Med Chem Quiz 9 (L33-35)
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
-sedative = drug that exerts calming effect
-hypnotic = drug that produces drowsiness and encourages onset and maintenance of sleep
-anxiolytic = drug that relieves excessive anxiety
Barbituates have a linear relationship when increasing dose with increased CNS depression (overdose more likely, benzodiazepines taper off)
Site of binding of barbituates not well defined
Many sedatives, hypnotics, and anti-anxiety drugs also have anti-epileptic activity
GABAa = post-synaptic inhibition
GABAb = pre-synaptic inhibition (slows release of neurotransmitters)
Both A and B produce CNS depression
Sedative and Hypnotic Agents Basic Overview
1. Two R’s at C-5-position needed for activity (replaces the 2 H's in barbituic acid)
2. ↑Number of C’s in C-5 R’s ⇒ ↑onset, ↓duration
-increase in lipophilicity (faster into CNS --> faster onset), but also permeates out of CNS quickly so metabolized quicker --> shorter duration
3. Polar groups, unsaturation, and branching on the C-5 R’s ⇒ ↑rate of metabolism, ↓duration
4. A methyl group @ N-1 position ⇒ ↑onset, ↓duration
5. Substitution on both nitrogens ⇒ loss of activity
6. Sulfur at the 2-position ⇒↑onset, ↓duration
-more lipophilic
Extra notes:
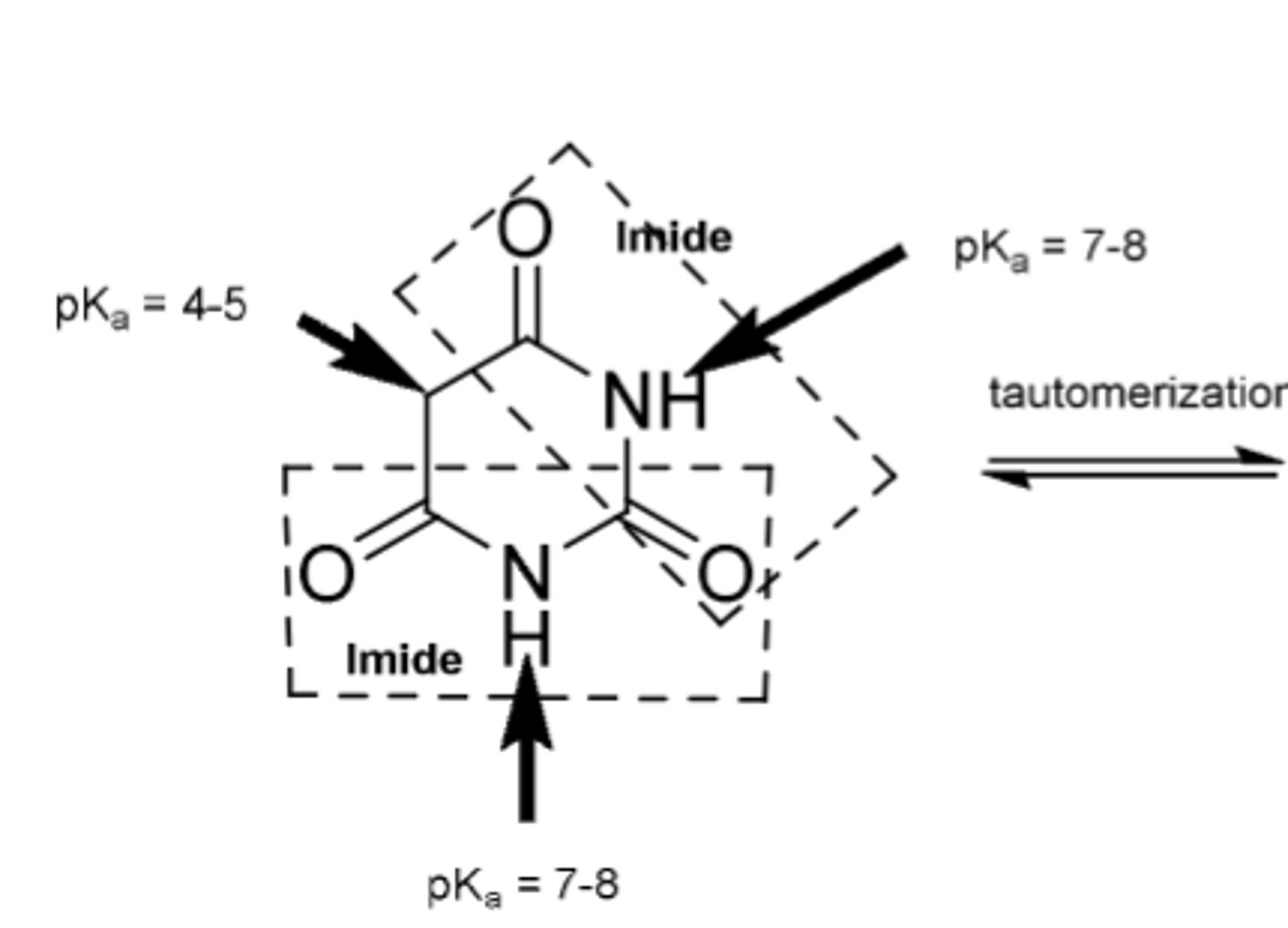
-barbituric acid has no CNS activity because its highly ionized
-tautomerizes to trihydroxy form
-NHs are acidic because they are between 2 carbonyls (so ionized)
-barbituates are enzymatic inducers
-continued use may lead to addiction
-primarily as anesthetics and anticonvulsants
SAR of barbituates
Ultra short acting (generally anasthetics) = less than 3 hours
Methohexital and Thiopental
Short acting (sedative hypnotic and preanesthetic) = 3-4 hours
Secobarbital and Pentobarbital
Intermediate Acting (sedative hypnotic) = 6-8 hours
Amobarbital and Butabarbital
Long Acting (anticonvulsant) = 10-16 hours
Phenobarbital
Barbituate Drugs
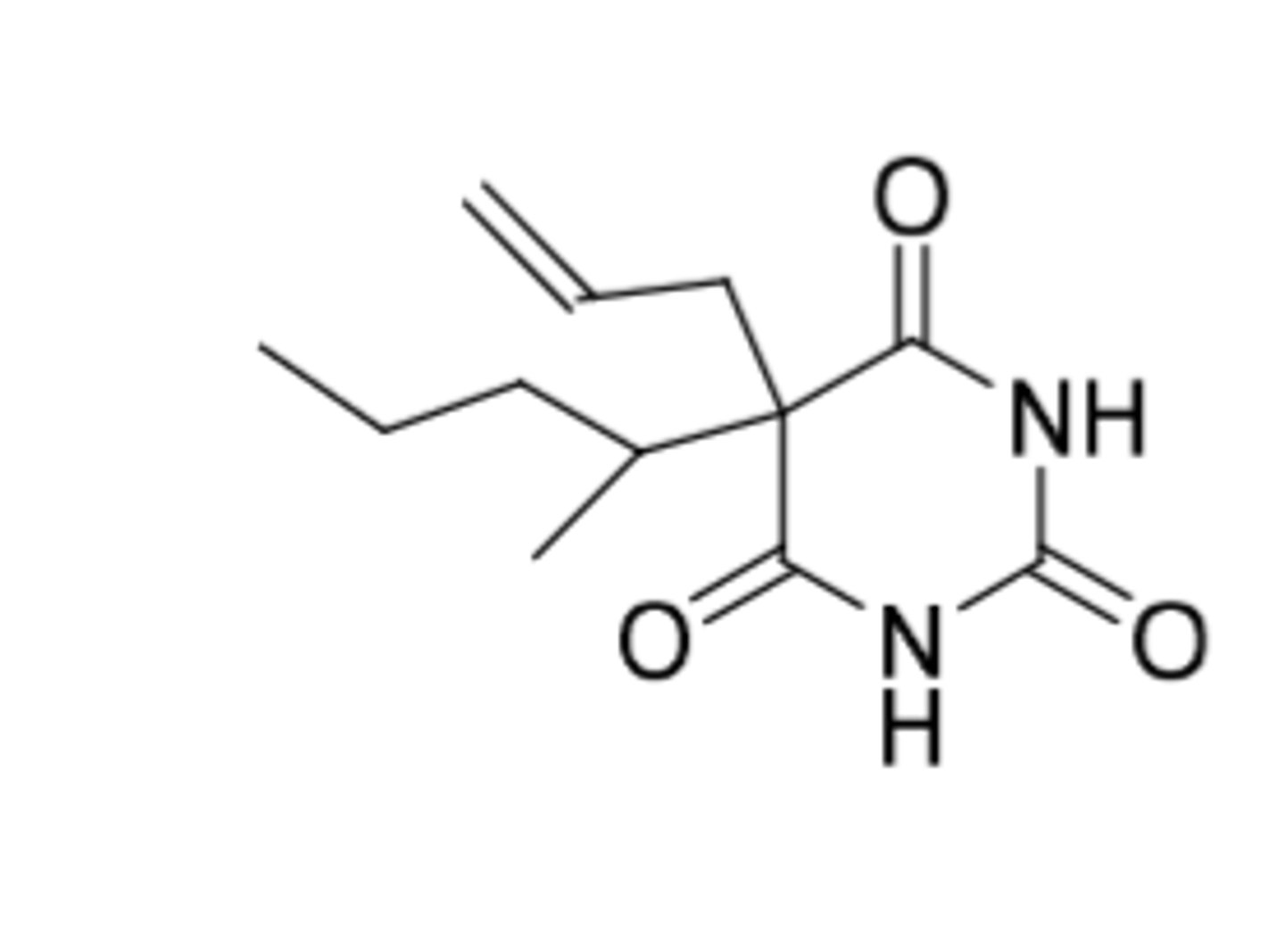
Secobarbital
-Used as hypnotic and preanesthetic
-C2
-Short-acting (duration = 3-4 hours)
-Onset = 10-15 min
-unsaturation and branching at 5 position lead to shorter duration (barbituate SAR)
