CEE 3 MODULE 1- MEDICINAL ORGANIC CHEMISTRY
1/194
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
195 Terms
incorporates different branches of chemistry
Medicinal Chemistry
any substance intended for the prevention, diagnosis, cure, mitigation, or treatment of symptoms of a disease or abnomarl condition
Drug
Drug can be classified according to its:
Origin
Medicinal use
Type of disease
Origin can be:
Natural compound
Synthetic compound
Semisynthetic compound
fights malignant cells/ tissues
Chemotherapeutic agent
Acts on various physiological functions of the body
pharmacodynamic agent
A type of disease that can be transmitted via person-to-person or outside agents
Infectious diseases
A type of disease that disorders of the human body are caused by genetic malfunction, environmental factors, stress, or old age
Non-infectious diseases
Pain alleviation, prevention of pregnancy, anesthesia
Non-disease
various chemical compounds are bound and produce a pharmacological response
Receptor
side chains on the surfaces of the cells were complementary to the dyes; gave way to SELECTIVE TOXICITY
Receptor Hypothesis
Who discovered the receptor hypothesis
Paul Ehrlich
The substrate is completely complementary in shape to the active site, so that it fits in perfectly
Lock and key theory
The substrate and active site are not completely complementary, but the enzyme conforms to the shape of the substrate
Induced fit theory
Maximum pharmacologic effect can be obtained if all the receptors are occupied; the pharmacologic effect of the drug depends on the percentage of the receptors occupied
Hypothesis of Clark
The effectiveness of a drug lasts as long as the receptor is occupied
Hypothesis of Ariens and Stephenson
Effectiveness of a drug does not depend on the actual occupation of the receptor, but on obtaining a PROPER STIMULUS
Hypothesis of Paton
Active-site-directed irreversible inhibition;
