Chapter 7 Aromatic Compounds (Part 1)
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
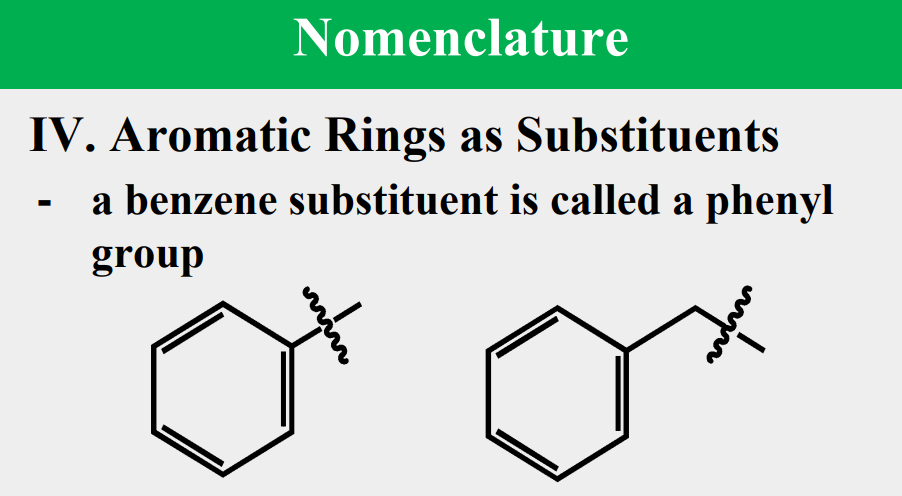
Benzene
_____________
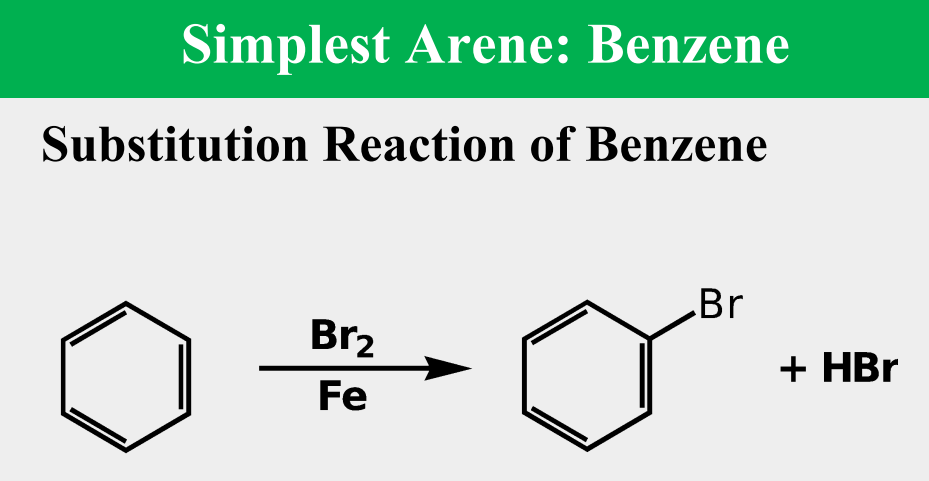
simplest aromatic hydrocarbon (arene)
undergoes substitution reactions and not addition reactions, which preserves the cyclic conjugation of the ring
Benzene
the carbon-carbon bonds have the same length (139 pm); intermediate between typical single and double bonds
single bond: 154 pm
double bond: 134 pm
