Equillibrium Lab
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Purpose
To determine the equilibrium constant (Kc) for the reaction with a series of Iron III and SCN- reactions
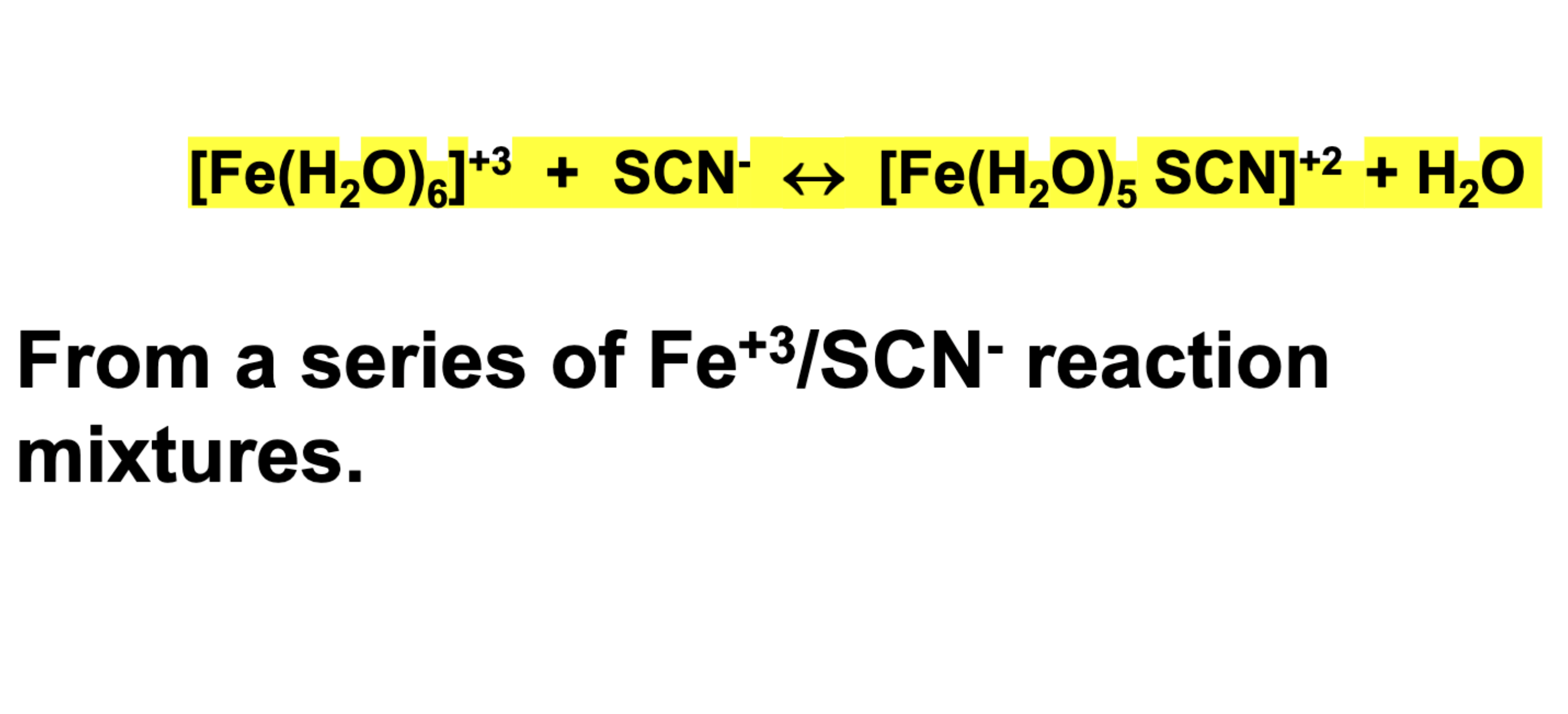
What is Kc?
Kc = [FeSCN²⁺] / ([Fe³⁺][SCN⁻])
What does K tell you when it’s greater than one?
K>1 means products are favored
What does K tell you when it’s less than one?
K<1 means reactants are favored
Where are the iron thiocyanite equations thrown away?
Blue label waste container.
What’s the difference between the initial [SCN-] and [SCN-] consumed?
[SCN-] present at equillibrium
What’s the difference between the initial [Fe+3] and [Fe+3]
consumed?
[Iron III] present at equillibrium
What is plotted in Beer’s Law curve?
Absorbance vs. Concentration
Purpose of Part I?
Create a calibration curve to find K
Purpose of part II?