Covalent bond formation between receptor and ligand
Hypothesis of Baker
The more potent enantiomer should be involved in a three-point fit to the receptor
Easson-Stedman hypothesis
It identifies what type of disease to expect
Target identification
Lead and natural sources from folklore medicine are assayed
Natural Product screening
All synthetic organic compounds available are tested in a pharmacological assay for a specific type of biological activity
Random screening
Focused approach on structural knowledge of the receptor or ligands to design, identify, or create a 'lead'
Rational drug design
Conducted for clinical candidates or drugs already in the market
Drug metabolism studies
defined as the concentration of the dissolved solute; a function of the presence of both lipophilic and hydrophilic features within its structure
Solubility
Used to describe the lipophilicity of a drug
Partition coefficient
Also known as hepatic metabolism, biotransformation, and detoxification
Metabolism
Breakdown of drug into smaller, more water-soluble compounds is called
Metabolites
The most important site for drug metabolism, well-perfused organ
Liver
Orally administered drugs that are absorbed into the bloodstream
First-pass effect (pre-systemic metabolism)
Also known as proagents, bio-reversible derivatives, congeners, and latentiated drugs
Prodrugs
A chemically modified inert precursor of the drug that, on biotransformation, liberates the pharmacologically active parent compound
Prodrugs
What phase of metabolism aims to increase polarity and promote excretion
Functionalization reaction
The most common and most important reactions
Functionalization reaction
Most common phase 1 reaction
Oxidation
CYP enzyme for paracetamol
CYP2A1
The most studied CYP enzyme
CYP2D6
most abundant CYP enzyme
CYP3A4
What phase 2 reaction causes gray baby syndrome
Glucuronidation
Most common phase 2 reaction for children
Sulfation
