)
•Worked in a mouse model with LCMV (lymphocytic choriomeningitis virus), which causes a fatal neurologic disease… death is a result of the strength of the immune response, not the virus itself
•Showed that T cells kill virus-ridden cells, but only if they receive TWO signals
•A molecule that tells the immune system that the infected cell is from the same individual (a protein, MHC antigen) AND A fragment of the antigen (in this case LCMV virus) that an infected cell displays on its surface telling the immune system that it is infected
What does the structure of a TCR in terms of how the TCR recognizes and binds antigen?
T cells carry out their effector functions by antigen-specific interactions with other cells in the body
T cells recognize antigen in the presence of host: a pathogen-derived peptide antigen BOUND by a human glycoprotein called MHC
So the ligand for the receptor is peptide + MHC at the cell surface
When these bind together, you have activation!
In short: T cells need to see antigen wrapped up in paper with a pretty bow on it to activate in the context of self!
How does it recognize and bind antigen differently from the BCR?
•TCR binds antigens specifically
•But, it sees a different type of antigen than Ig
•Ig binds an epitope of an intact pathogen directly
•The TCR binds part of an antigen that has been processed and sent to it on a silver platter. So, it’s picky in that way.
•The antigen has to be presented by another cell (& another cell that is self)
•There is always cell to cell contact
How is diversity in the TCR generated?
•Similar to what happens with the BCR!
•Happens BEFORE antigen exposure in the thymus, where T-cells mature*
•RSS, RAG, & P&N Nucleotide additions are involved
•But, NO somatic hypermutation or class switching
Is the total diversity of TCRs greater than, less than, or equivalent to the diversity that can be generated in the BCR?
BCR Higher Diversity: BCRs (immunoglobulins) have higher diversity because they undergo somatic hypermutation (point mutations) in the germinal centers, allowing for affinity maturation, which TCRs do not.
Additional Mechanisms: BCRs also use class-switch recombination to alter constant regions, allowing for more variation in effector function.
TCR Limits: While TCR diversity is vast, it is mostly established during initial development, whereas the BCR repertoire continues to evolve and diversify upon antigen encounter.
LESS THAN
What is the TCR complex?
a specialized surface protein complex on T lymphocytes that recognizes antigen peptides bound to Major Histocompatibility Complex (MHC) molecules. It comprises an αβ (or γδ) heterodimer responsible for antigen recognition, non-covalently associated with CD3 chains (epsilon, gamma or epsilon, delta) that transduce activation signals to the cell.
Why does the TCR have to associate with CD3 to do its thing?
The T cell receptor (TCR) must associate with CD3 because the TCR itself lacks the intracellular signaling domains necessary to activate the T cell. The TCR alpha and beta chains only recognize antigen, while the CD3 complex transduces this recognition into internal cellular signals
What is the difference between the αβ and 𝛾𝛿 TCRs?
•αβ T cells are more well studied: they recognize antigen in the context of self
•They are major players in adaptive immunity and are the more abundant class of TCR (95%)
•This restriction is not the case for 𝛾𝛿 T cells, which recognize a wider variety of antigen
•They can’t recognize self from non-self
•It also has properties that are characteristics of lymphocytes in innate immunity (similar to the NK cell)
•𝛾𝛿 probably evolved first and then got more sophisticated
No T cells express both!
How does the TCR see antigen in the context of self?
•Antigen Processing via the Degradation of Pathogens and their Products
•Uptake and killing of bacterial pathogens by macrophages and dendritic cells, resulting in detritus
•Peptides from the degradation of viral proteins in infected human cells
•Antigen Presentation
•After the antigen in processed, it has to be brought to the cell surface
•Binds to MHC** and is carried to the cell surface
•Display of peptide antigens on the surface of human cells in a way that they can bind a TCR
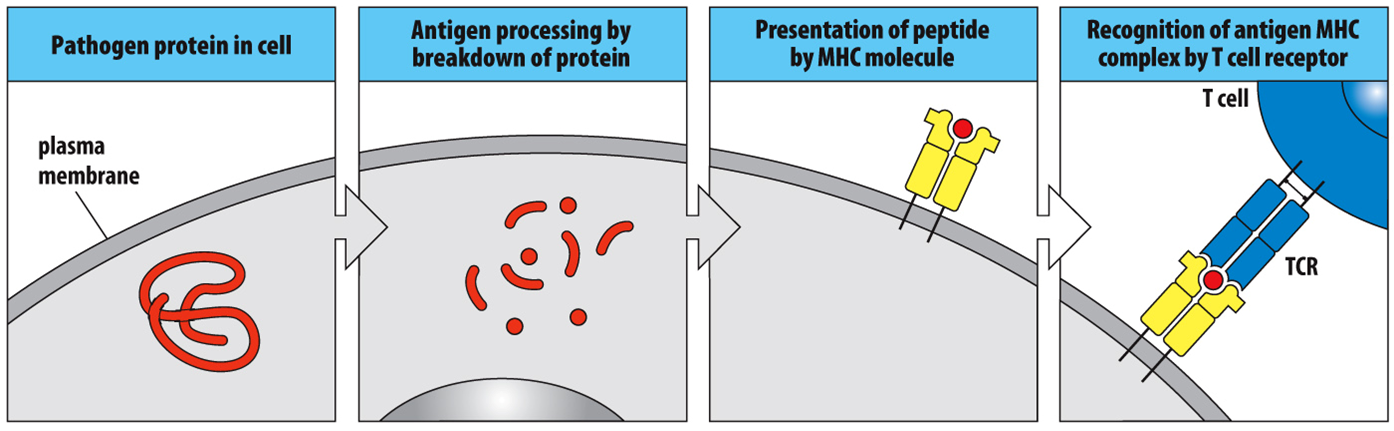
What happens when a TCR binds antigen in the context of self?
