CHE 0200-Midterm 2 TopHat
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
Seven people stand on a scale and have a total weight of 1280 lb. A specific property called pep is defined as the number of people divided by mass. Pep is an intensive property.
A. True
B. False
True
A flowing open system cannot be at thermodynamic equilibrium; it can only be at steady state.
A. True
B. False
True
How many variables must be specified to fix the thermodynamic state of liquid water in equilibrium with its vapor?
A. 0
B. 1
C. 2
D. 3
B. 1
How many variables must be specified to fix the thermodynamic state of liquid water in equilibrium with a mixture of water vapor and nitrogen?
A. 0
B. 1
C. 2
D. 3
C. 2
If the ideal gas law holds for (diatomic) gases up to (RT ∕ P) > 5 L∕mol at room temperature (25℃), up to what pressure can you use the ideal gas law for diatomic gases?
A. 0.4 bar
B. 5 bar
C. 42 bar
D. 496 bar
B. 5 bar
An ideal gas is to be heated in a constant volume container at P = 1.5 bar and T = 21℃ until the pressure has doubled. What temperature will it attain?
A. 21 C
B. 42 C
C. 315 C
D. 588 C
C. 315 C
What is the reference state used for specific internal energy in the steam tables?
A. Water at the critical point (647.3 K, 220.48 bar)
B. Water at 25 C and 1 atm
C. Saturated liquid water at the triple point (273.16 K, 0.006113 bar)
D. Saturated vapor at 100 C and 1 atm
C. Saturated liquid water at the triple point (273.16 K, 0.006113 bar)
The molar translational kinetic energy of gas molecules____________.
A. 3RT/2
B. 2RT/3
C. RT/2
D. Dependent on the size of the molecule and the number of its translational degrees of freedom
A. 3RT/2
At the same temperature and for a pure species, the internal energy of a gas is _____ that of a solid.
A. less than
B. greater than
C. equal to
D. Incomparable to
B. greater than
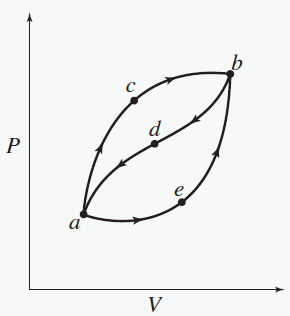
When a system is taken from state a to state b along path acb, 100 J of heat flows into the system and the system does 40 J of work. How much heat flows into the system along path aeb if the work done by the system is 20 J? Neglect kinetic and potential energy changes.
A. 40 J
B. 60 J
C. 80 j
D. 140 j
C. 80 J
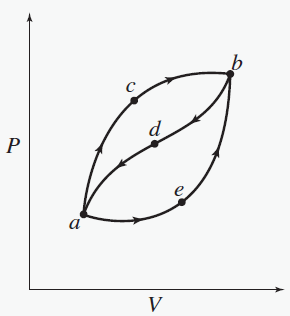
When a system is taken from state a to state b along path acb, 100 J of heat flows into the system and the system does 40 J of work. The system returns from b to a along path bda. If the work done on the system is 30 J, does the system absorb or liberate heat? How much? Neglect kinetic and potential energy changes.
A. absorbs heat, 30 J
B. absorbs heat, 70 J
C. liberates heat, 90 J
D. liberates heat, 130 J
C. liberates heat, 90 J
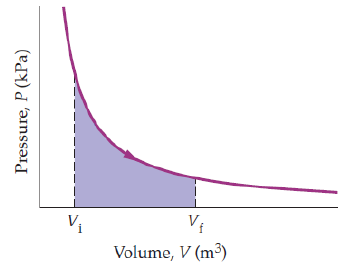
The process shown is an isothermal expansion of a gas on a P-V diagram. What does the area under the curve between Vi and Vf represent?.
A. The change in internal energy of the gas during the expansion
B. The flow work associated with gas entering and leaving a control volume
C. P ΔV work done by the gas during a reversible expansion
D. P ΔV work done on the gas during a reversible expansion
C. P ΔV work done by the gas during a reversible expansion
While the gas is expanded, a heat exchanger provides 7 J of heat to the system and 24 J of work is transferred. Assuming the gas behaves as an ideal gas, what happens to the temperature? Assume:
A. Temperature remains constant (isothermal)
B. Temperature increases
C. Temperature decreases
D. Cannot be determined without knowing the gas type
C. Temperature decreases
You find an old thermodynamics text with an equation ΔU = m C (T2−T1). When is this expression valid?
A. An isobaric process where C is temperature dependent
B. An isobaric process where C is temperature independent
C. An isochoric process where C is temperature dependent
D. An isochoric process where C is temperature independent
D. An isochoric process where C is temperature independent
Calculate the heat (J) required to raise the temperature of 1 mol of methane from 260 to 600 °C in a steady-flow process at a pressure sufficiently low that the ideal-gas state is a suitable approximation for methane.
