Oxides
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
What does diatomic molecule mean?
It means that it consists of two atoms
What does diatomic anion imply in oxygen chemistry?
The anion must be O2(-) (superoxide) or O2(2-) (peroxide). NOT O(2-) (oxide, monatomic)
What ions are present in KO2?
K(+) and O2(-)
Why is O2 in KO2 NOT -4?
Because O atoms are covalently bonded. Charge is delocalised over the O2 unit. The species is O2(-), not 2 x O(2-)
What is the formula, geometry, charge and bond in oxide?
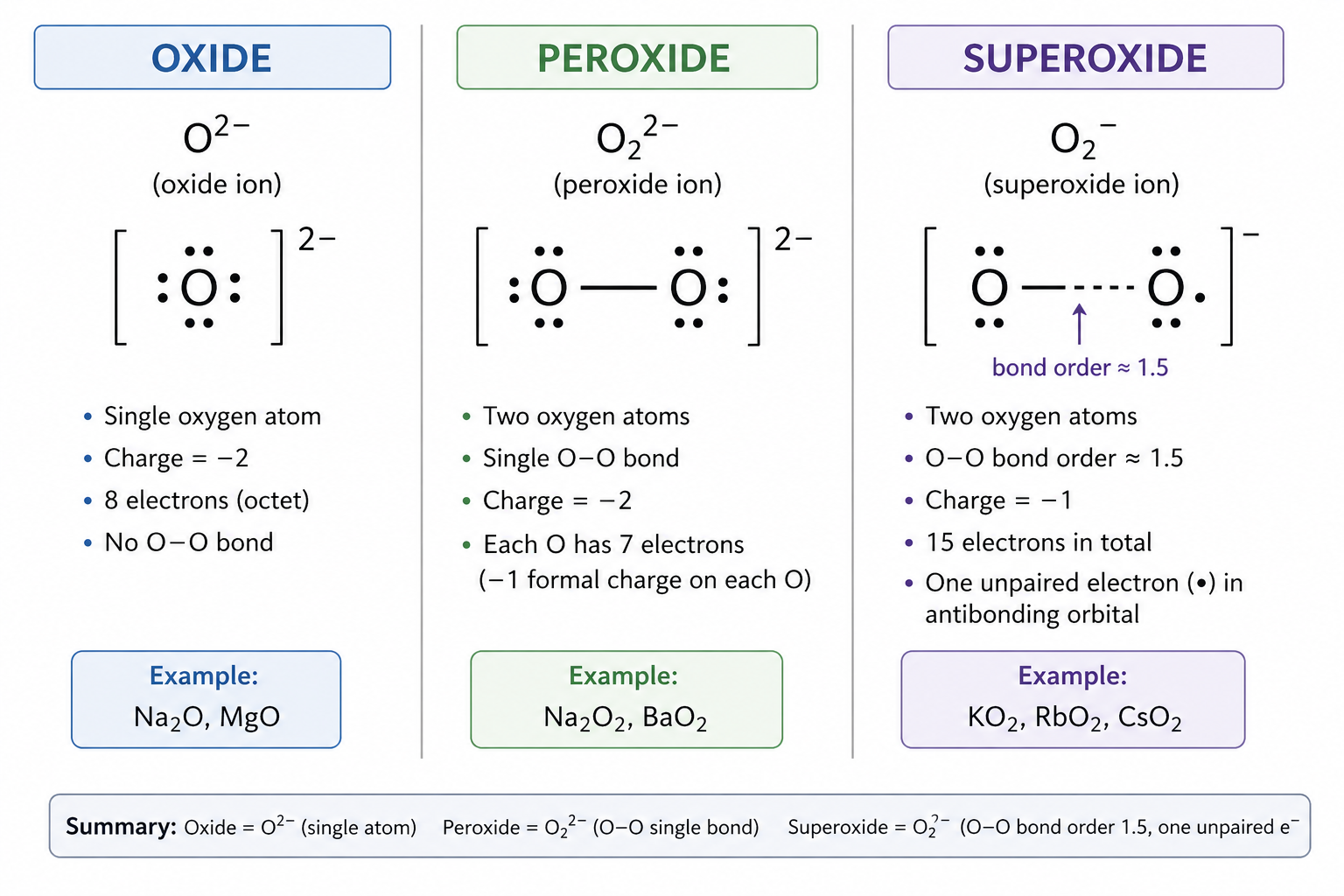
Why is O2(-) paramagnetic?
It has one unpaired electron in an antibonding pi* orbital
Why does potassium form KO2 (superoxide)?
Because large K+ stabilizes large, diffuse anions. And it favours O2(-) over O(2-)