Benzene
the electron density in all six carbon-carbon bonds is identical
Benzene
Simplest Arene
Benzene
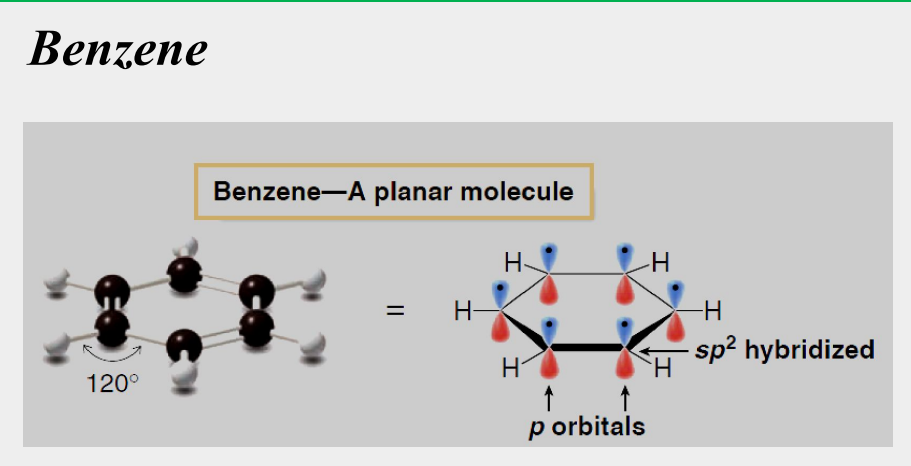
planar
all C-C-C bond angles are 120°
Benzene

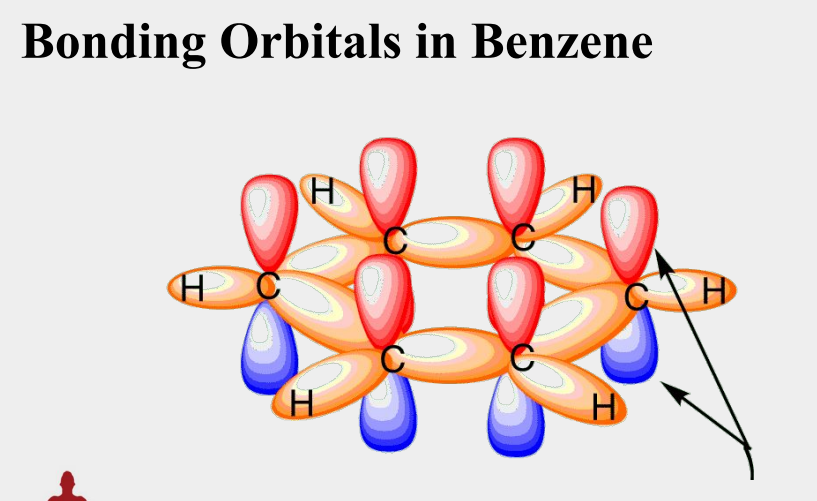
all six carbon atoms are sp2 hybridized
each carbon has a p orbital perpendicular to the plane of the ring
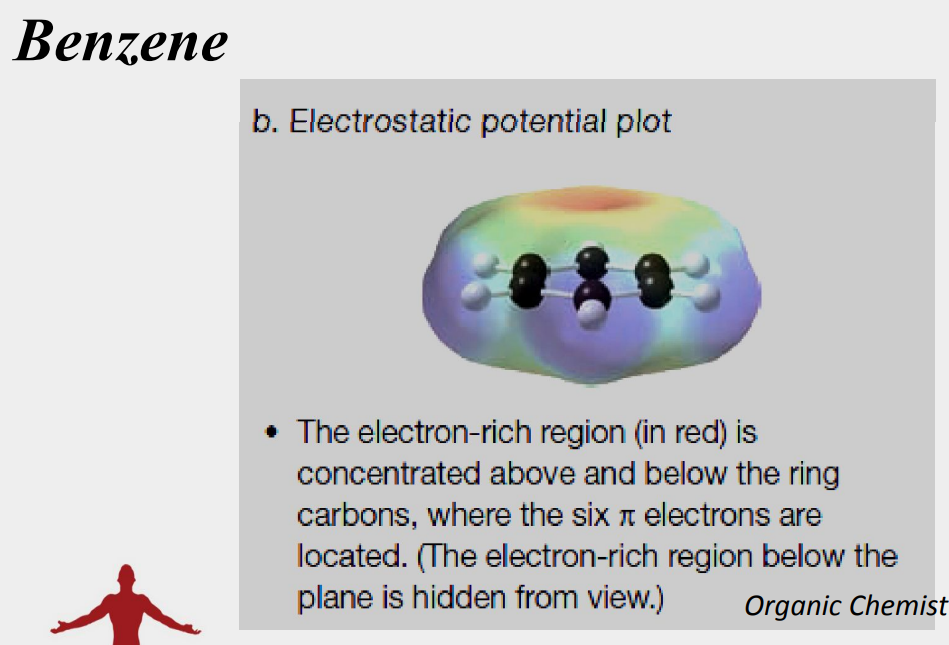
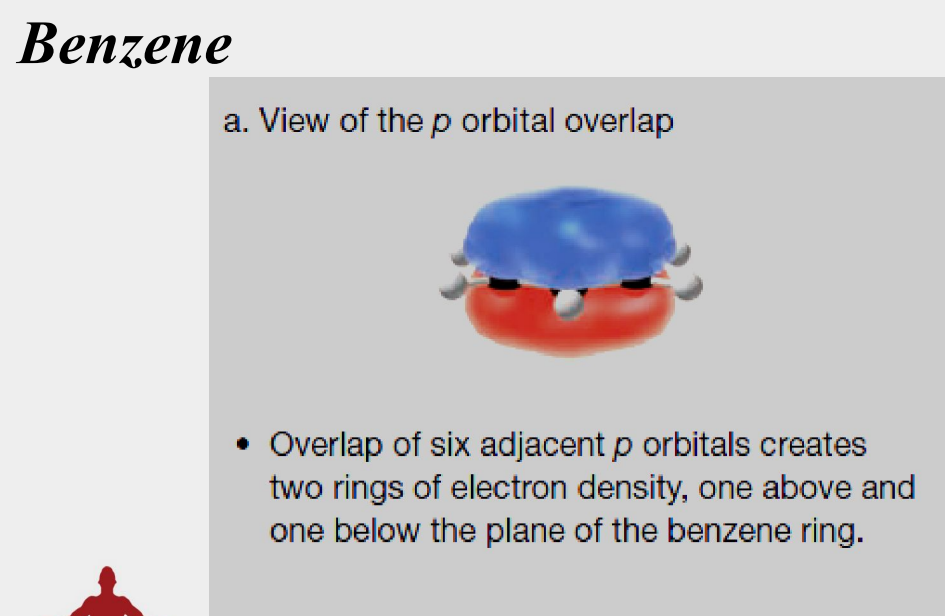
planar
conjugation (double bonds alternate in the ring)
4n+2 π