19778.0 J
What follows from the definition of standard enthalpy of formation for a species composed of only one element in its naturally occurring (standard) form at 298 K and 1 bar?
A. It is always positive
B. It is always negative
C. It is zero
D. It depends on the element's molar mass
C. It is zero
What is the standard enthalpy of formation of liquid water, H2O(l), at 298 K and 1 bar (using NIST WebBook data)?
A. +286 kJ/mol
B. -286 kJ/mol
C. 0 kJ/mol
D. -572 kJ/mol
B. -286 kJ/mol
How would the adiabatic temperature change if excess air was used?
A. Increases
B. Decreases
C. Stays the same
D. It depends on the amount of excess air
B. Decreases
When applying the first law to a system where a chemical reaction occurs, what modification is required to the first-law equation itself?
A. Add an equilibrium term
B. Add a heat-of-reaction term
C. Nothing changes
D. Add a separate "chemical energy" term to account for bond energies
C. Nothing changes
An ideal gas expands from 2 bar to 1 bar adiabatically but irreversibly. Compared to an adiabatic, reversible process from 2 bar to 1 bar, the final temperature will be _____________ for the irreversible process.
A. higher
B. lower
C. the same
D. it depends on K
A. higher
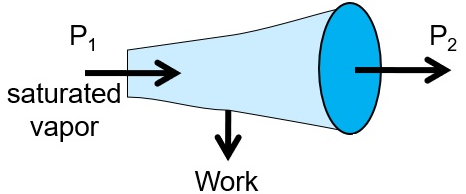
A saturated vapor is fed to a turbine: The exit from the turbine is ______________.
A. saturated vapor
B. superheated vapor
C. vapor-liquid mixture
D. any of the above
C. vapor-liquid mixture
Which statements are true for an ideal Carnot heat-engine cycle operating between a hot reservoir at TH and a cold reservoir at TC ? (Select all that apply.)
A. The Carnot cycle consists of four reversible processes: two isothermal and two reversible adiabatic (isentropic).
B. Among all heat engines operating between the same TH and TC, the Carnot cycle has the maximum possible thermal efficiency (no engine can exceed it).
C. Over one complete cycle, the net work output equals the net heat input.
D. A Carnot cycle can be executed only in an open system (a closed system cannot execute a Carnot cycle).
A. The Carnot cycle consists of four reversible processes: two isothermal and two reversible adiabatic (isentropic).
B. Among all heat engines operating between the same TH and TC, the Carnot cycle has the maximum possible thermal efficiency (no engine can exceed it).
C. Over one complete cycle, the net work output equals the net heat input.
What is the coefficient of performance for a Carnot refrigeration cycle that removes 30 kJ/s of heat from a cold reservoir and transfers 50 kJ/s of heat to a hot reservoir?
A. 0.5
B. 0.67
C. 1.5
D. None of the above
C. 1.5
A solid phase change takes place for iron at 75°C. The phase transformation from the a phase to the b phase is endothermic. The entropy of the iron _______.
A. increases
B. decreases
C. remains constant
D. depends on the history of treatment of the iron
A. increases
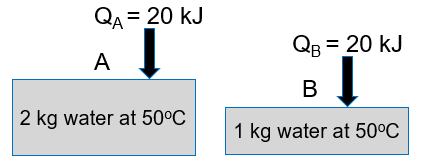
Container A has 2 kg of water, and container B has 1 kg of water. Both containers are initially at 50°C. To each container, 20 kJ of heat is added. Which container has the greater total (not per kg water) entropy change?
A. A
B. B
C. same entropy change for both
D. need more information
A. A
What is the change in entropy when 1 mole of saturated ethanol vapor condenses at its normal boiling point? hvap = 38.56 kJ/mol and Tb = 78.2 C
A. 0.11 kJ/(mol K)
B. - 0.11 kJ/(mol K)
C. 0.49 kJ/(mol K)
D. - 0.49 kJ/(mol K)
B. - 0.11 kJ/(mol K)
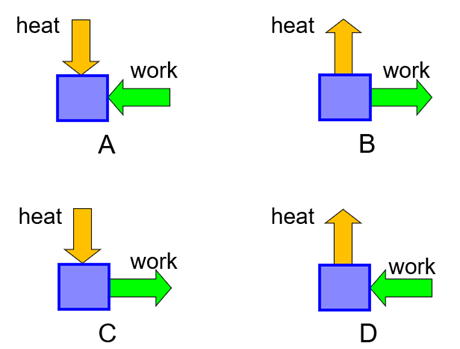
Which of these processes could run under continuous, steady-state operation?