Designed to find relationships between chemical structure and biological activity of studied compounds
Structure-activity relationship (SAR)
Molecular framework defining the important or necessary components responsible for the biological activity of the compound
Pharmacophore
Groups that have nearly equal molecular shape and volume, approximately same distribution of electronm and has the same physiochemical properties
Bioisosterism
Replaces certain number of atoms, valency, degree of unsaturation, and aromaticity with its biological properties still the same
Classical bioiosteres
comprises groups which are structurally similar but DO NOT meet the steric and electric requirements
Non-classical bioiosteres
any ring system that are made up of carbon atoms and at least one othe element (sulfir, nitrogen, or oxygen)
Heterocyclic compound
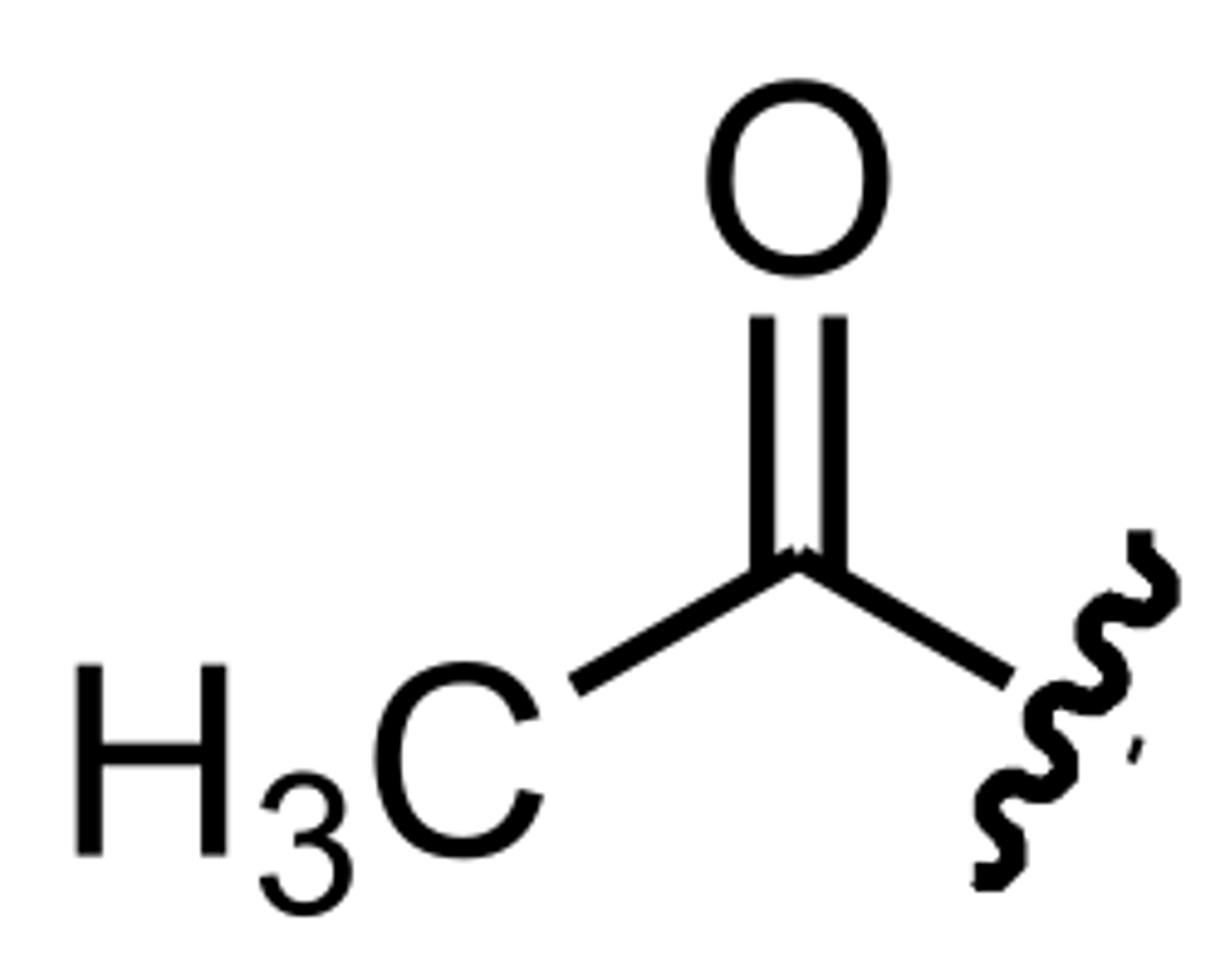
Acetyl
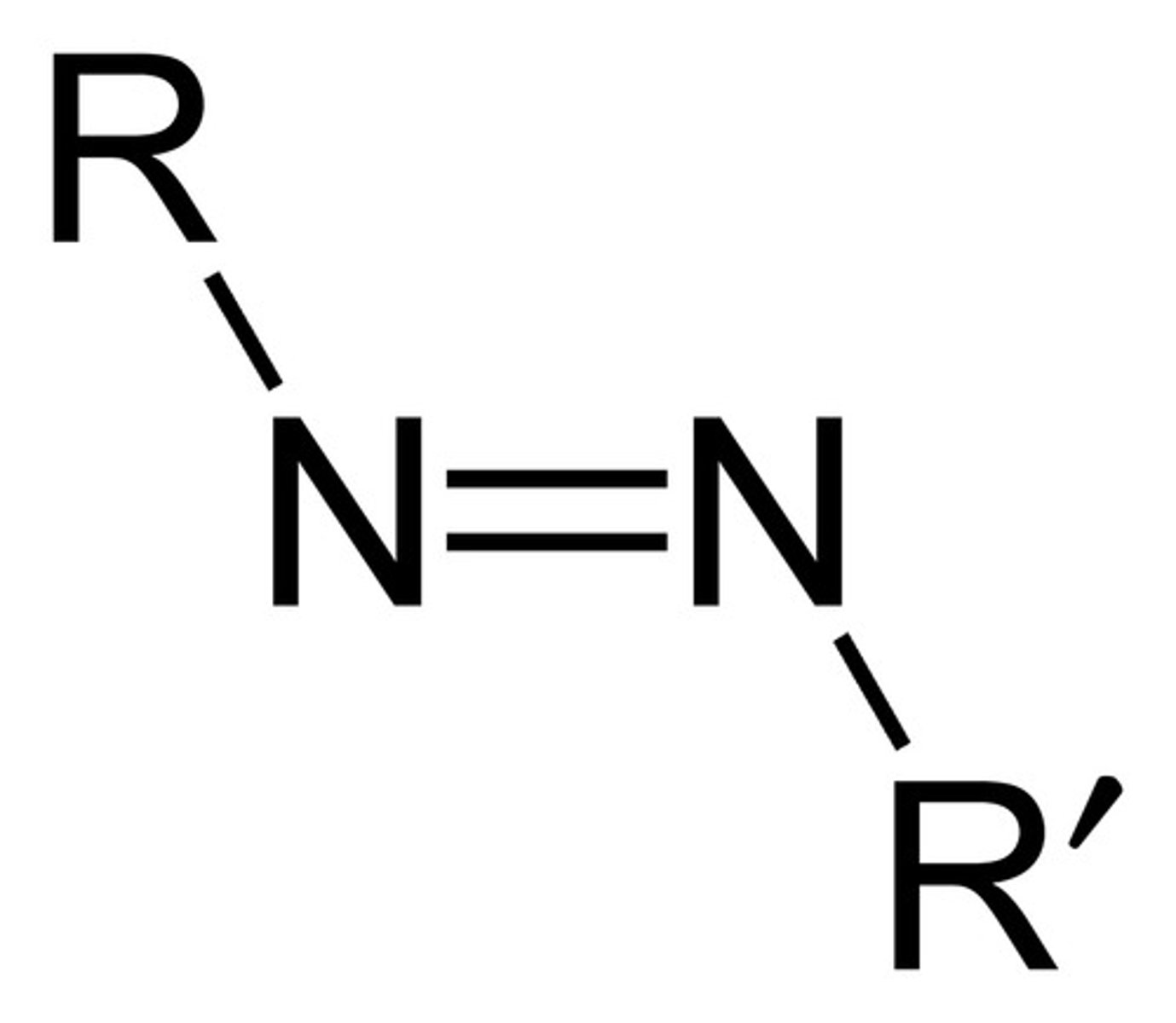
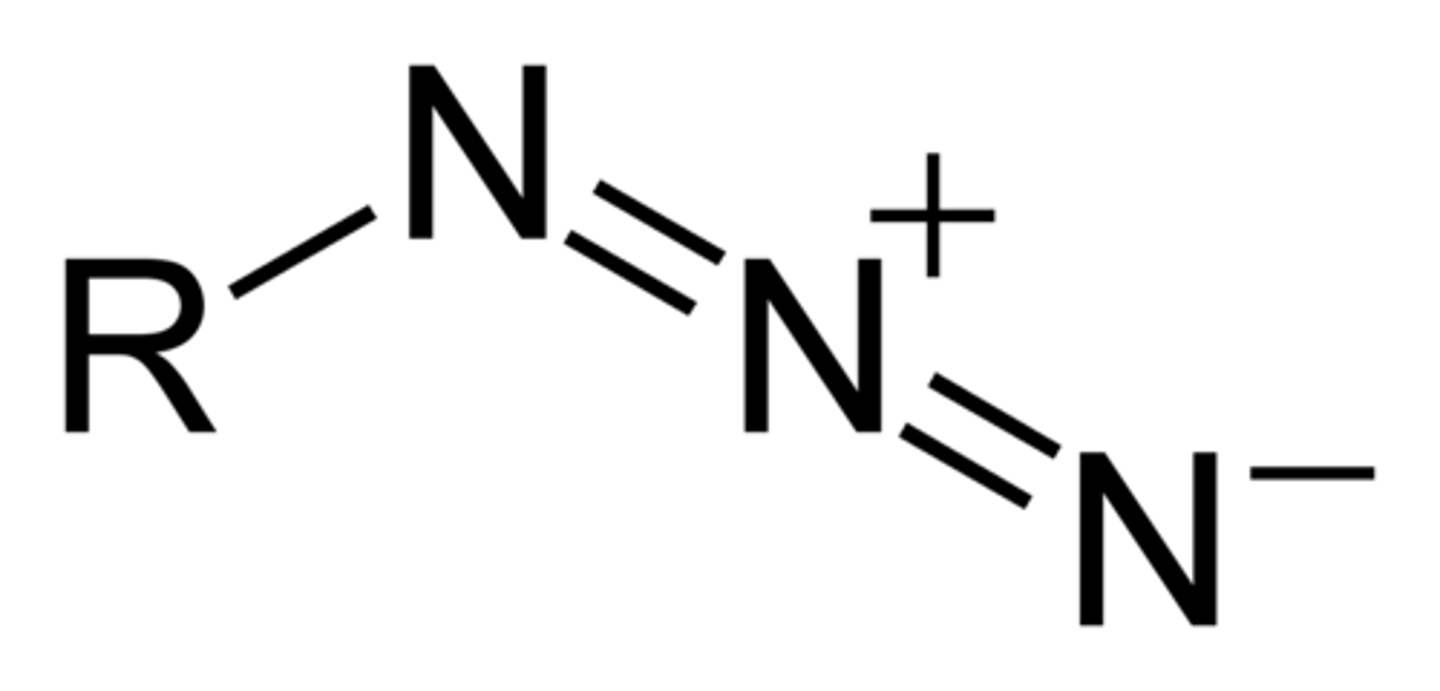