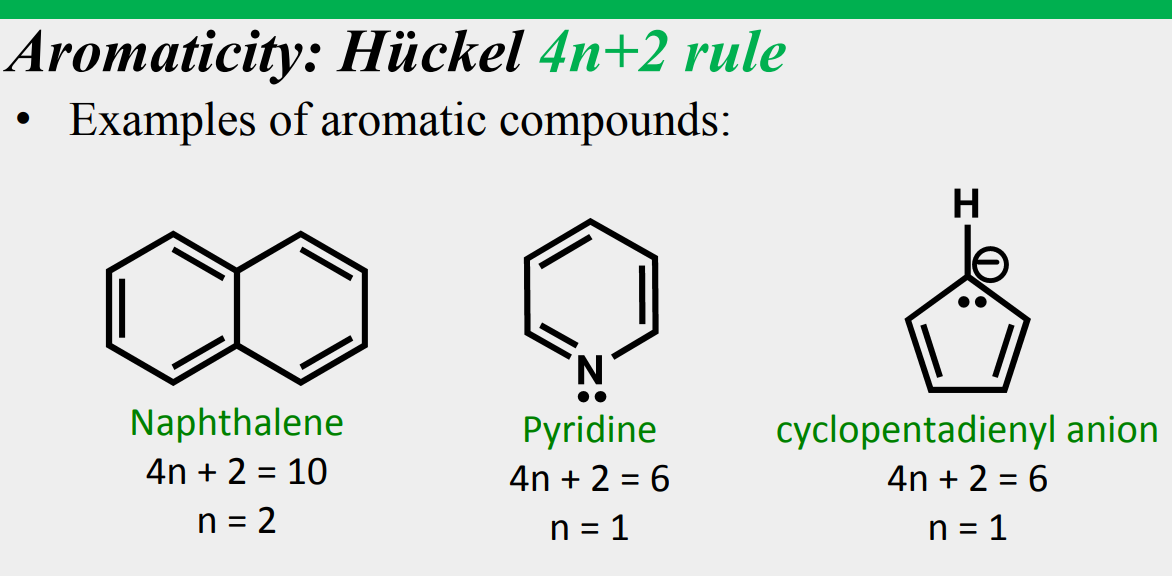
All are flat, conjugated, and n is an integer
Note: An integer is a whole number that does not include fractional or decimal parts, consisting of positive numbers, negative numbers, and zero (e.g., -5, 0, 10).
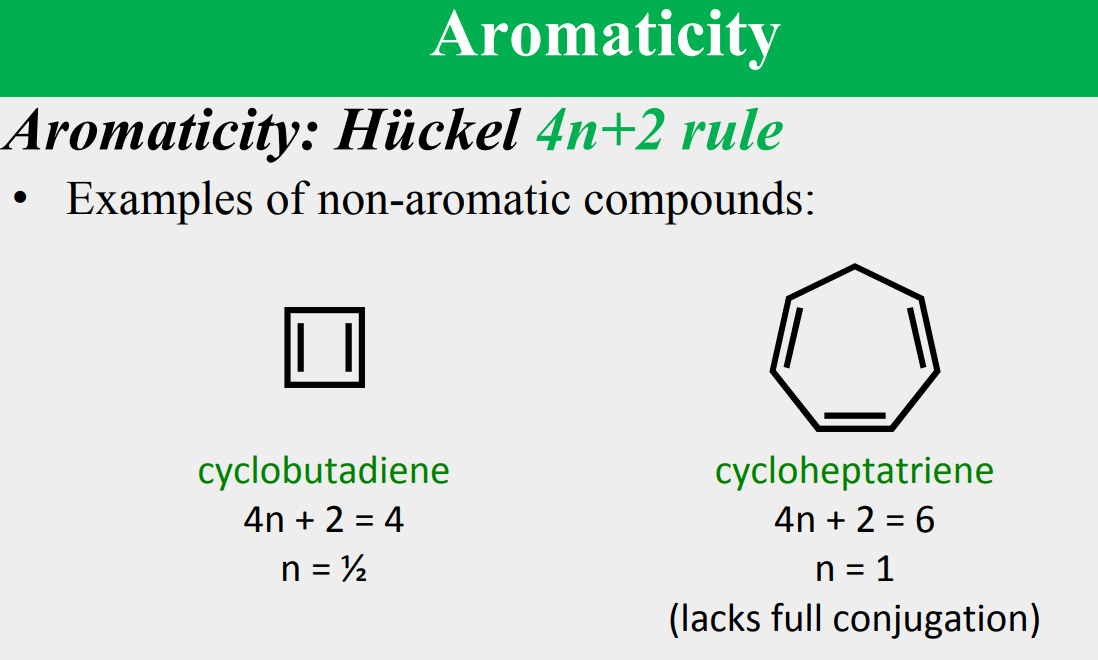
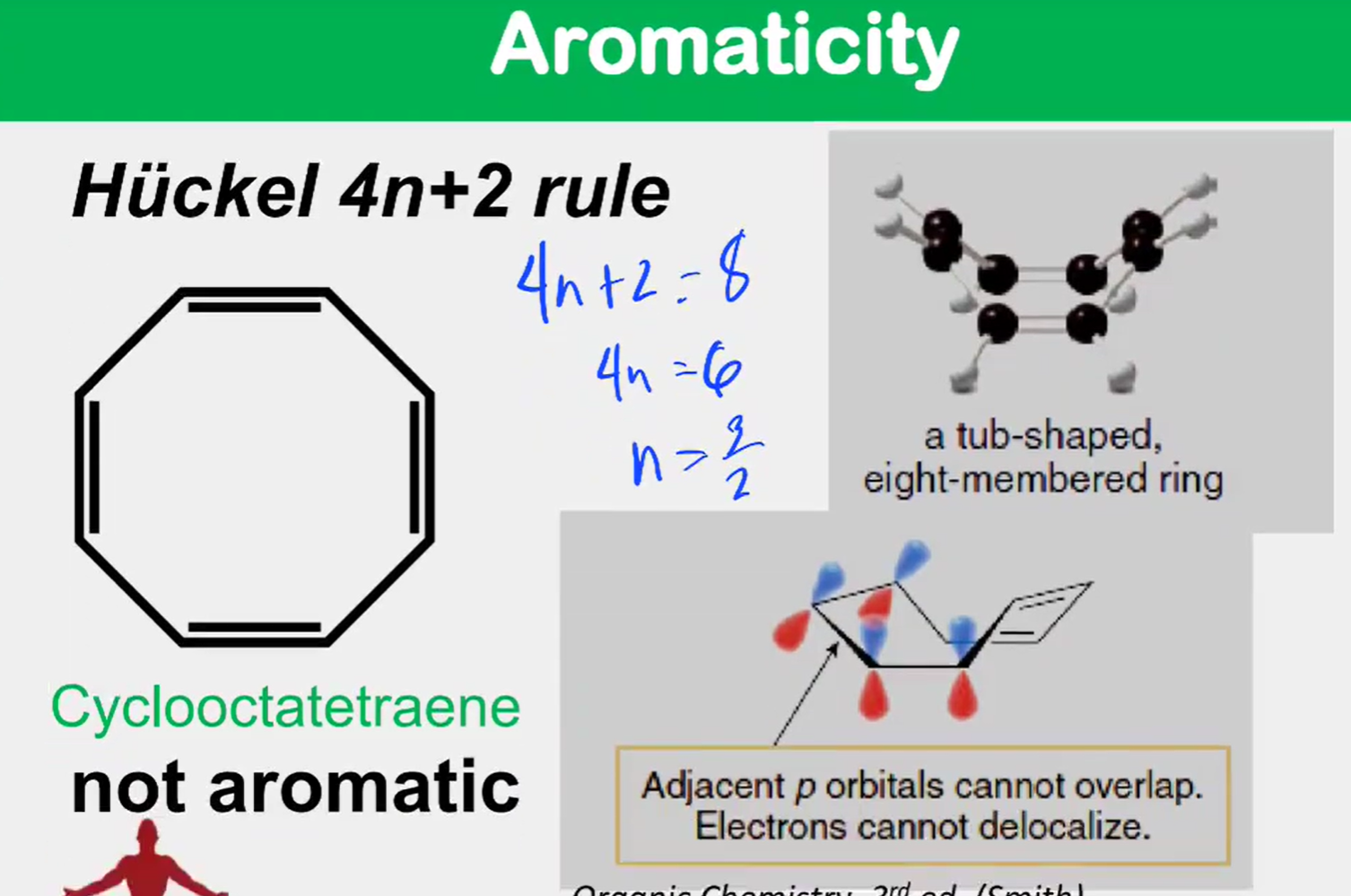
Aromaticity: Hückel 4n+2 rule
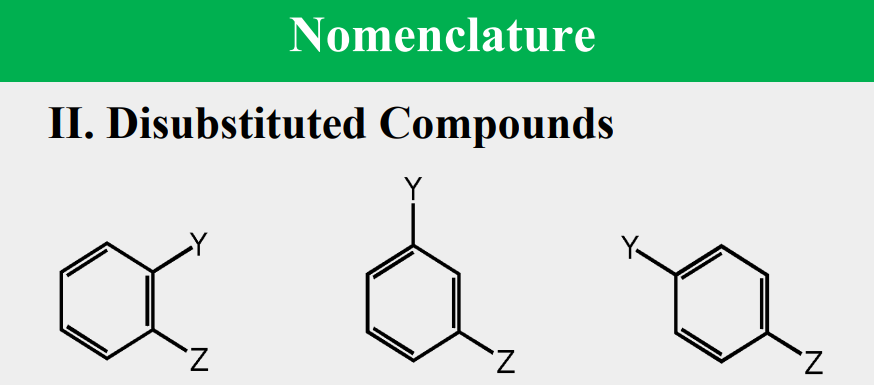
a molecule is aromatic only if it has a ____________, monocyclic system of _____________ with a total of ____________ electrons, where n is an integer