T cells are activated
What are the two classes of MHC molecules that present antigen?
MHC Class 1 and MHC Class 2
What is the difference in the structure and function of each MHC?
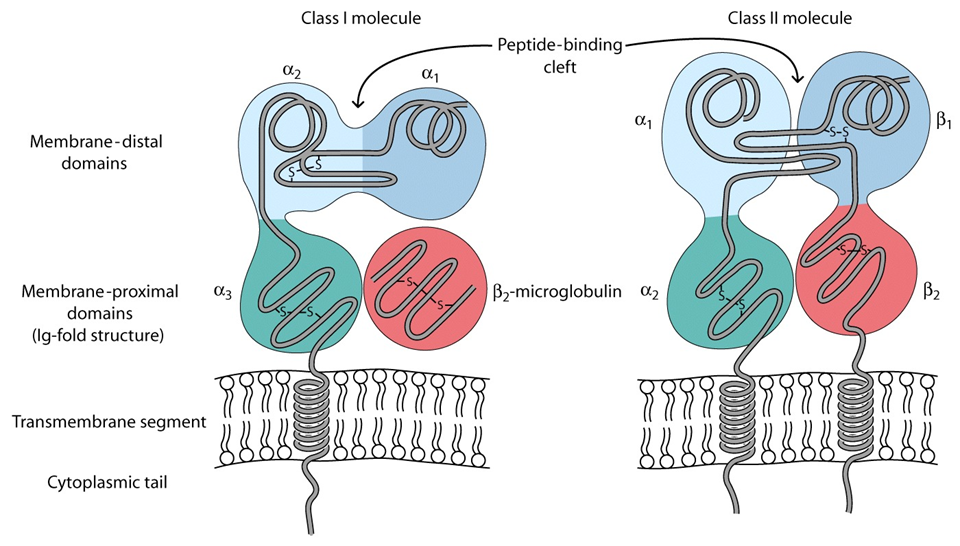
•Both are protein chains embedded in the membrane with a peptide binding cleft where the antigen (red dot) goes
•MHC Class I is made of a large and small polypeptide chain, only one of which is anchored in the cell membrane
•MHC Class I has…
•An α chain with 3 domains (α1, α2, α3… one long protein) associated with a β2 macroglobulin, which helps prop up the chain
•It is found in all nucleated cells
•MHC Class II has…
•Two molecules (α & β) that form one long protein
•These molecules come together to form the antigen-binding cleft
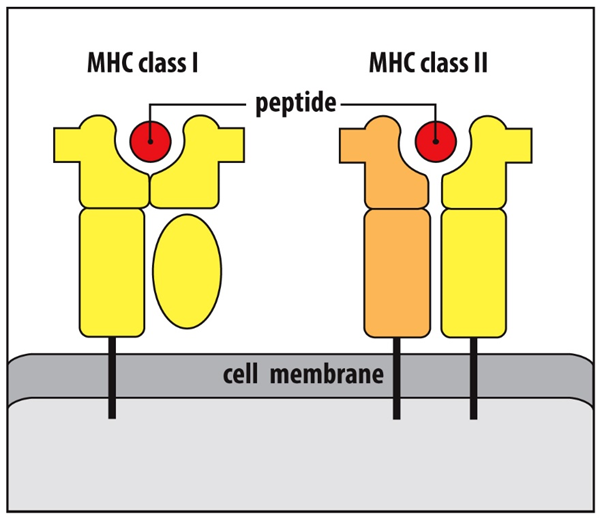
What cell types express MHCI vs MHCII?
•ALL nucleated cells (except RBC) have MHC Class I
•Professional antigen presenting cells (i.e., dendritic cells, macrophages, and B cells) ALSO have MHC Class II
What are the CD8 and CD4 co-receptors? Do they bind antigen?
•Corresponding to the two classes of MHC are two types of effector T cells
•The cytotoxic T cell, which recognizes antigens presented by MHC Class I molecules, thus defends against intracellular pathogens
•The helper T cell, which recognizes antigens presented by MHC Class II molecules and defends against extracellular infection
•Each class of MHC also expresses surface glycoproteins that serve as co-receptors, cooperating with the TCR in the recognition of peptide: MHC complexes
•But, they DO NOT bind antigen!
What type of T cell is a CD8 T cell? What is its effector function?
•CD8 is found on cytotoxic T cells
•These cells can physically kill cells with intracellular pathogens
CD8 binds to Class I MHC, but does not bind antigen
What type of T cell is a CD4 T cell? What are its possible effector functions?