Azo
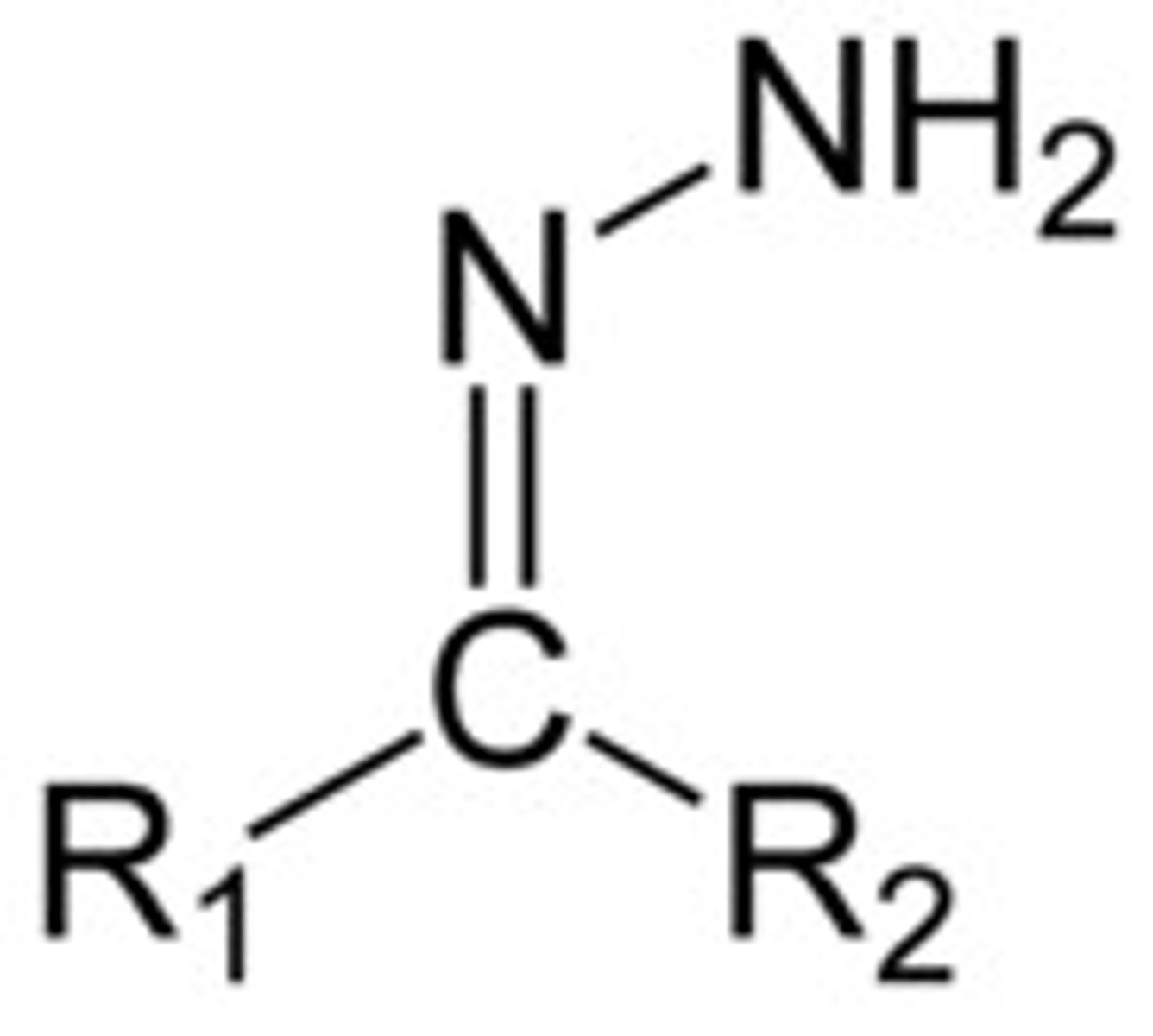
Has an amino group attached to Nitrogen
Hydrazone
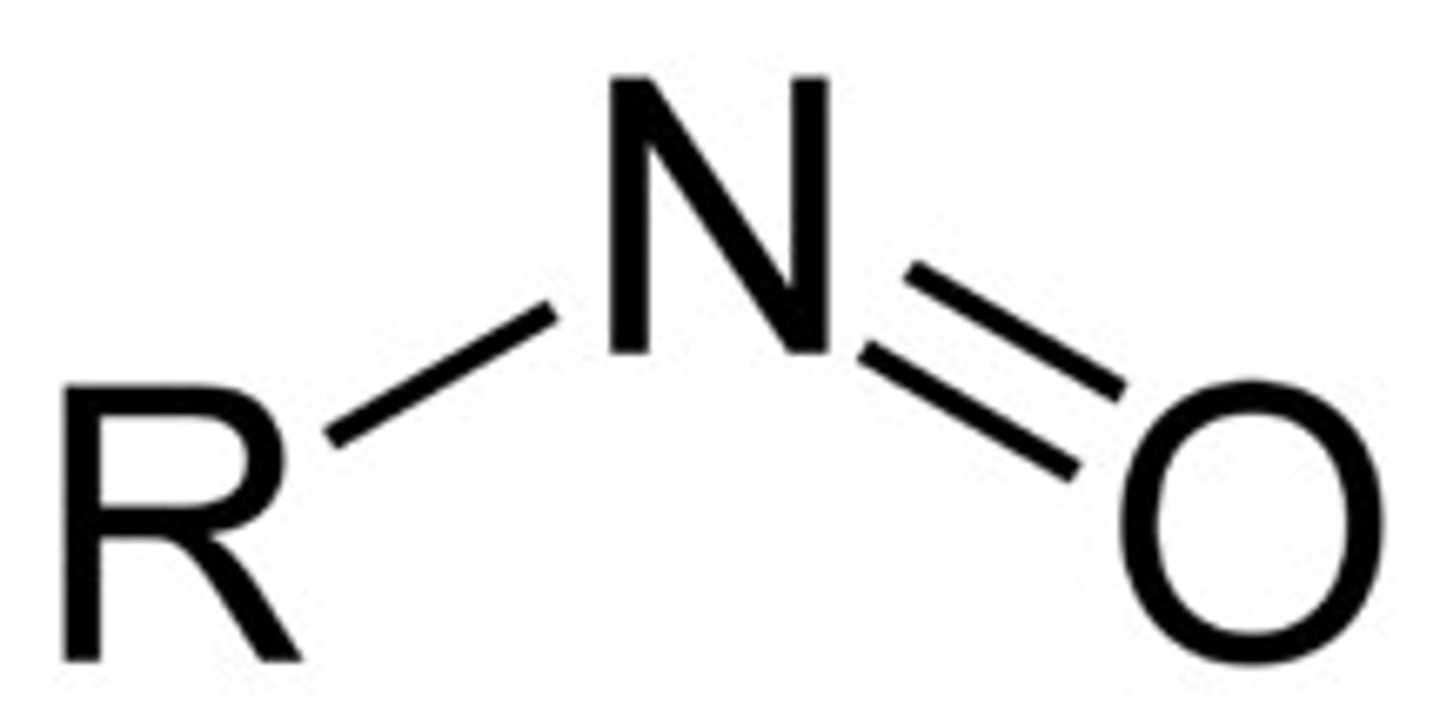
Nitroso
Nitro
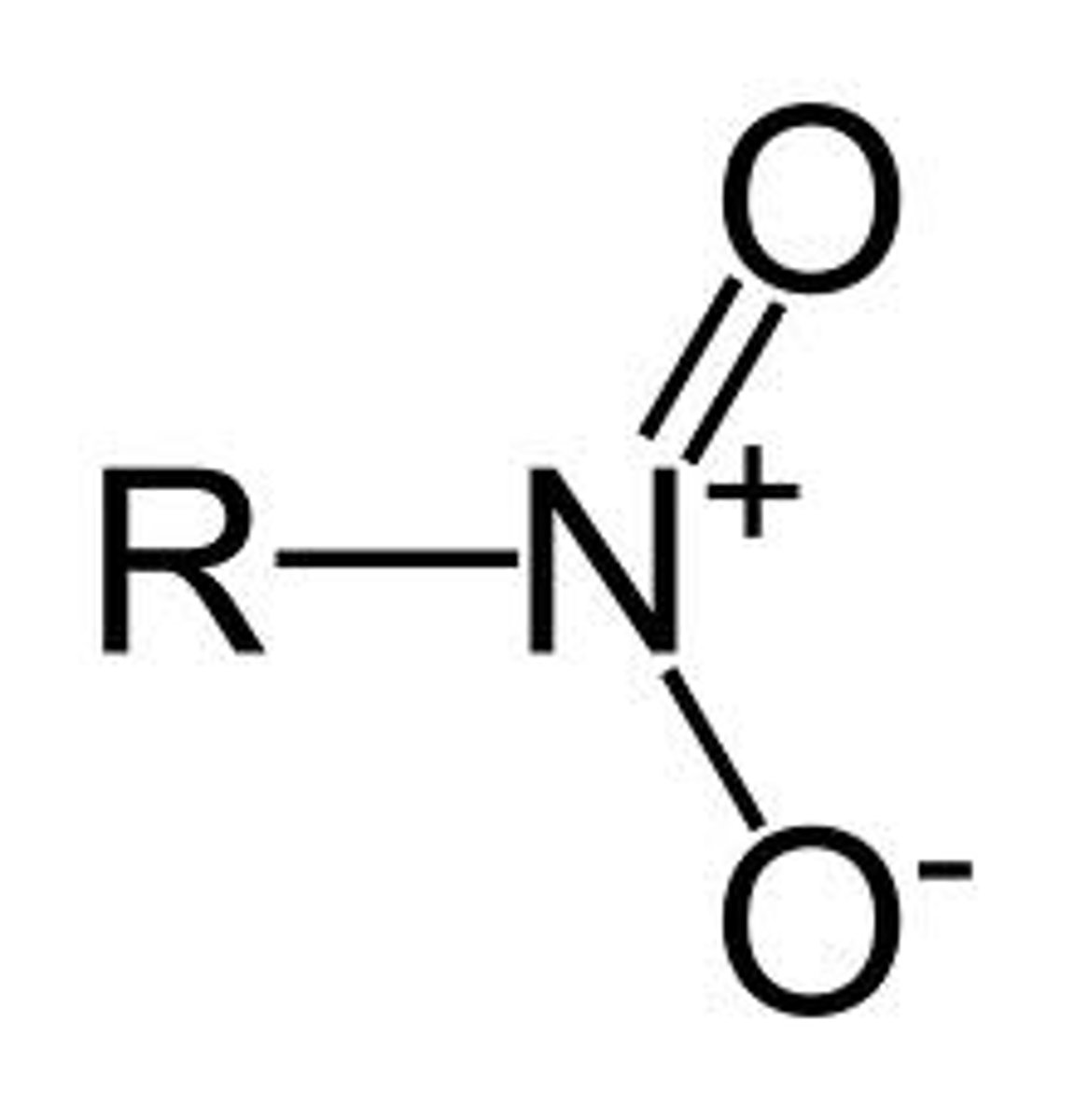
Azido
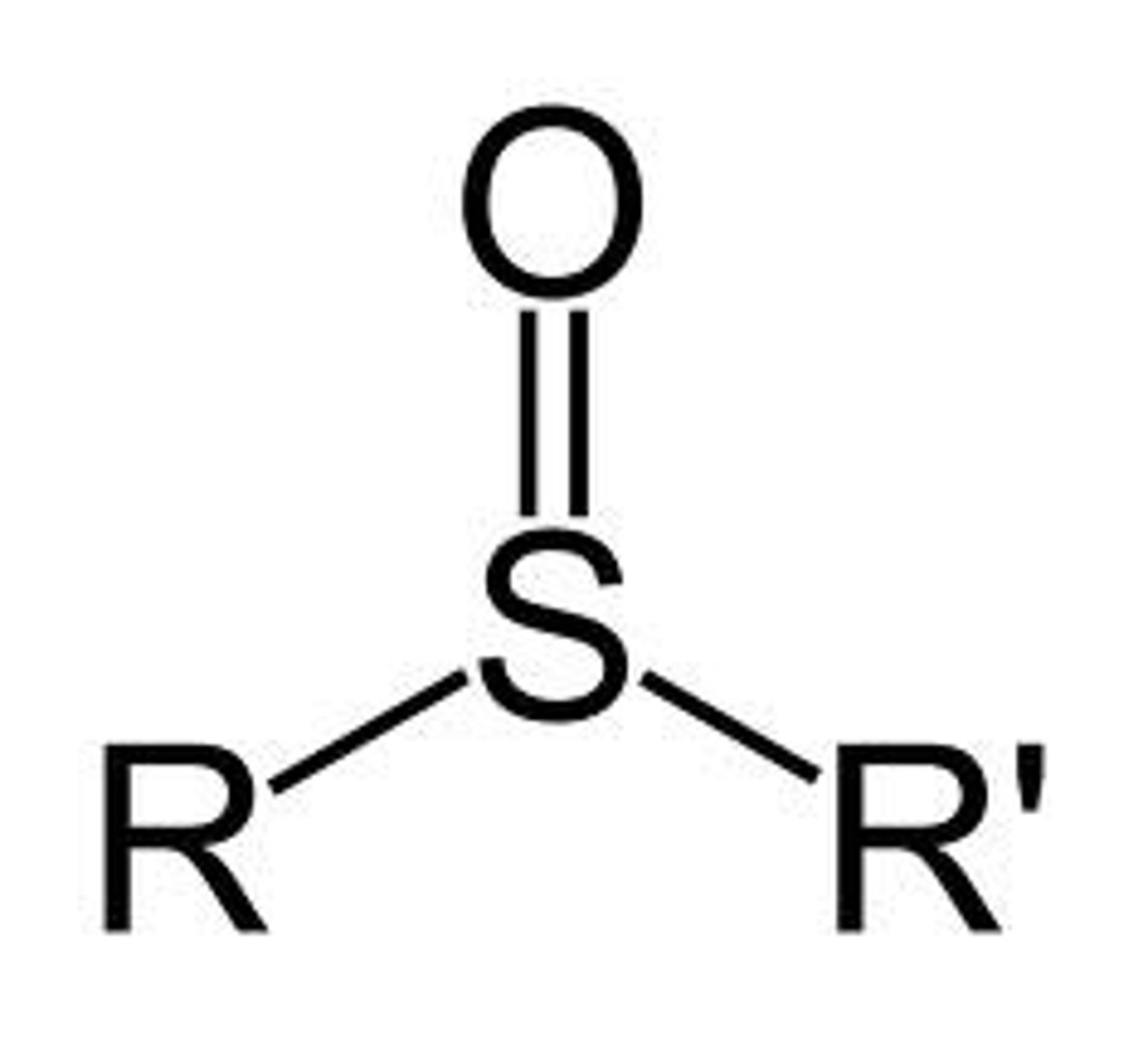
Sulfoxide
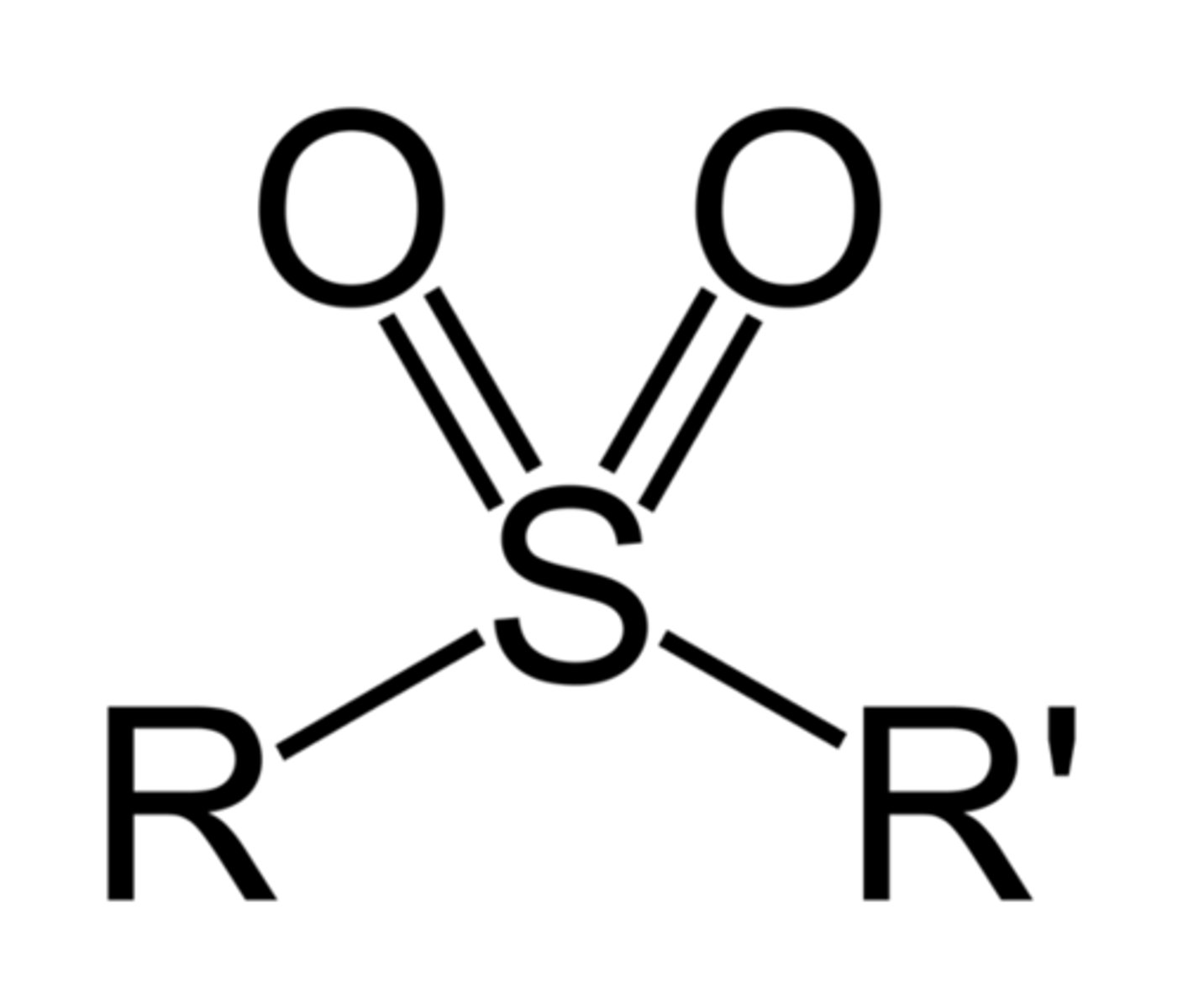
Sulfonyl
"NOO"
Carbamate
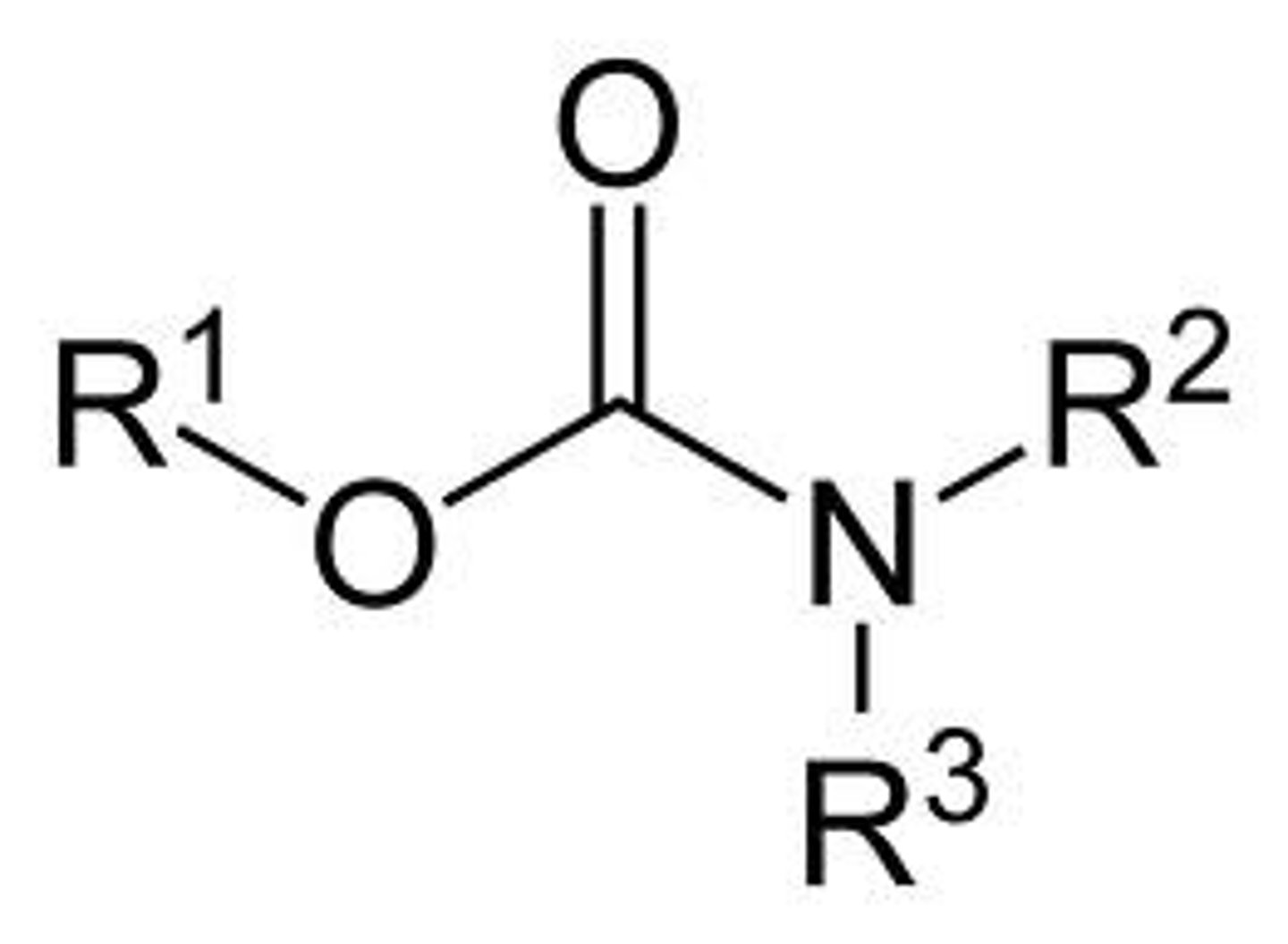
"Non" has 2 amino group
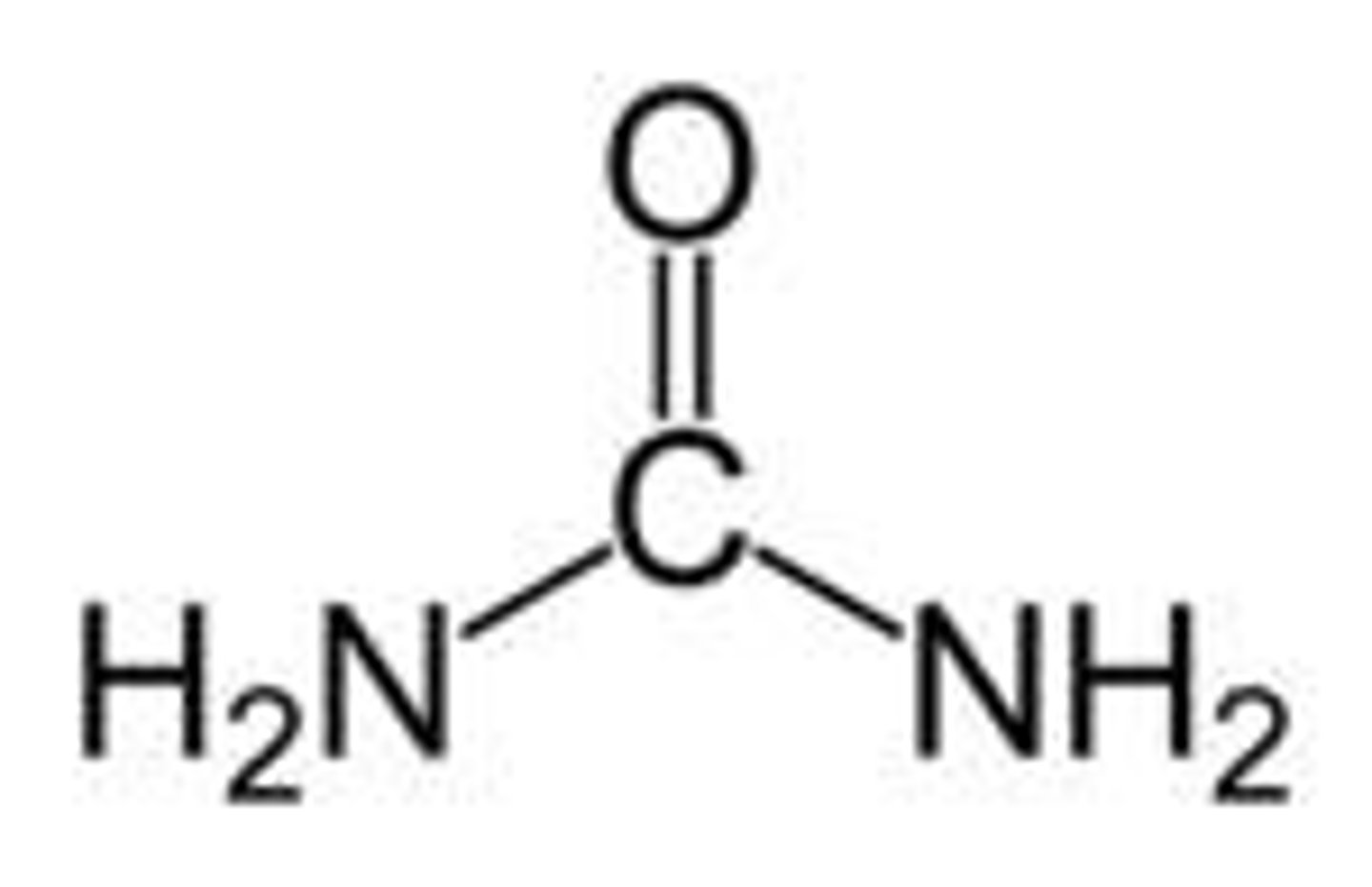
Urea
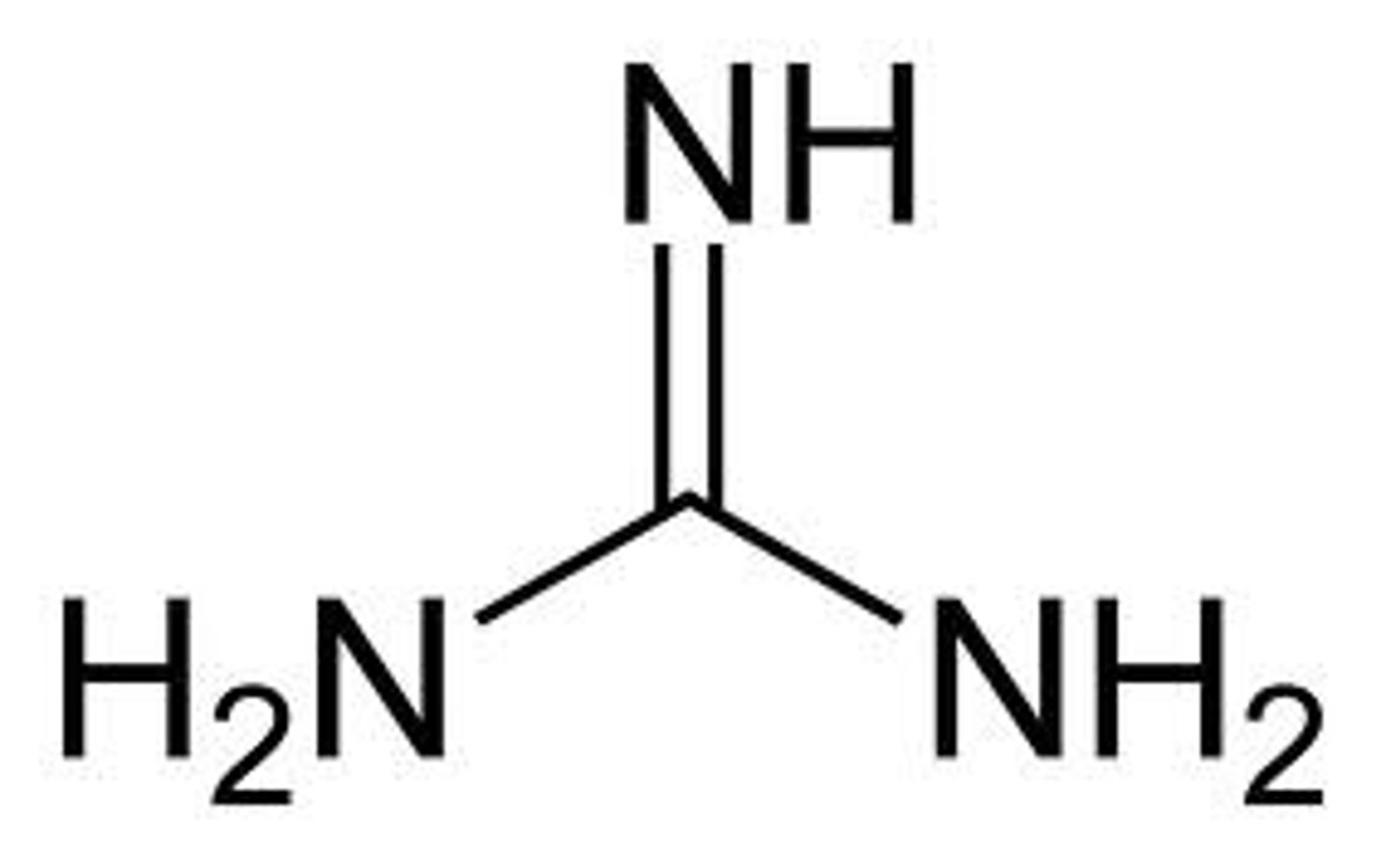
"NNN"
Guanidine
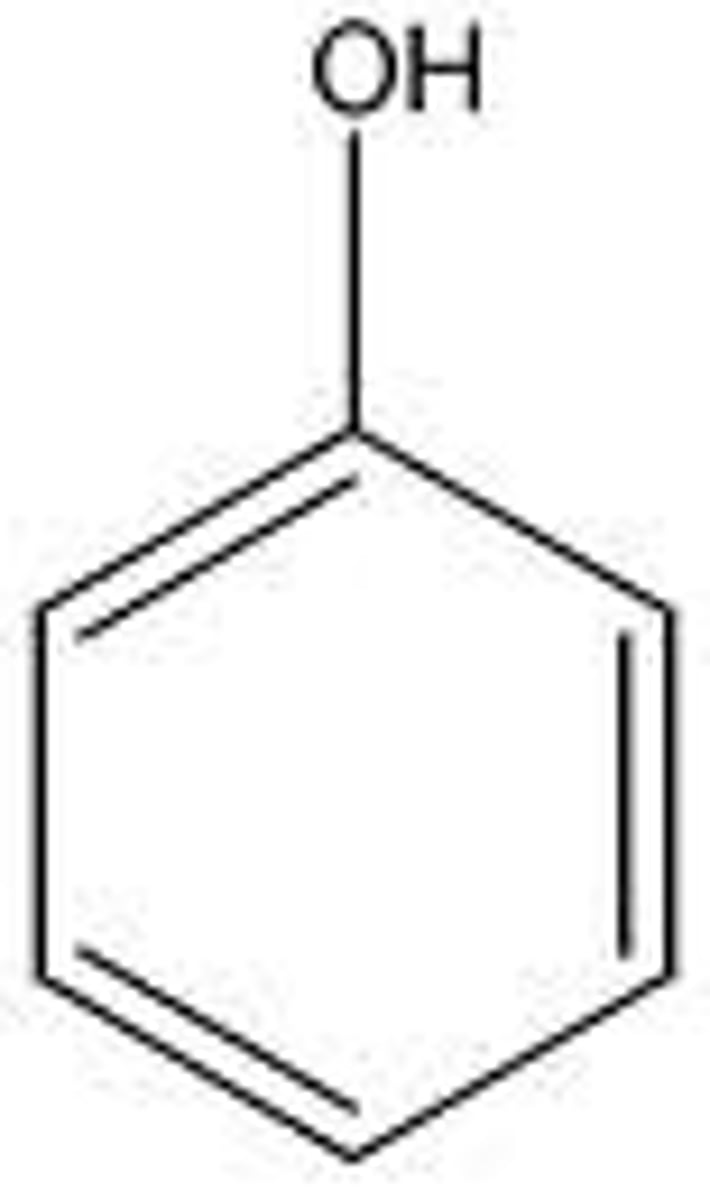
Phenol
catechol
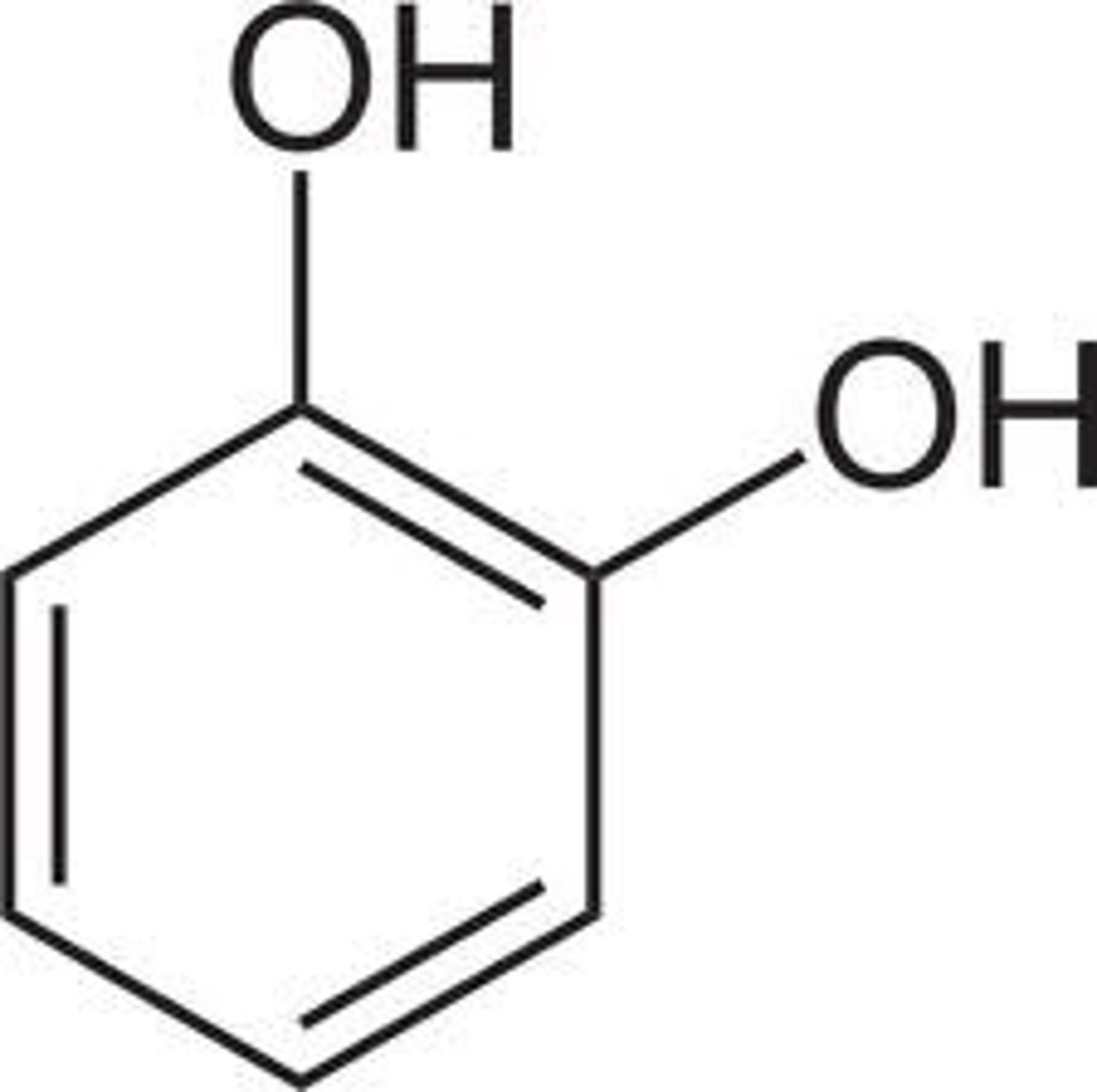
Resorcinol
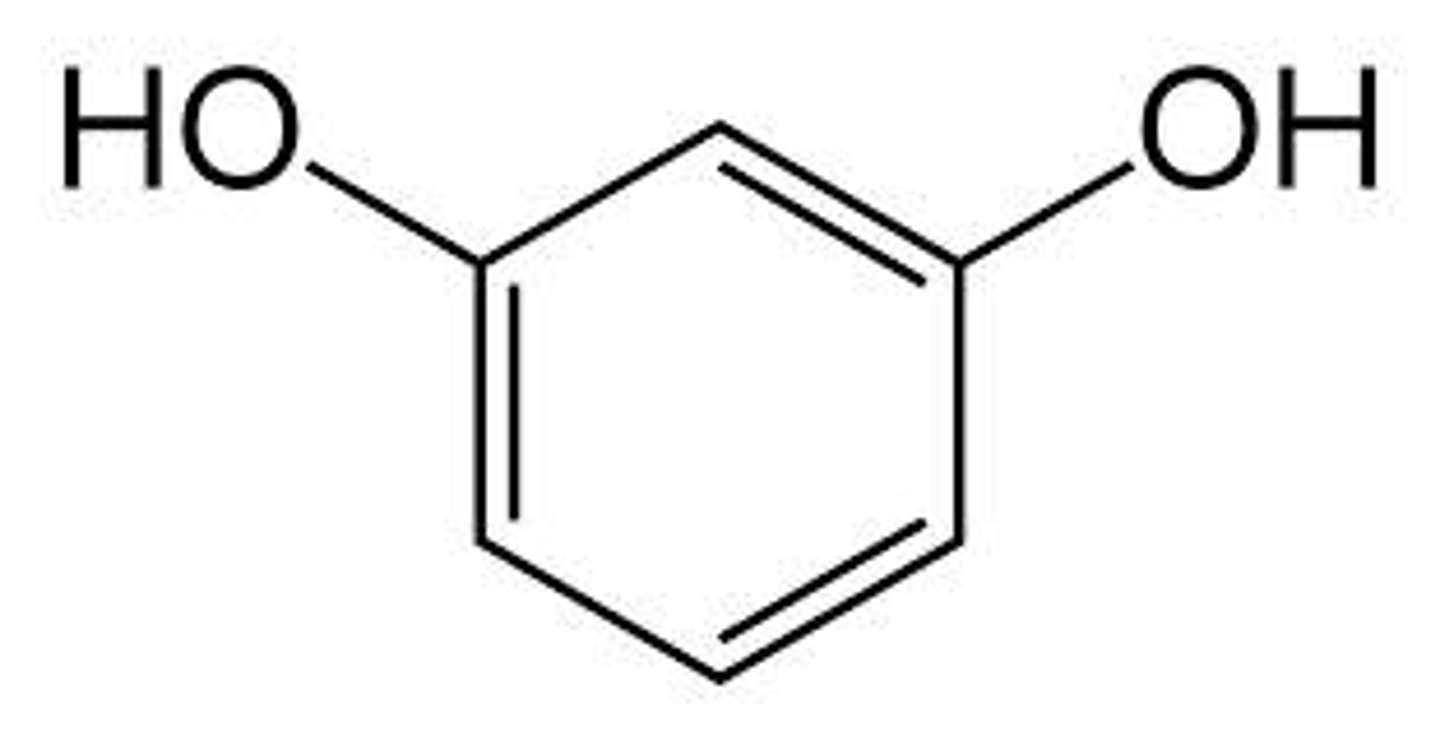
Hydroquinone
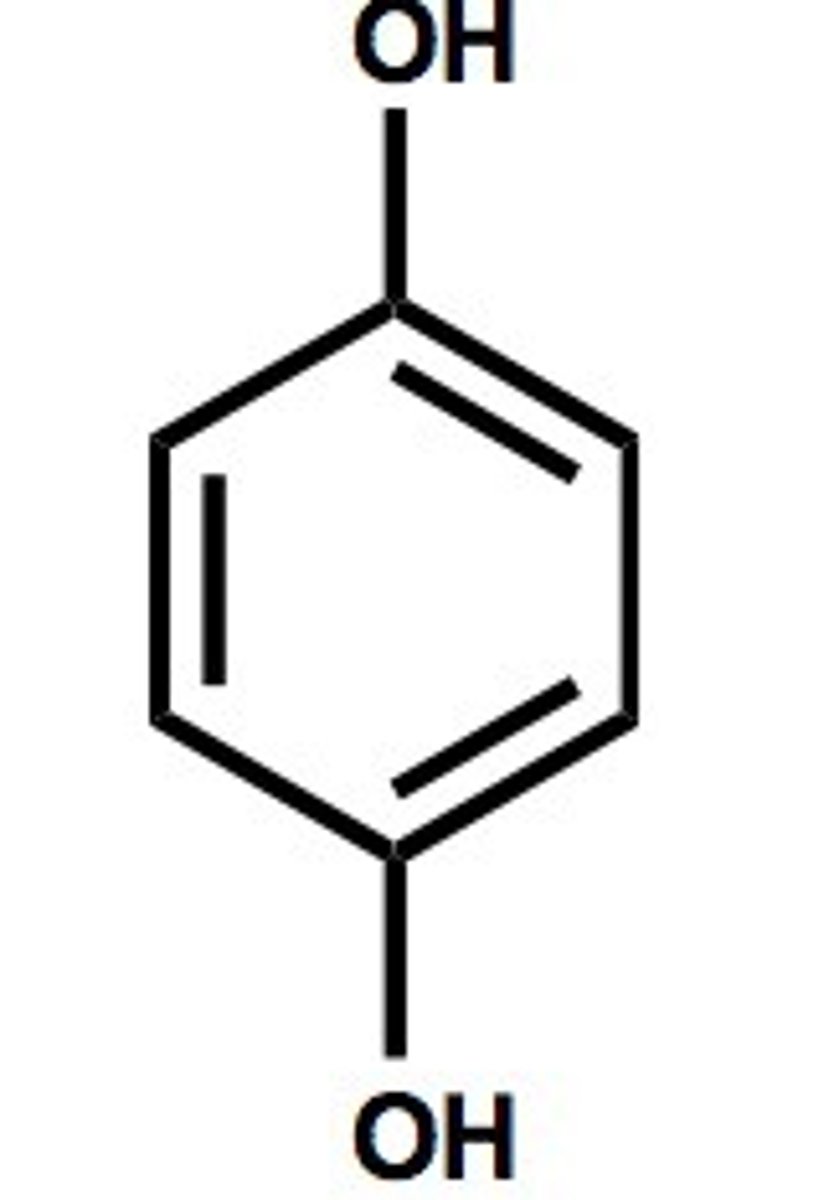
Toluene
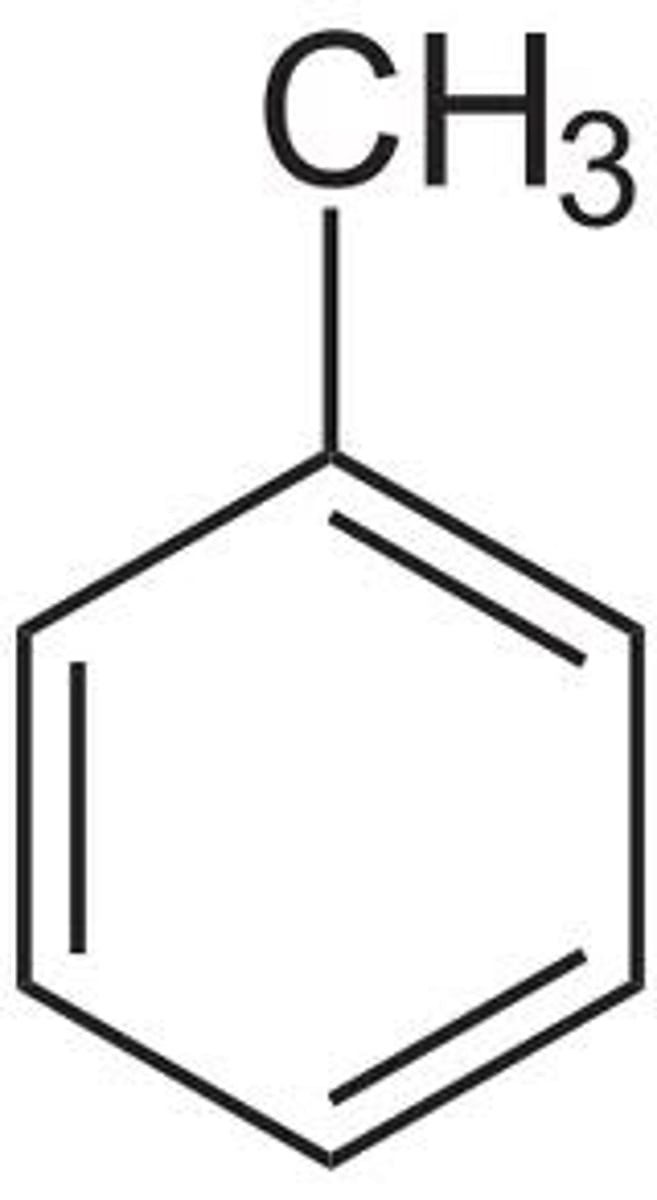
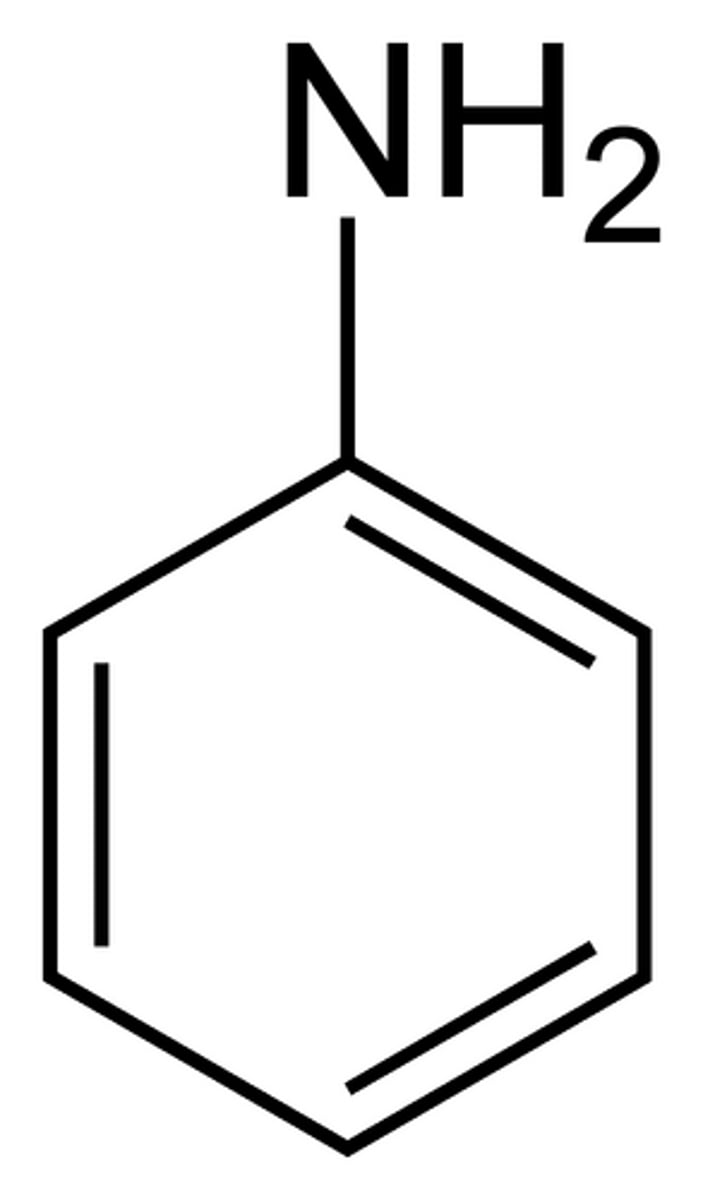
Aniline
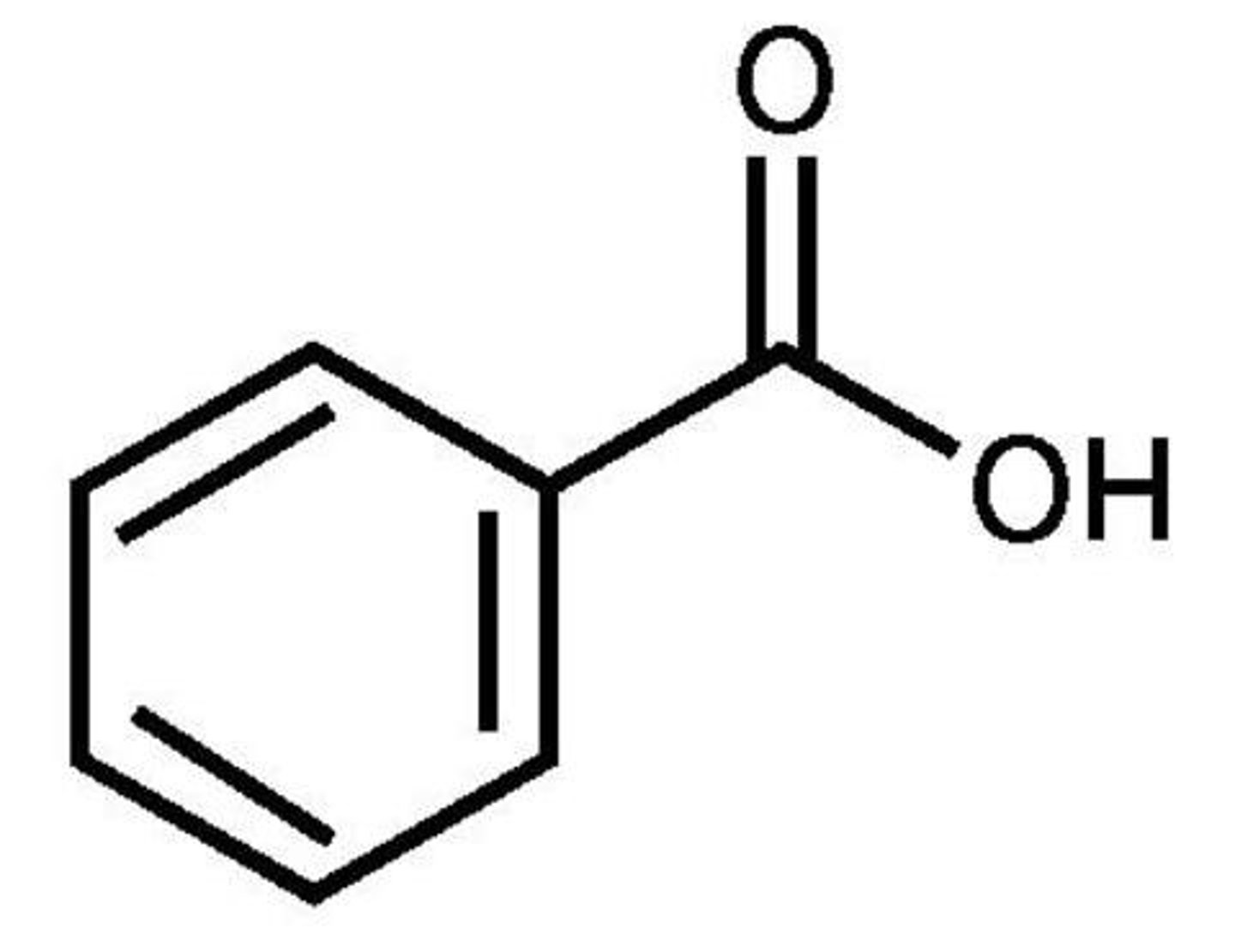
Benzoic acid
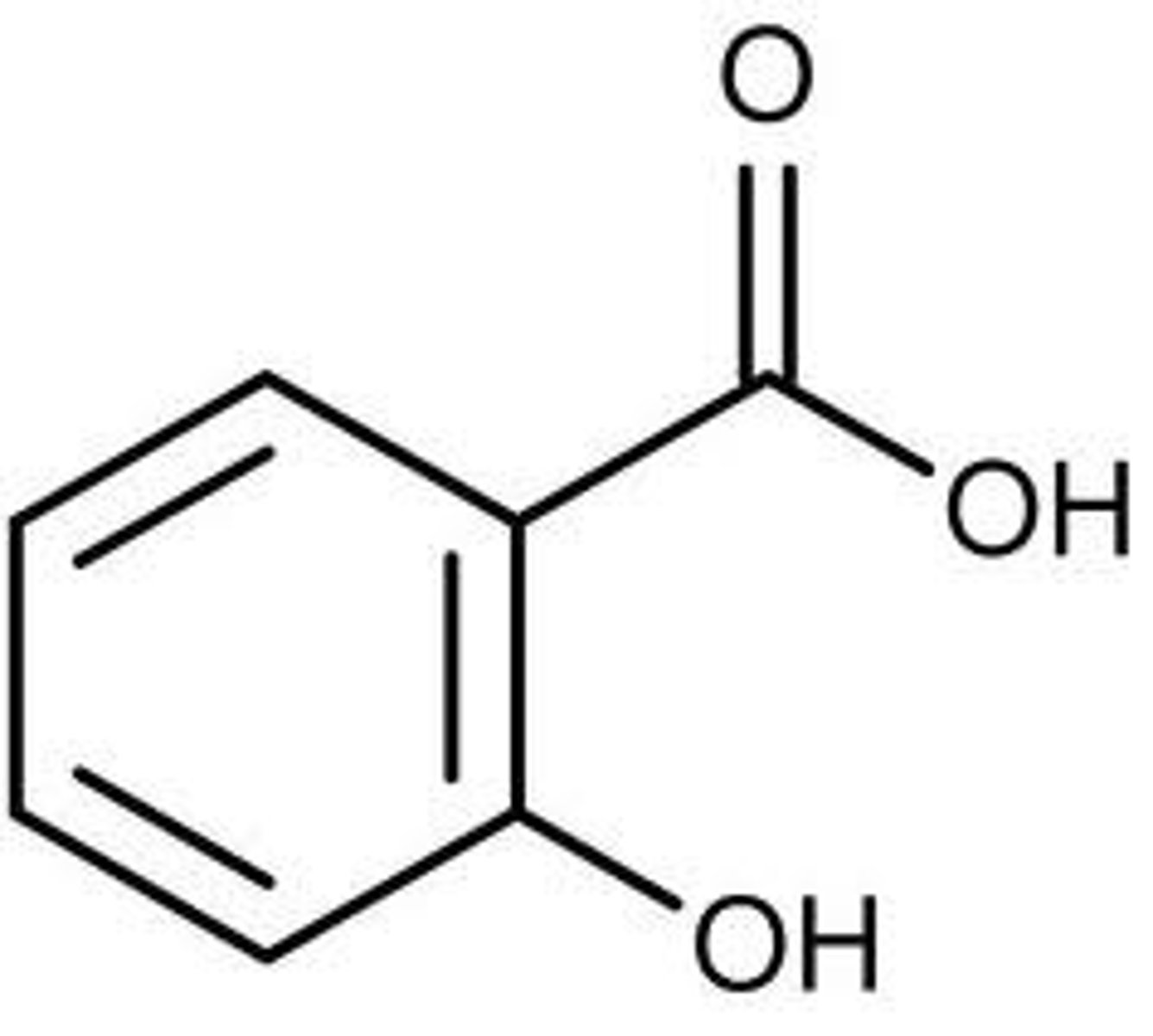
Salicylic acid
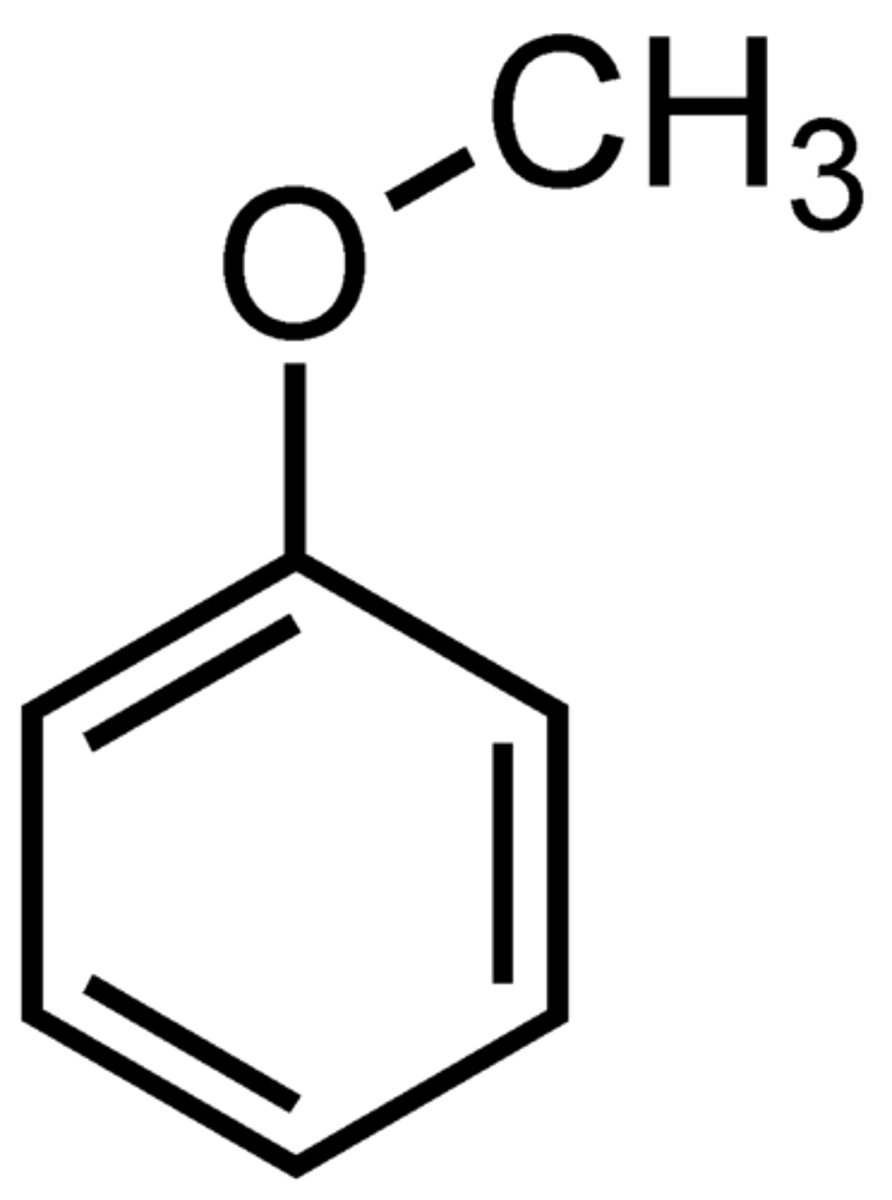
Anisole
Cresol