Are these aromatic and why or why not?
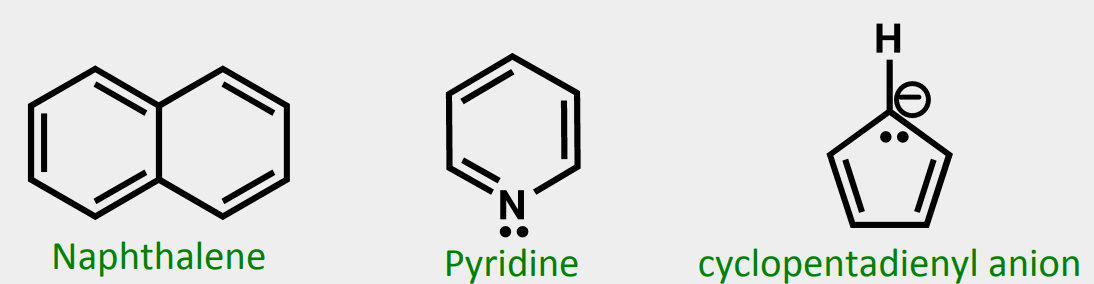
Are these aromatic and why or why not?

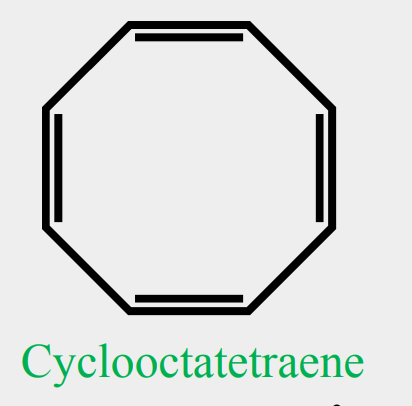
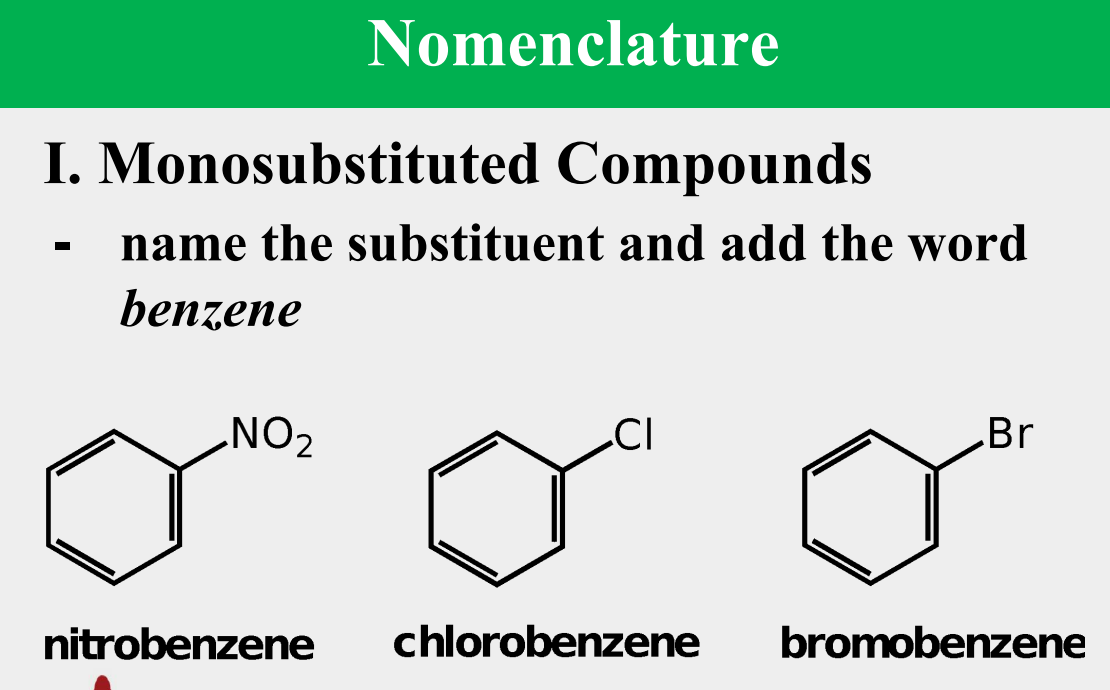
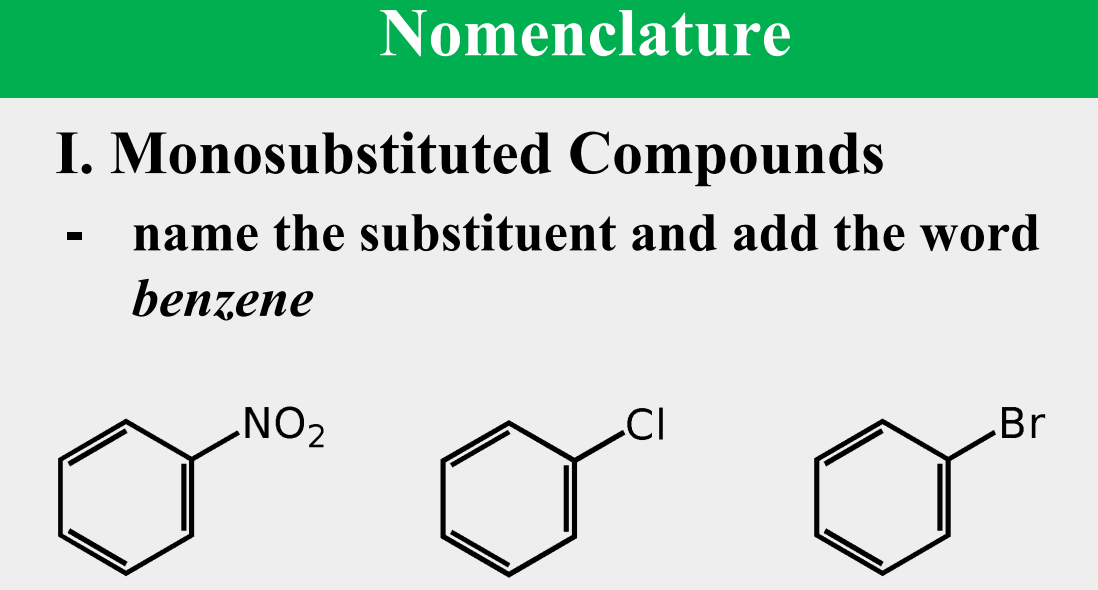
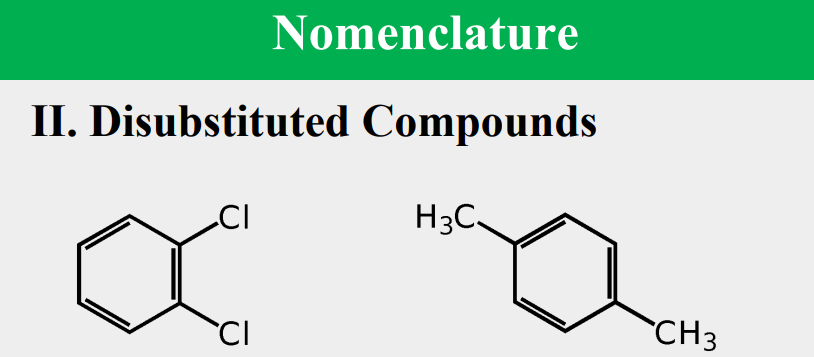
Provide the common name.
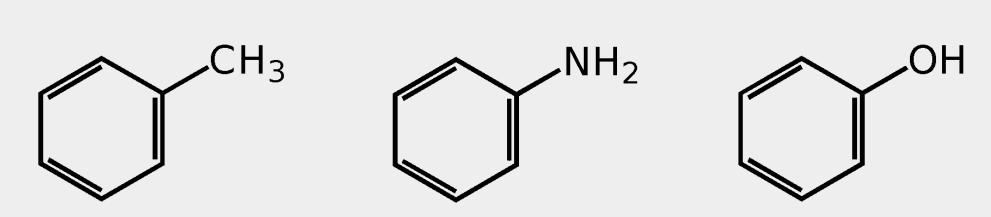
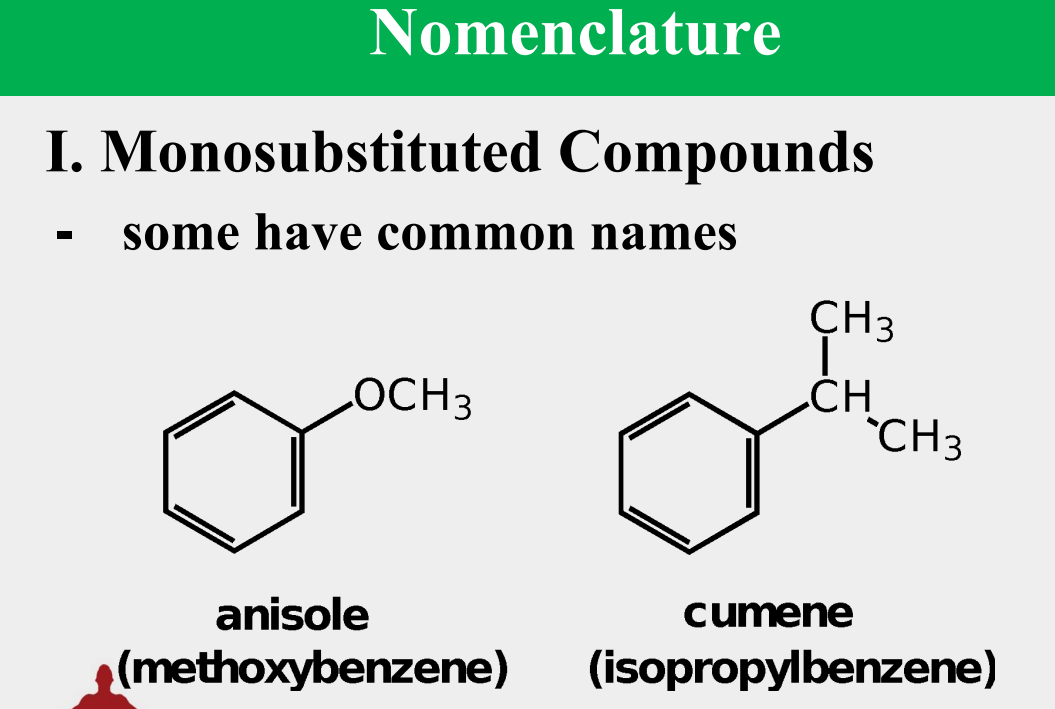
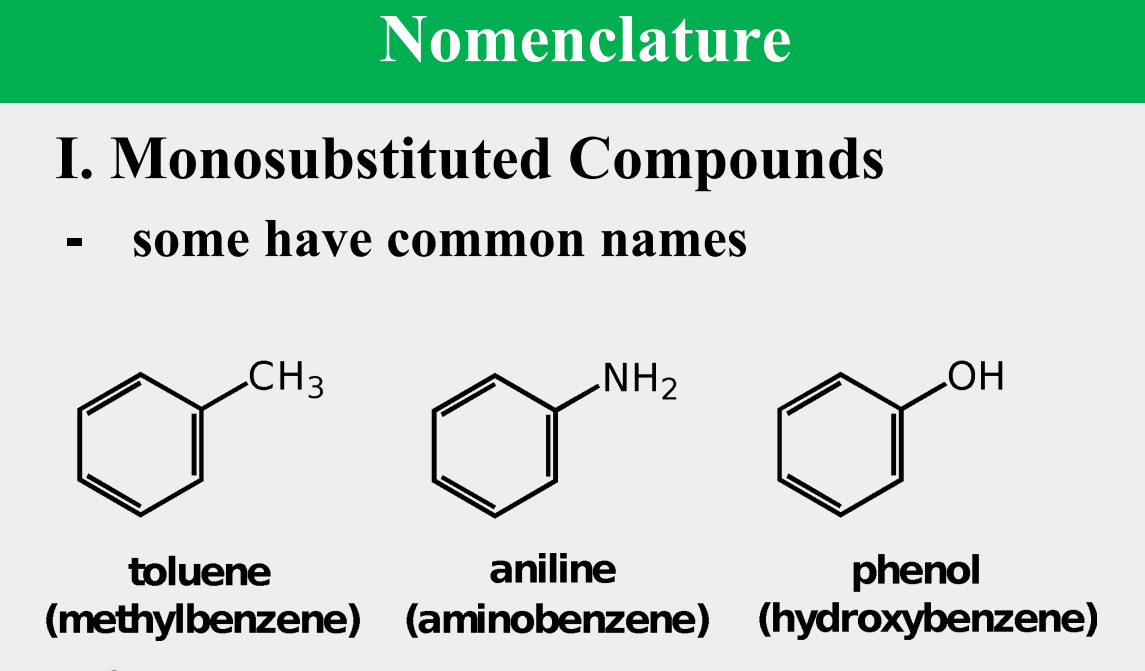
Provide the common name.