•CD4 is found on T-Helper cells
•These T cells tell other cells of the immune system what to do (in helping them do their jobs)
•CD4 binds MHC II
When the MHC-Co-Receptor complex binds antigen (in the context of self), what different things could happen?
(L) Binding causes signal transduction that results in killing the virus-infected cell
(M) Binding causes signal transduction that turns on genese that express cytokines (TNF-alpha & CXCL8
(R) Binding tells the B cell to differentiate into a plasma cell and start to secrete antibodies that are specific to the antigen; whether the B cell differentiates or not depends on how the foreign antigen appears to it- it does not always require T cell help! This is just another pathway!! REDUNDANCY!!! = ROBUST RESPONSE!!!!!
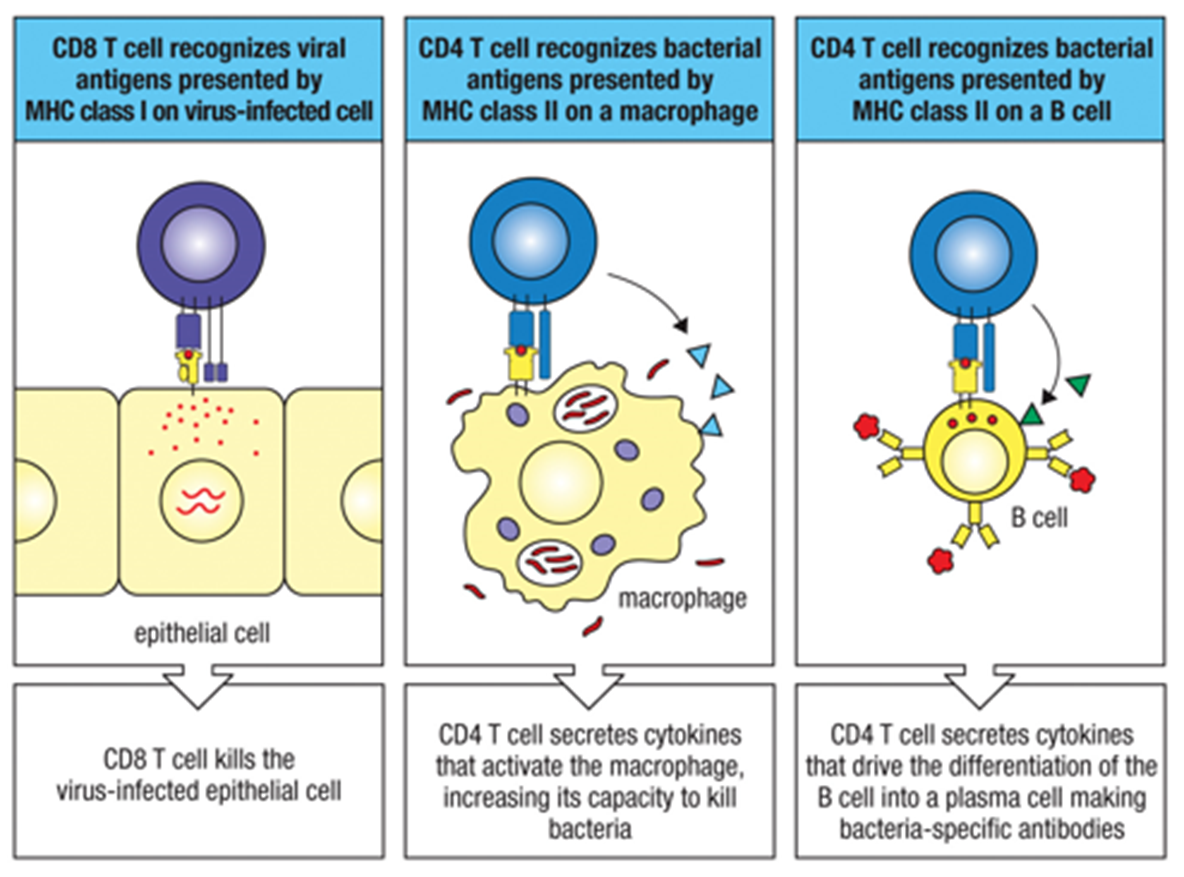
3 options: CD8 T cell kills the viral infected cell after binding antigen presented on MHC class 1, CD4 T cell secretes cytokines to tell other effector cells how to do their jobs (increases their capacity to kill bacterial infection), OR CD4 T cell secretes cytokines that drive a B cell to differentiate into a plasma cell.
What fits in the MHC cleft?
•MHC proteins can bind peptides of various specificities*
•Constraints are based on the structure of the antigen cleft on MHC
•If it fits, it binds!
