Chapter 3 AP Bio
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
What are enzymes, and what are some of their characteristics?
Enzymes are macromolecules
Biological catalysts that speed up biochemical reactions
Are proteins (made out of amino acids) — active site gets its properties from amino acids and enzymes are made out of proteins.
Maintained Tertiary shape
Have an active site
What does the active site interacts with, and what are some of their characteristics?
Active Site Interacts with the Substrate
Substrate = molecule that interacts with an enzyme by the active site to create active-substrate complex.
The Active Site has
Unique shape/size
Can have chemical changes or not
Physical/chemical properties of the substrate must be compatible with the active site
Slight changes (induced fit) can occur to align with the substrate.
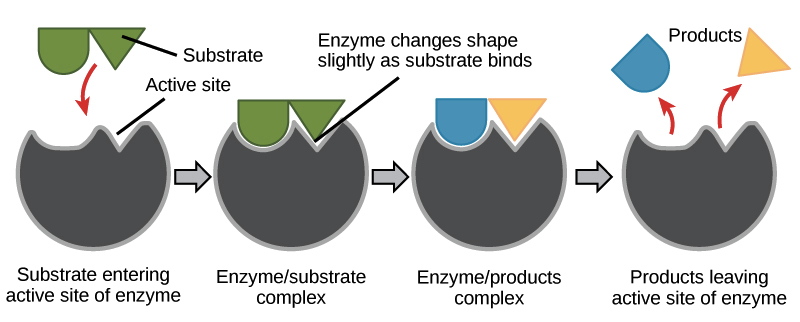
Enzymes are ____________ and can facilitate _______ or _______ reactions
Reusable
Not chemically change by the reaction
Cells maintain a specific enzyme concentration
Synthesis; digestion
Enzyme structure is…
Specific where each enzyme facilitates one type of reaction
What do enzymes affect?
Enzymes affect the rate of biological reactions
What do all biochemical reactions require? The enzyme has what no effect to what specific energy?
All biochemical reactions require activation energy or the initial starting energy
The enzyme does not have any effect on the free energy (Triangle G) as the reactants are converted to products
We can see that the amounts of energy that's required to do a reaction is greatly lowered when there is an enzyme present
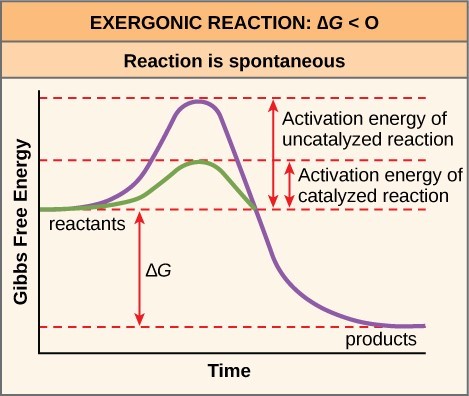
What do some biochemical reactions result in?
Some biochemical reactions result in net release or net absorption of energy
Reactions resulting in a net release of energy require less or more activation energy?
Reactions resulting in a net release of energy require less activation energy
How do enzymes accerlate the rate of reactions
Enzymes lower the activation energy requirement of all enzyme-meditated reactions, accerlating the rate of reactions.
What happens when there is a change in the molecular structure of an enzyme
Change to the molecular structure = Loss of enzyme function
Enzymes have unique functional 3D shapes
Changes in the shape is called denaturation, it is caused by
Changes in enviromental temperature
Changes in enviromental pH
Can Enzyme denaturation sometimes be irreversible or reversible?
Enzyme structures can sometimes be irreversible (catalytic ability of enzyme is loss) or reversible (regain catalytic ability)
Environmental temperature: What does optimum temperature mean?
Range in which enzyme-mediated reactions occur the fastest.
Reaction rates change when optimum temperature aren’t maintained
What happens when there is an increase in temperature?
Increase in temperature results an increase in reaction rates by speeding molecular movement and frequency of enzyme-substrate collisions
Also results in enzyme denaturation
What happens when there is a decrease in temperature?