Metabolism:
-alpha oxidation (allylic)
-epoxidation (can cause inactivation of CYPS) then hydrolysis
-omega-1 oxidation
Pentobarbital
-Used as hypnotic seddative and preanesthetic
-C2
-Short-acting (duration = 3-4 hours)
-Onset = 10-15 min
-unsaturation at 5 position leads to shorter duration (barbituate SAR)
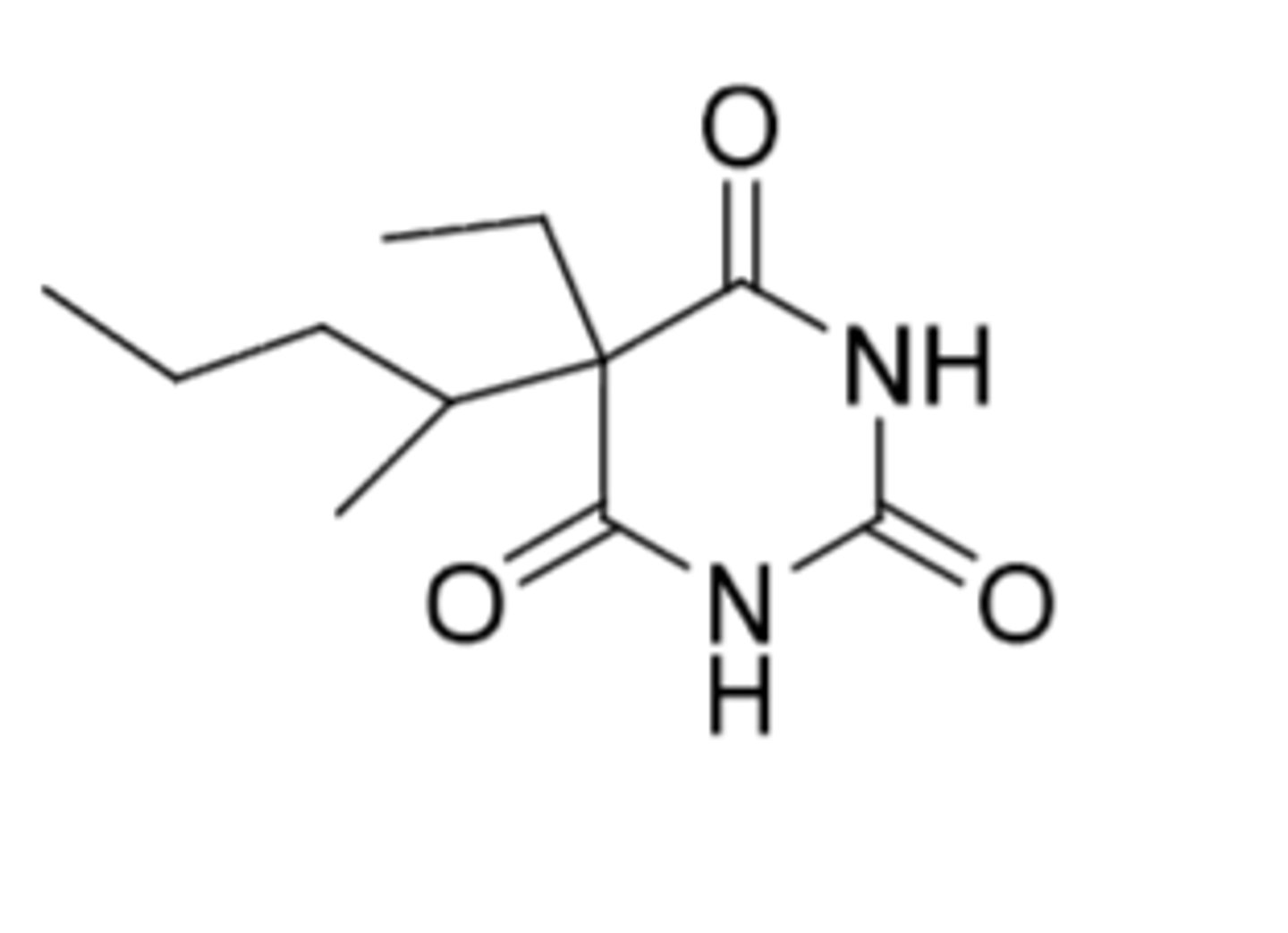
Amobarbital
-intermediate-acting (duration = 6-8 hours)
-onset = 45-60 min
-C2
-sedative and hypnotic
Metabolism (hydroxylations):
-omega-1 oxidation or omega-oxidation
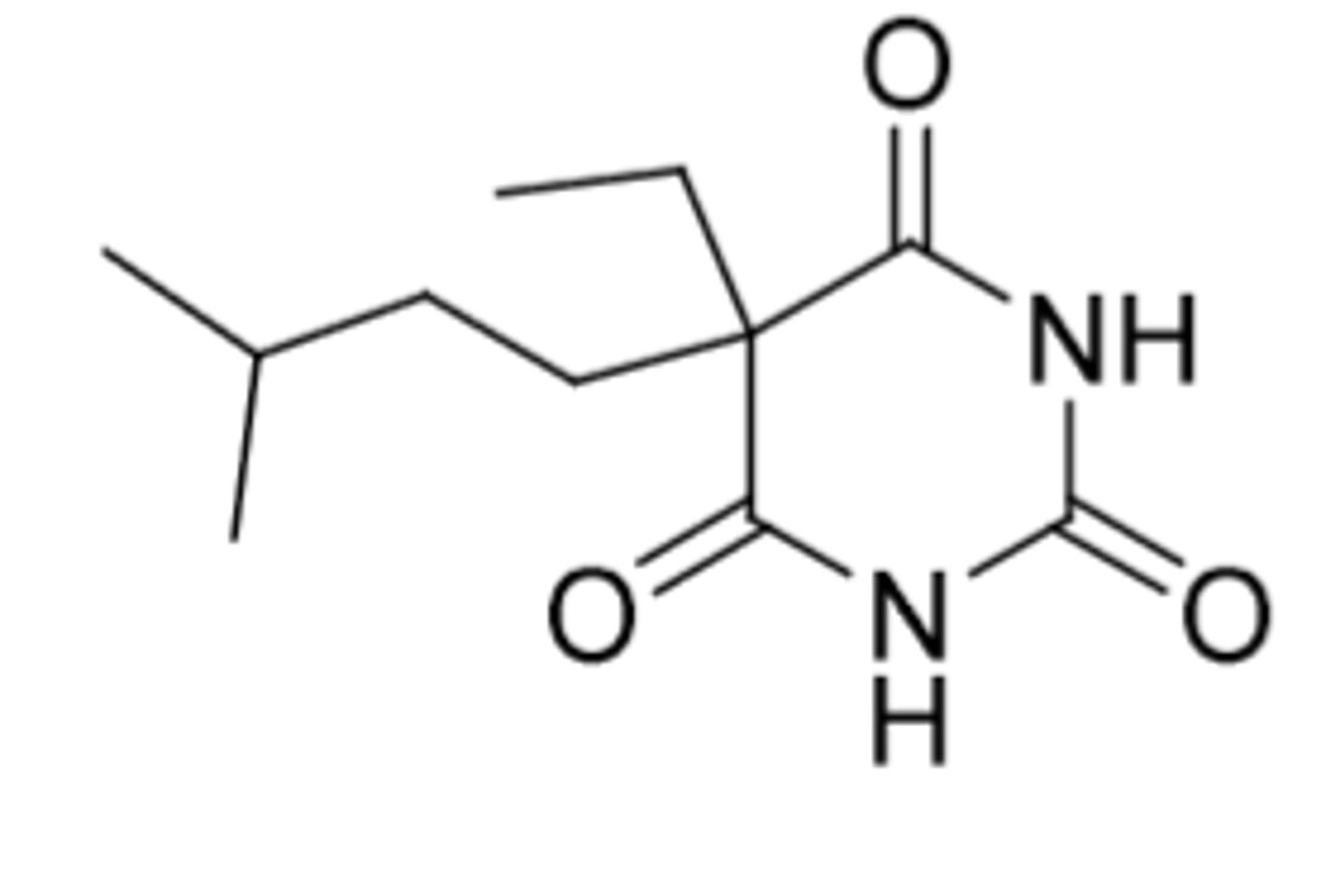
Butabarbital
-intermediate-acting (duration = 6-8 hours)
-onset = 45-60 min
-C3
-sedative and hypnotic
-just has one less carbon than amobarbital
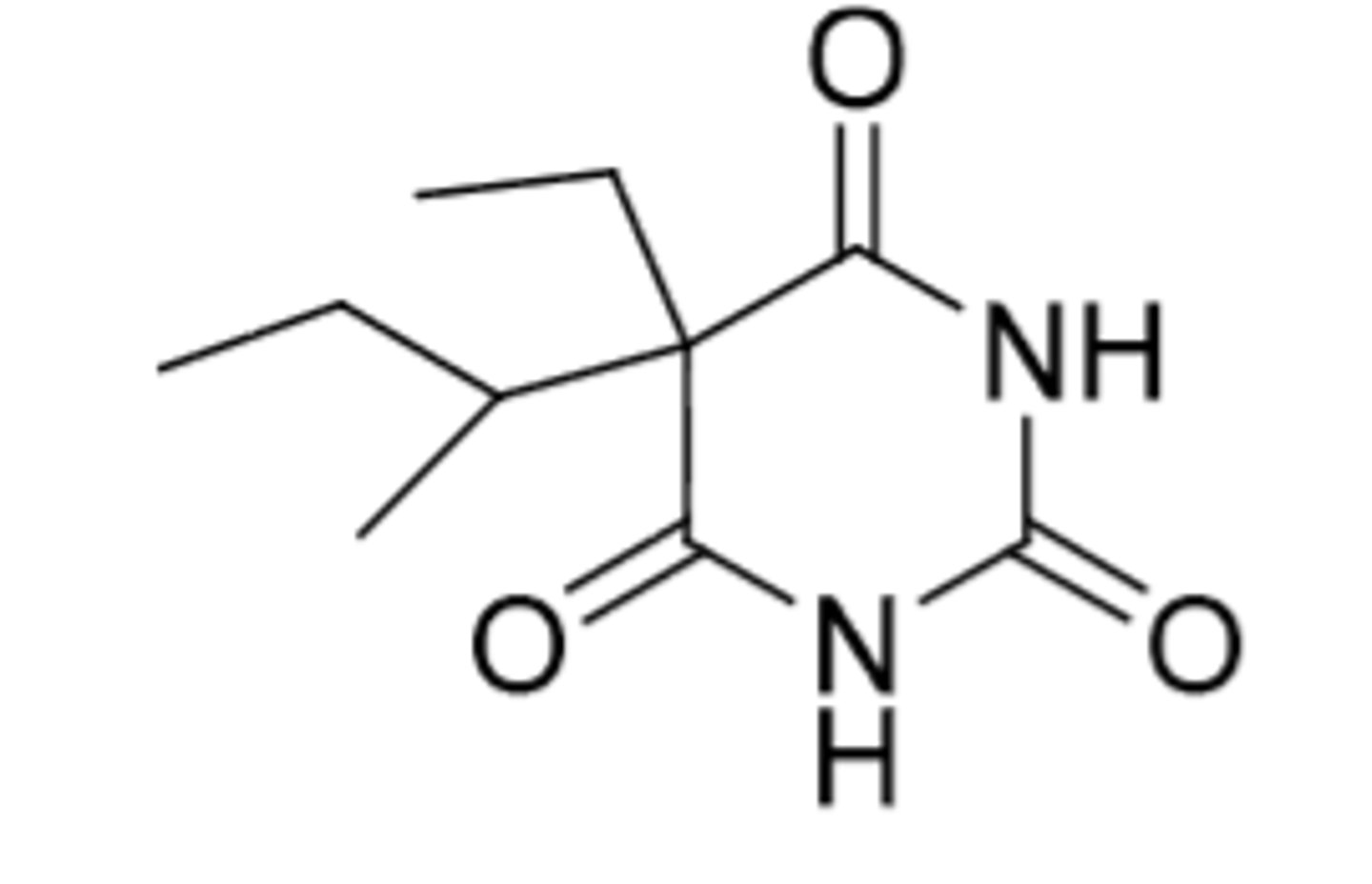
Phenobarbital
-long-acting (Duration = 10-16 hours)
-onset = 30-60 min
-used as anti-convulsant (treatment of seizures)
Metabolism:
-oxidative hydroxylation at para position on phenyl ring
-glucuronidation of one NH (turns into chiral center --> preference of S metabolite over R)
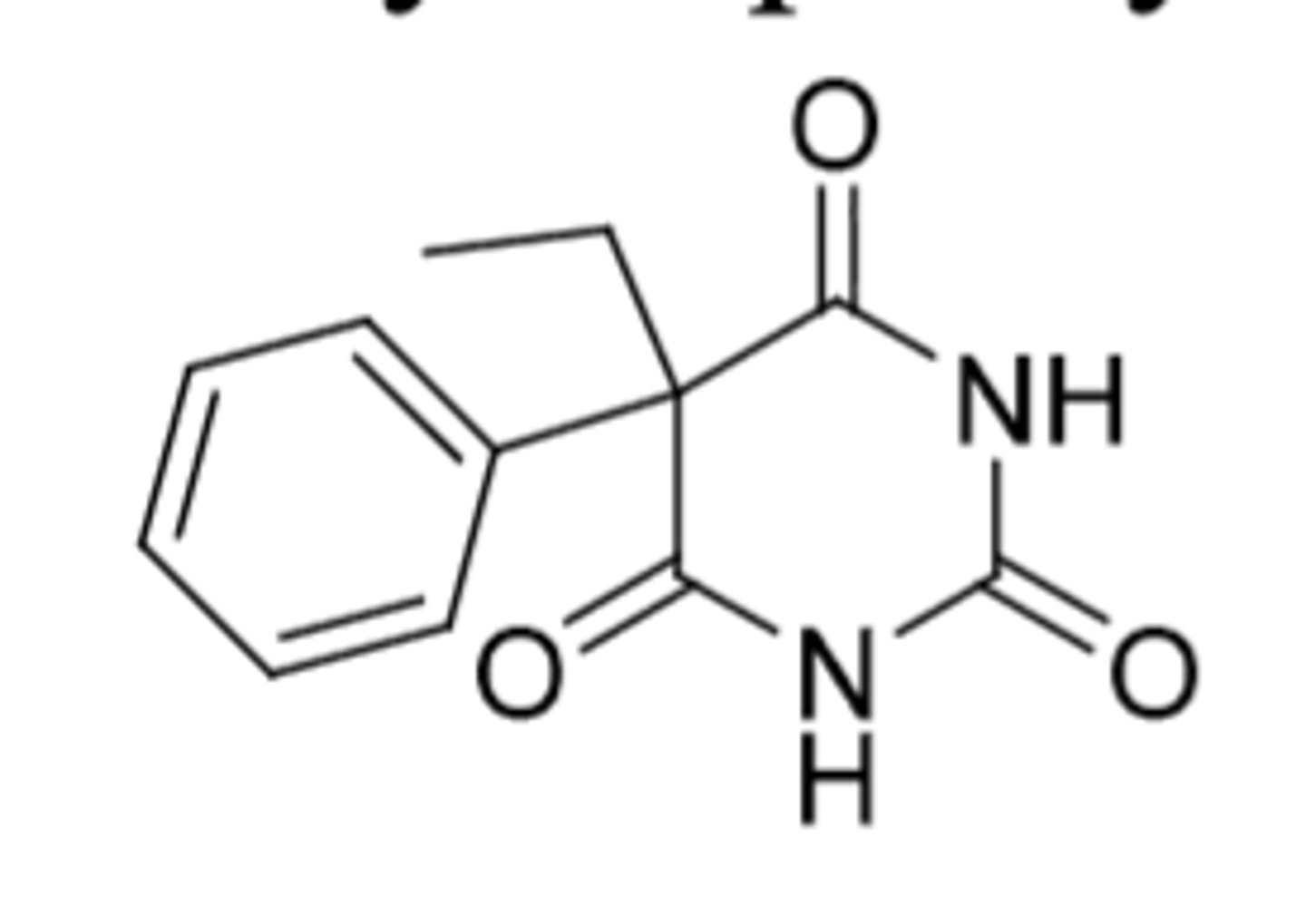
Chloral Hydrate
-C4
-weakly acidic
-Reduced to an active metabolite or oxidized to inactive metabolite
-Drawbacks: bitter taste (take w/ fruit juice) and gastric irritation
-sedative and hypnotic, can suppress and prevent alcohol withdrawal symptoms
-can be synergistic w/ alcohol (leading to misuse)
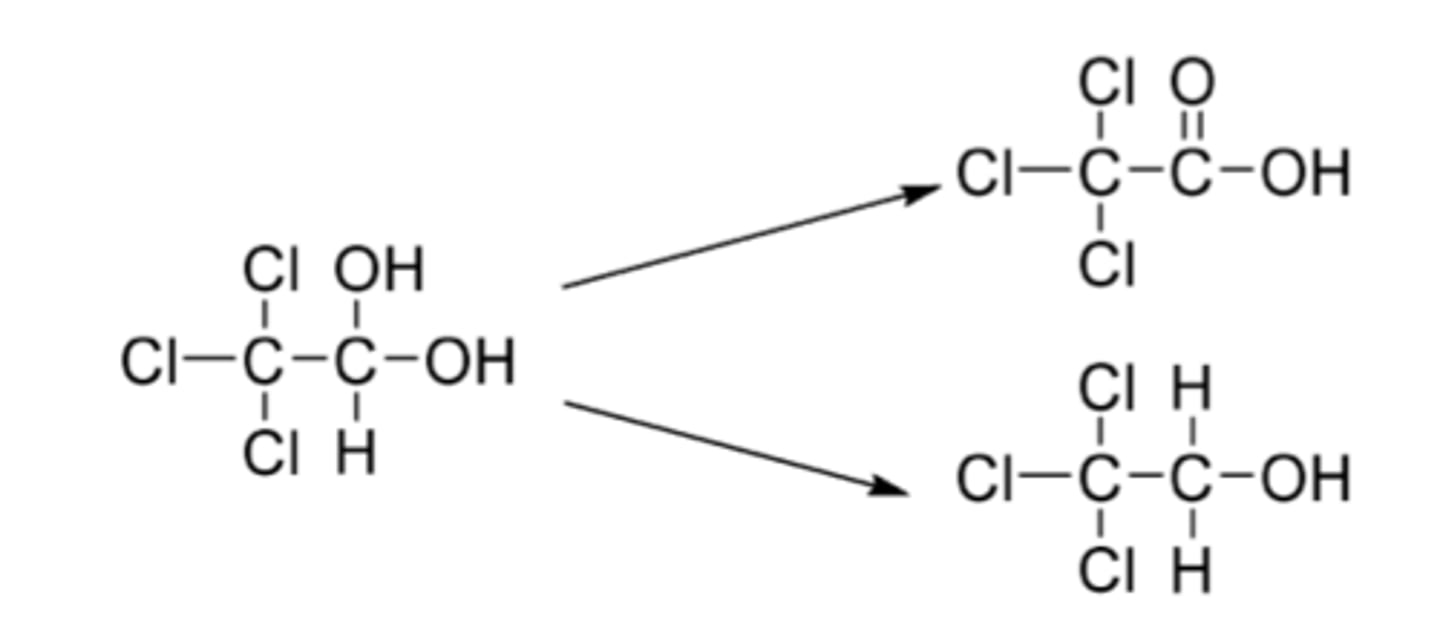
Diphenhydramine
-OTC first generation antihistamine
-safe in children over 6
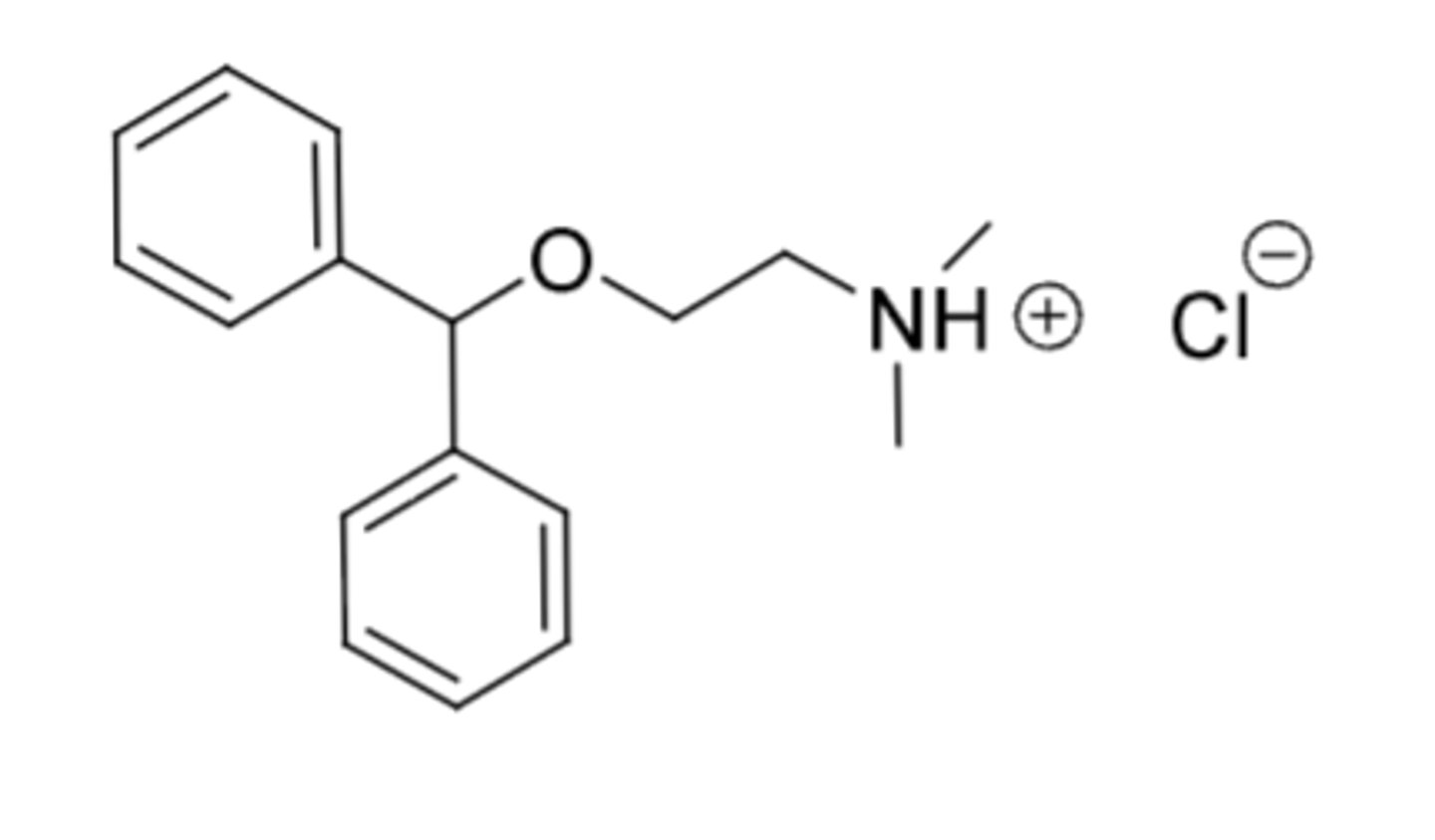
Zolpidem Tartrate (Ambien)
-known to be used in military for pilots to induce sleep
The 3-Z drugs:
-mimic benzodiazepines by selectively binding to type 1 BZ binding sites (associated with sleep and hypnotic effects)
-C4
-extensively metabolized after administration (very little eliminated unchanged)
-used for short term treatment of insomnia
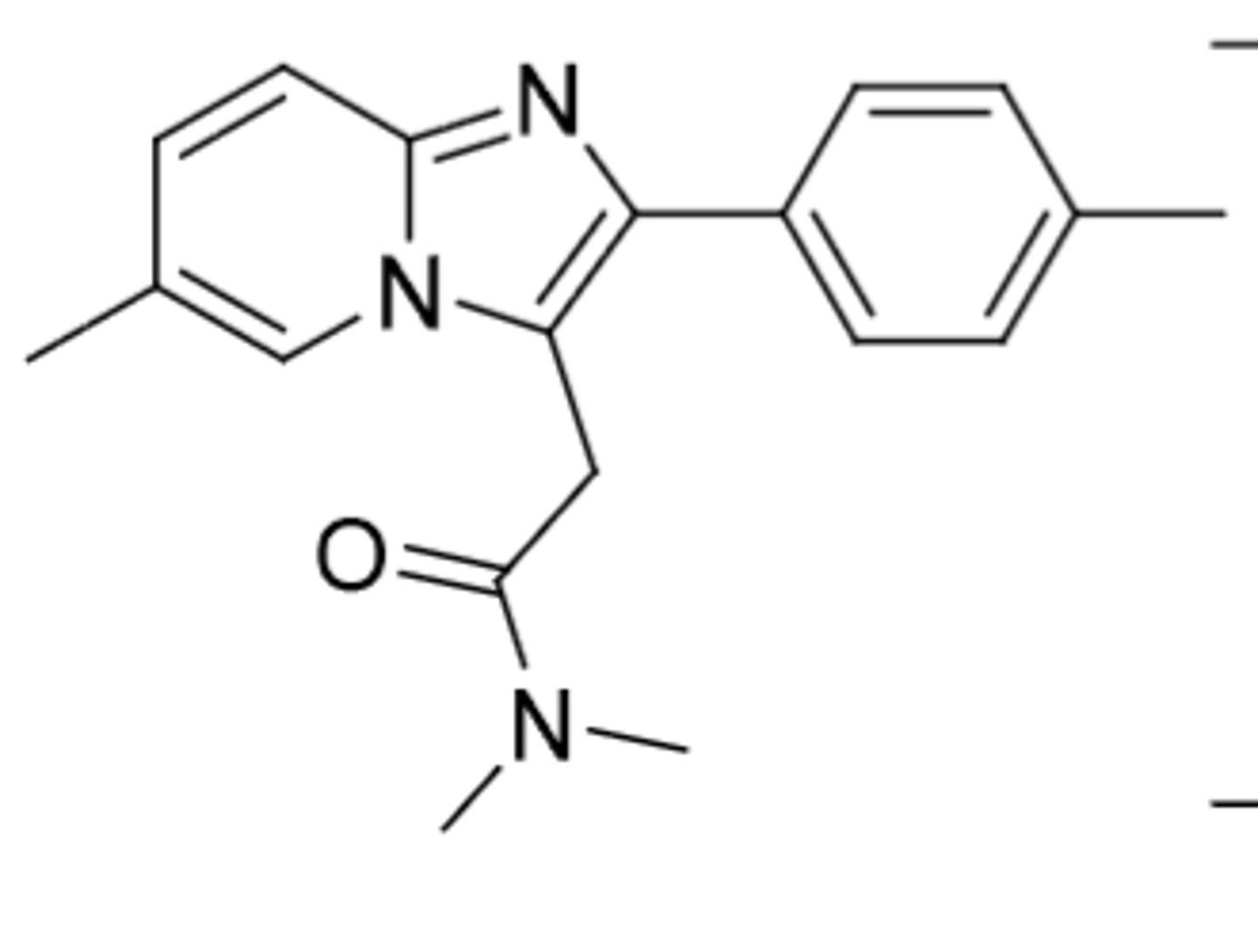
Zaleplon (Sonata)
The 3-Z drugs:
-mimic benzodiazepines by selectively binding to type 1 BZ binding sites (associated with sleep and hypnotic effects)
-C4
-extensively metabolized after administration (very little eliminated unchanged)
-used for short term treatment of insomnia