Determine equilibrium concentrations and calculate Kc
Procedure Part 1
1.Added 10.0mL of Iron III solution to 6 50mL volumetric flasks with a 10.0mL graduated cylinder
2.Pipetted 1.0-5.0mL of 0.0020M KSCN to flasks B-F
3.Added 0.1M HNO3 to each volumetric flask until 50.0mL, covered with parafilm, and mixed
4.Transferred solutions to cuvettes and measured absorbances at 447nm with the spectrophotometer
Procedure Part II
1.Added 5.0mL of 0.0020M Fe(NO3)3 to test tubes A-F
2.Added 1.0-5.0mL of 0.0020M KSCN to test tubes B-F in increasing order
3.Added HNO3 to test tubes A-E to add to 10.0mL, covered with parafilm and inverted to mix
4.Transferred solutions to cuvette and measured absorbances at 447nm with the spectrophotometer
What settings is the spectrophotometer?
447nm
What was the purpose of KSCN (the source of SCN-)?
In part 1, determines the known concentrations of FeSCN+2 and
In part 2, establishes the diff equillibrium mixtures to calculate Kc
What was the concentration of the Fe(NO3)3 stock solution for the calibration curve(Part 1)?
0.2M
What was the concentration of the KSCN stock solution?
0.002M for part 1 and 2
What was the concentration of the Fe(NO3)3 stock solution part two?
0.002M, the same as KSCN
Why does initial [SCN-] = [FeSCN²⁺]?
There’s more iron III so all of SCN- reacts completely
Use dilution to calculate initial [SCN-]/[FeSCN+2] for calibration curve
C1 = 0.002M KSCN V1 = Volume of KSCN added (mL)
C2 = X [SCN-] V2 = 50.0mL
![<p><span style="background-color: transparent; font-family: "Times New Roman", serif;">C<sub>1</sub> = 0.002M KSCN V<sub>1</sub> = Volume of KSCN added (mL)</span></p><p><span style="background-color: transparent; font-family: "Times New Roman", serif;">C<sub>2</sub> = X [SCN-] V<sub>2</sub> = 50.0mL</span></p>](https://assets.knowt.com/user-attachments/71a4a96b-dbb0-4612-b7b0-f28002e35394.png)
Calculating initial [Fe+3] for calibration curve
1.Molarity x Volume = Moles
2.Moles/Total Volume = Molarity
Molarity of Fe(NO3)3 Stock Part 1 (0.2M) x 10.0mL = molsFe(NO3)3
Fe(NO3)3 mols/ 50.0mL total volume solution = [Fe+3]
![<p>1.Molarity x Volume = Moles</p><p>2.Moles/Total Volume = Molarity</p><p></p><p>Molarity of Fe(NO<sub>3</sub>)<sub>3 </sub>Stock Part 1 (0.2M) x 10.0mL = molsFe(NO<sub>3</sub>)<sub>3</sub></p><p>Fe(NO<sub>3</sub>)<sub>3 </sub>mols/ 50.0mL total volume solution = [Fe<sup>+3</sup>]</p>](https://assets.knowt.com/user-attachments/60530f65-6242-4522-8ab1-7d718c1b4709.png)
What calculations are for part 1?
Equilibrium [FeSCN+2] = initial [SCN-], initial [Fe+3]
Why is the calculated initial [Fe+3] constant for each table?
We added the same amount of Fe+3 to every solution each part of the experiment, so it’s the same concentration
Using 0.0020MFe+3, 10.0mL in each flask part 1 and then 5.0mL each tube part 2.
What calculations are in part II?
initial [Fe+3]
initial [SCN-]
equillibrium[FeSCN+2]
then calculate [Fe+3] and [SCN-] at equillibrium with an ICE box
How to find [FeSCN+2] with the graph for part 2?
Absorbance/slope
Equation for ice box
[Fe+3] + [SCN-] → [FeSCN+2]
How to find Fe+3] and [SCN-] with icebox
We know:
[FeSCN+2] at equilibrium and all the initial concentrations
then add/subtract to find the rest of the concentrations at equilibrium
Use K = FeSCN+2]
[Fe+3] [SCN-]
Calculating initial [Fe+3] in part 2
[Fe+3] = (M stock x mL added/ Total Volume
0.0020M Fe(NO3)3 × 5.0mL/ 10.0mL = 0.0010M
Calculating initial [SCN-] in part 2
C1 = 0.002M KSCN
V1 = Volume of KSCN added(mL)
C2 = X [SCN-] we want to find
V2 = 10.0mL total volume
What does the spectrophotometer detect?
FeSCN+2 complex
Role of HNO3
To maintain an acidic environment for the reaction
Why keep total volume the same in each trial?
So concentration differences are only due to reactants, not dilution
Does K have units?
No