Whether it will bind or not depends on the size of the antigen
MHC Class 1 Antigen Processing
•Angry foreign protein in the cell gets broken down (chopped up) in an activated proteasome
•TAP proteins transport the protein fragments of the broken down pathogen into the ER and place them inside of MHC Class I
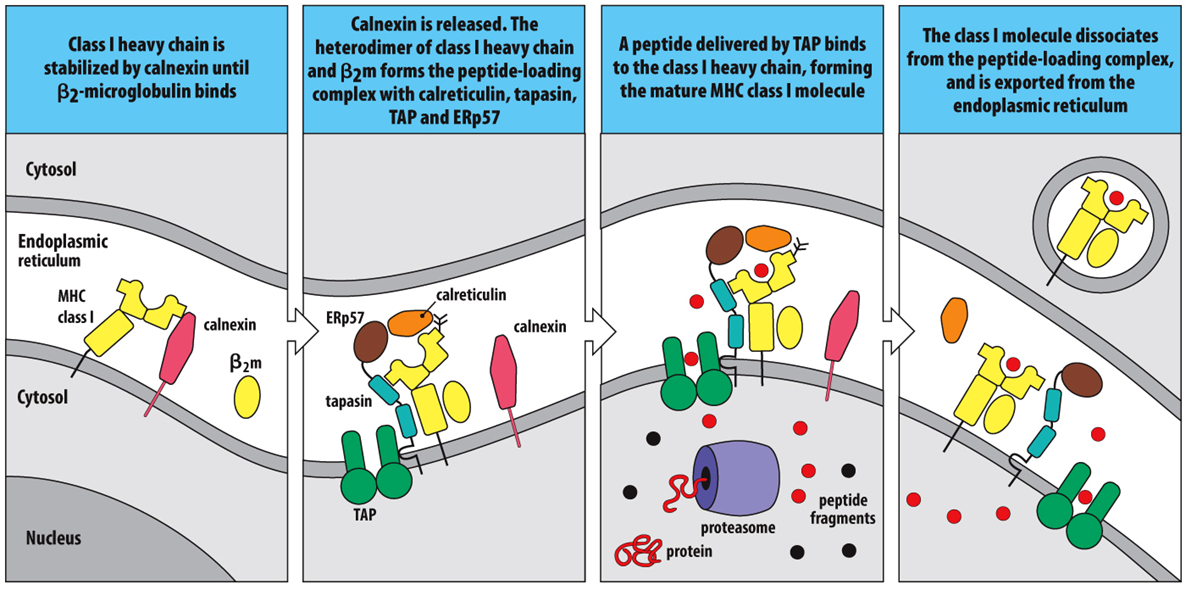
MHC Class 1 Loading
1)MHC Class I is stabilized by calnexin; it stays there until β2 microglobulin comes over and associates (binds) with MHC I
2)Calnexin is released; this helps to form a complex (TAP + tapasin + other molecules + MHC I) that allows the antigen to be loaded*
3)Processed antigen is transported from the cytoplasm to the ER via binding to TAP and transport across the membrane & is placed inside the MHC I cleft
4)The loaded MHC I dissociates from the peptide loading complex and is exported from the ER, destined for the cell surface
MHC Class 2 Antigen Processing
1)Antigen (red squiggle) is taken up from the extracellular space into intracellular vesicles
2)In early endosomes of neutral pH, endosomal proteases are inactive
3)Onset of respiratory burst! Acidification of vesicles activates proteases to degrade antigen into peptide fragments (red dots)
4)Vesicles containing fragments fuse with vesicles containing MHC II molecules … loading!
•Gets its antigen in the cytoplasm!
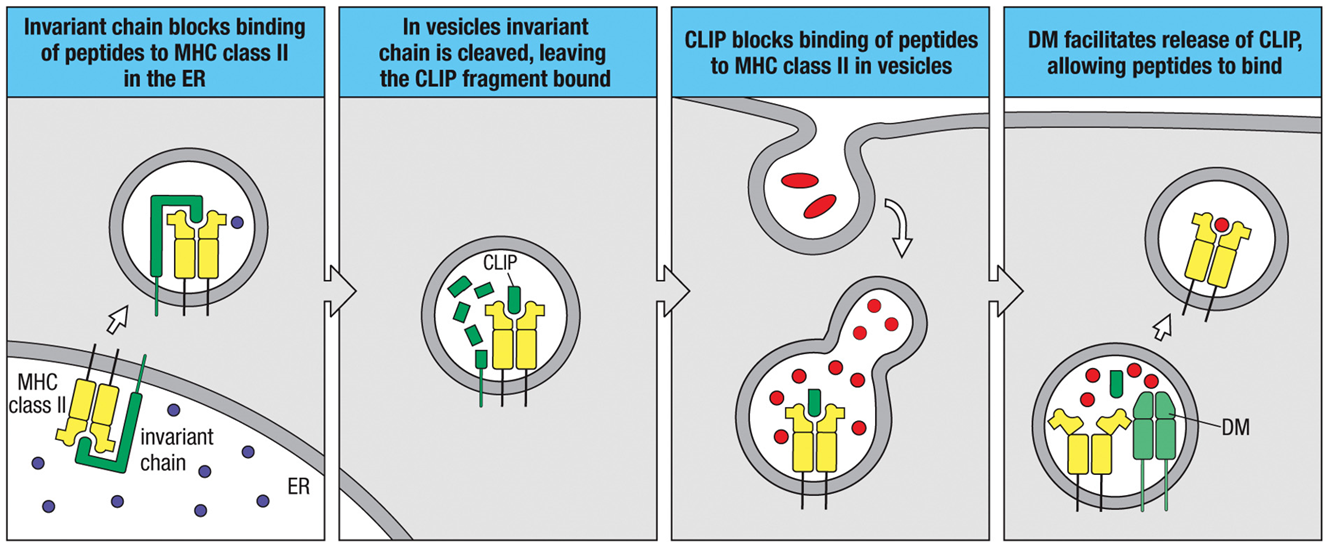
MHC Class 2 Antigen Loading
1)In the ER, when MHC II is translated, it gets associated with an invariant chain, a molecule that is there to keep the antigen cleft open and prevent stuff from binding*
2)The whole complex leaves the ER, and the invariant chain gets chopped up until all that’s left is a tiny piece called CLIP
3)CLIP travels with MHC II through the cytoplasm until…
4)DM (that’s already in the vesicle) allows CLIP to be released and an antigen fragment takes its place
5)Everything goes to the surface of the cell
MHC Class 1 Antigen Presentation
The peptide is loaded onto the MHC Class I molecule within the ER, then transported to the surface.