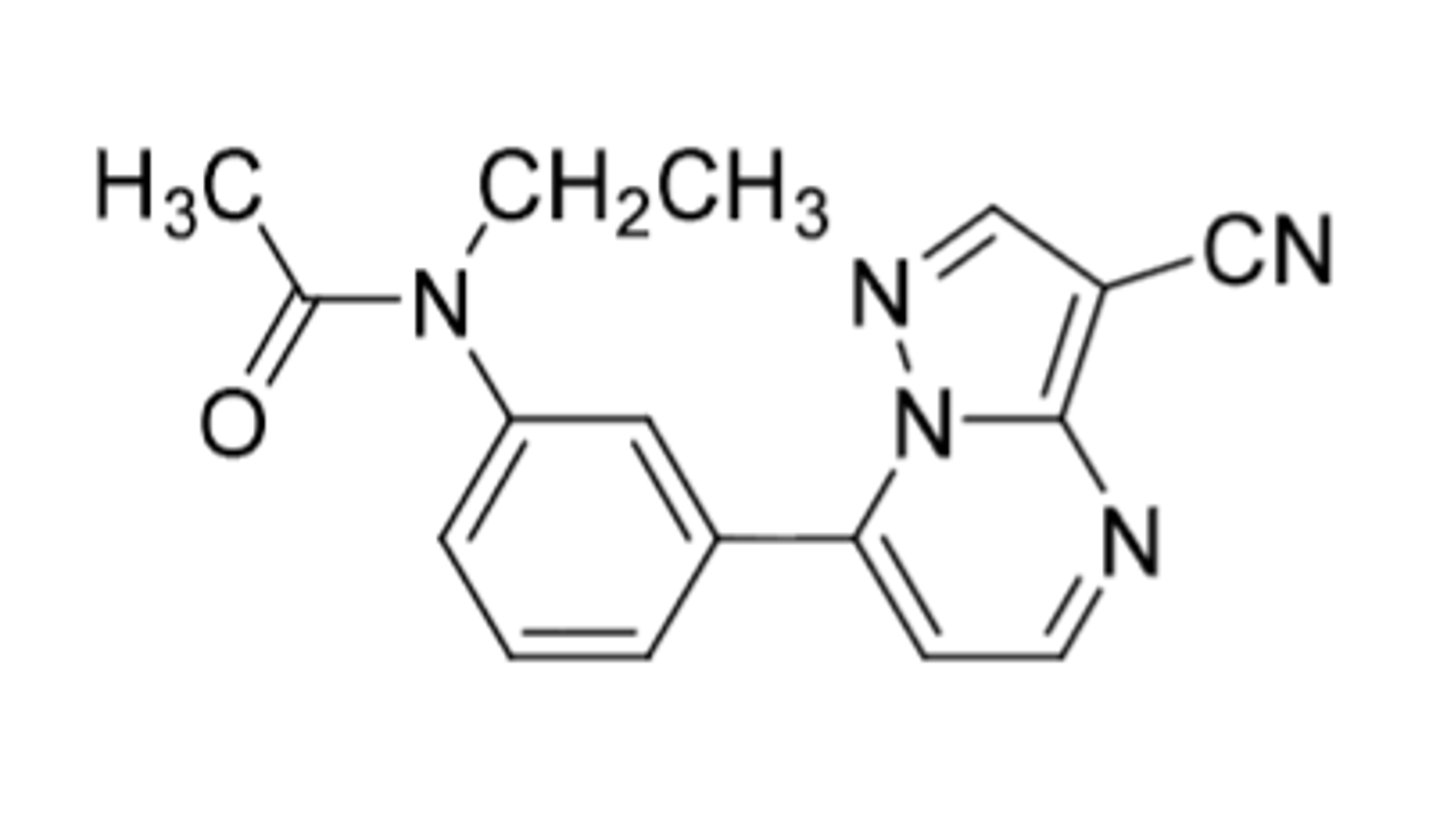
Eszopiclone (Lunesta)
The 3-Z drugs:
-mimic benzodiazepines by selectively binding to type 1 BZ binding sites (associated with sleep and hypnotic effects)
-C4
-extensively metabolized after administration (very little eliminated unchanged)
-used for short term treatment of insomnia
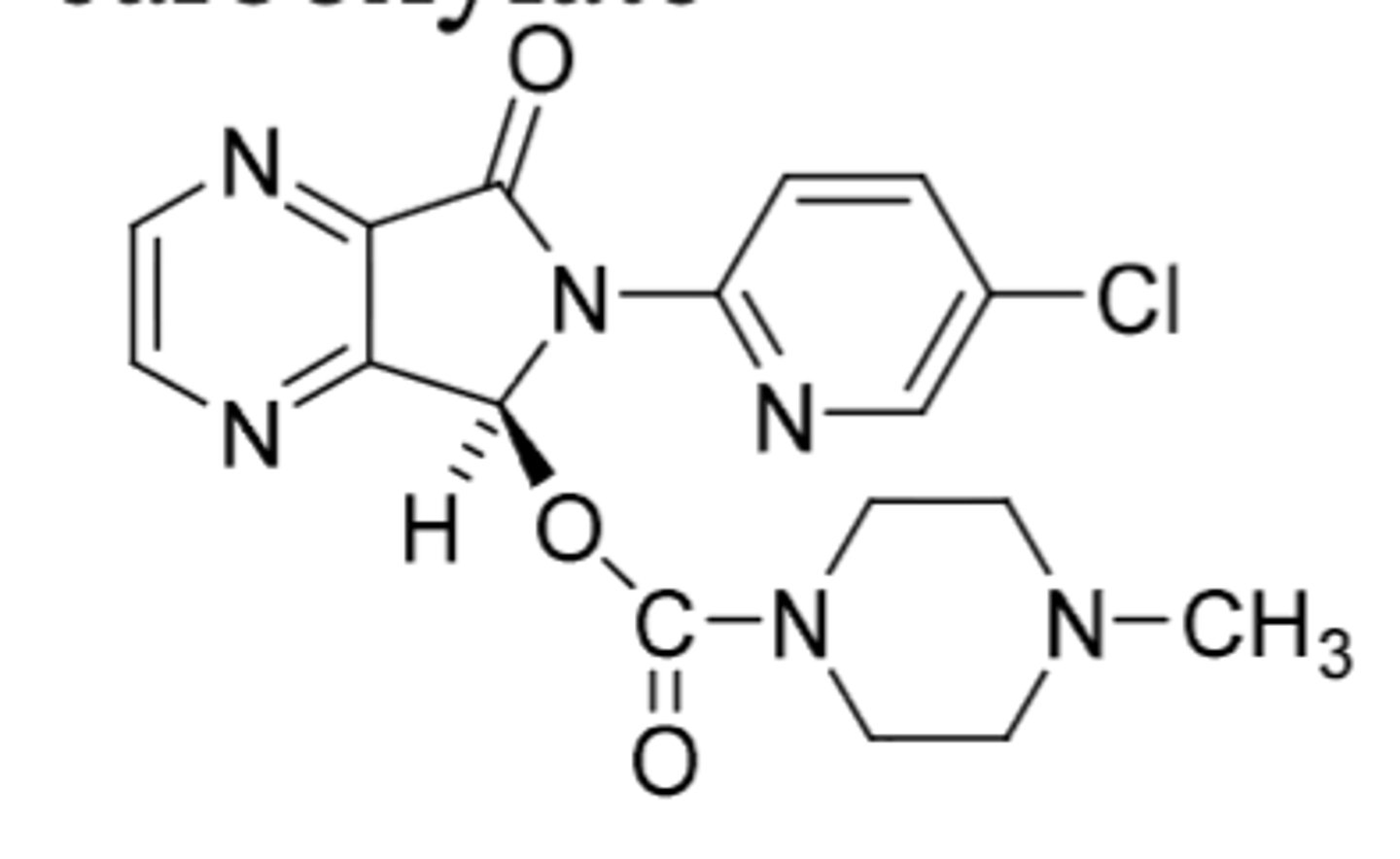
Dexmedetomidine
-selective alpha-2 agonist (prevents release of NE and ATP to cause CNS depression)
-used as sedative in ICU for intubated or ventilated patients as infusion or injection; regional or general anesthetic
-also exists as film formulation for buccal or sublingual administration
-NOT controlled
-t1/2 = 2h (extensive metabolism)
Ramelteon (Rozerem)
-for insomnia (can be used for long term treatment)
-NOT controlled
-mimics melatonin, binding to MT1 and MT2 receptors
-sleeping aid without potential for abuse, no withdrawal symptoms
-t1/2 = 1-2.6 hours and F= >84% (longer and higher than melatonin)
-Problem: has first pass metabolism (only 1.8% reaching systemic cicrculation)
-DDI: fluoxamine (SSRI) is a strong CYP inhibitor --> AUC increase by 170 fold when used together (do NOT use rameleton and fluoxamine)
-major metabolite: hydroxylation --> has 1/10 activity but concentration can be 20-100x the parent drug (a lot of ramelteons effect from this)
Structure compared to melatonin
-methoxy in ring = conformational restriction
-replacing N with C = bioisosteric replacement
-ethyl group instead of methyl
Melatonin has poor and variable BA (F=10=56%), short t1/2 (20-60min)
MT1 --> sleep
MT2 --> circadian rhythm
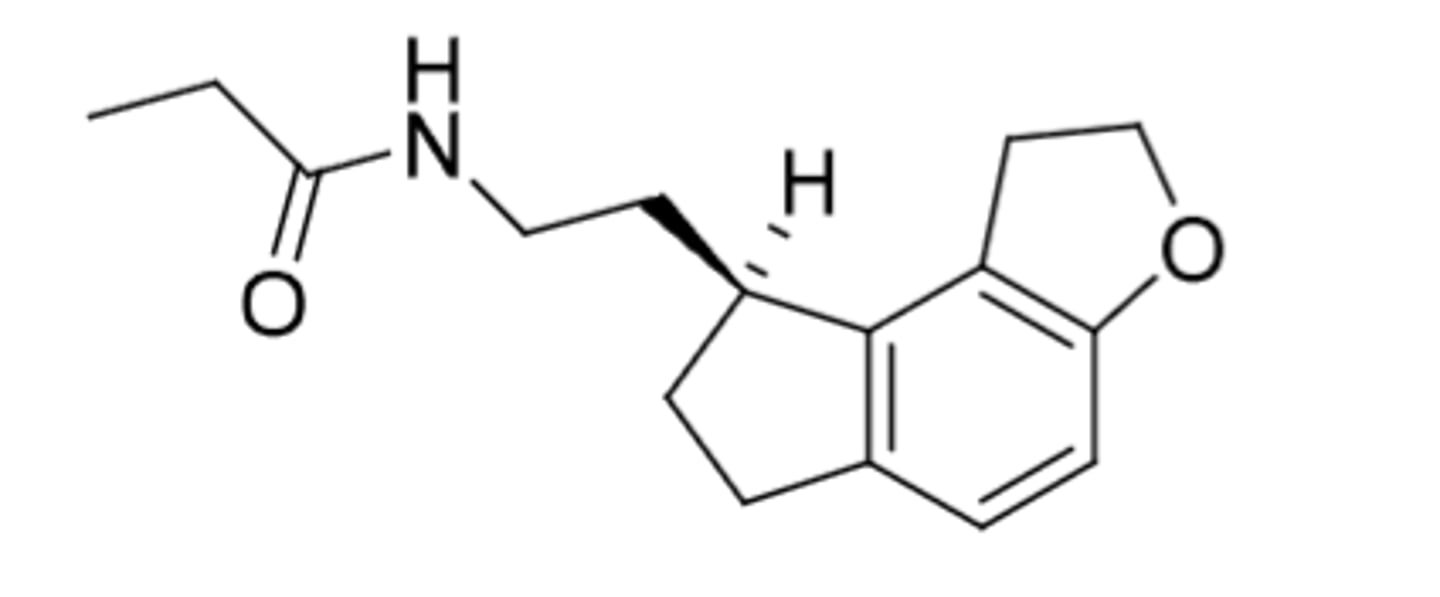
Tasimelteon
-Preferentially binds to MT2 for circadian rhythm control
-not controlled
-t1/2=1-2 hours
-extensive metabolism
-cyclopropyl = conformational restriction
Indications:
1. Non-24 Hour Sleep Wake Disorder (N24HSWD) (usually a condition in totally blind people)
2. Night time sleep disturbance in Smith-Magneis Syndrome
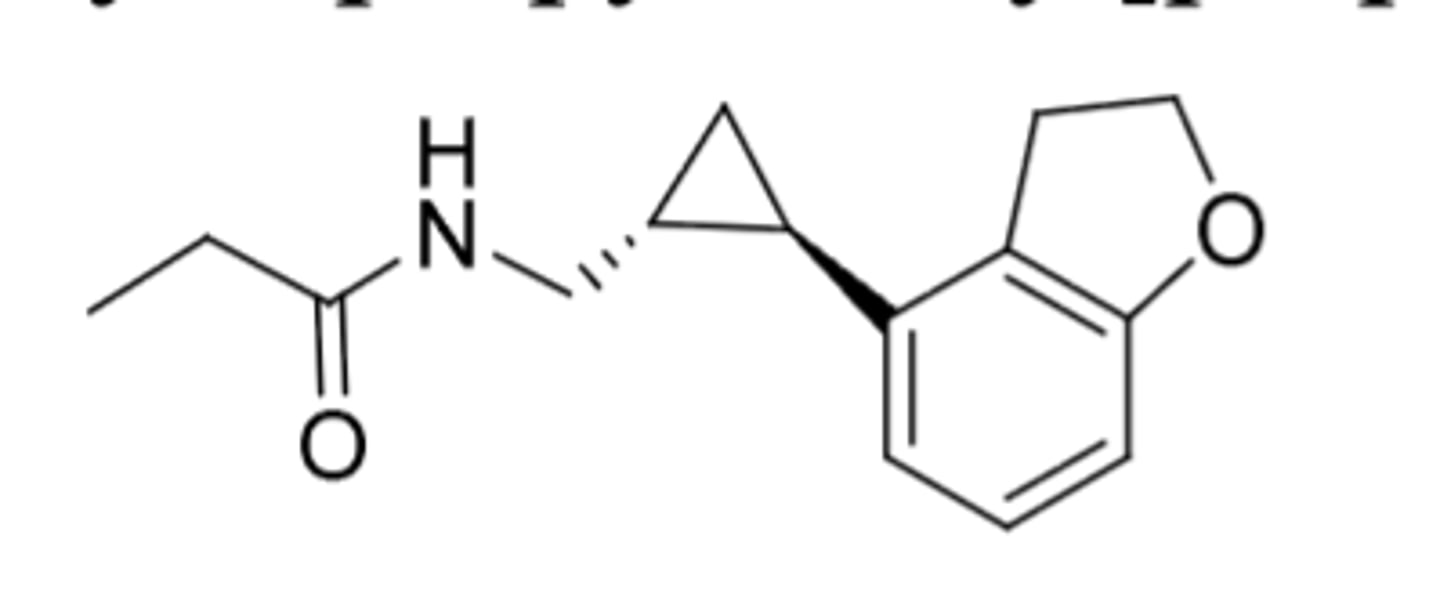
Suvorexant
-t1/2 = 12h
-10 mg daily
All "rexants" properties:
-DORA = dual orexin receptor antagonists
-orexin is a neuropeptide hormone in CNS to control and keep people awake