A. A
B. B
C. C
D. D
D. D
An ideal gas is expanded isothermally from 0.5 mol/L to 0.09 mol/L in a reversible process. The entropy of the system:
A. increases
B. decreases
C. stays the same
D. more information is needed to answer
A. increases
Our analysis assumed the turbine was reversible. However, the actual expansion through the turbine produces -2.1 MW of power. What is the isentropic efficiency for the process? (Power =-2.81 MW)
A. 0.35
B. 0.70
C. 0.75
D. 1
C. 0.75
In an adiabatic heat exchanger, the amount of lost energy is ............., and there is ............... of exergy.
A. 0, gain
B. 0, loss
C. positive, gain
D. positive, loss
E. 0, no change
B. 0, loss
44 g CO2 are placed in a 0.23-liter container at 26 ℃ and 6 MPa. Should the ideal gas approximation be used?
A. Yes
B. No
B. No
For pure CH4, which of the following cases has the lower magnitude of the second virial coefficient B?
A. 300 K, 10 bar
B. 300 K, 20 bar
C. B is the same for both cases
D. Not enough information is provided
C. B is the same for both cases
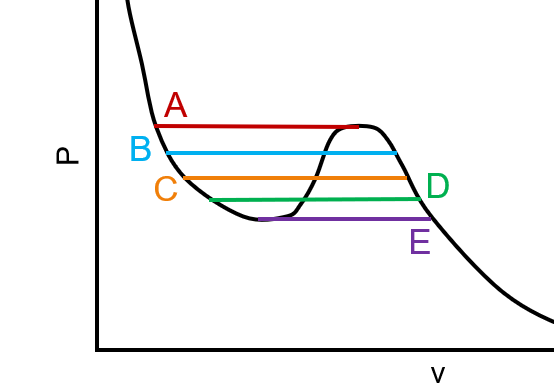
An isotherm for a cubic equation of state is shown below. Which line corresponds to a vapor-liquid equilibrium?
A. A
B. B
C. C
D. D
E. E
C. C
A hole is drilled into an aluminum plate. The plate is placed in a furnace and heated to a high temperature. If the hole is measured very accurately, the area of the hole
A. increases
B. decreases
C. stays the same
D. changes depending on the hole size
A. increases
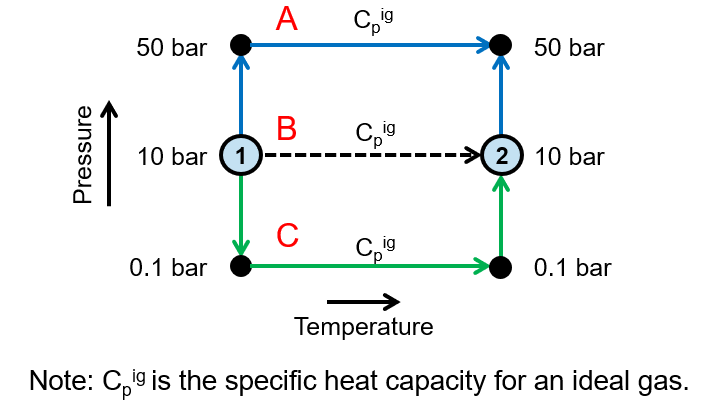
Which pathway from state 1 to state 2 will give the most accurate change in enthalpy?
A. Pathway A
B. Pathway B
C. Pathway C
D. Same for all pathways
C. Pathway C
Which of the following has the enthalpy departure function that is largest in magnitude?
A. Methane at 170℃ and 1 bar
B. Methane at 250℃ and 1 bar
C. Methane at 170℃ and 10 bar
D. Not enough information provided
C. Methane at 170℃ and 10 bar
Which of the following has the enthalpy departure function that is largest in magnitude?
A. Methane at 200℃ and 1 bar
B. Water at 200℃ and 1 bar
C. They have the same magnitude
D. Not enough information provided
B. Water at 200℃ and 1 bar
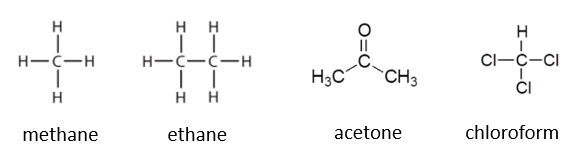
Which of the following has the entropy departure function that is largest in magnitude?