Result: The cell is recognized and destroyed by CD8+ T cells.
MHC Class 2 Antigen Presentation
The peptide-MHC Class II complex is transported to the cell surface.
Result: The APC activates CD4+ T cells, which initiate further immune responses.
What is cross-presentation? Why does it happen?
•Cross-presentation* enables extracellular antigens to be presented by MHC I: the viral antigens are brought into the APC by the MHC II pathway, but cross over to the MHC I pathway to be loaded
•Occurs in many cell types, but very important in dendritic cells, which initiate the primary T cell response
REMEMBER: A strategy of viruses is to lower MHCI expression on infected cells so that the immune system won’t recognize the cell is infected… cells like NK cells look for the absence of MHCI to know when to kill a cell (absence of expected) BECAUSE absence of expected means something is off
CROSS PRESENTATION IS IMPORTANT because it allows APCs (like dendritic cells) to load antigen into MHCI and subsequently activate CD8 T cells (killer T cells)
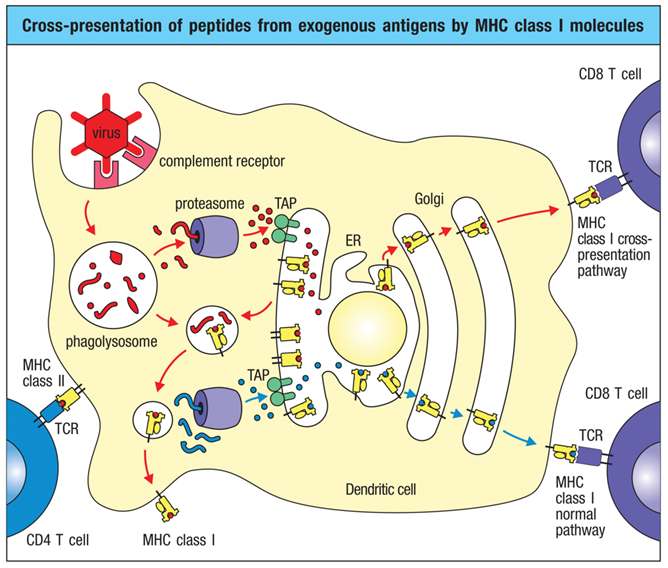
Why do we have two different strategies re: MHC?
We have two different strategies for DIVERSITY!
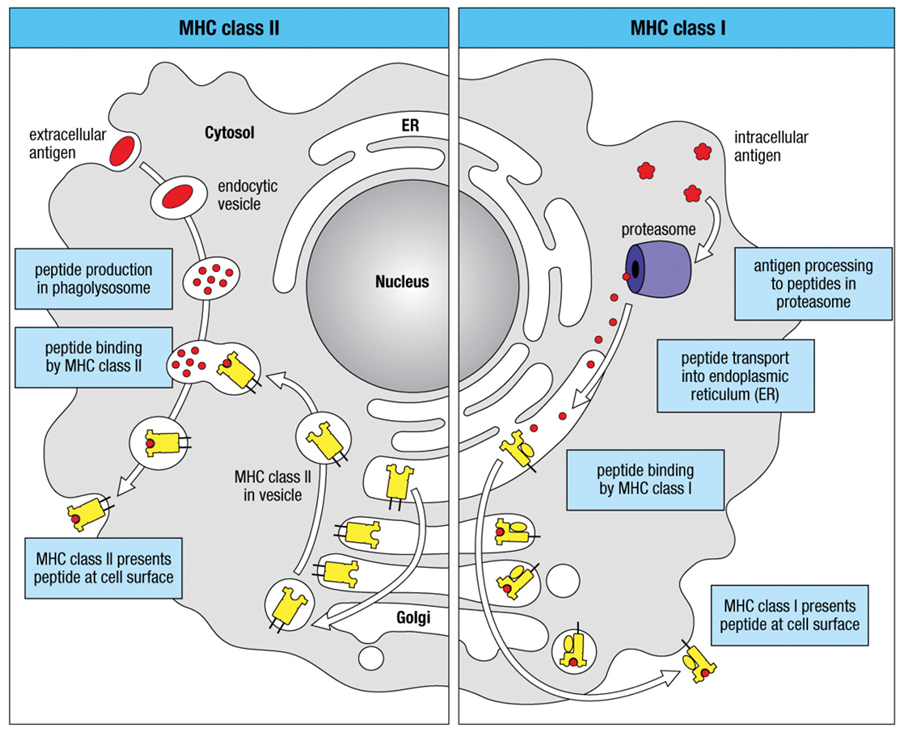
Are there problems with antigen processing?