-Antagonism of orexin OX1R and OX2R blocks the excitatory transmission of these receptors
-orexin A and B are excitatory neuropeptides that work on OX1R and OX2R
-OX1R (orexin A preferentially binds) and OX2R (orexin A and B binding)
-Taken 30 minutes before bedtime
-Used for treatment of insomnia (second line; first line is cognitive behavior therapy)
-food effect: food can delay onset (don't take with food or after meal)
-all C4
Lemborexant
-t 1/2 = 17-19h (longest of rexants)
-5 mg daily
All "rexants" properties:
-DORA = dual orexin receptor antagonists
-orexin is a neuropeptide hormone in CNS to control and keep people awake
-Antagonism of orexin OX1R and OX2R blocks the excitatory transmission of these receptors
-orexin A and B are excitatory neuropeptides that work on OX1R and OX2R
-OX1R (orexin A preferentially binds) and OX2R (orexin A and B binding)
-Taken 30 minutes before bedtime
-Used for treatment of insomnia (second line; first line is cognitive behavior therapy)
-food effect: food can delay onset (don't take with food or after meal)
-all C4
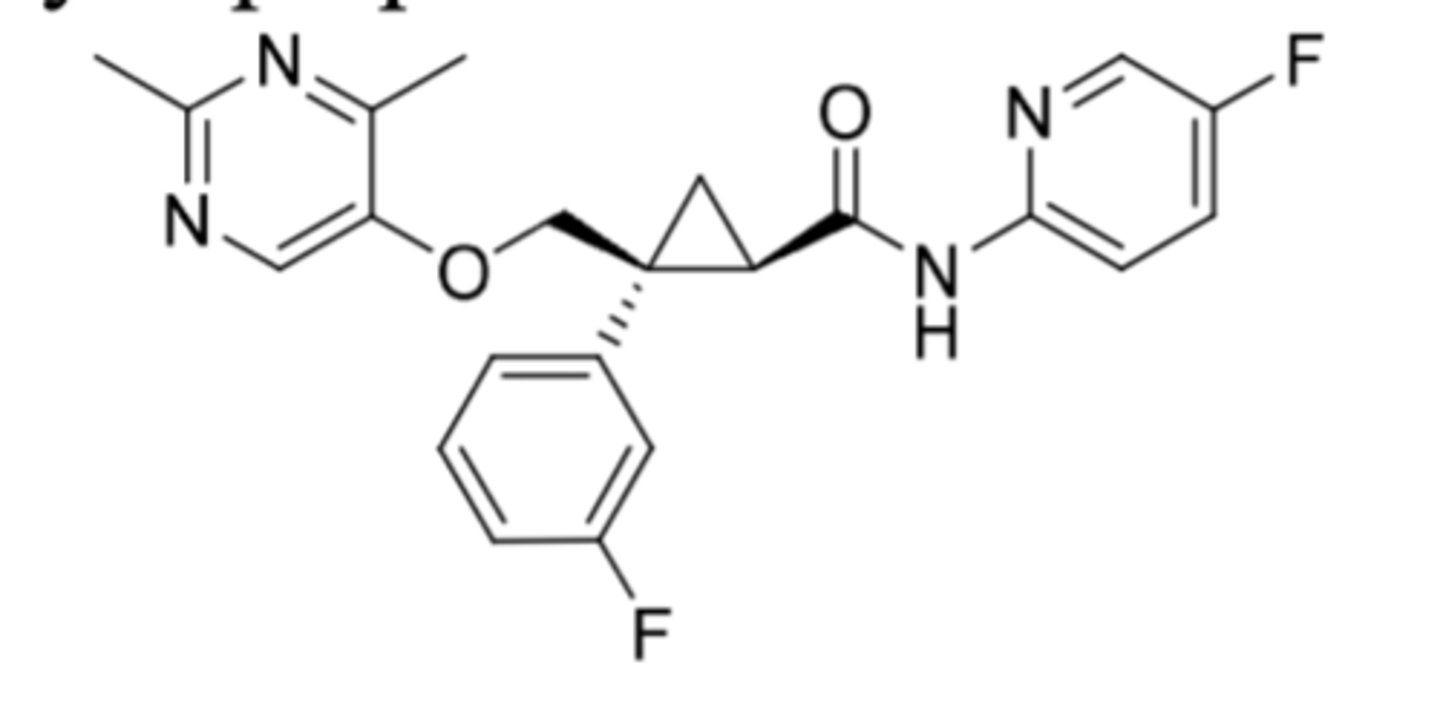
Daridorexant
-t1/2 = 8h
-25-50 mg daily
All "rexants" properties:
-DORA = dual orexin receptor antagonists
-orexin is a neuropeptide hormone in CNS to control and keep people awake
-Antagonism of orexin OX1R and OX2R blocks the excitatory transmission of these receptors
-orexin A and B are excitatory neuropeptides that work on OX1R and OX2R
-OX1R (orexin A preferentially binds) and OX2R (orexin A and B binding)
-Taken 30 minutes before bedtime
-Used for treatment of insomnia (second line; first line is cognitive behavior therapy)
-food effect: food can delay onset (don't take with food or after meal)
-all C4
Used as anxiolytics, sedative-hypnotics, anesthetics, skeletal-muscle relaxants, and antiepileptic drugs
In general do not promote the metabolism of other drugs (unlike barbiturates)
All benzodiazepines have muscle relaxant properties and all are C4
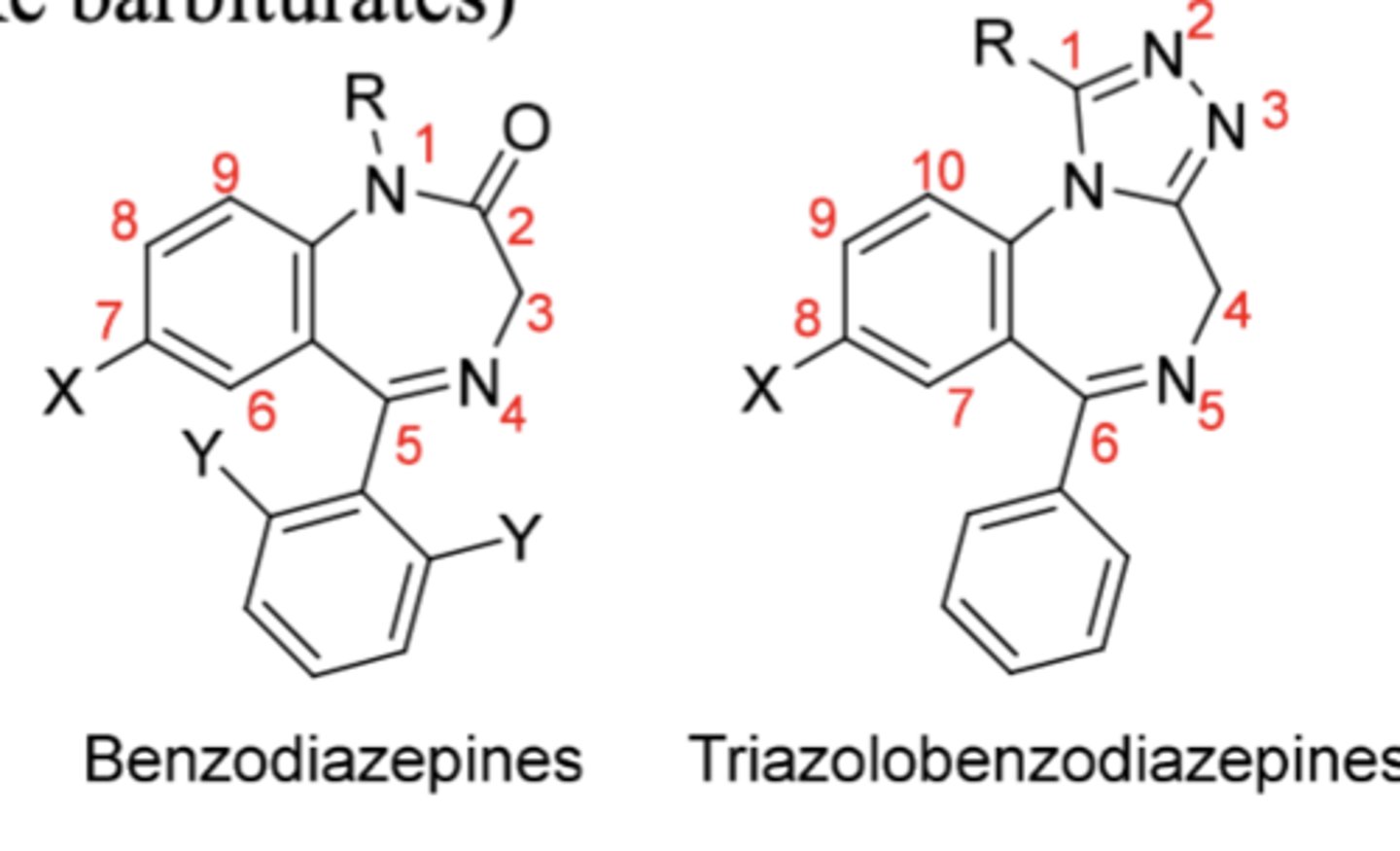
SAR:
1. An electron-withdrawing group @ 7-position (or 8 in triazolo) required for activity. Stronger withdrawing groups result in better activity. X is usually a Cl or NO2.