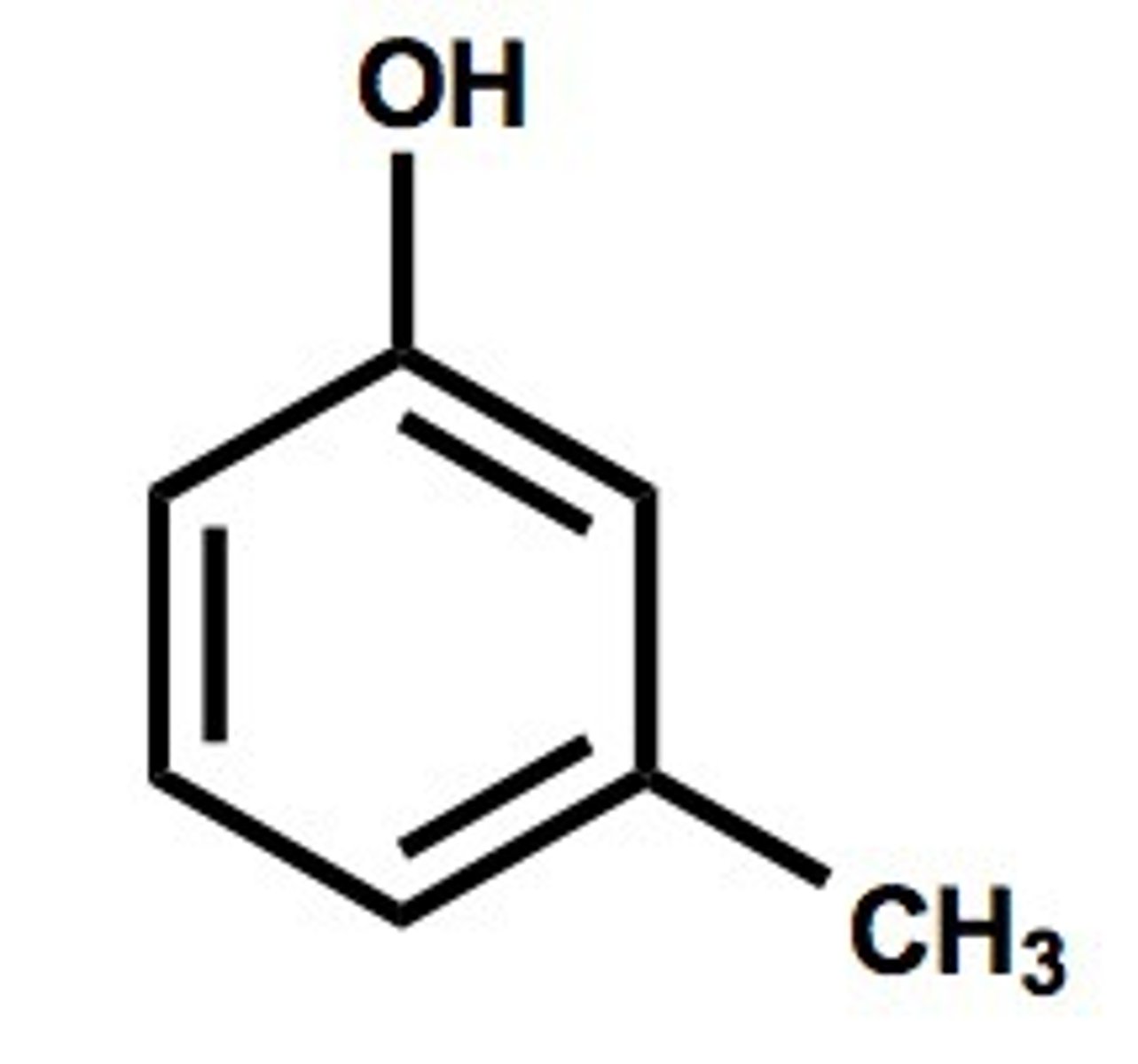
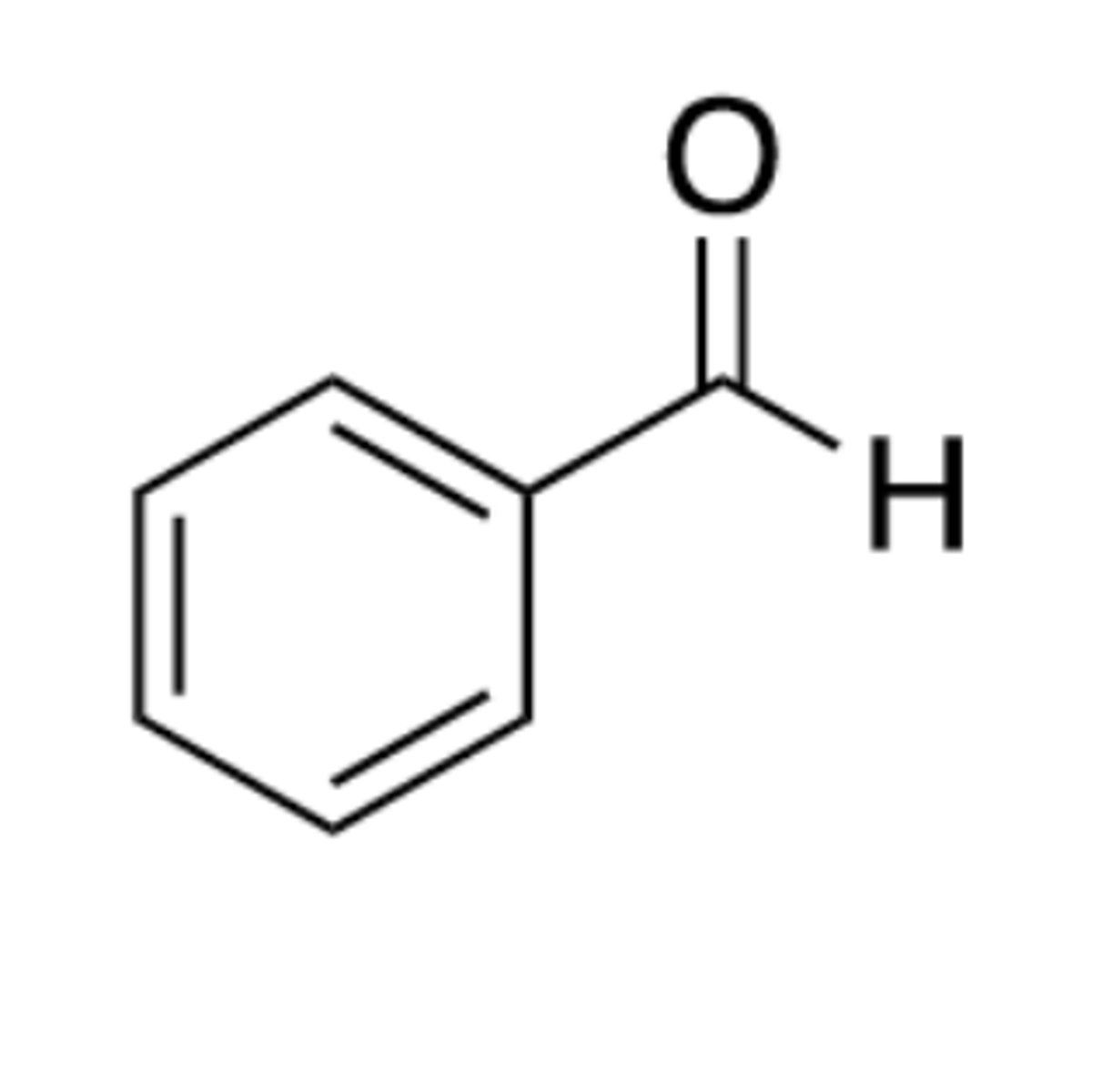
Benzylaldehyde
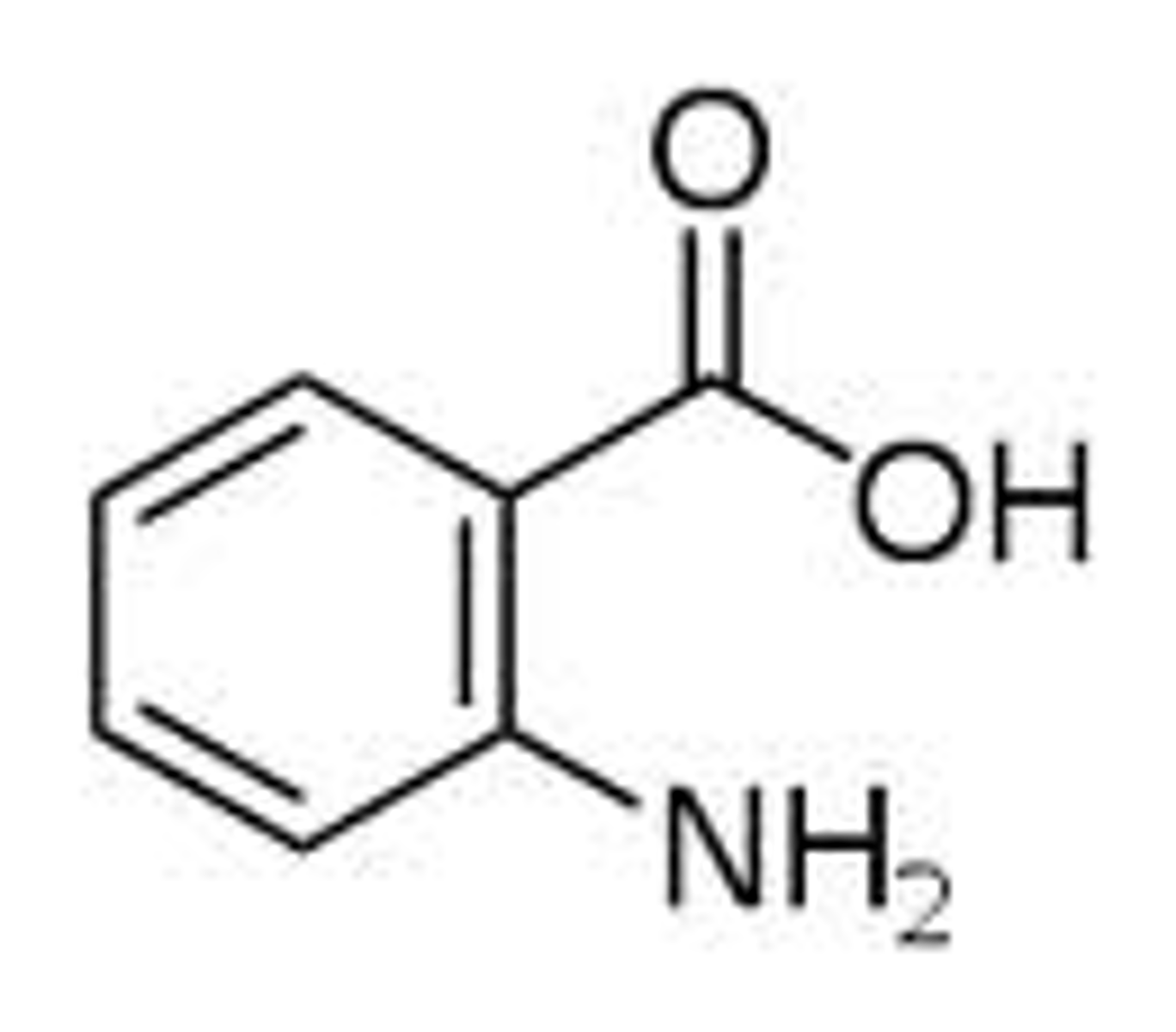
Anthranilic acid
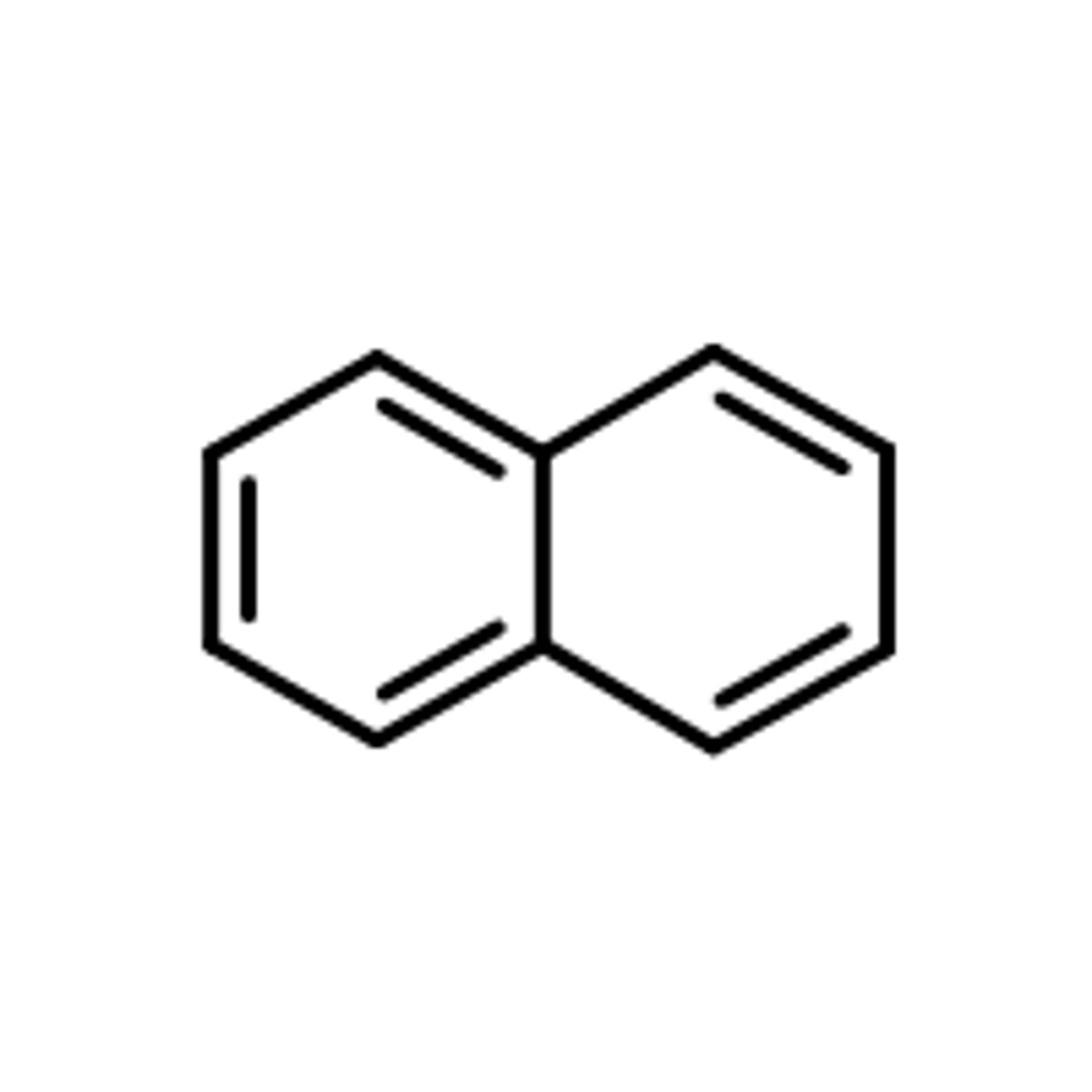
Naphthalene
Antracene
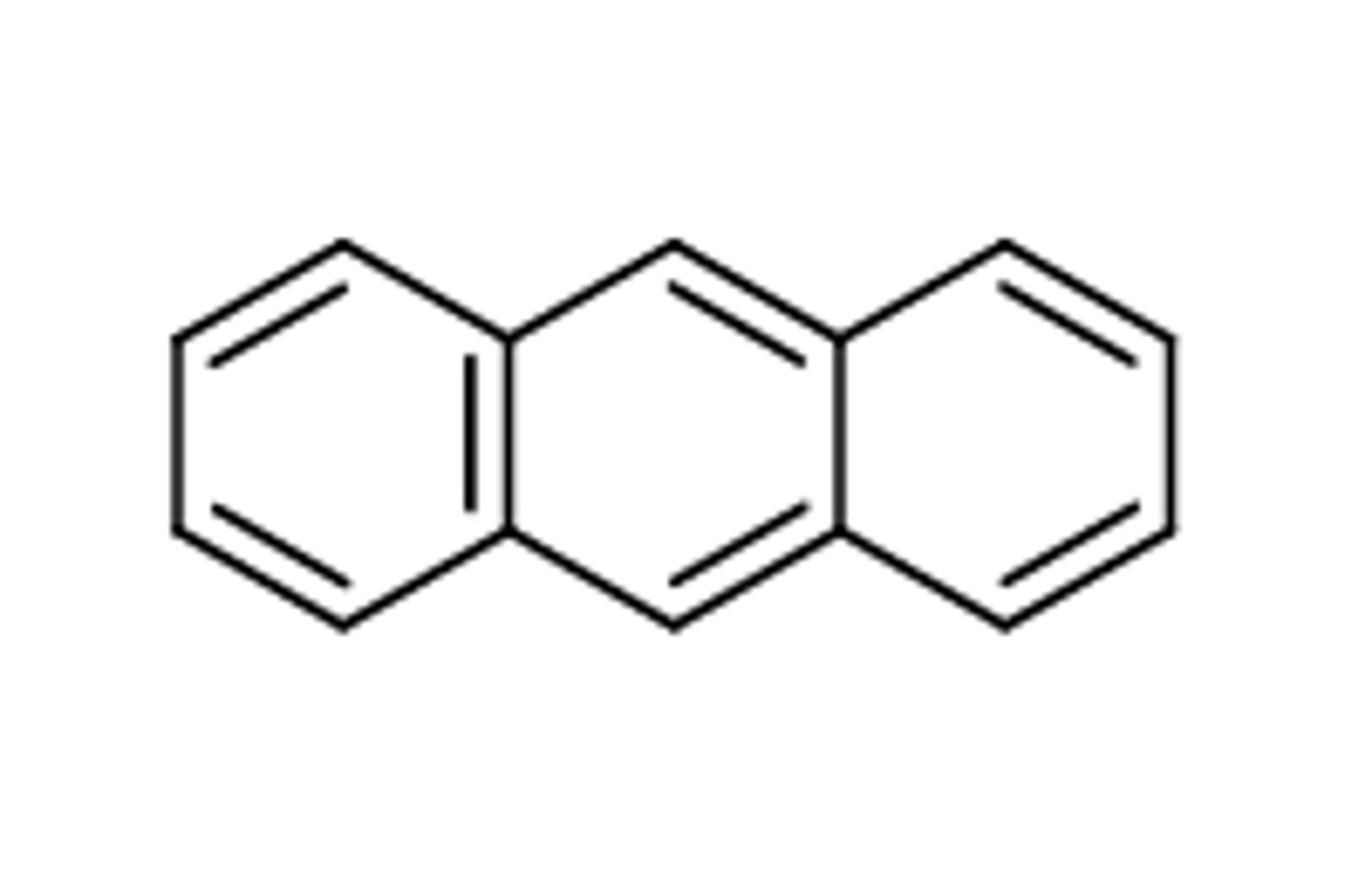
"bent"
Phenanthrene
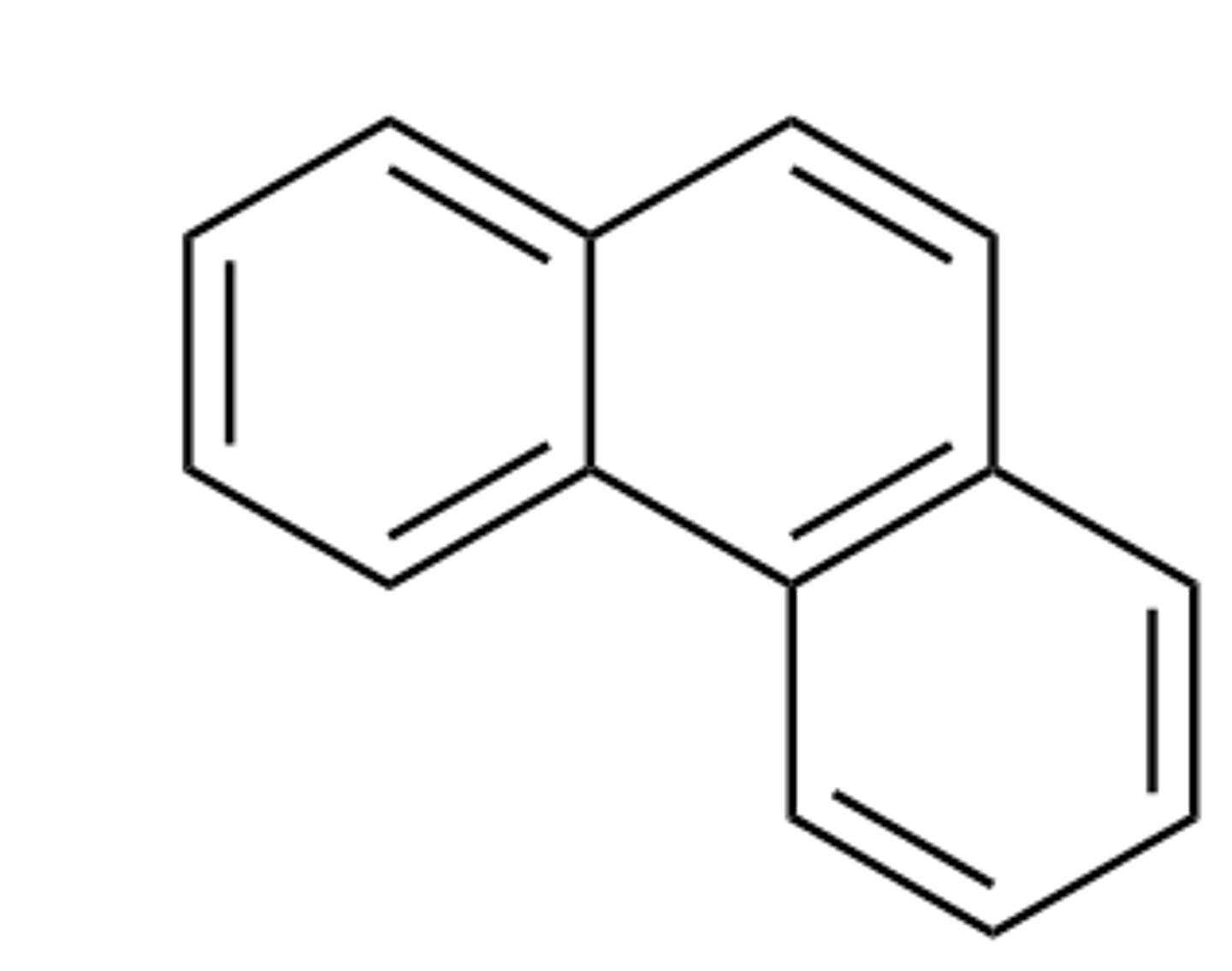
"Aromatic"
Pyrrole
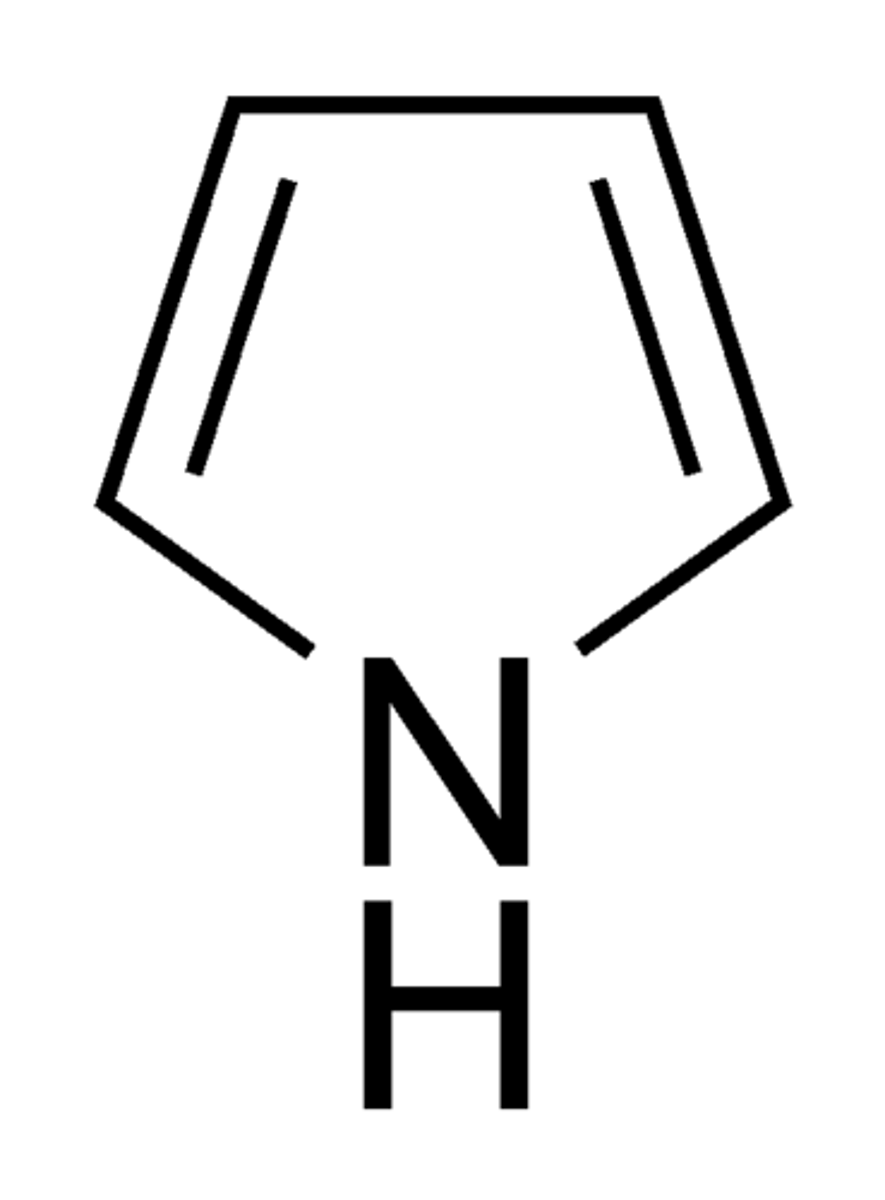
Non-aromatic
Pyrrolidine
p6
Pyran
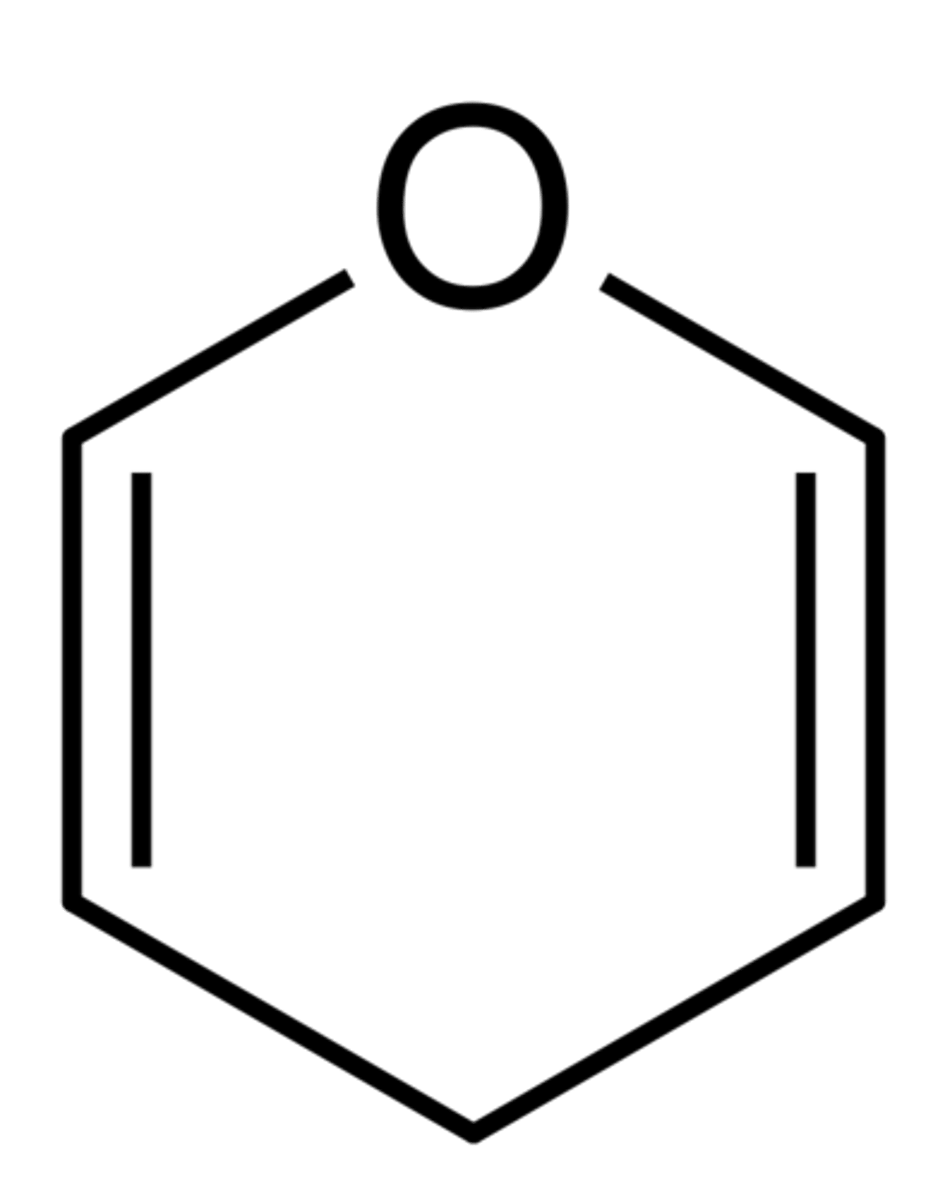
Thiophene
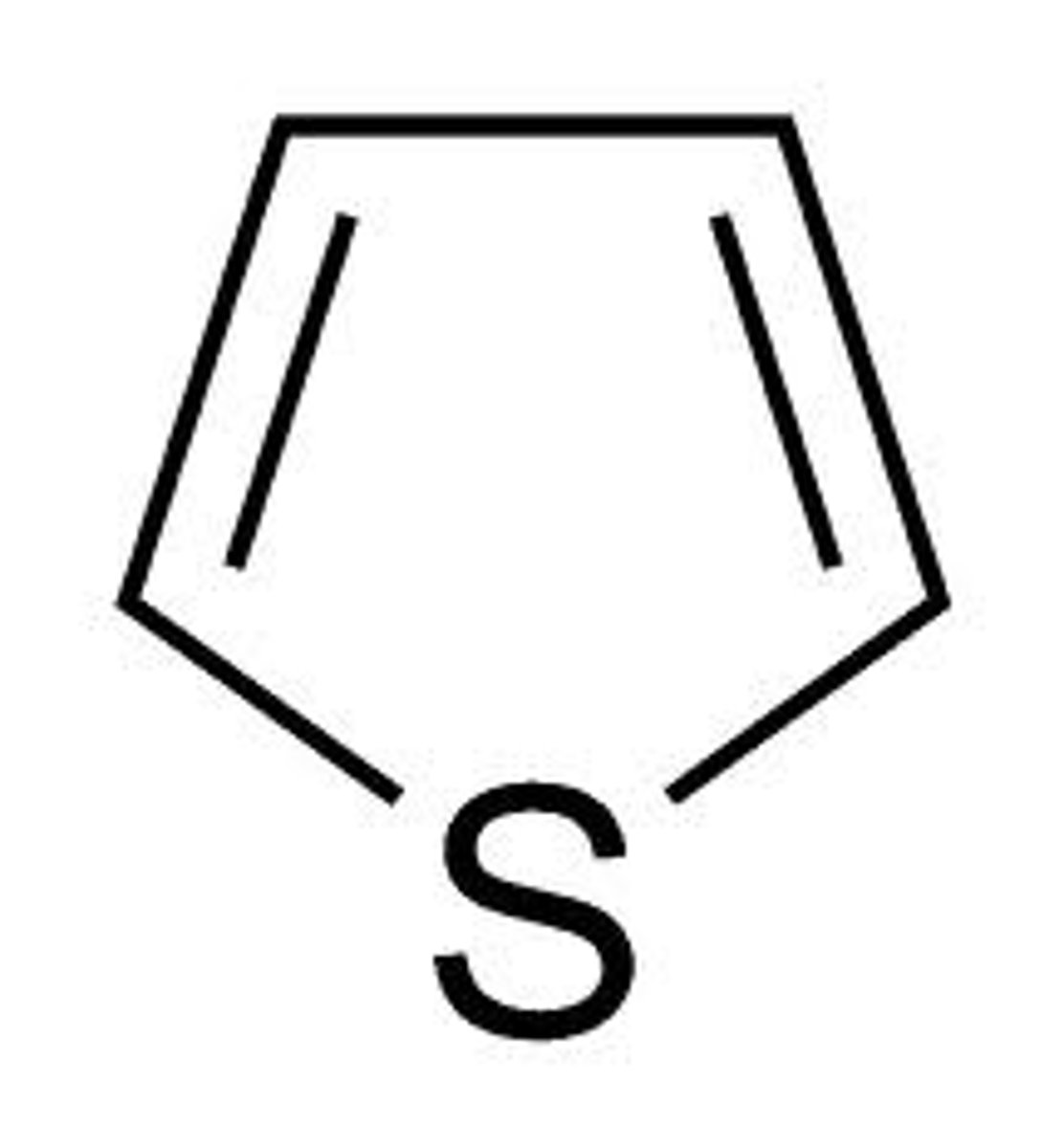
Imidazole
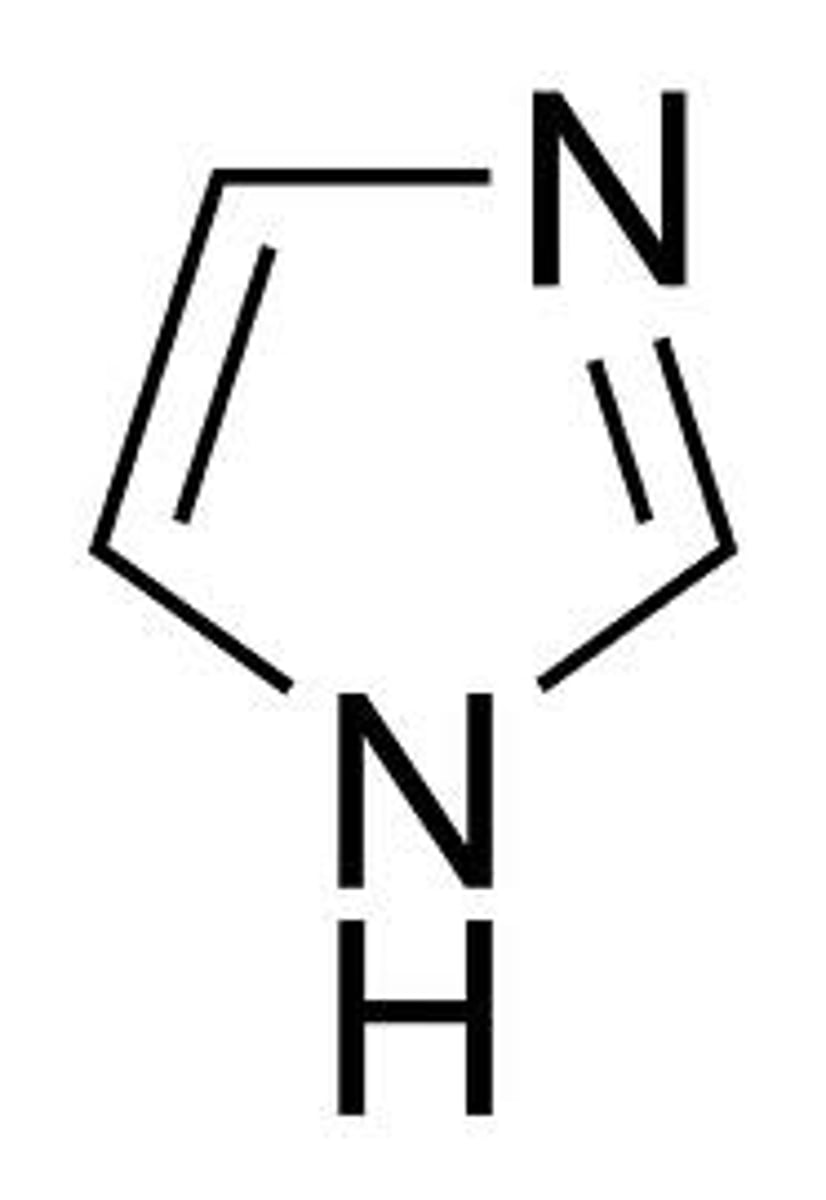
Thiazole
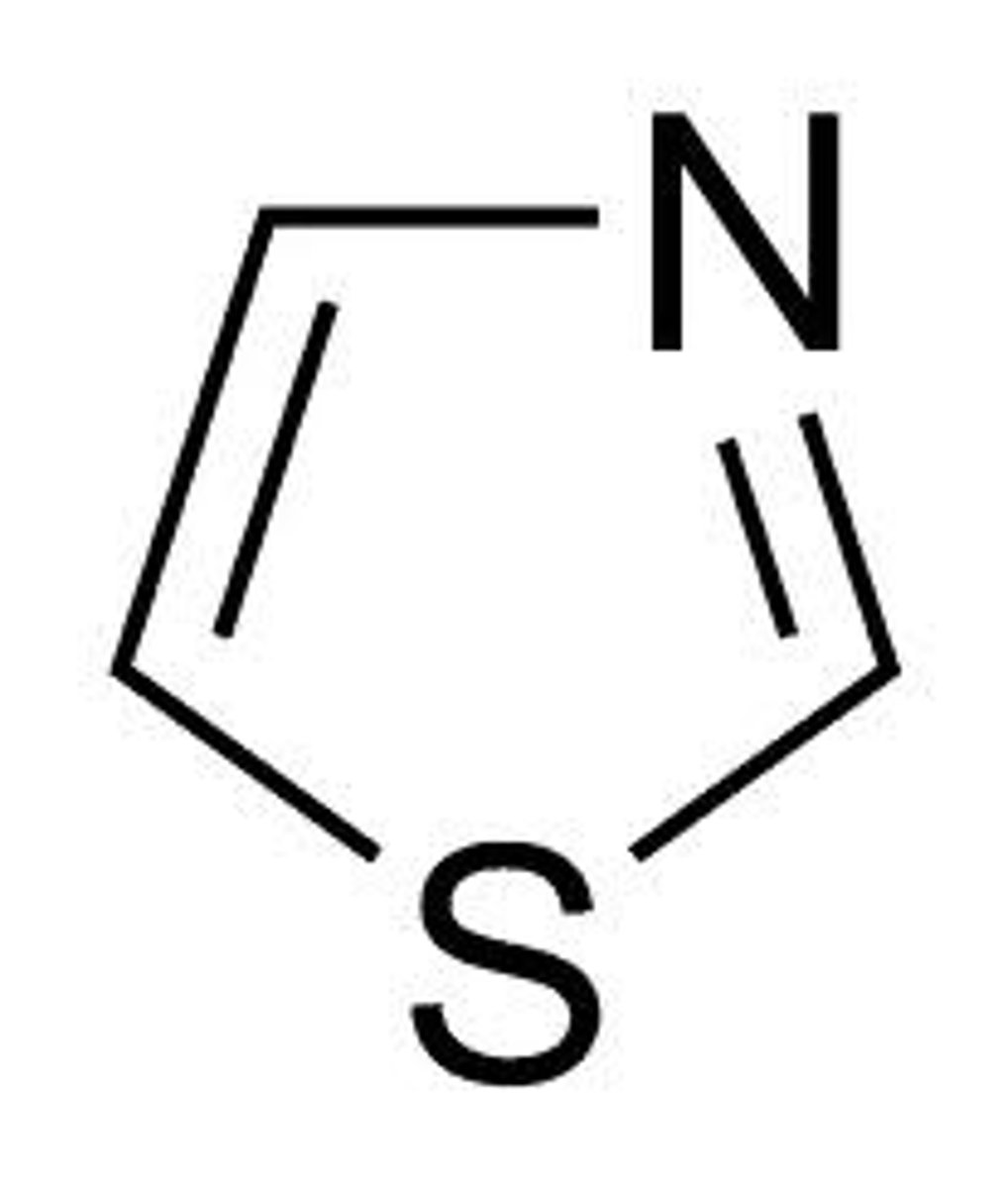
"five-ran"
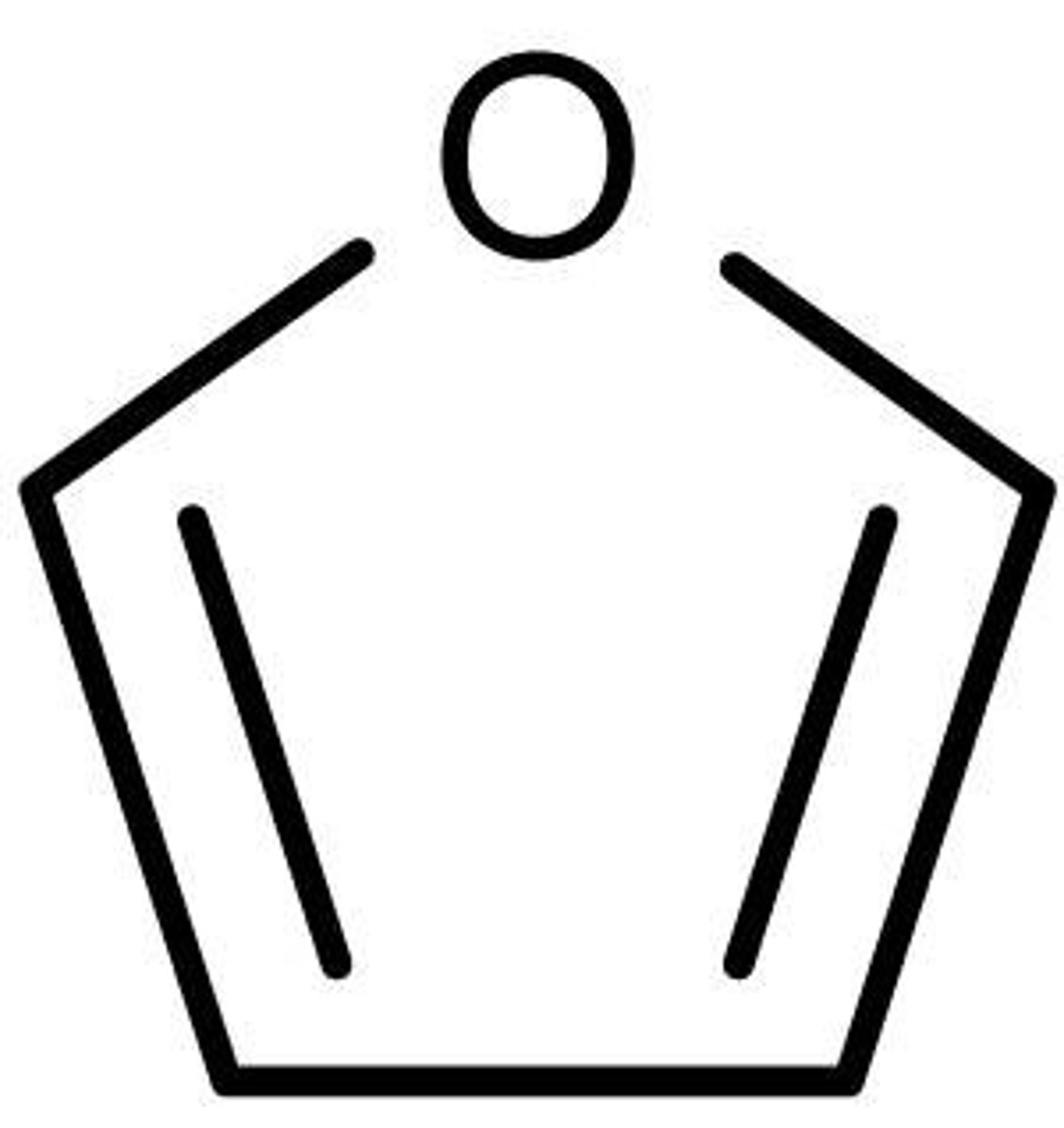
Furan
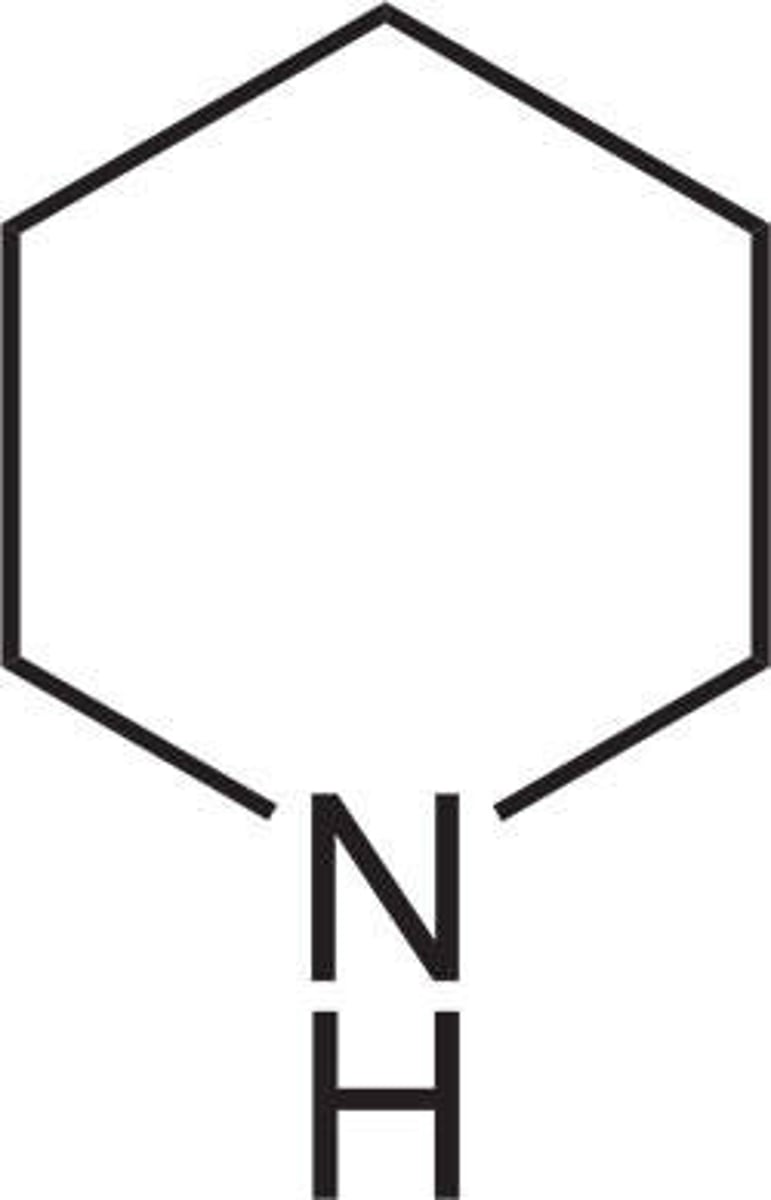
Piperidine
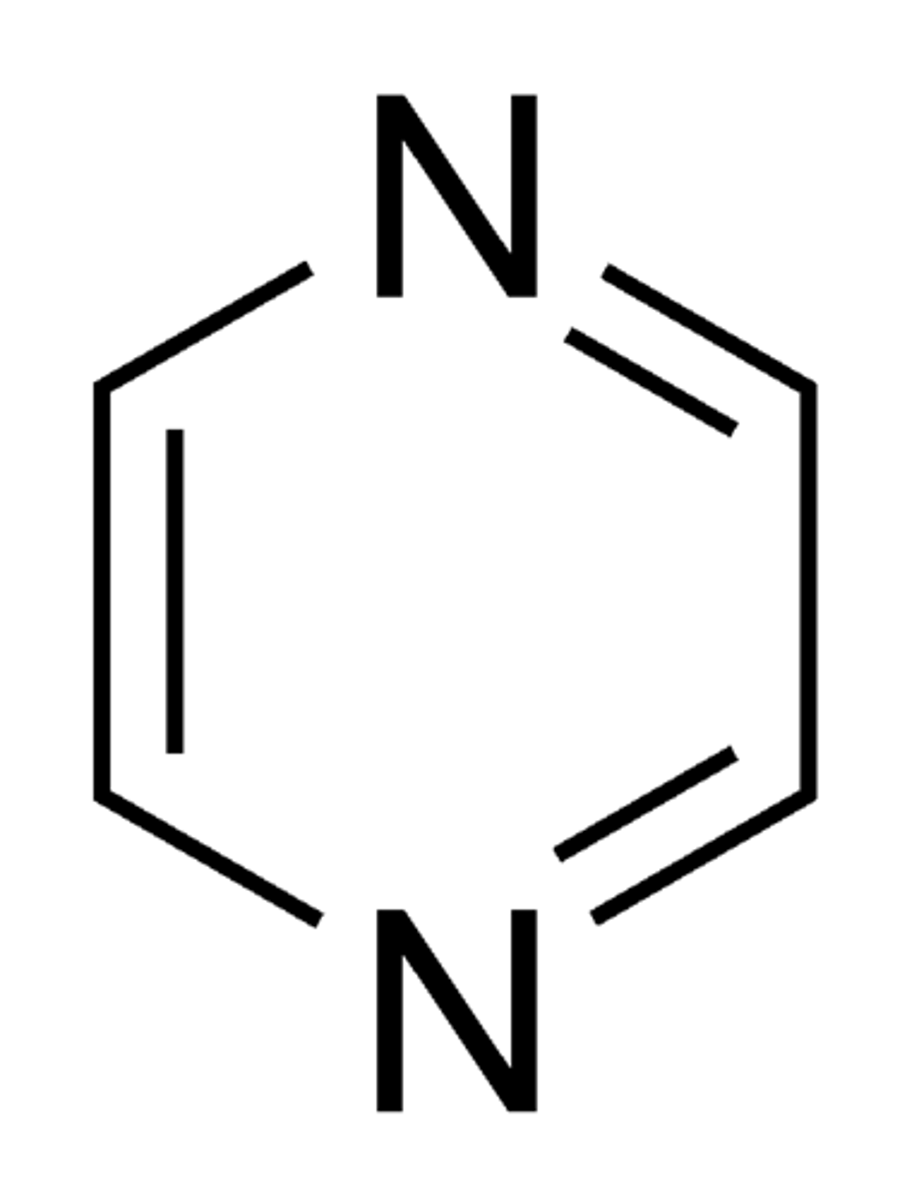
pyrazine
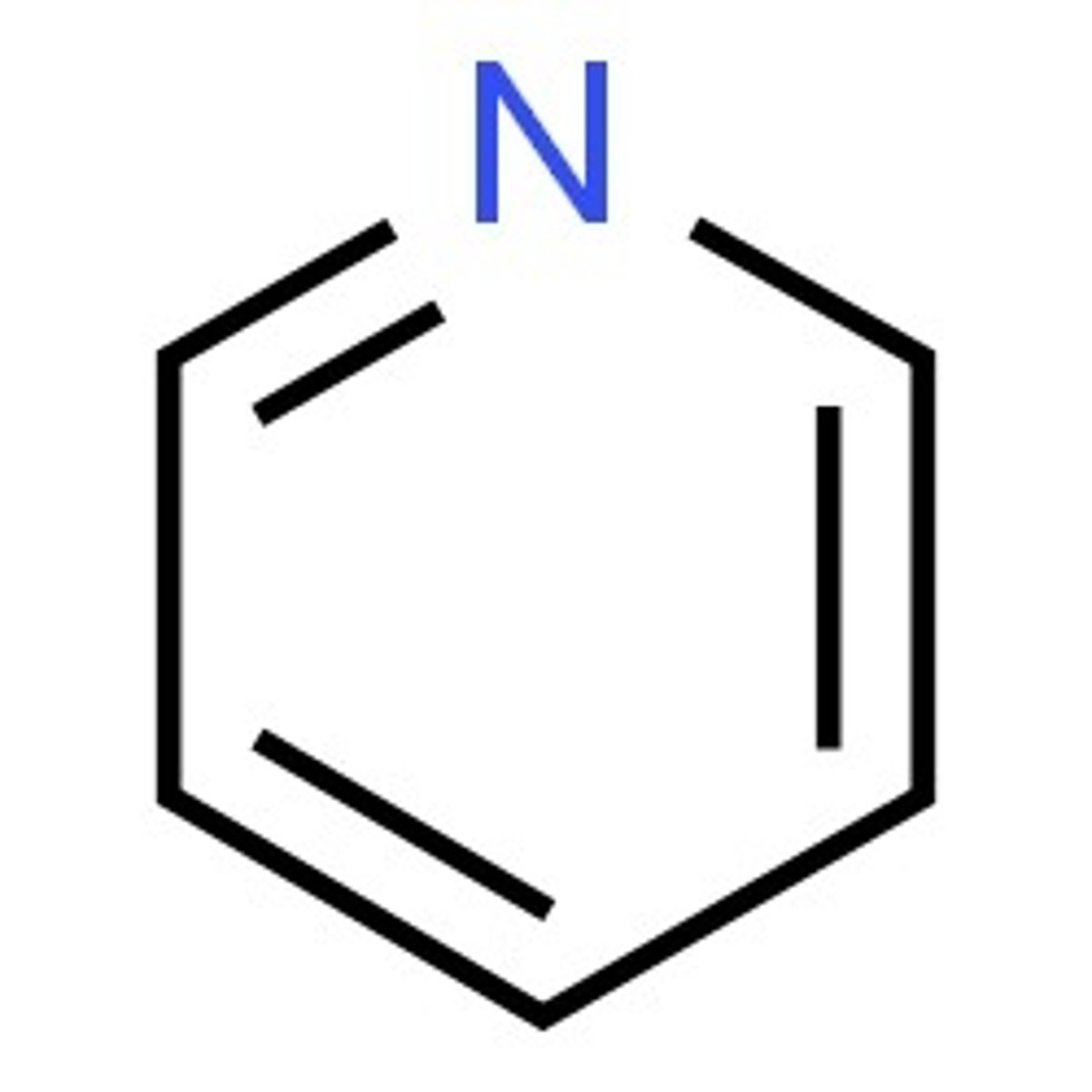
Pyridine
pyrimidine
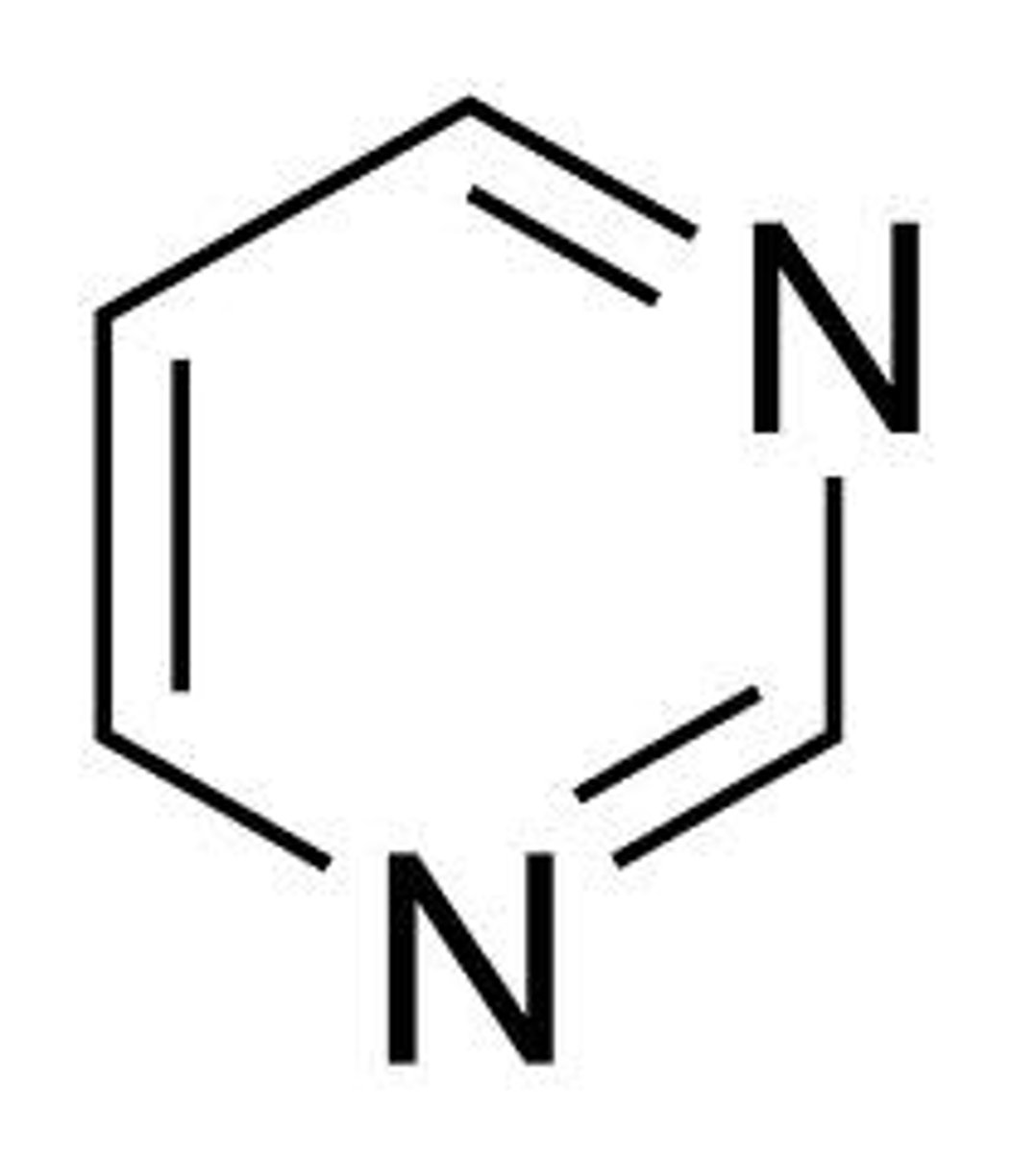
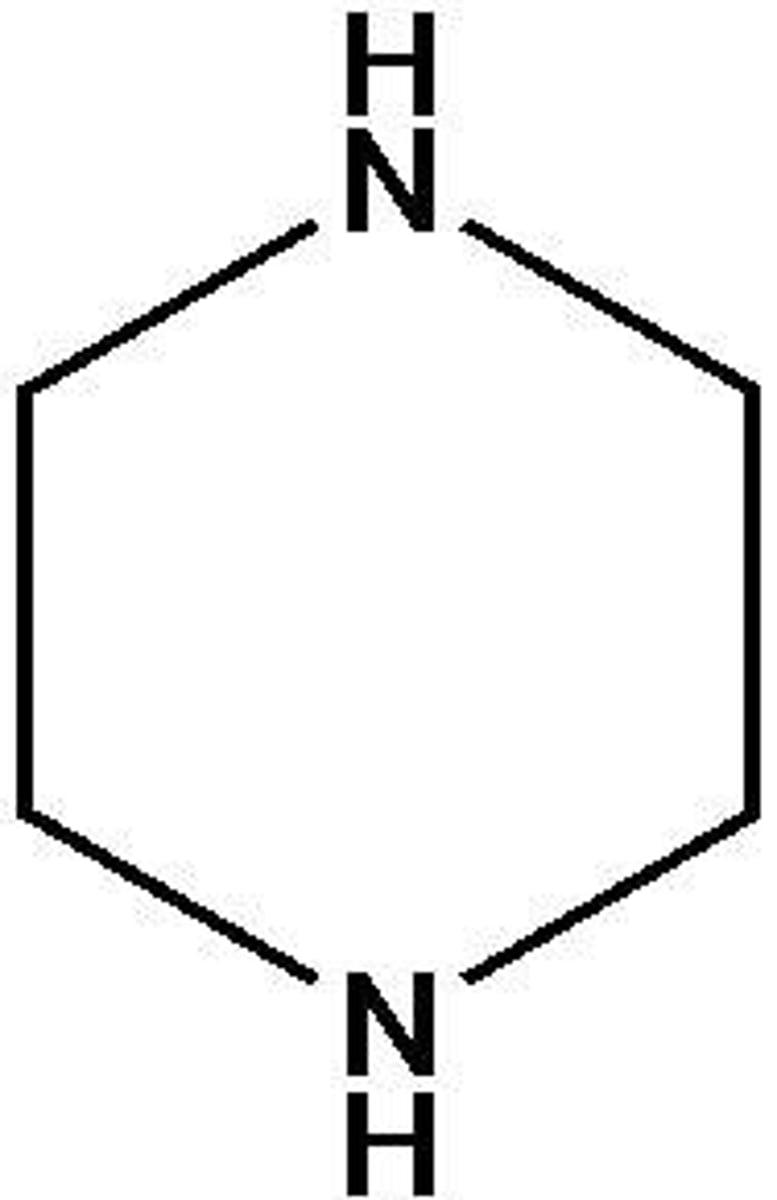
Piperazine
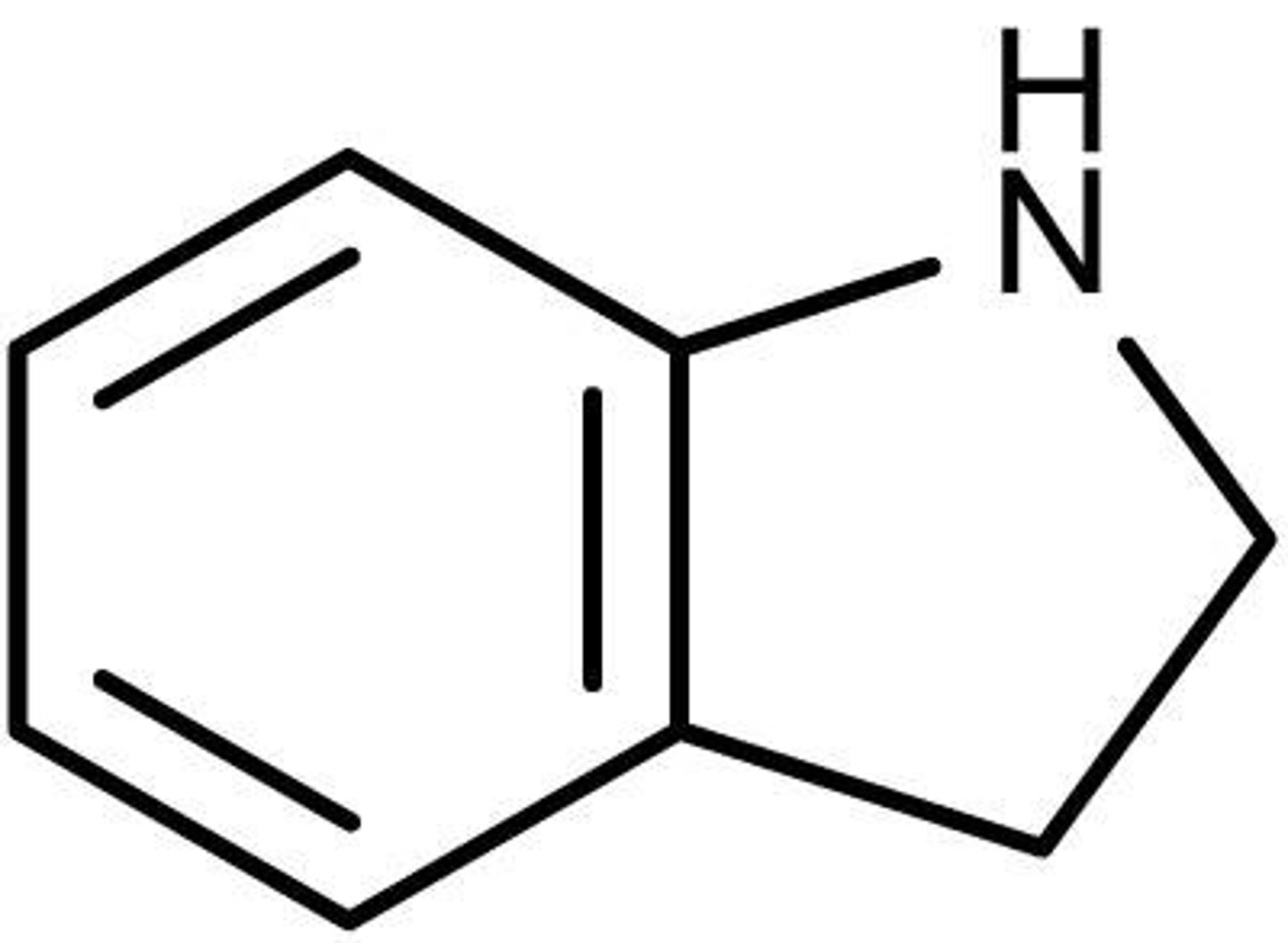
Indoline