•Yes, there can be!
•These reactions happen even in the absence of antigen… what if MHC binds host tissue?
•Possible & in rare cases could elicit an autoimmune response
•What if the pathogen resists intracellular degradation (like tuberculosis)?
•No processing, no presentation by MHC II; so not as strong of an immune response
•But, overall, it’s a good surveillance system for both intracellular and extracellular pathogens so that your T cells can create very specific responses
How was the MHC discovered?
•By accident!
•1954: Richard Herrick was dying of kidney failure and was put on a dialysis machine
•He was referred to John Merrill, a transplant doctor who had 9 failed attempts at kidney transplantation
•He believed that it was not his technique that was causing the issue, but it was some hereditary component that was influencing the success of the transplants
•Herrick had an identical twin brother, and they did a transplant from the healthy to the sick twin
•Richard lived & thrived: this was the first successful solid organ transplant
ie: both had the same MHC so the organ was not rejected
What is the MHC? Where is it located?
•The Major Histocompatibility Complex
•A tightly linked cluster of genes that is present in all mammals
•In humans, it is 4 million base pairs of DNA on the short arm of chromosome 6
•Is divided into three regions (below)
•Also called the HLA (Human Leukocyte Antigen) Complex
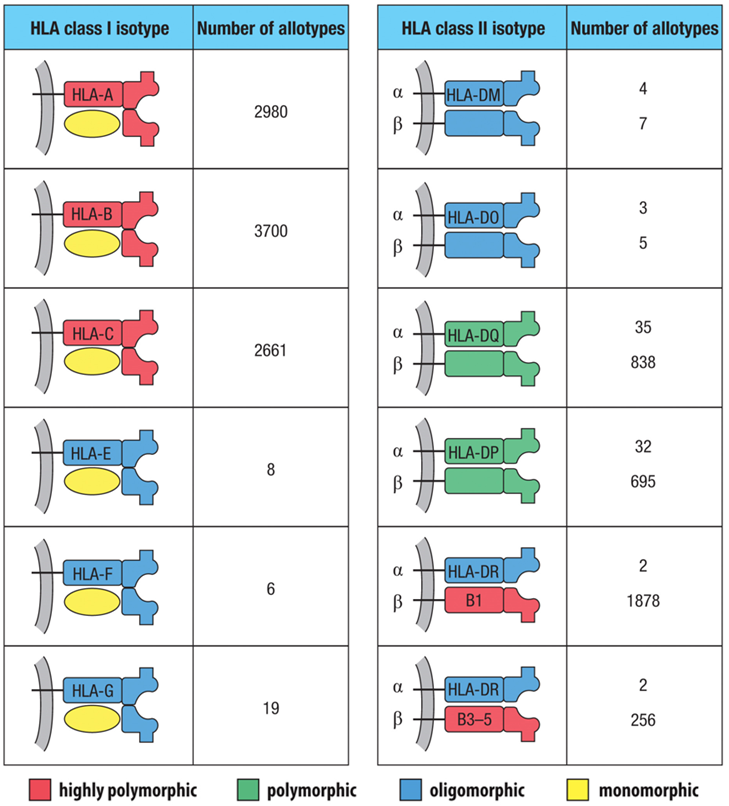
THE MOST DIVERSE SET OF GENES IN THE HUMAN GENE POOL
•The gene products play a role in intracellular recognition of self vs. non-self (i.e., self-discrimination)
•It influences the number of antigens to which a person can respond
•Different MHC combinations make people respond to infectious diseases differentially (i.e., stronger vs. weaker responses)
•It a the region of the human genome that is strongly and frequently correlated with human disease and its therapy
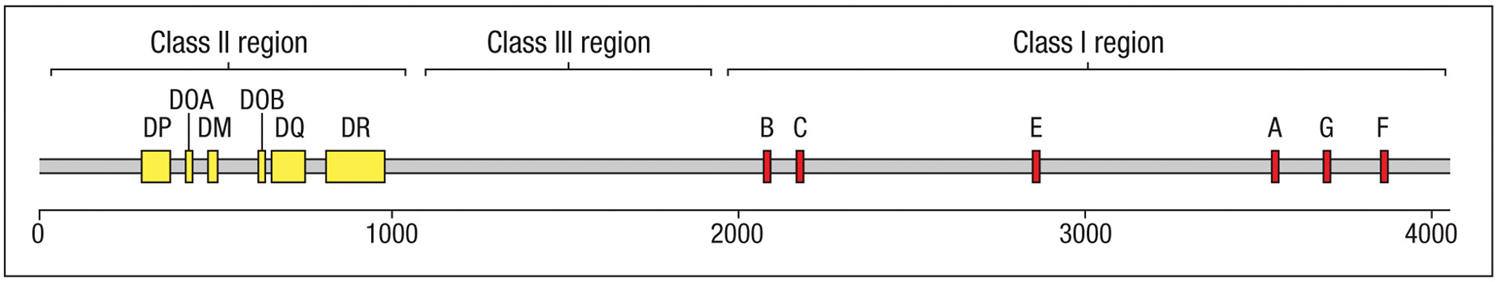
How many different genes are found in the class 1 region?