2. A phenyl @ position 5 needed for good activity.
o Electron-withdrawing Y (Cl or F) at o-position or both o-positions ⇒ ↑activity.
o A substituent at the p-position ⇒ ↓activity
o hydroxylation unlikely if EWG on phenyl
3. Saturation of 4,5-double bond (imine) ⇒ ↓activity
4. A hydroxyl @ 3-position is tolerated
5. A carbonyl at 2-position optimal for activity
6. Small R @ 1-nitrogen for maximal activity
o If too large --> prodrug (needs to be removed for activation)
Benzodiazepine SAR
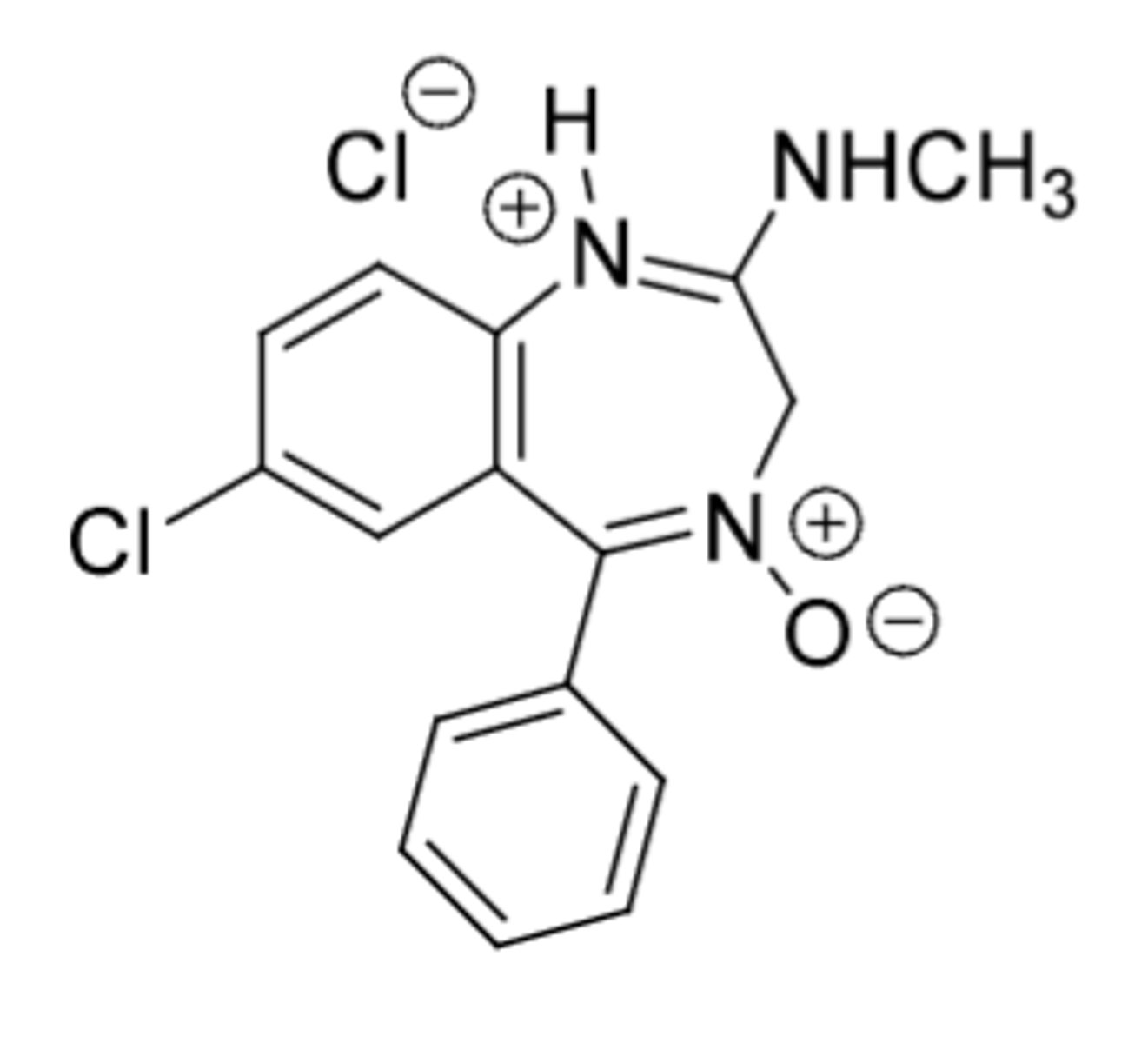
Chlordiazepoxide Hydrochloride
-anxiolytic, sedative
Metabolism:
-demetyhlation
-alpha-hydroxylation (at 3 position) --> active metabolite
-anoxide can be reduced to imine (Active)
-hydroxylation on phenyl
Diazepam (Valium)
-for relief of anxiety and systematic relief of acute alcohol withdrawal as a skeletal muscle relaxant; can be used for the treatment of seizures
-Relieves skeletal muscle spasms, tetanus, spasticity caused by cerebral palsy and paraplegia
-prolonged use may lead to dependence
-t1/2 = 60-72 hr
-nordiazepam (demethylated active metabolite) t1/2 can be as long as 200h
Metabolized to temazepam (which is an active drug itself)
-alpha-hydroxylation at 3 position
-temazepam is used clinically as hypnotic agent
-t1/2 = 4-18h
Temazepam is metabolized to oxazepam (which is an active drug itself)
-demethylation
-t1/2 = 6-8h
-used clinically treatment of anxiety and alcohol withdrawal symptoms
-can be used in elderly patients w/ liver dysfunction because there is no further metabolism that occurs
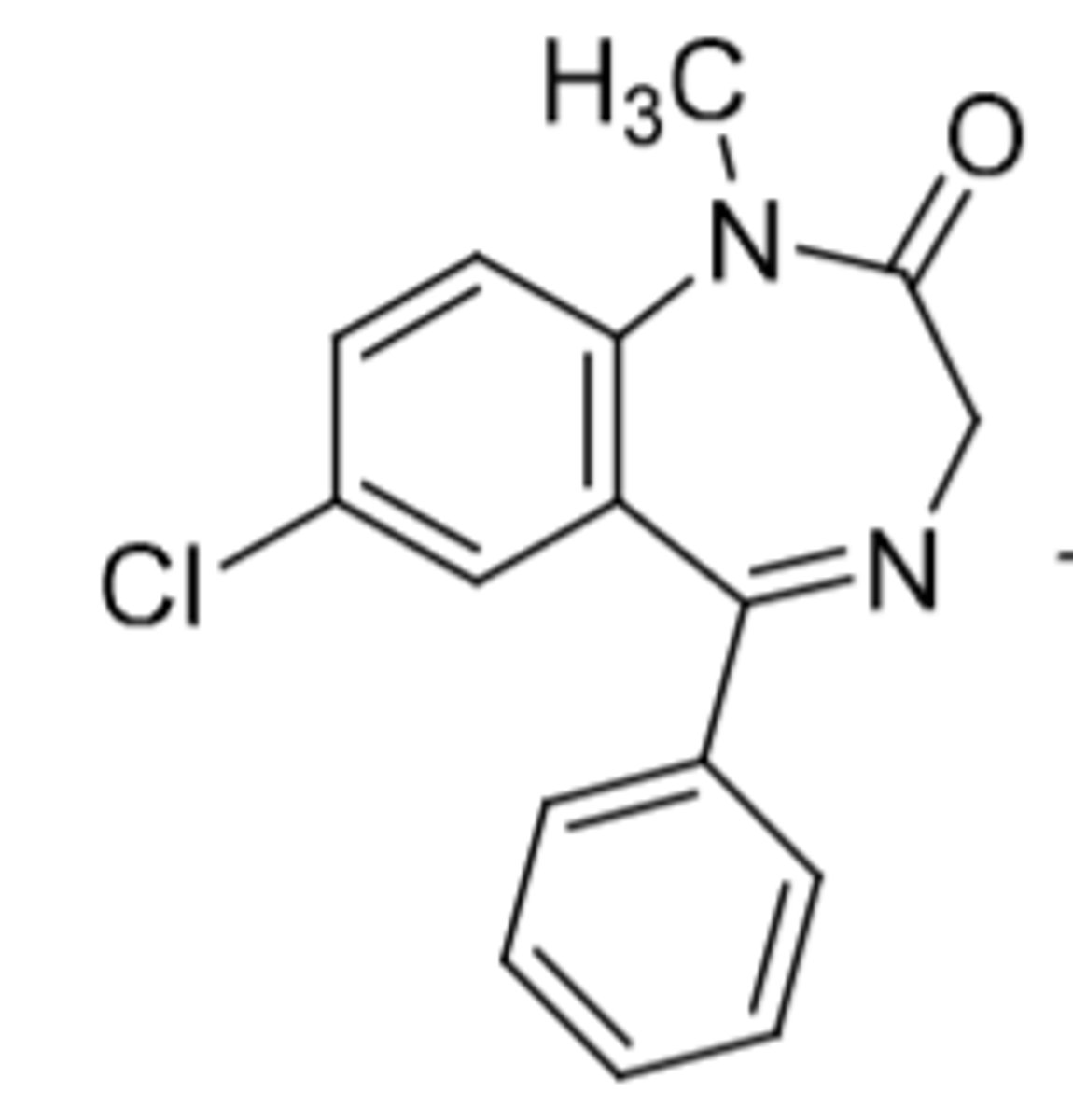