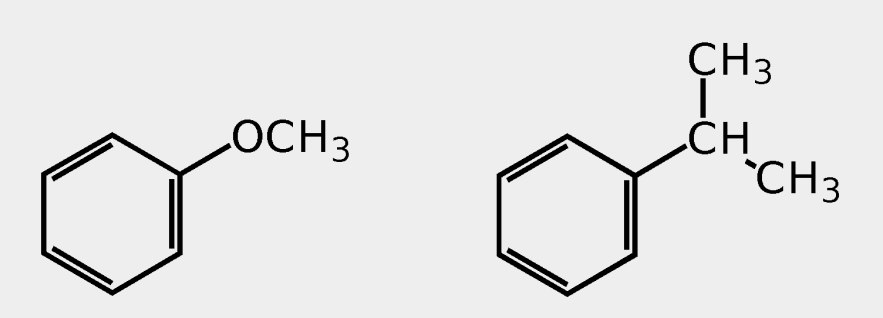
Provide the common name.
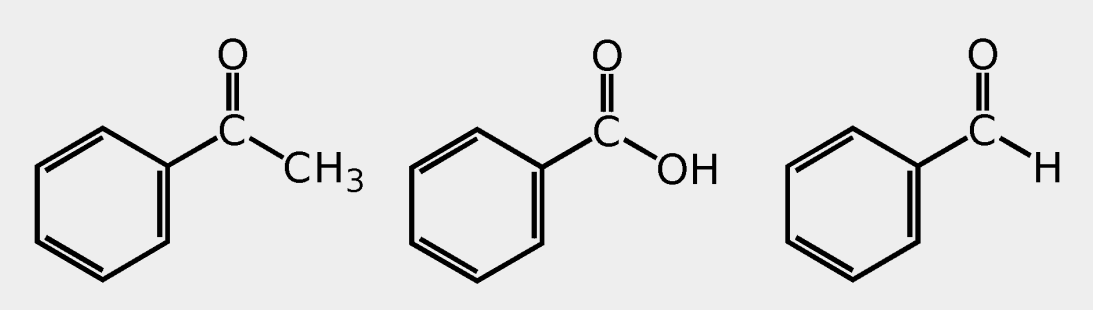
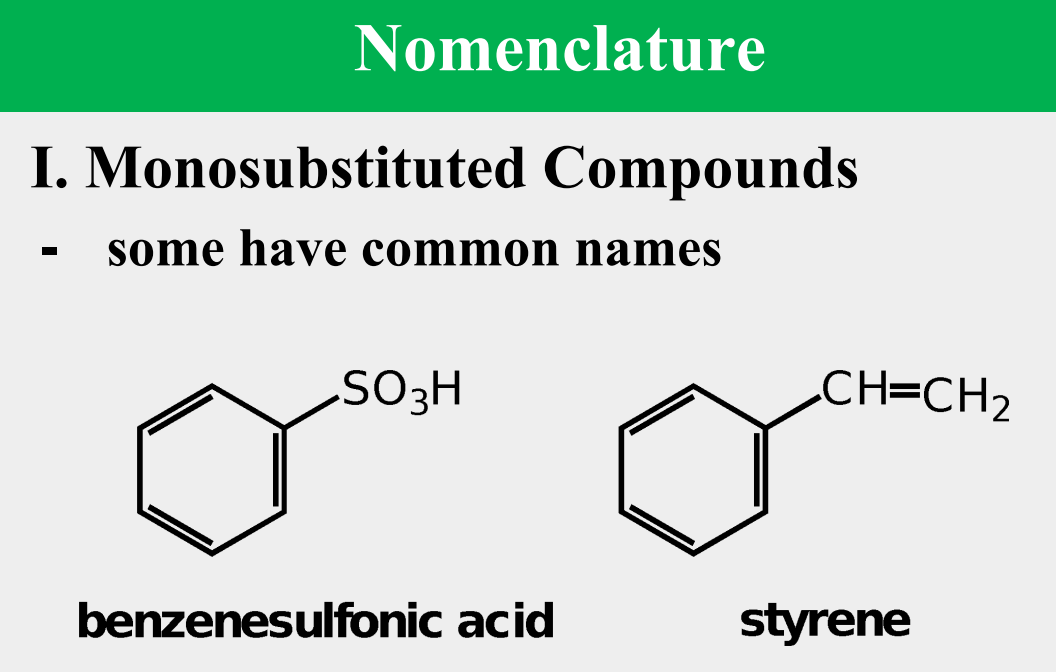
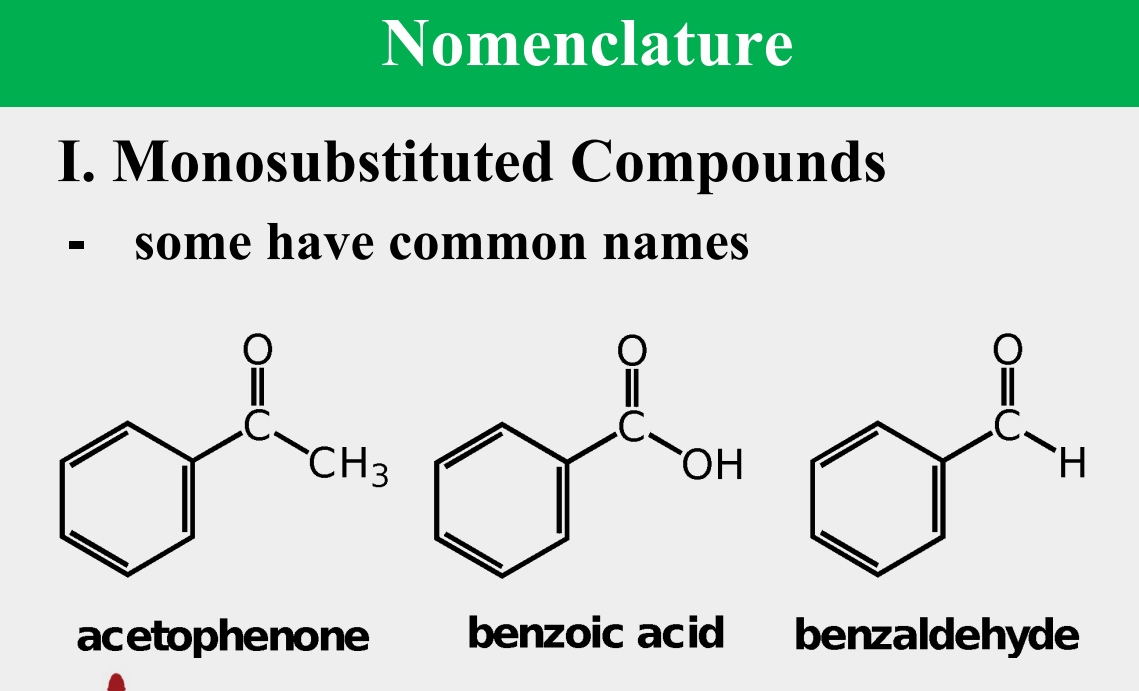
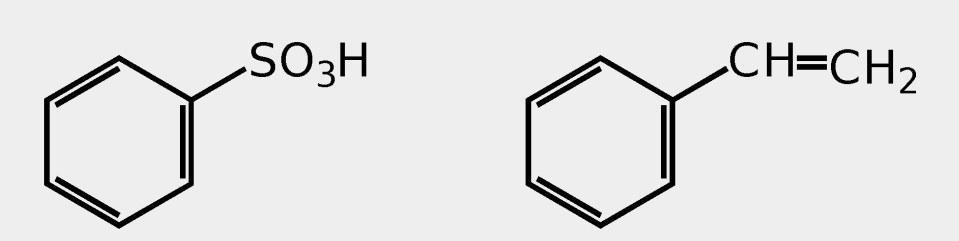
Provide the common name.