A decrease in temperature slows down reaction rates by decreasing the frequency of enzyme-substrate collisions.
No denaturation occurs
What does optimum pH means?
Range in which enzyme-mediated reactions occur the fastest
For example, Pepsin works best with a lower pH (acidic)
Changes in pH outside the range will slow/stop enzyme activity
Enzyme denaturation can occur as a result
Small changes in pH values equate to large shifts in H-ion concentration
Changes in H concentration can disrupt H bonds that maintain enzyme structure
What happens when there is an initial increase in subtraste concetration?
An initial increase in substrate concentration results in an increase in reaction rate
More substrate = More chance to collide with enzyme
Substrate Saturation will occur
Results in no further increase in rate
Reaction rate will be constant if saturation levels are maintained.
What happens when there is an increase concentration of products?
An increased concetration of products decreases opportunity for addition of substrate
More products in an area = lower chance of enzyme-substrate collisons
Slows down reaction rates
How does changes in enzyme concentration impact reaction rate?
Less enzyme = slower reaction rate
Less opportunity for substrates to collide with active sites
More enzyme = faster reaction rate
More opportunity for substrates to collide with active sites
What are competitive inhibitors and what is the impact they have on reactions?
Competitive inhibitors are molecules that can reversibly or irreversiblely bind to the active site of an enzyme
Competes with normal substrate for enzyme’s active site
An increase in inhibitor concentration = Slow subtrate concentration reactions
A decrease in inhibitor concentration = Normal substrate concentration reaction
Regions than the active site which molecules can bind to in an enzyme is called
Allosteric Site
What are some key ideas about non-competitive inhibitors?
Binds to the allosteric site instead of the active site
Binding causes conformational shape change
Biding prevents enzyme function because the active site is no longer available
Reaction rate decreases
Increasing substrate cannot prevent effects of non-competitive inhibitor binding
What do all living things required?
All living things require energy
Sunlight is the main energy input
Autorophs capture energy from physical (sunlight) or chemical sources and transform them to usable energy source.
What happens to the energy during every energy transformation?
During every energy transformation process, some energy is lost as heat (unusable)
Life requires a ____________, and does not violate the ________________
Highly ordered system (energy), first and second law of thermodynamics
First law: Energy can never be created nor destroyed.
Second law: Energy transfer increases the disorder of the universe (entropy) and usually flows from hot to cold objects.
What are some of the key characteristics about energy transfers in organisms?
Every energy transfer increases the disorder of the universe (entropy)
Living cells are not at equilibrium; constant flow of in/out of materials
Cells manage energy resources by energy coupling
Energy Coupling: Process in a pathway that the released energy may be paired with another pathway or subsequent reaction that requires energy in order to reduce lost energy.
Explain the metabolic pathways of energy
Within a chemical pathway such as energy, the product of 1 reactant can serve as a reactant in a subsequent reaction.
This allows for a more efficient transfer of energy
How does these features support the concept of common ancestry
Certain metabolic pathways (e.g. glycolysis) are common among all living things (e.g. Bacteria, Archaea).
What is photosynthesis and its chemical equation?
Series of reactions in plants and plant-like organisms that uses CO2, H2O, and energy from light to produce O2 and carbohydrate that can be used or stored
6CO2 + 6H2O —- C6H12O6 + 6O2
Explain the evidence about photosynthetic prokaryotes
Photosynthesis evolved from prokaryotes (particularly cyanobacteria) and scientific evidence supports the claim that prokaryotic (cyanobacterial) photosynthesis was responsible for the production of an oxygenated atmosphere.