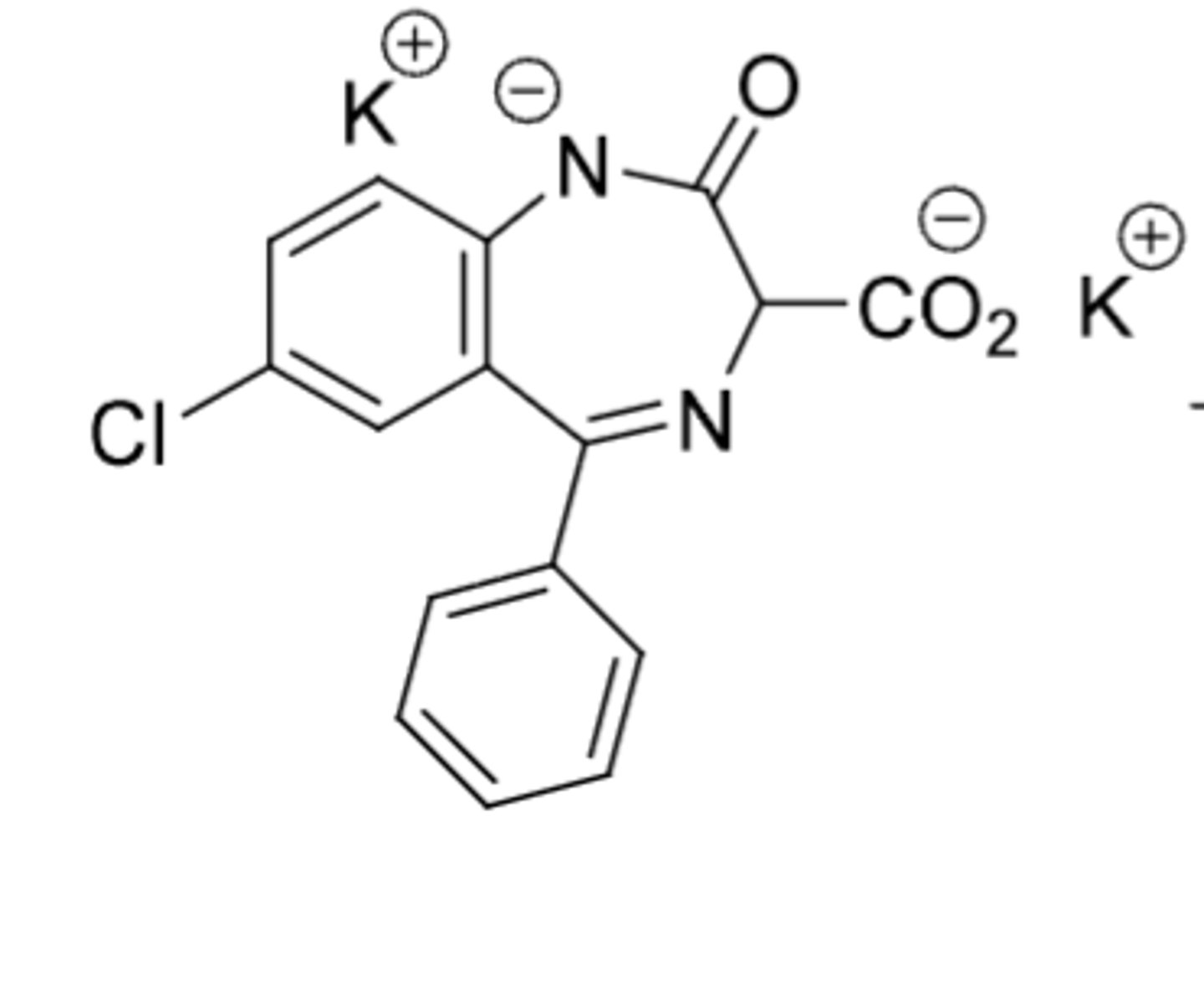
Clorazepate Dipotassium
-prodrug for nordiazepam due to COOH at 3 position (needs to be removed by decarboxylation to get activated)
-used for anxiety, treatment of alcohol withdrawal symptoms, and epilepsy
-nordiazepam half life = 36-200h
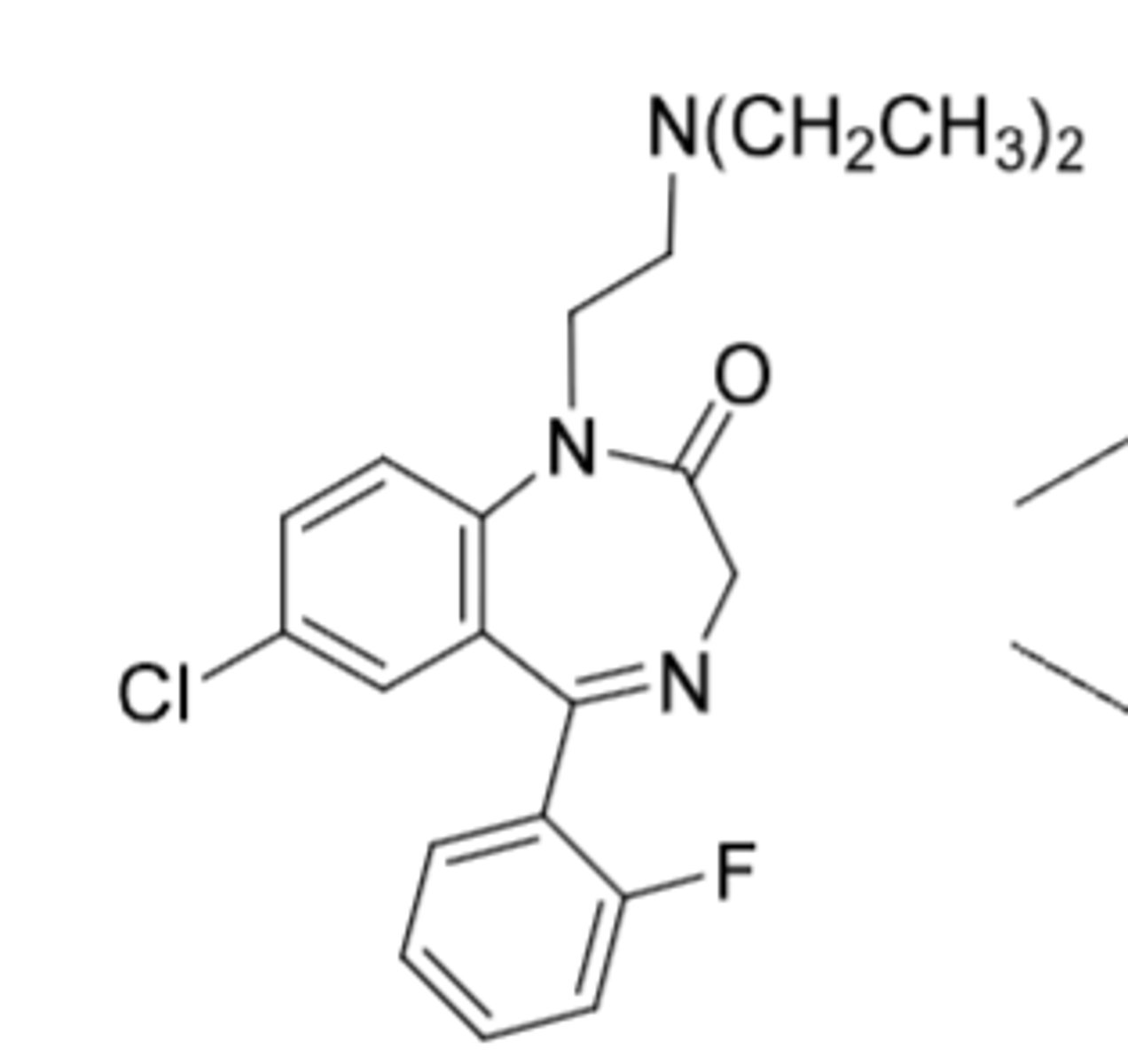
Flurazepam
-prodrug (bulky R group at 1 position - removed metabolically through oxidative dealyklation)
-N-desalkyl major active metabolite --> t1/2 = 47-100h, good sedative and hypnotic activity (can help normal REM cycles for sleep)
-used for sleep inducing activity
-Same structure as nordiazepam with additional Fluorine (EWG) on phenyl (provides metabolic resistance to hydroxylation)
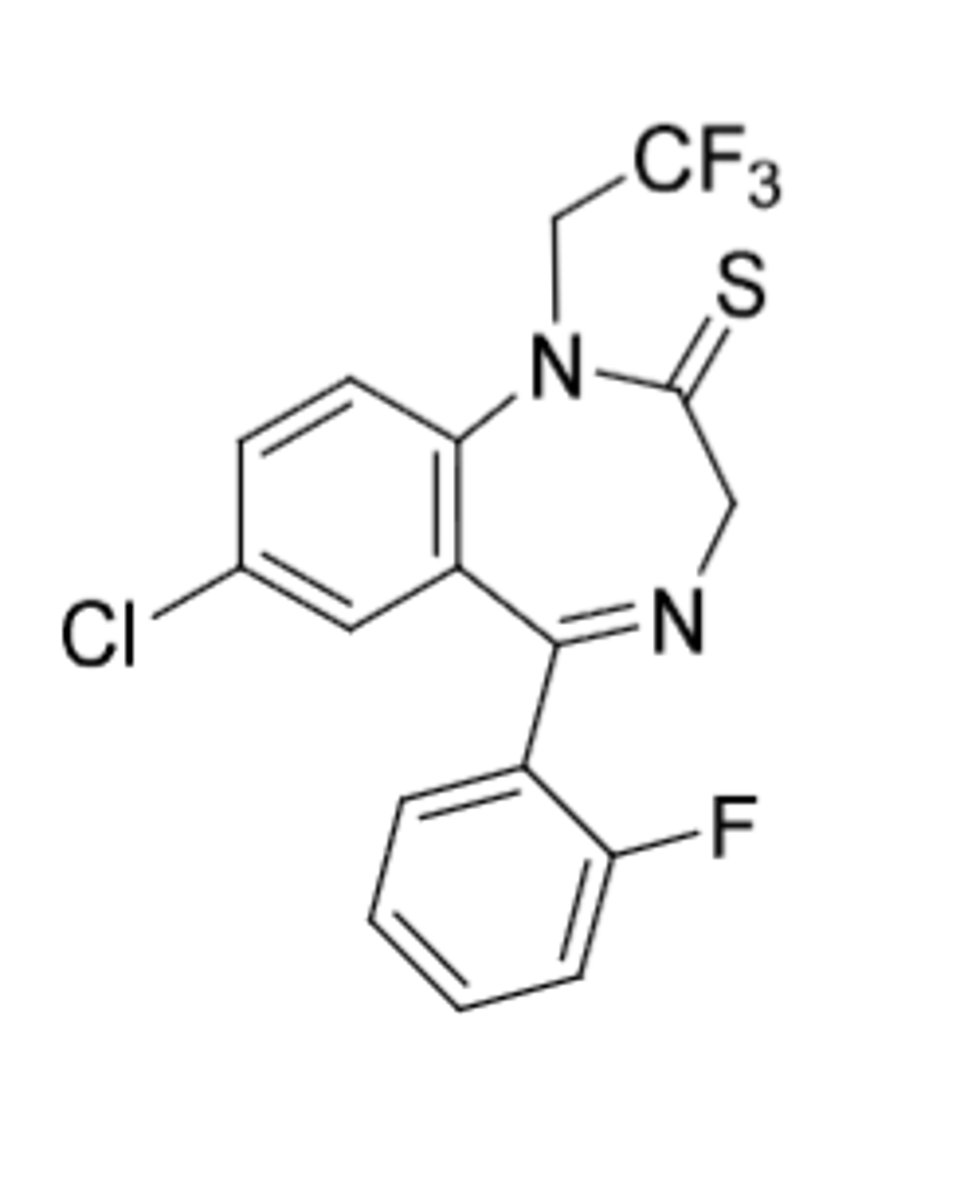
Quazepam
-Sulfur at 2 position (more lipophilic)
-for patients with insomnia
-N-desalkyl metabolite (t1/2 = 47-100h) (same metabolism as flurazepam but its not a prodrug because parent drug is active --> CF3 is small enough R group)
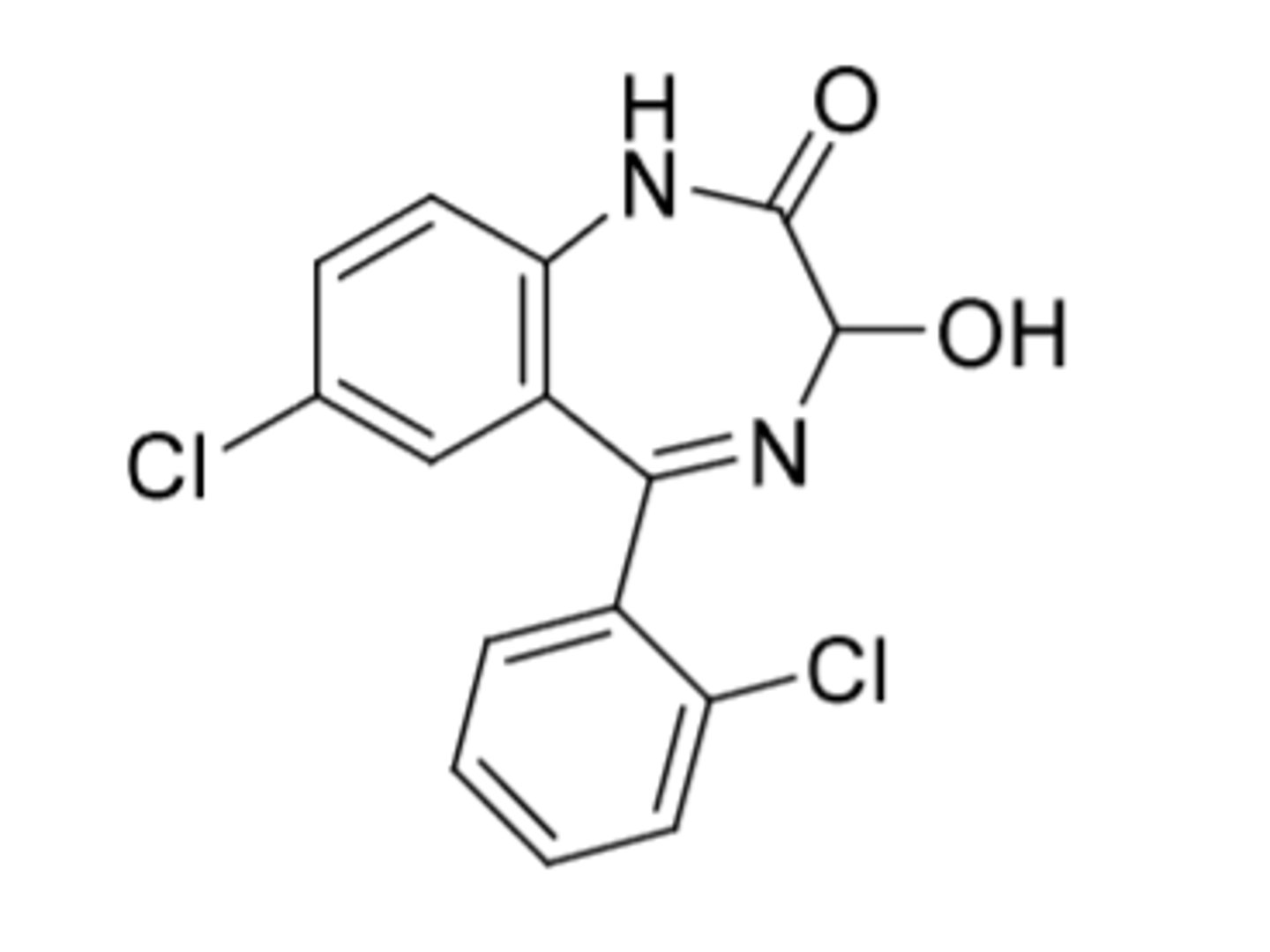
Lorazepam
-Used to treat anxiety and as anticonvulsant
-treatment of choice for delirium tremors (DTs), which is a severe form of alcohol withdrawal syndromes
-Cl on both phenyls (prevent hydroxylation)
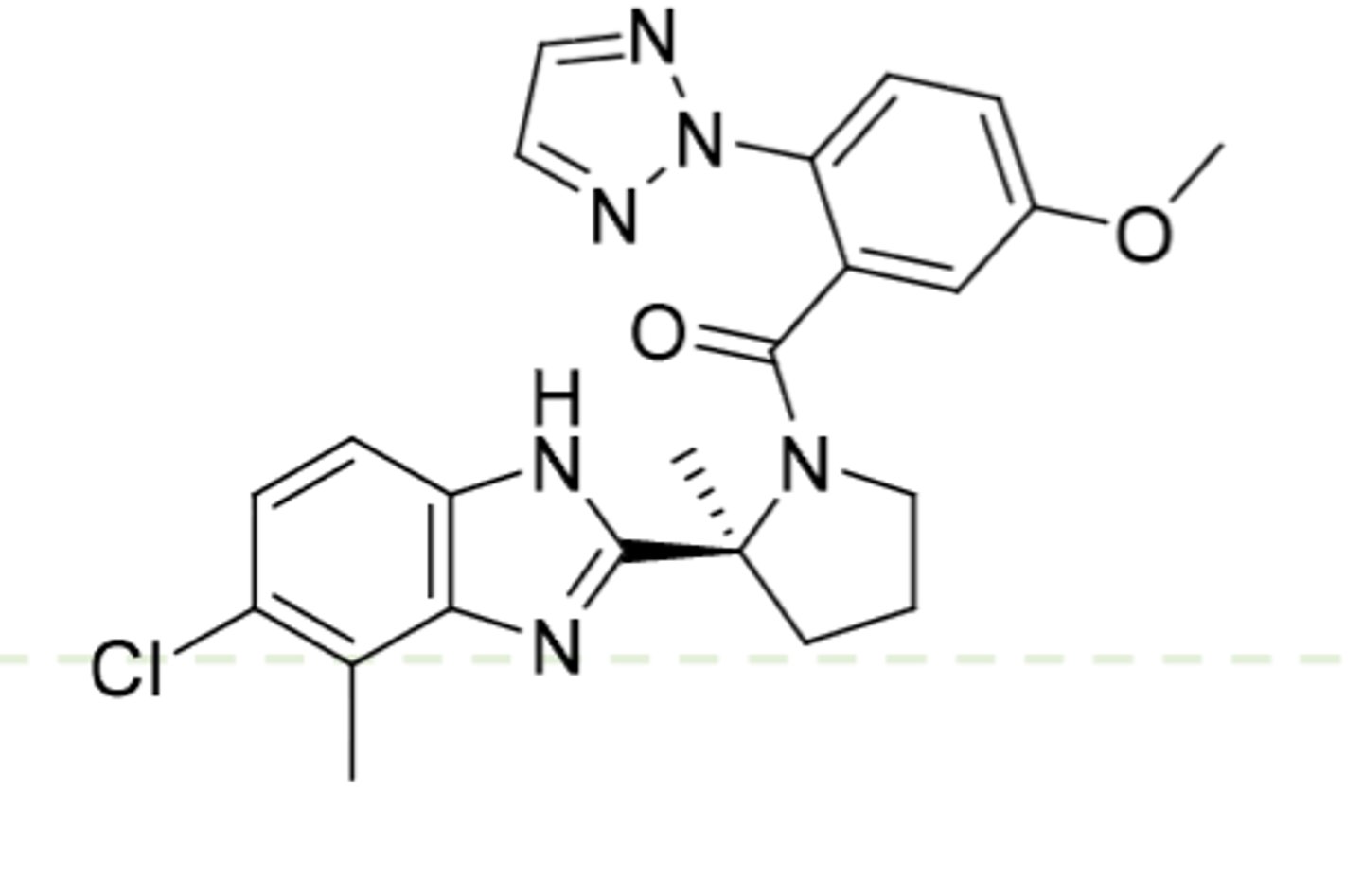
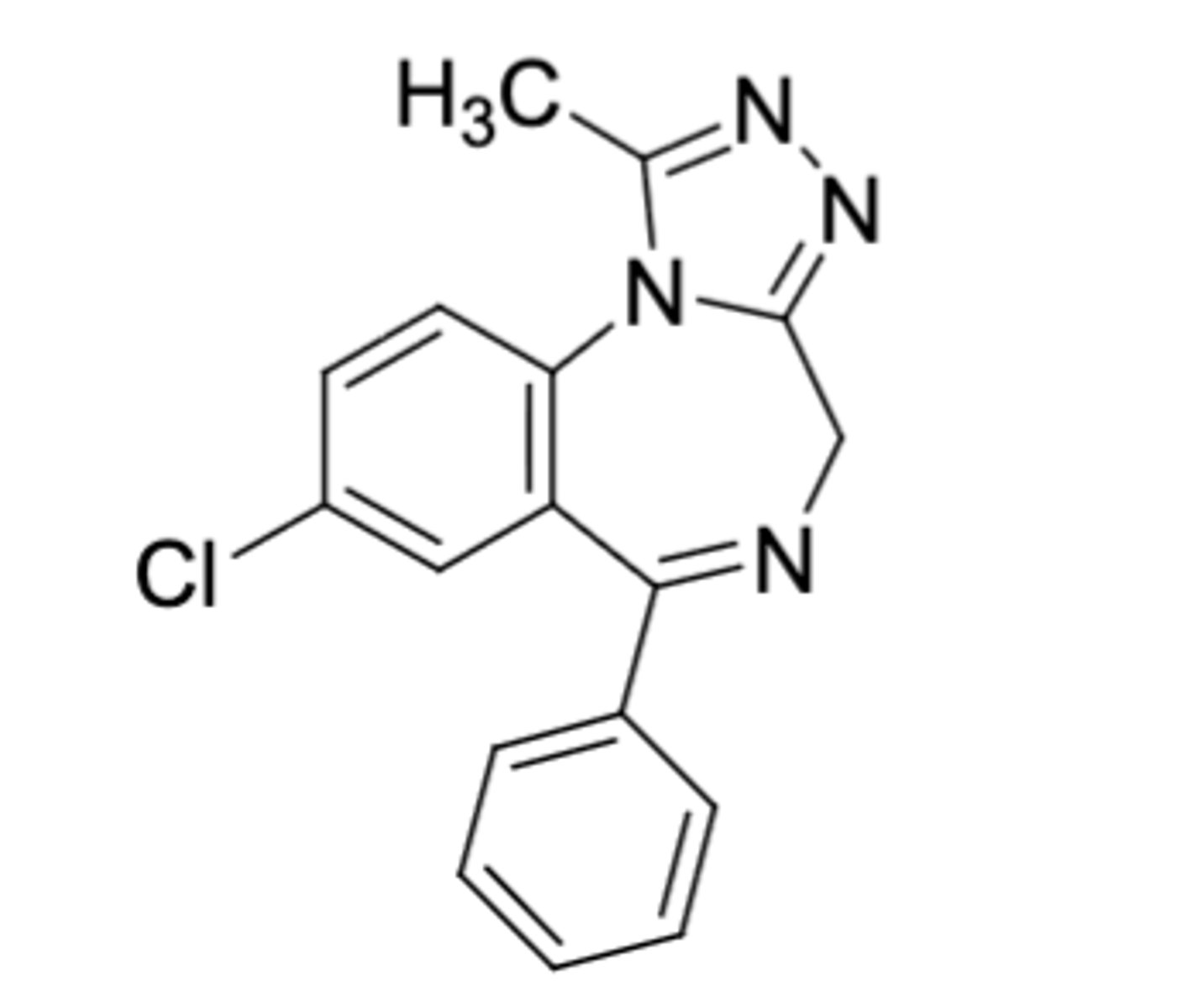
Alprazolam
-Triazolobenzodiazepine
-CH3 on triazolo ring (more lipophilicity), no second Cl on phenyl
-0.25-0.5 mg TID
-for panic disorder and anxiety
-also for agorophobia, which is fear of public spaces
Triazolobenzodiazepine potency: Triazolam > Alprazolam > Estazolam