A. 50 mol% methane mixed with 50 mol% ethane
B. 50 mol% acetone mixed with 50 mol% chloroform
C. They have the same magnitude
D. Not enough information provided
B. 50 mol% acetone mixed with 50 mol% chloroform
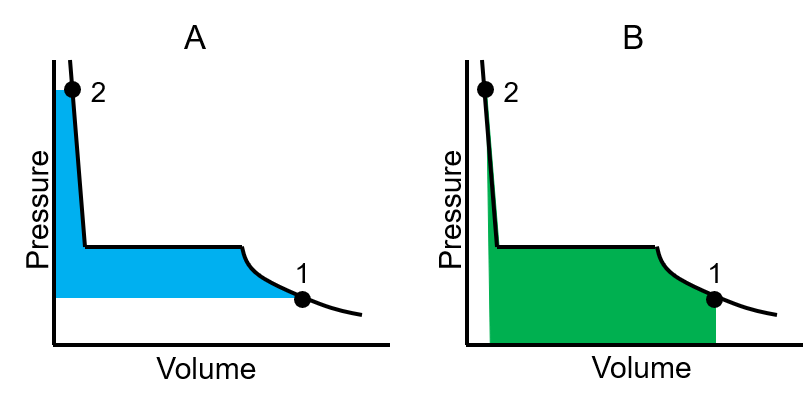
As the pressure increases from point 1 to 2, which figure shows an area proportional to the change in Gibbs free energy?
A. A
B. B
C. Both are correct
D. None of them
A. A
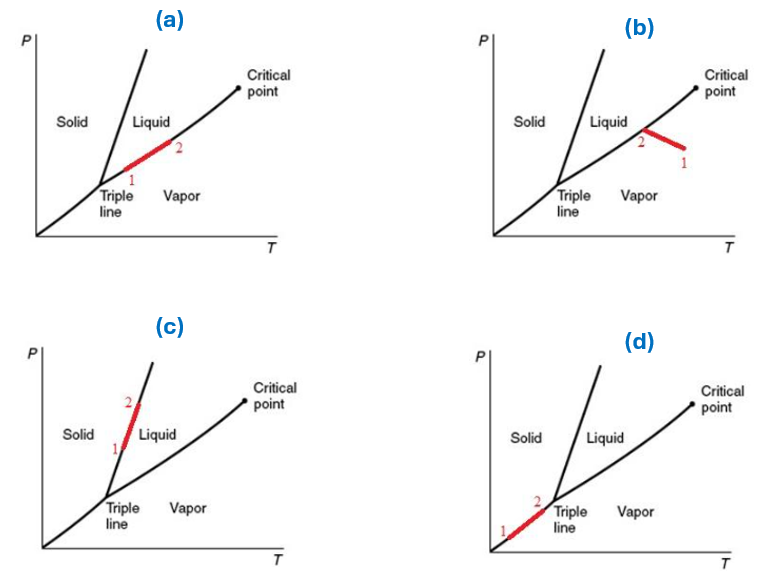
Several changes of state from state 1 to state 2 are shown on diagrams below. For which diagrams can the Clapeyron equation be applied?
A. Only (a)
B. Only (b)
C. All of them
D. All of them except (b)
E. Not enough information provided
D. All of them except (b)
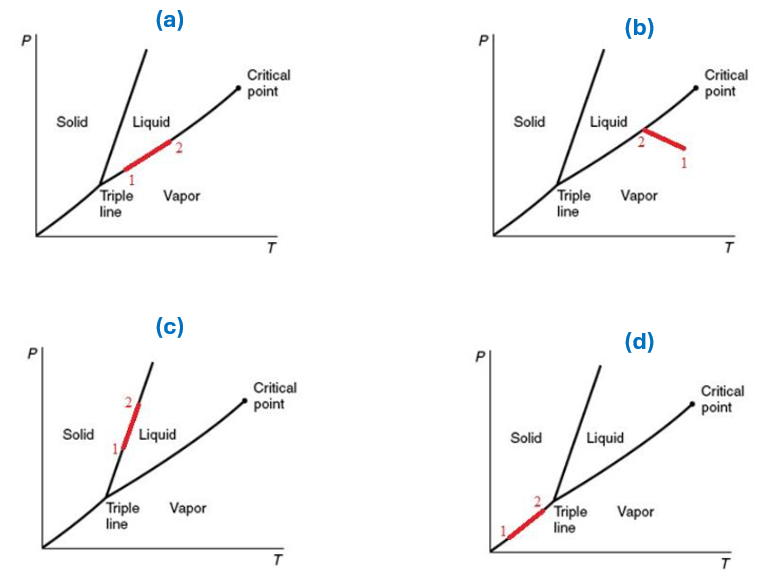
Several changes of state from state 1 to state 2 are shown on diagrams below. For which diagrams can the Clausius-Clapeyron equation be applied?
A. Only (a)
B. Only (b)
C. All of them
D. All of them except (b)
E. Not enough information provided
A. Only (a)