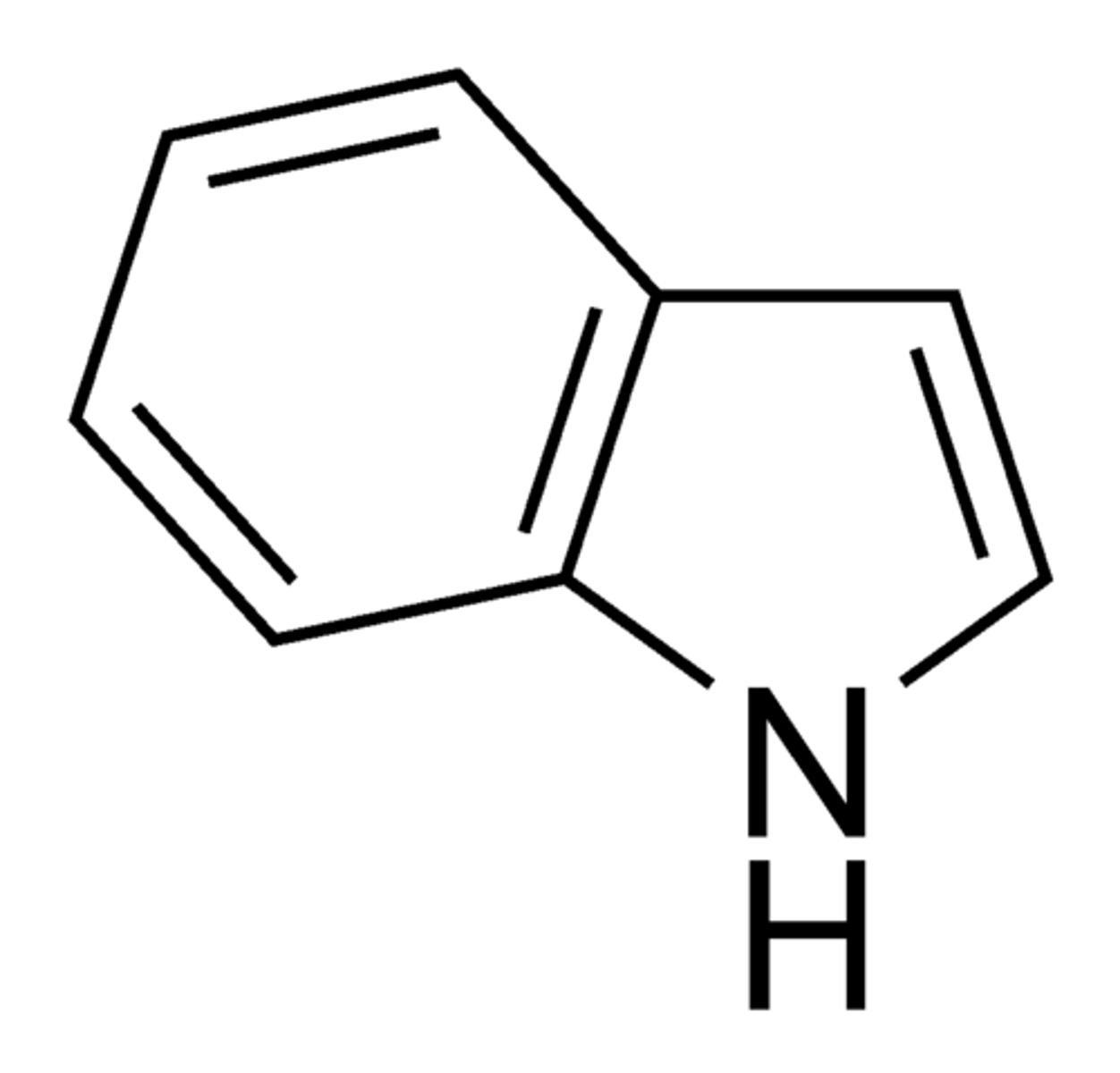
Indole
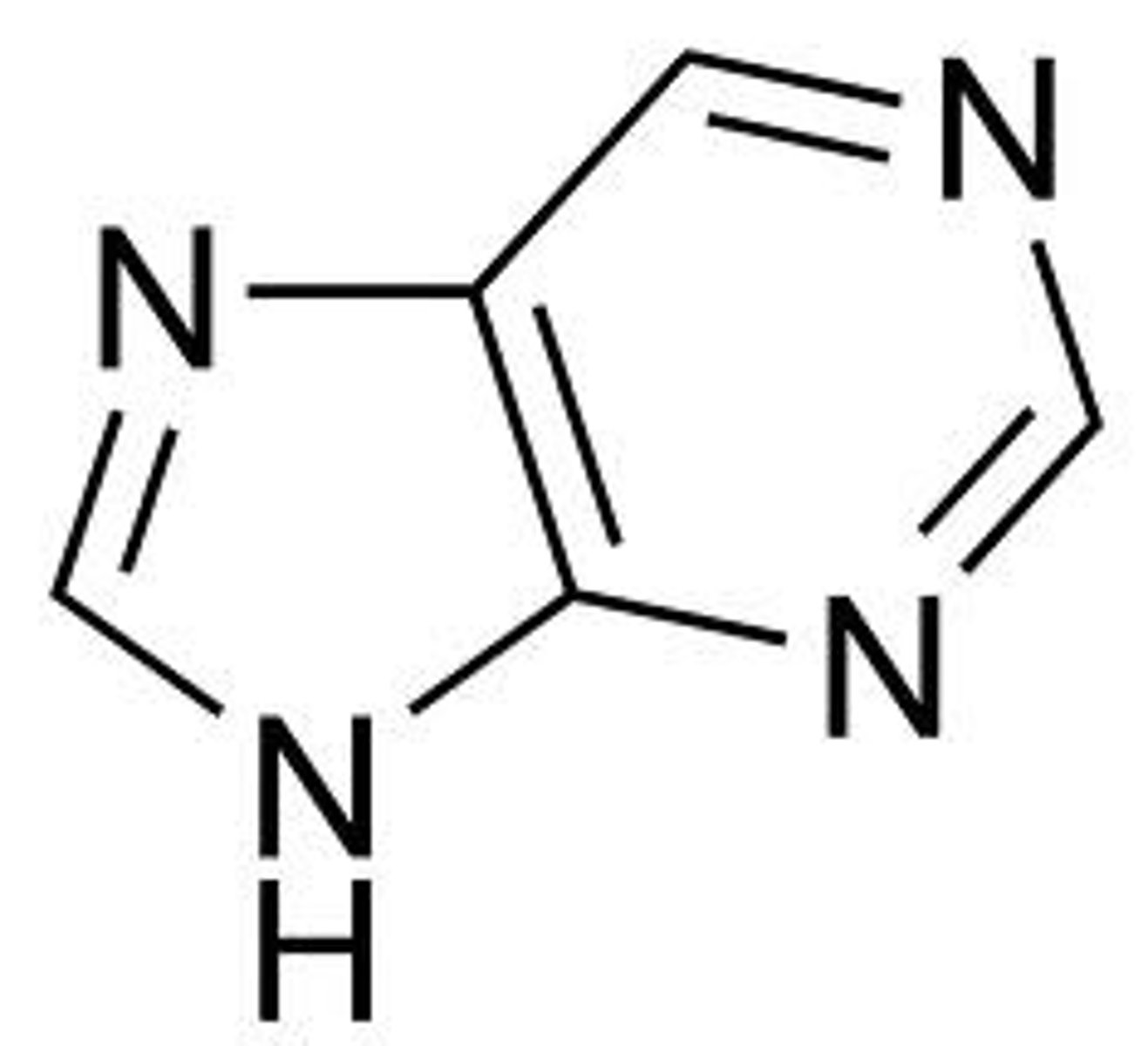
Purine
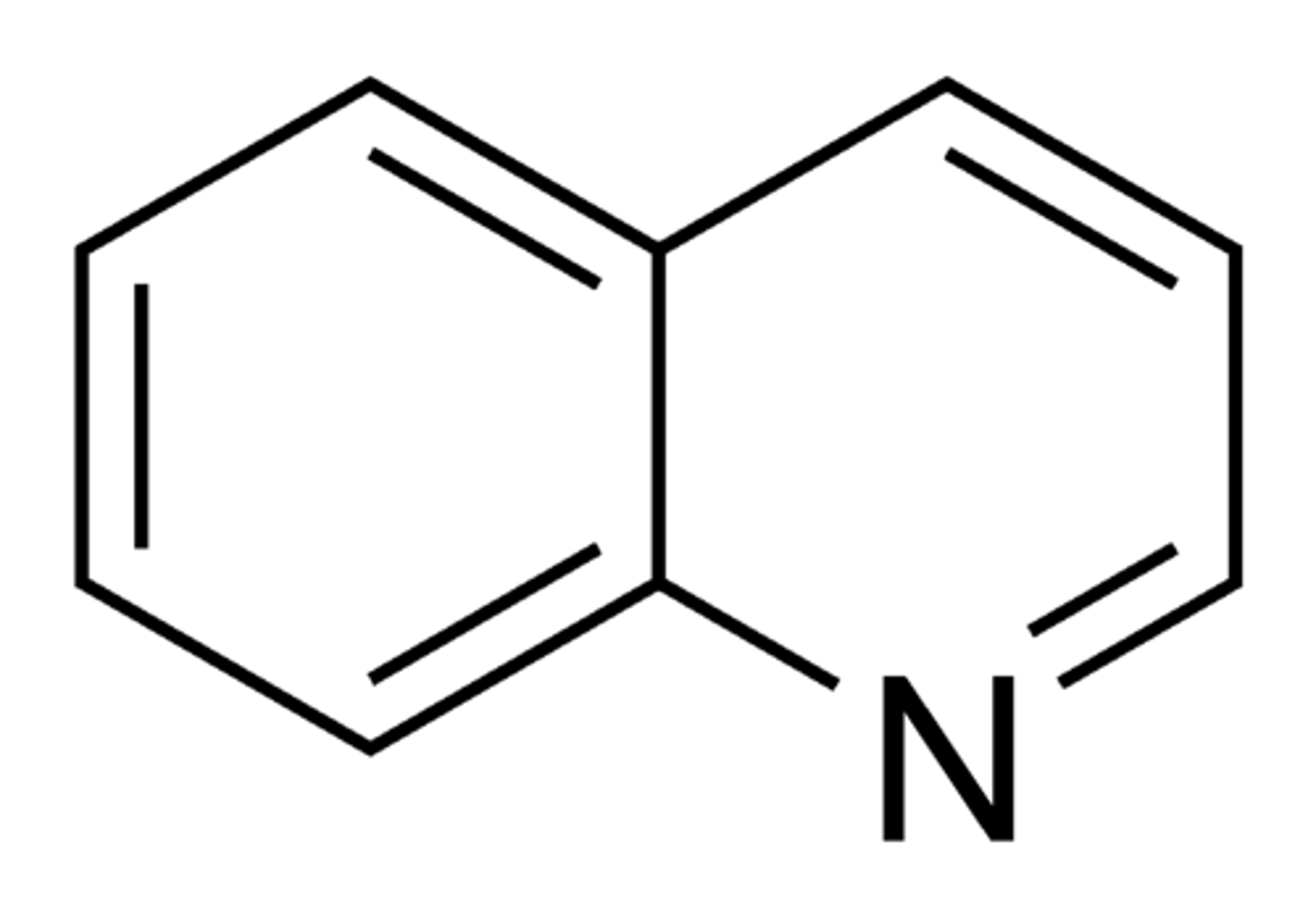
Quinoline
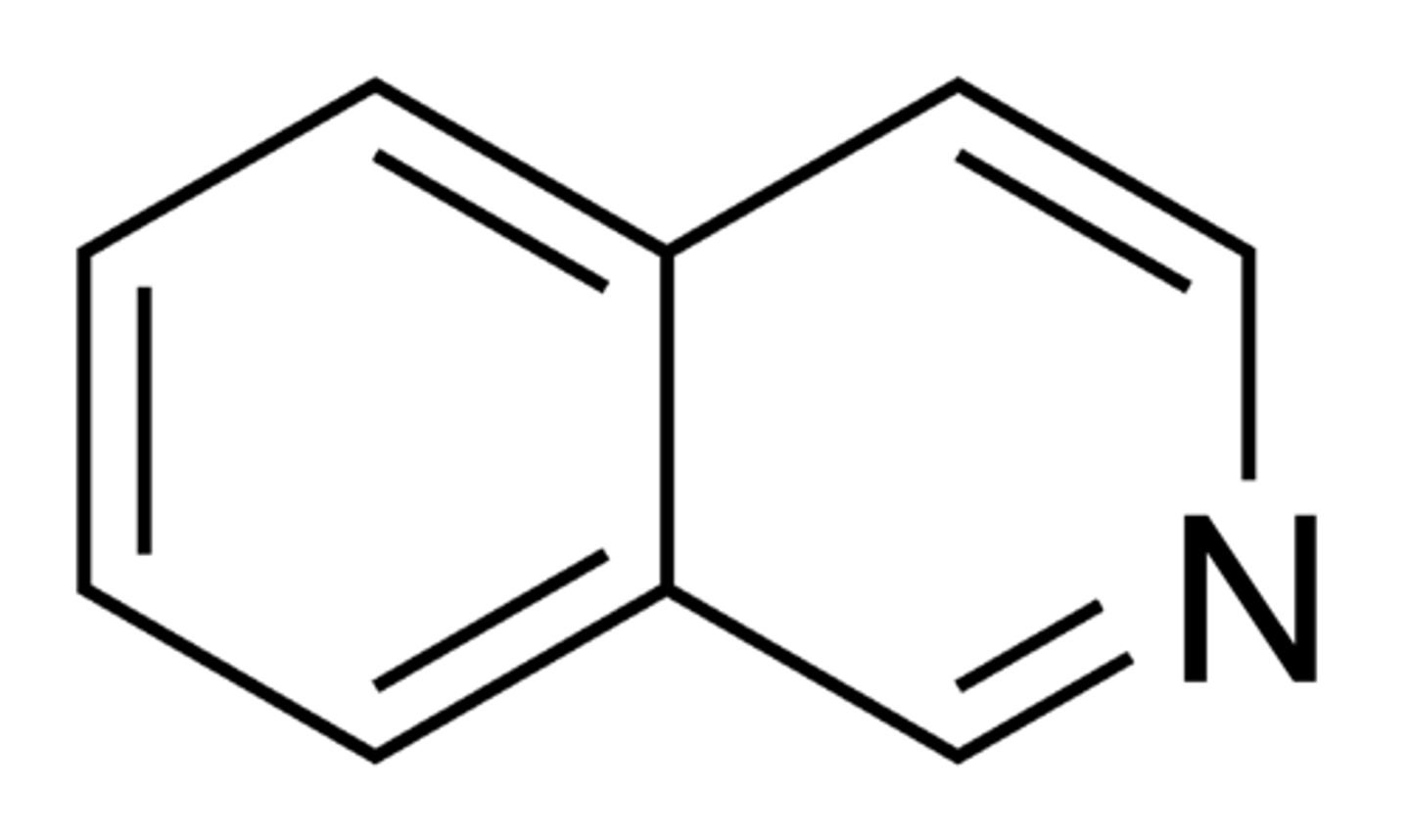
Isoquinoline
A core molecular framework that can bind to multiple biological targets
Privileged scaffold
A compound that kills (-cidal) or inhibits the growth (-static) of microbes when applied to living tissue
Antiseptic
destruction of a pathogen to an inanimate object
Disinfectant
MOA: protein denaturation
Alcohol
rendered unfit for drinking
Denatured alcohol
Used to sterilize heat-sensitive equipment
Ethylene oxide
Formalin, methanal; embalming fluid and disinfectant
Formaldehyde
Used to sterilize heat-sensitive equipment; used in medical mission in remote areas
Glutaraldehyde
alcohol that is suitable for surgical instrument
Isopropyl
External use alcohol
Ehyl alcohol
remains the standard to which the activity of most germicidal substances is compared
Phenol