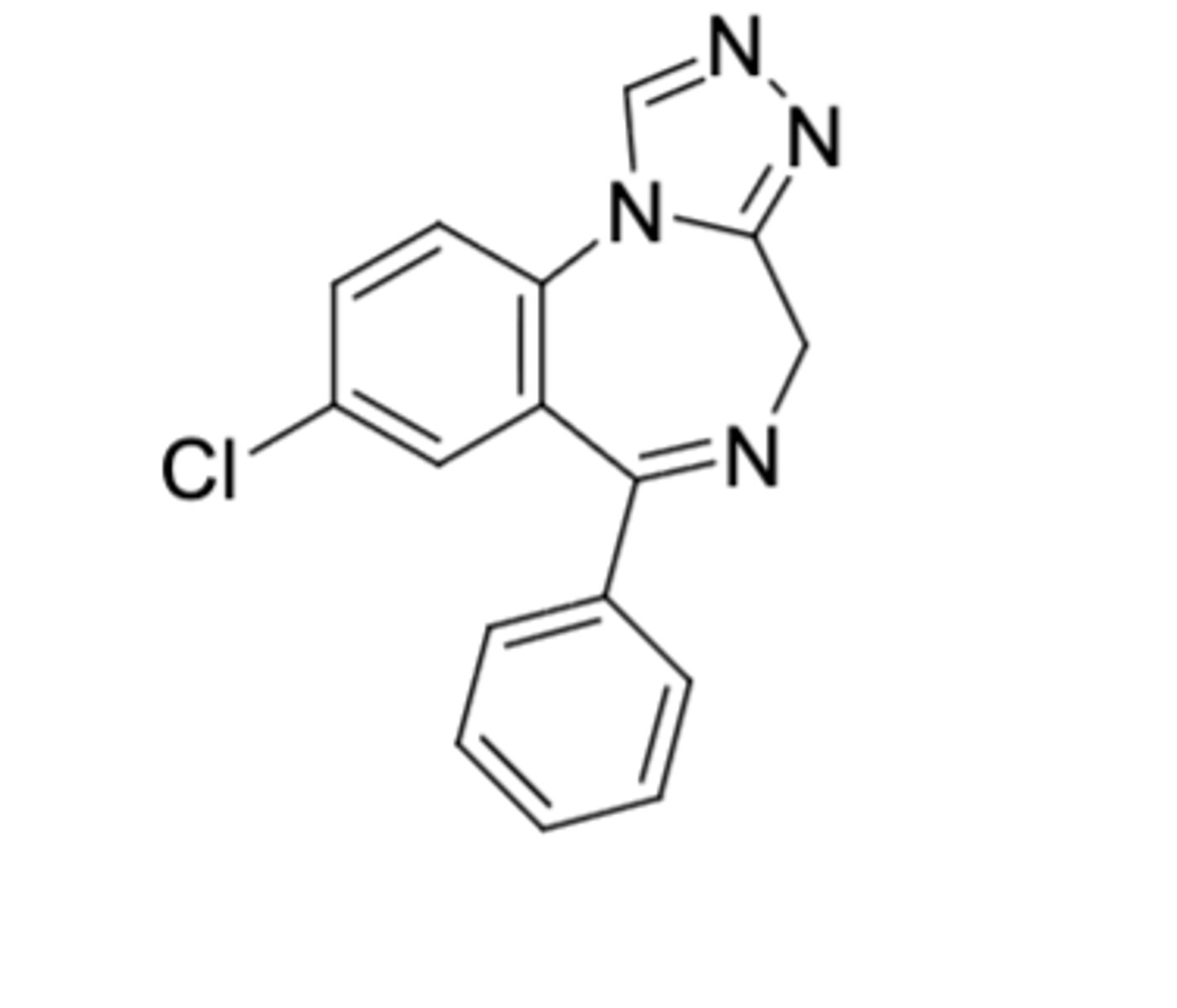
Estazolam
-Triazolobenzodiazepine
-No CH3 on triazolo ring (more lipophilicity) and no second Cl on phenyl
-1-2mg at bedtime
-for insomnia
Triazolobenzodiazepine potency: Triazolam > Alprazolam > Estazolam
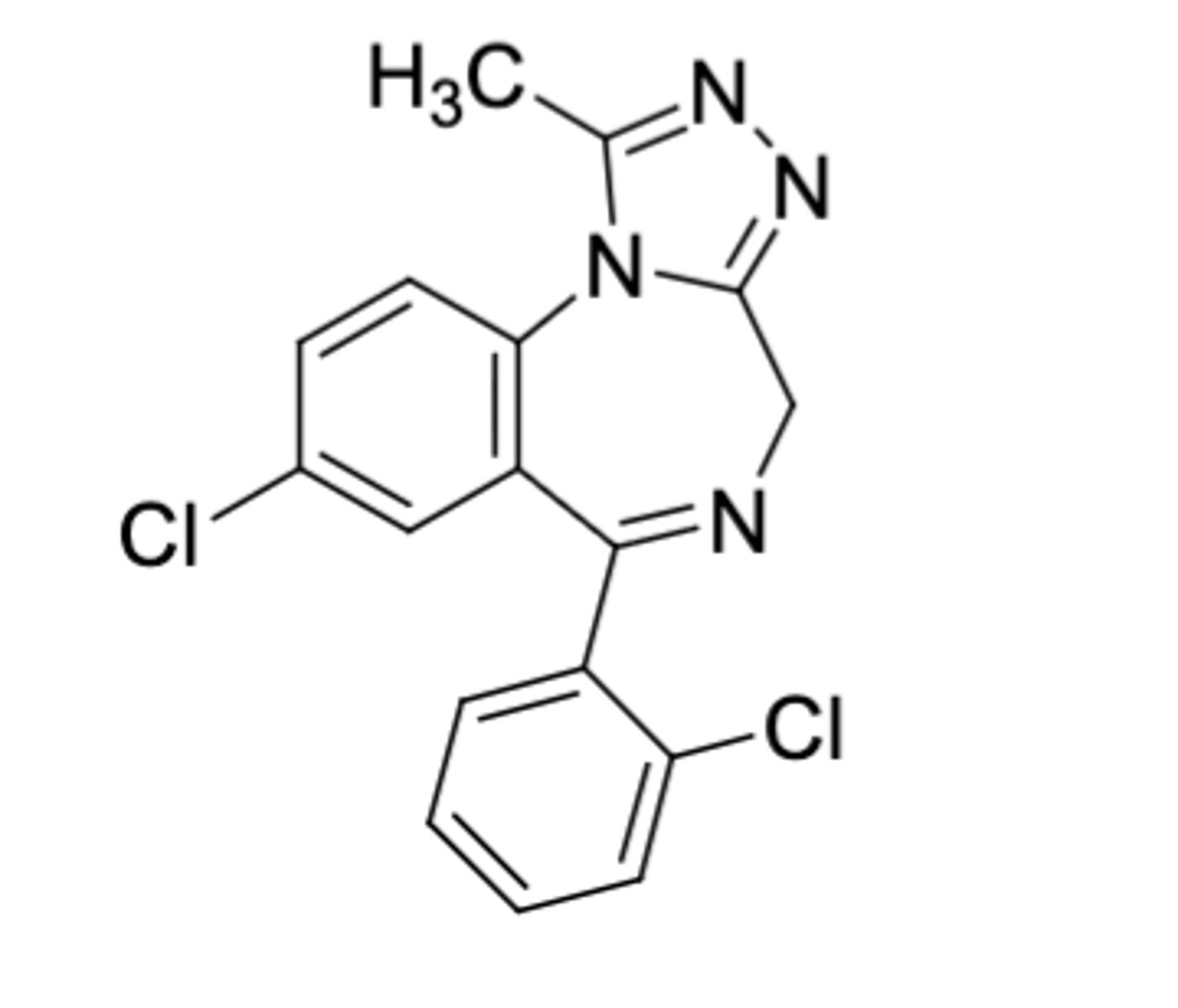
Triazolam
-Triazolobenzodiazepine
-CH3 on triazolo ring (more lipophilicity) AND second Cl on phenyl
-0.25 mg at bedtime for insomnia
-known to be associated with greater frequency of side effects (rebound anxiety, amnesia) because most potent
-withdrawn in other countries but still used in U.S. w/ low doses
Triazolobenzodiazepine potency: Triazolam > Alprazolam > Estazolam
Flumazenil
-Benzodiazepine receptor antagonist
-Soft drug
-ester group allows for ester hydrolysis
-will bind to receptor without CNS depressive effects to prevent benzodiazepines in patients system from activating GABAa receptors
-usually given IV (need to be administered quickly) --> 200 mcg over 1-2 min until effects in patient (max dose is 3 mg/hr)
-very short t1/2 --> 7-15 min in brain, 20-30 min systemic, 40-80 min terminal
-sometimes if you stop giving it, patients may fall back into CNS depression (so may have to readminister)
-No phenyl ring at 5 position which is required for activation of receptor
-side effects: associated with occurence of seizures
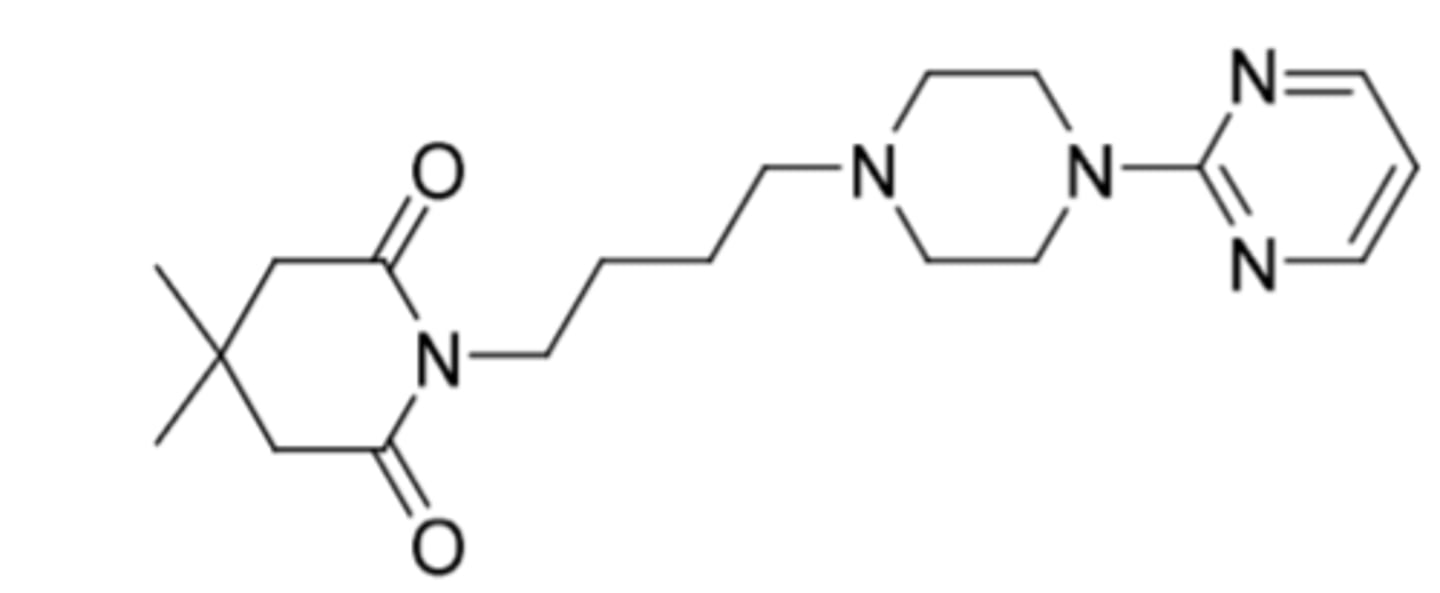
Buspirone (Buspar)
-5HT1A receptor agonist
-partial agonist at PRE-synaptic 5HT1A
-full agonist at POST-synaptic 5HT1A
-used for anxiety (no anticonvulsant or muscle relaxant properties)