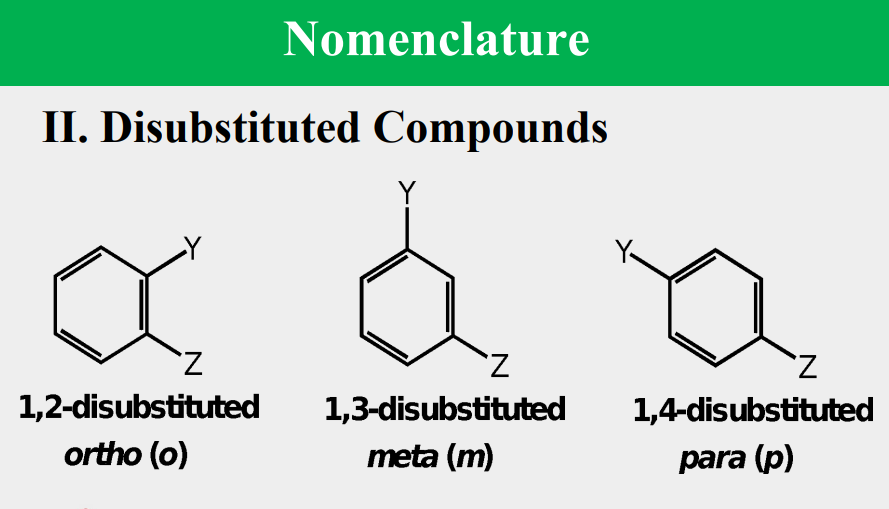
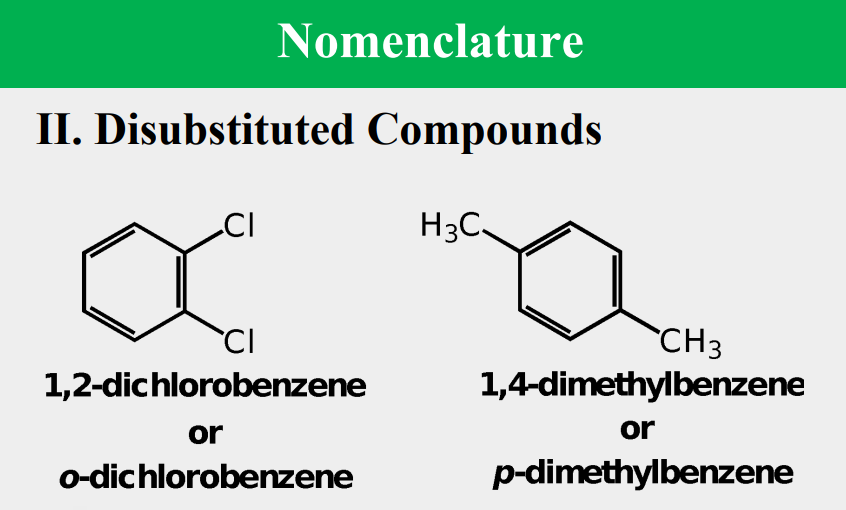

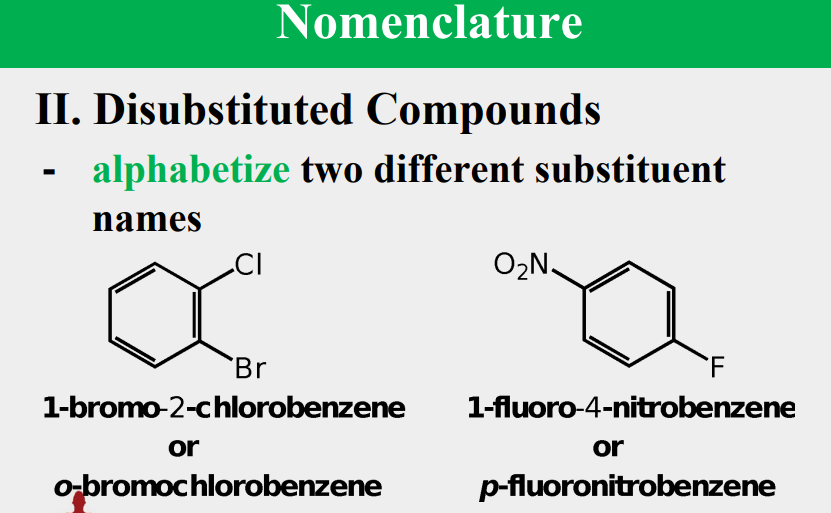
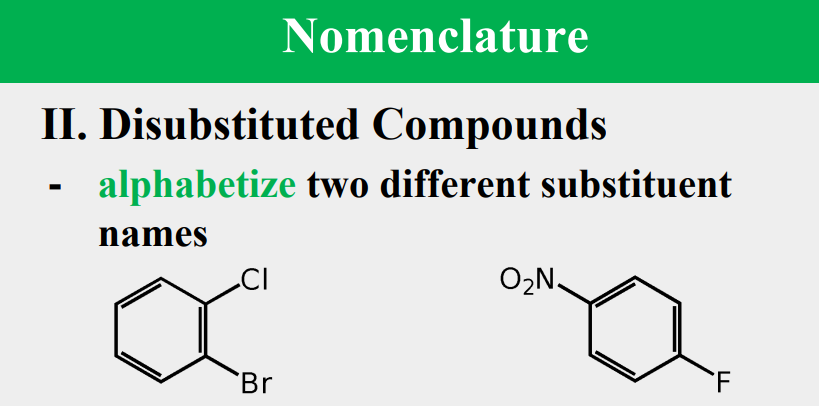
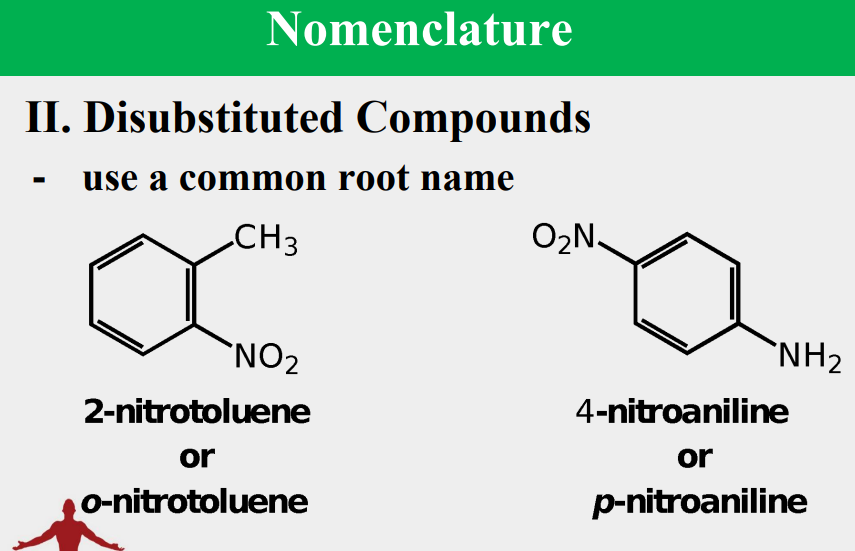
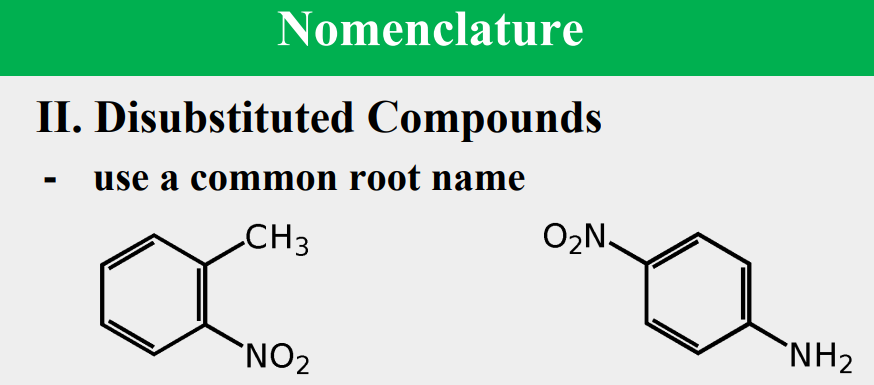
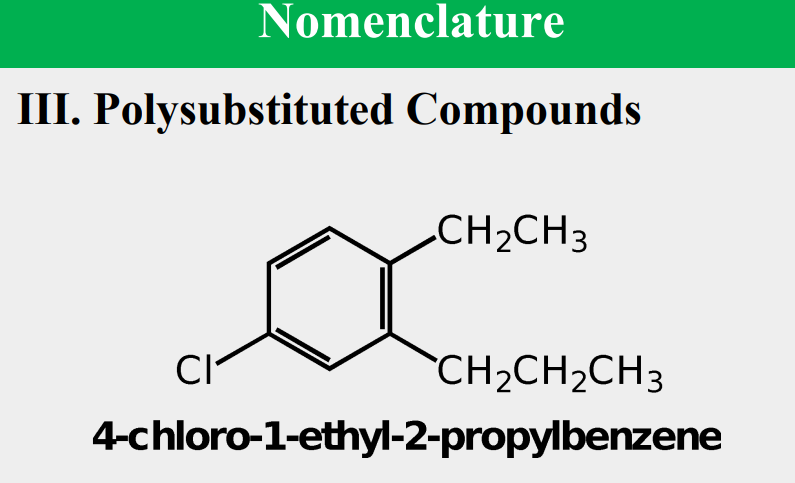