six expressed HLA class I genes + 11 nonfunctional class I genes and gene fragments
How many different genes are found in the class 2 region?
contains the expressed class II genes + 5 nonfunctional class II genes
How many genes are found in the class 3 region?
no class I or II genes, but dense with other genes that encode for things like complement proteins (C2 and C4) & inflammatory cytokines
What do class I, II, and III genes do?
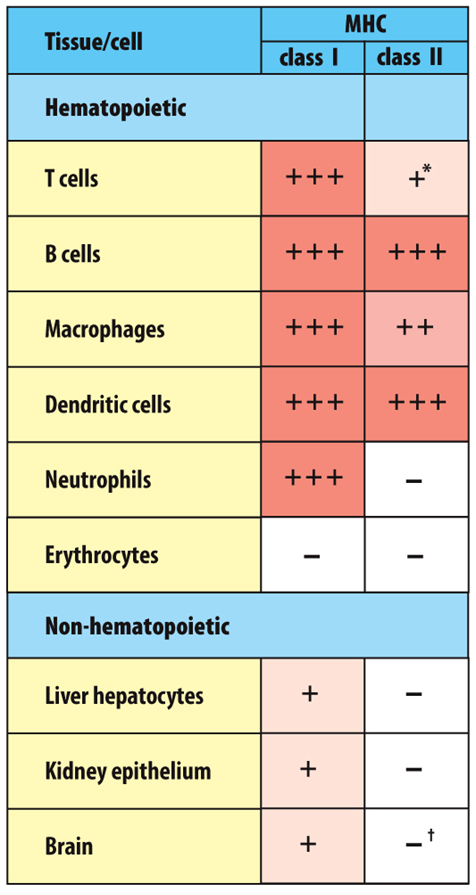
What does a class 1 gene do?
•Glycoproteins expressed on the surface of nearly all nucleated cells
•Major function is to present antigen to cytotoxic T cells (CD8)
What does a class 2 gene do?
•Glycoproteins expressed on all antigen presenting cells
•Major function is to present antigen to T helper cells (CD4)
What does a class 3 gene do?
Other immune functions: complement and inflammatory cytokines
What does diversity in MHC come from? Why is this important?
•Not as great as B and T cell receptors
•Coded for by conserved (stable) genes:
•Your MHC haplotype: expression of at least 3 different class I molecules and 4 different class II molecules
•The presence of multiple alleles at a given genetic locus means that many alternative forms exist
Your genetics determines what you express in your cells!
Describe how natural selection can influence the MHC haplotypes that are present in a population over time.
Balancing selection (the heterozygous advantage)
•For an individual, having multiple HLA class I and class II genes is an advantage
•They contribute different peptide-binding specificities, allowing for a greater number of pathogen-derived peptides to be presented during infection
•This improves the immune response by increasing the number of activated pathogen-specific T cells
Directional selection
•Imposed by epidemics
•Favors particular HLA class I or class II alleles or combinations of alleles at the expense of others
•Antigen presentation by particular MHC allotypes is advantageous, so selection favors* those, increasing their frequency
selection favors= more likely to survive, reproduce, & genes move on to next generation within population
REMEMBER THE BIG FOUR ACT AT THE POPULATION LEVEL (GENE POOL), NOT ON INDIVIDUALS
What are the consequences of natural selection on MHC haplotypes?
Extreme Polymorphism: The MHC is the most polymorphic region in vertebrates.
Long-Term Survival: This constant selection ensures the population retains the ability to adapt to new or evolving pathogens (e.g., pathogens cannot easily wipe out the entire population).
Disease Susceptibility: Specific, fixed, or highly common MHC haplotypes can affect the population's susceptibility to autoimmune diseases, as pathogen-driven selection can create a trade-off where advantageous resistance genes also promote autoimmunity.
Trans-species Polymorphism: Some MHC haplotypes are so ancient and well-selected that they are shared between species.