-NOT controlled (no sedative effects, no dependence or withdrawal)
-moderate D2 affinity
-after cleavage, 1-PP potent alpha-2-antagonist (promotes release of NE)
-t1/2 = 2-3h
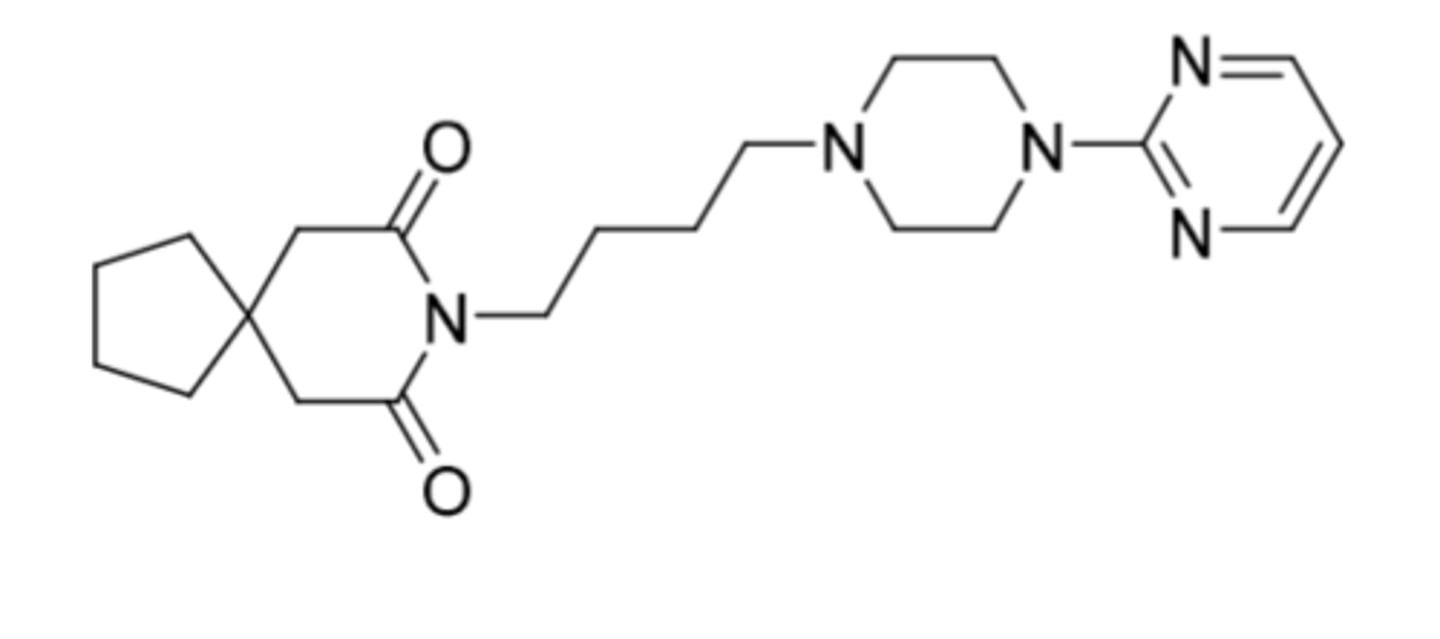
Gepirone
-same structure as buspirone but removed 2C on end ring
-approved for MDD
-5HT1A partial agonist
-No D2 affinity
-not controlled
-produces 1-PP metabolite (strong alpha-2 antagonism promoting release of NE)
-Exxua = ER formulation for QD dosing
-t1/2 = 5h
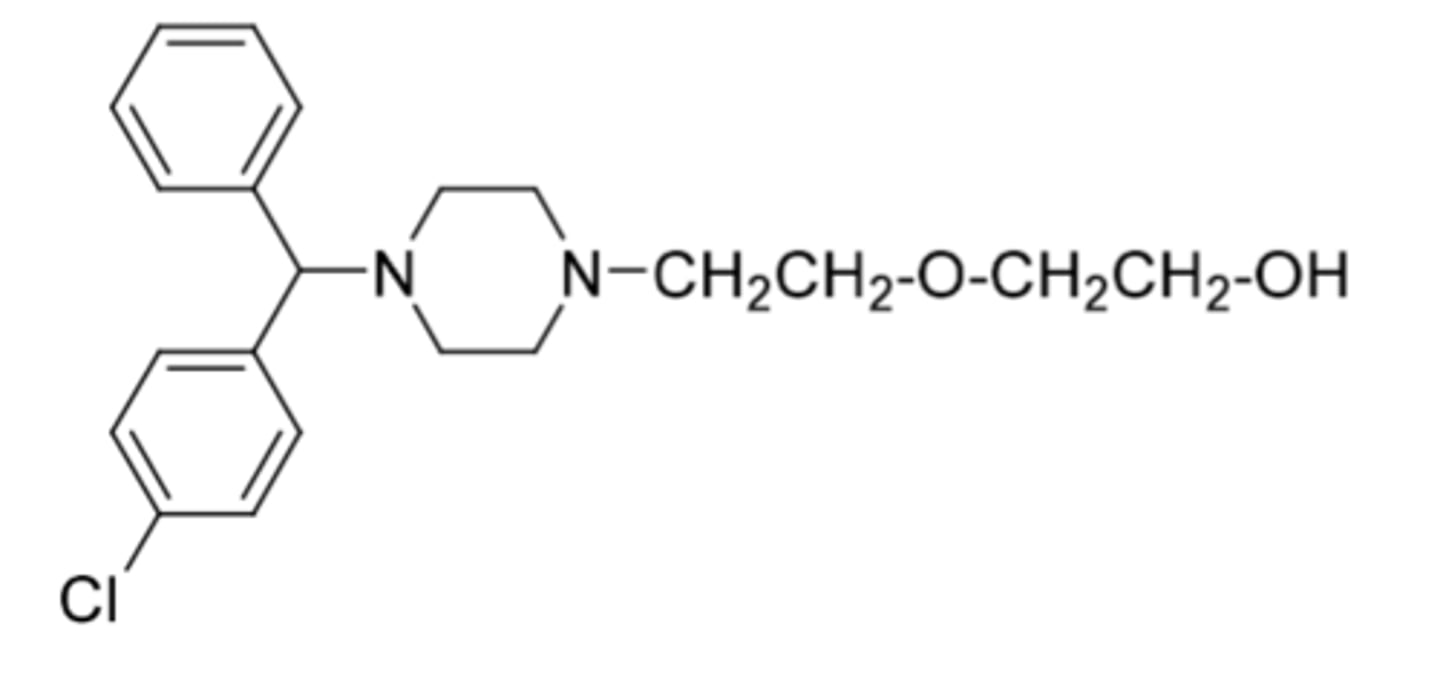
Hydroxyzine
-first generation antihistamine (H1 antagonist)
-not controlled
-used as mild tranquilizer to reduce anxiety
-side effect: teratogenic activity
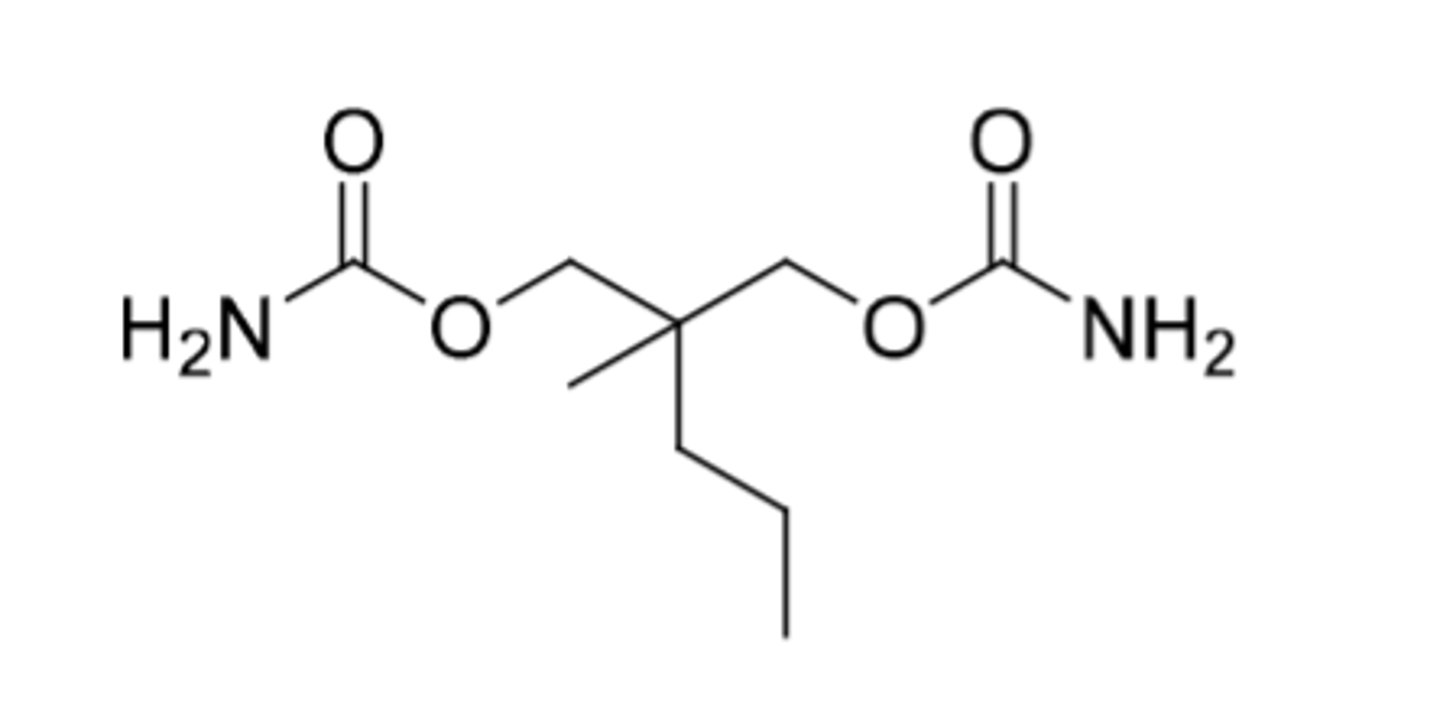
Meprobamate
-Miltown brand name
-omega-1 oxidation
-used to control anxiety
-"happy pill"
-mechanism not known
-can lead to physical dependence (C4)
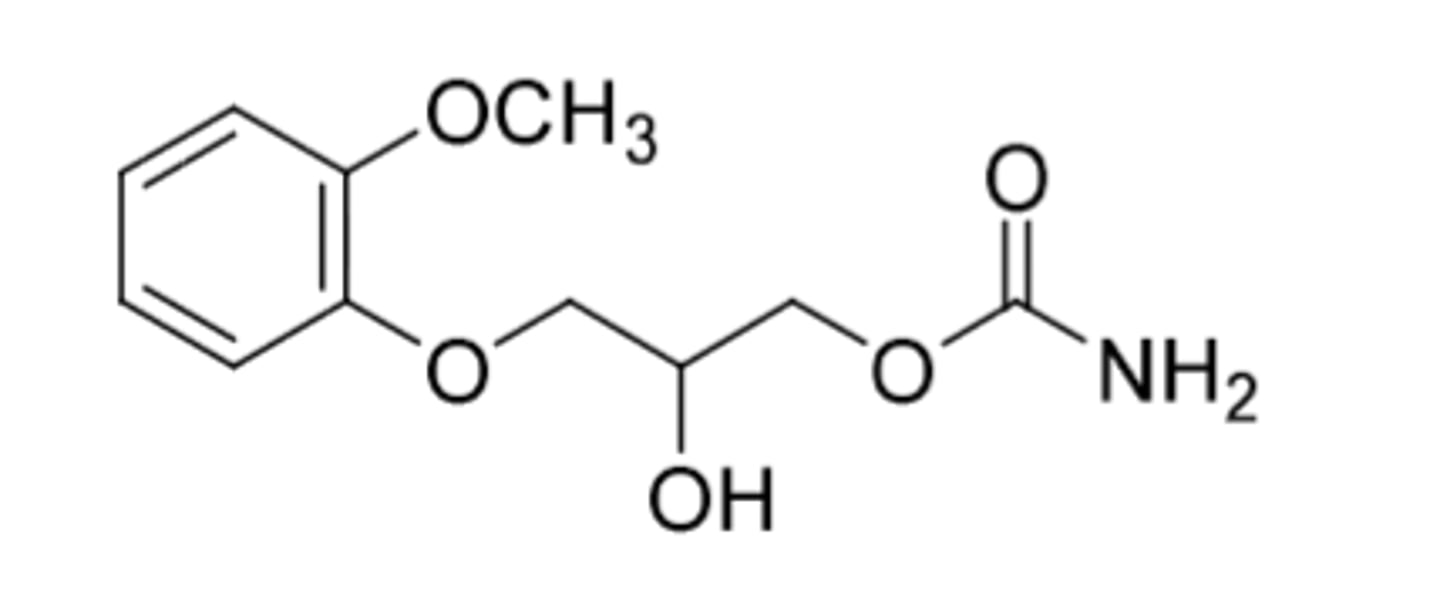
Methocarbomal (Robaxin)
-skeletal muscle relaxant
-used to relieve discomfort associated with acute and painful musculoskeletal conditions; can also be used to control neuromuscular aspects of tetanus
-carbamate of guafenesin (an expectorant)
Often used in combo with analgesics
-with acetominophen --> Robexacet
-with ibuprofen --> Robax Platinum
-with aspirin --> Robaxisal
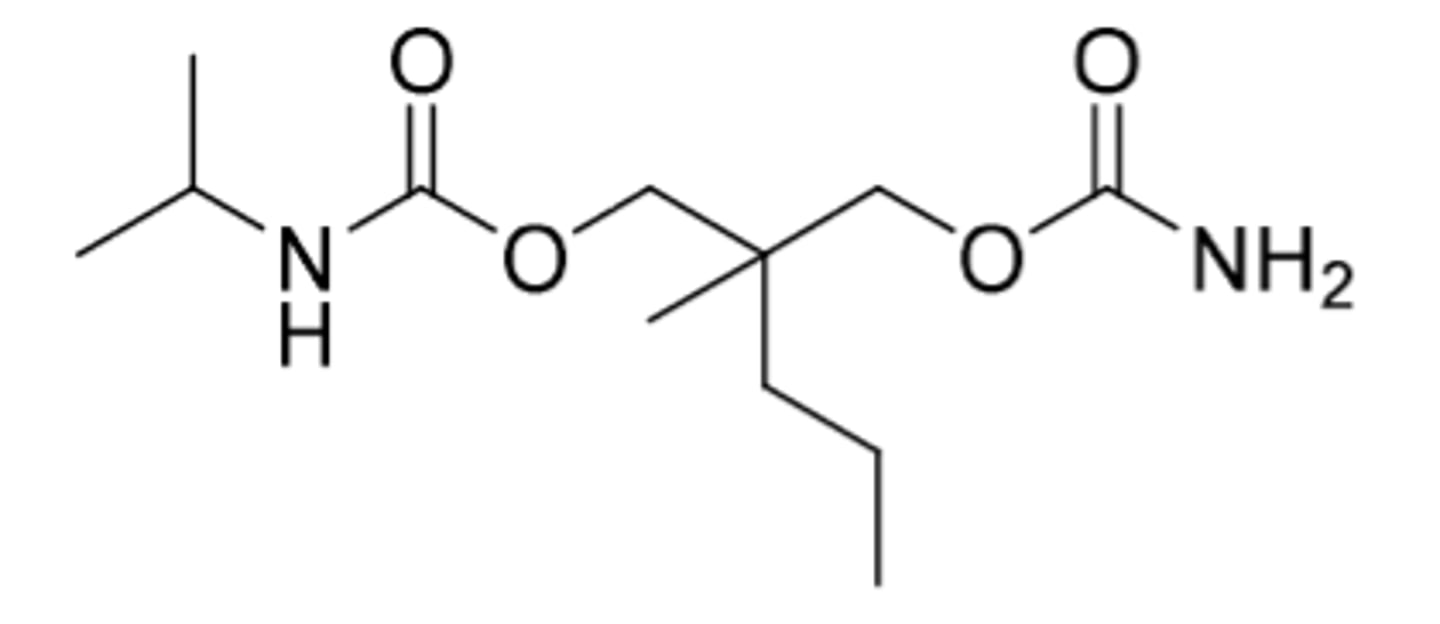
Carisoprodol
-skeletal muscle relaxant
-similar to meprobamate structure (metabolically turns into meprobamate
-CNS depression, abuse potential (C4)
-Works directly on spinal cord area, not directly on skeletal muscle cells
Still learning (6)
You've started learning these terms. Keep it up!