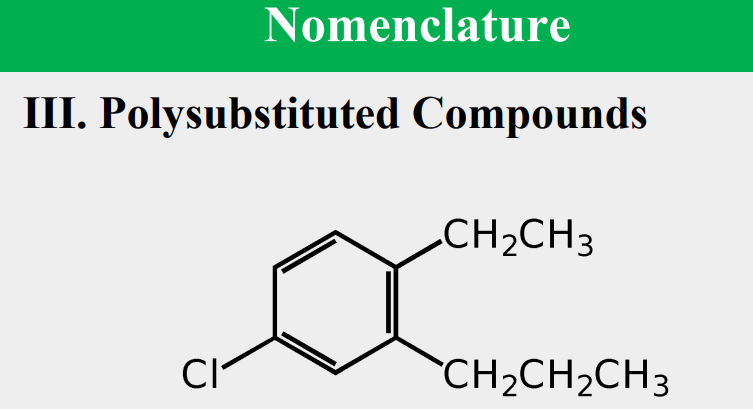
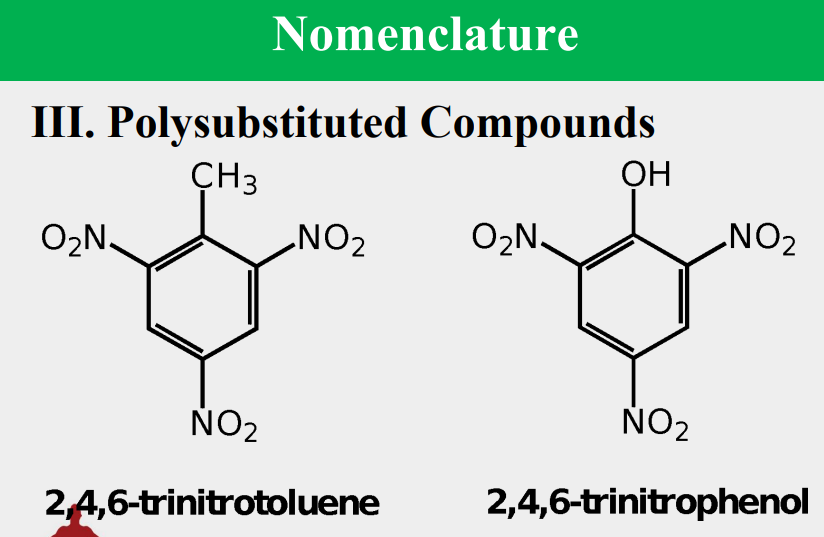

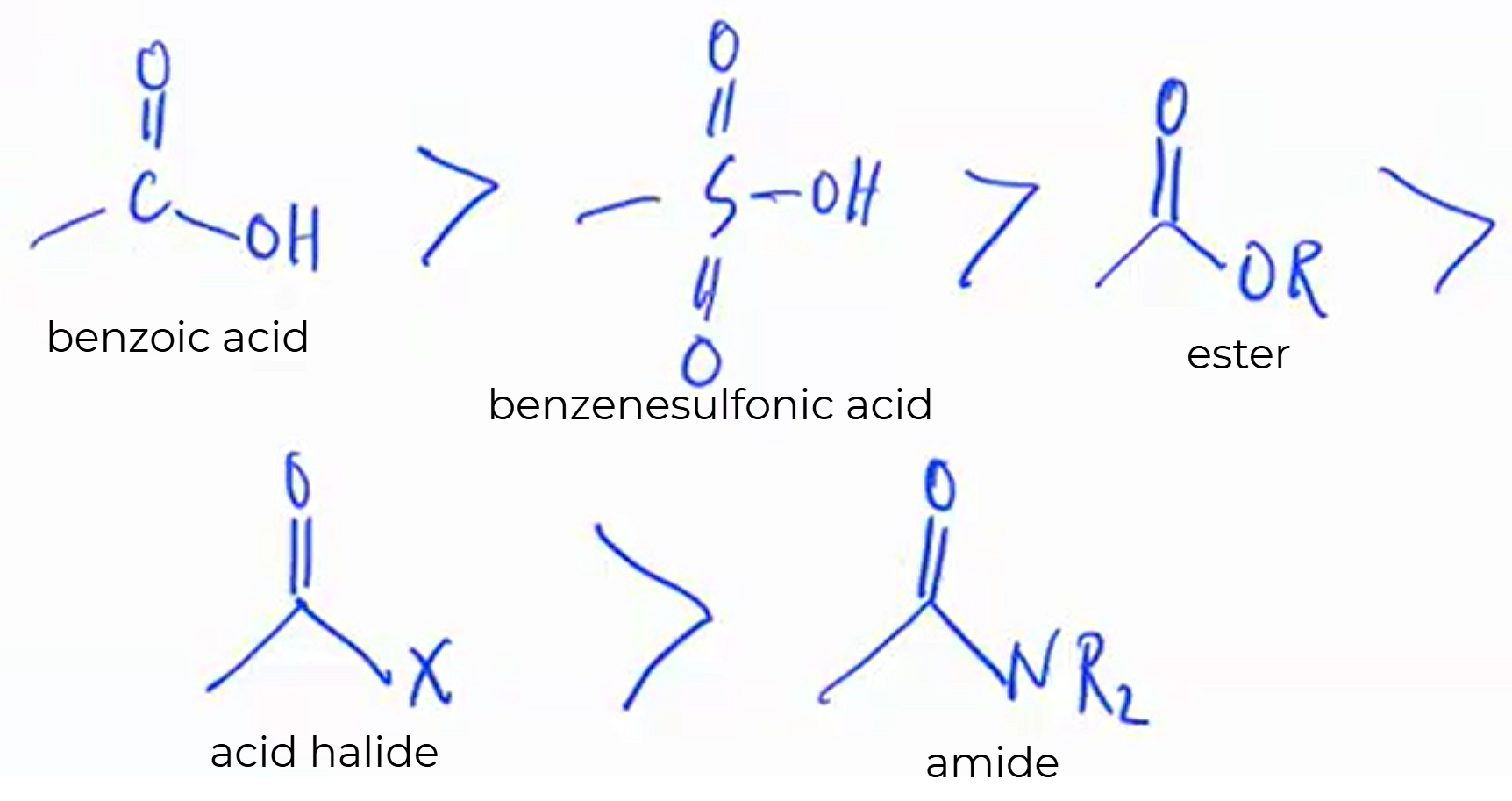
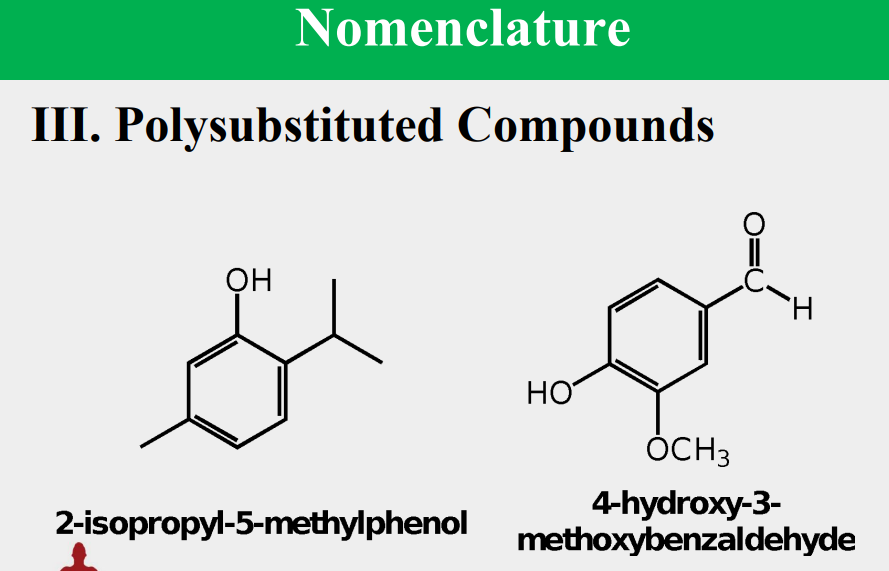
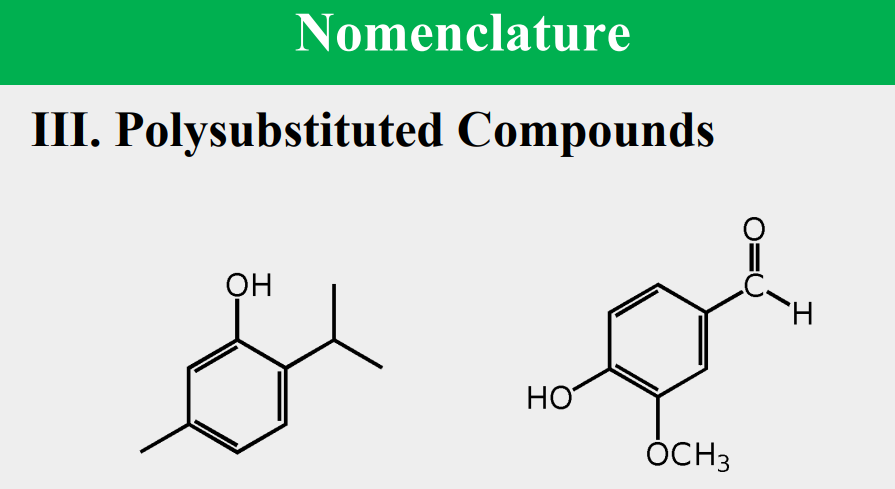
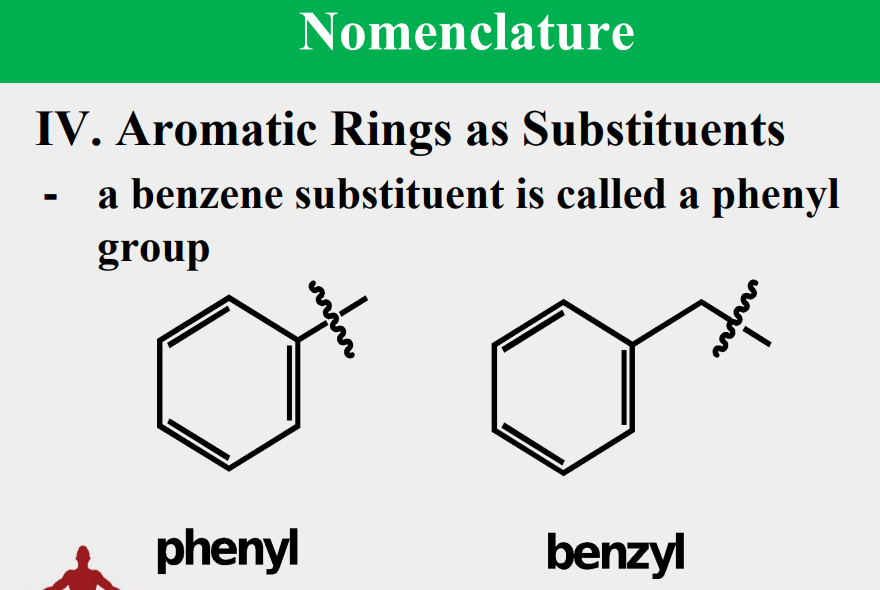