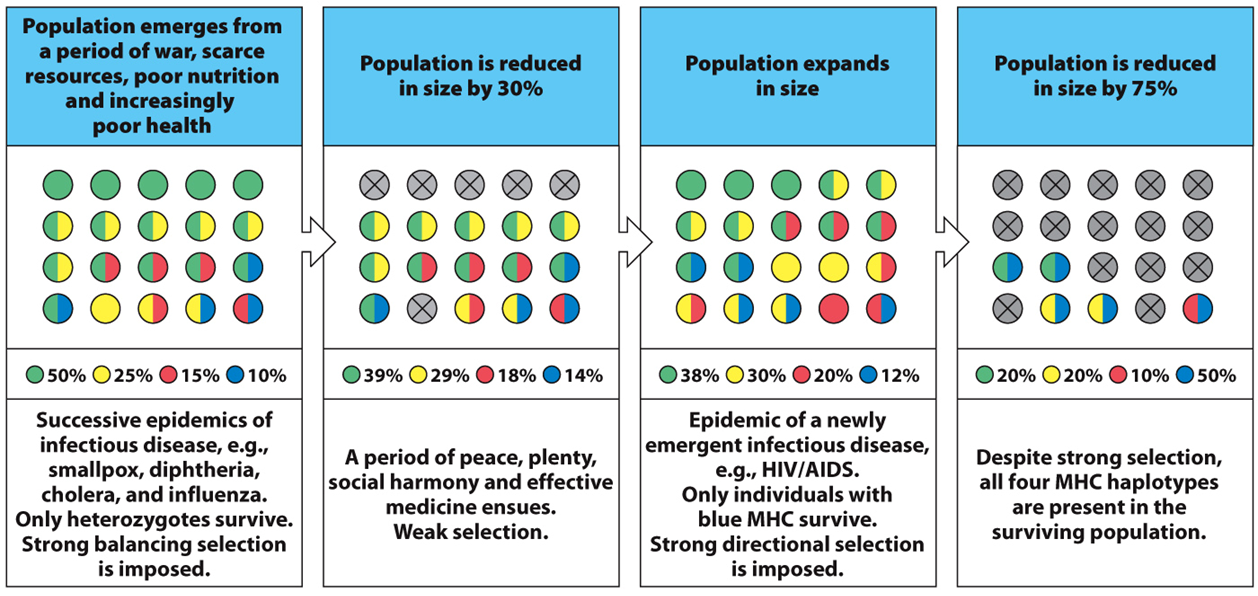
Example of haplotype selection
•4 different haplotypes (green, yellow, red, & blue) with given frequencies
•Panels 1 → 2: The population experience a period characterized by balancing selection arising from successive epidemic infections. Heterozygotes survive & haplotype frequencies shift. The remaining population goes through a period of calm and health and starts to recover.
•Panels 3 → 4: Then, DISEASE! Nasty emerging zoonotic!! One haplotype is favored (i.e., blue) and only those individuals with it emerge from the epidemic. You see directional selection, but all four haplotypes are retained.
•ex in history: black death plague
Why does your haplotype matter (functionally)?
It is very important in terms of how you fight disease; it sets a genetic predisposition to disease
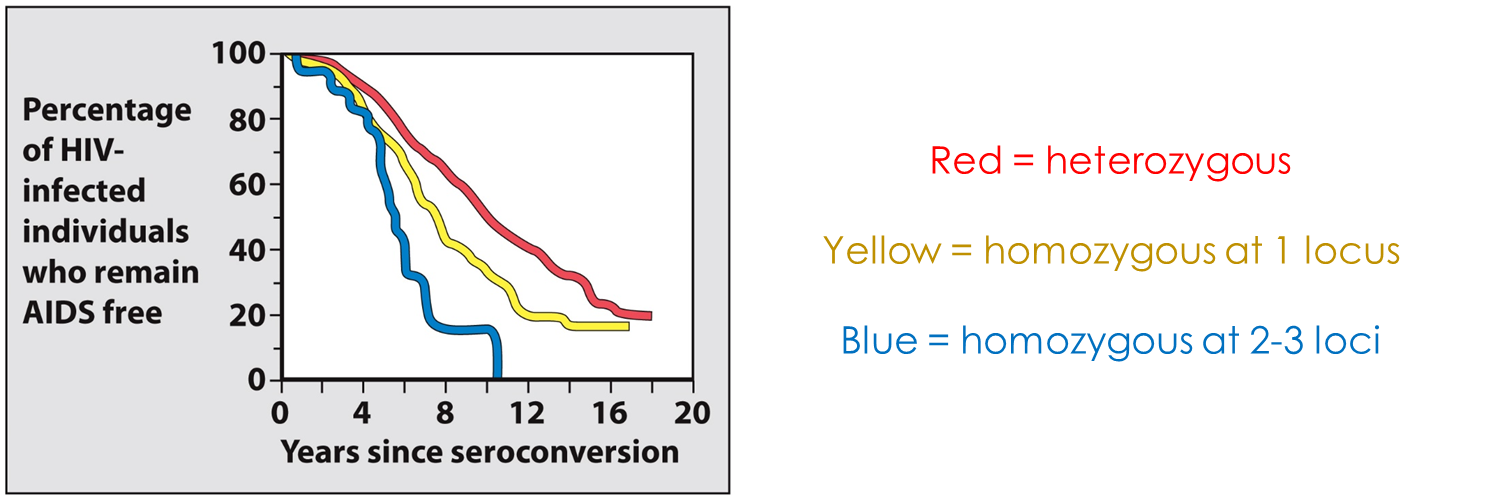
•More diverse = better chance of being a long-term non-progressor (i.e. remaining AIDS free)