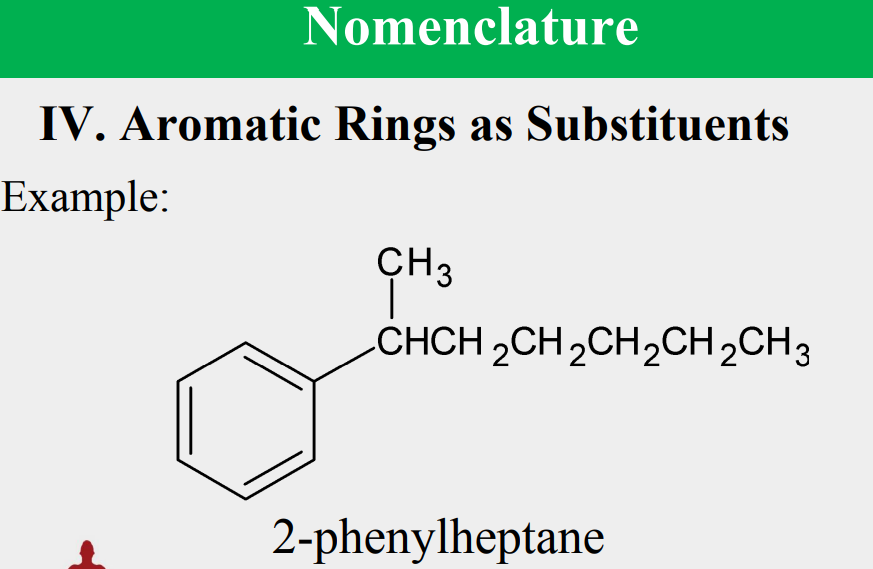
Given the following, rank them and draw the substituents:
benzoic acid
benzenesulfonic acid
ester
acid halide
amide
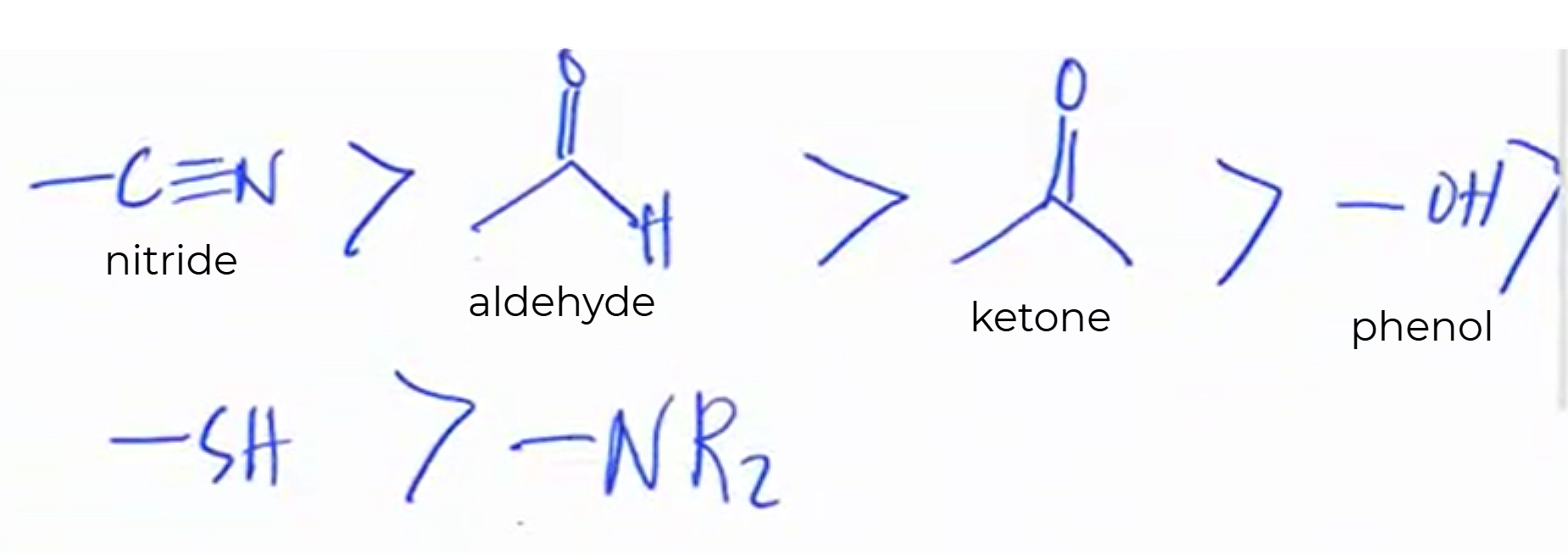
Given the following, rank them and draw the substituents:
nitride
aldehyde
ketone
phenol
SH
NR2