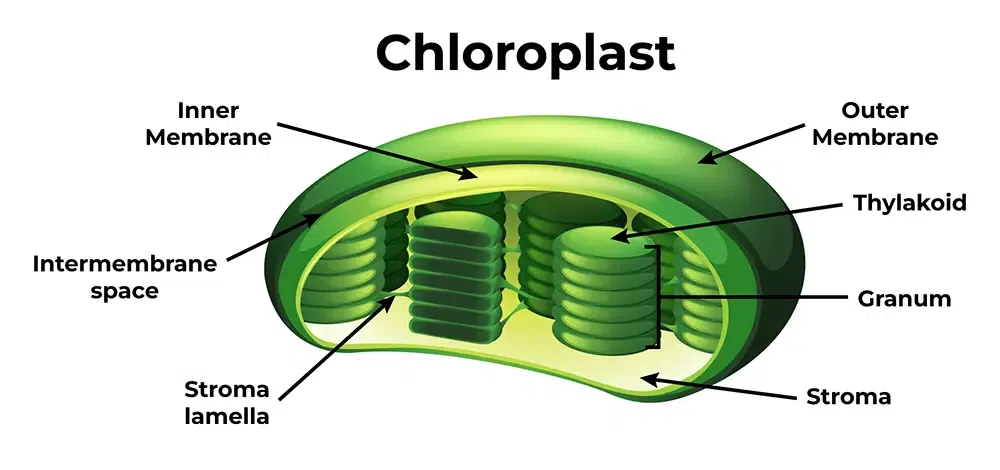
What are the 2 key structures of the chloroplast and determine their key roles in photosynthetic reactions
Stroma and thylakoids are found within the chloroplast:
Stroma is the fluid within the inner chloroplast membrane and outside the thylakoid
Carbon-fixation cycle (Calvin Cycle) occur in stroma
Thylakoids are organized into stacks called grana
Light dependent reactions occur in the grana
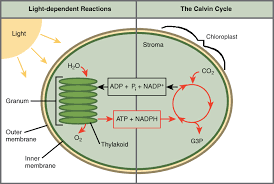
Light dependent reactions (In thylakoids)
Metabolic pathways that capture light energy through chlorophyll pigments to produce ATP (cellular energy) and NADPH (electron carrier/”reducing agent”)

Explain the steps of Light Reactions
1) Chlorophyll in Photosystem ll on the thylakoid membrane absorbs light energy through pigments, which “excites” the electron to a high-energy state.
Note: Energy from electrons will be used to establish a proton gradient and to reduce NADP+ to NADPH.
2) Electrons are replaced by water molecules which are split to form H+ ions and O2 (hydrolysis)
Note: The hydrogens from the splitting are released into the thylakoid space which creates a proton gradient
3) An electron transport chain begins, where the excited electron is passed along proteins in the thylakoid membrane, pumping H+ ions into the thylakoid.
4) The H+ (proton) concentration gradient between the thylakoid and stroma is used by ATP synthase to phosphorylate ADP + P into ATP via chemiosis
Note: ATP synthase is permeable to H+ ions and while H+ ions pass through, it spins while making ATP.
Uses Passive Transport
5) Photosystem l re-excites the electron by absorbing another photon of light
6) The electron is passed to NADP reductase, which adds a H+ ion to produce NADPH
Energy from ATP and reducing properties of NADPH powers production of carbohydrates in the Calvin cycle.
Photosystem ll and l are located where and what are they connected by?
Photosystem l and ll are embedded in the thylakoid membrane of chloroplasts
Connected by the transfer of electrons through an ETC.
What is the difference between oxidation and reduction reactions and how does it apply to photosynthesis?
Reduction reactions mean gaining electrons while oxidation reactions mean loosing electrons
LEO the lion goes GER
Carbon dioxide is being reduced (gaining electrons) to glucose. Happens in Calvin cycle stages.
Water is being oxidized (losing electrons) to oxygen. Happens in light independent reactions

Calvin-Cycle (In Stroma)
The energy captured in the light reactions and transferred to ATP and NADPH powers the production of carbohydrates from carbon dioxide in the Calvin cycle.
In basic terms: Uses products from lights reactions (ATP, NADPH, and CO2) to produce carbohydrates (glucose).
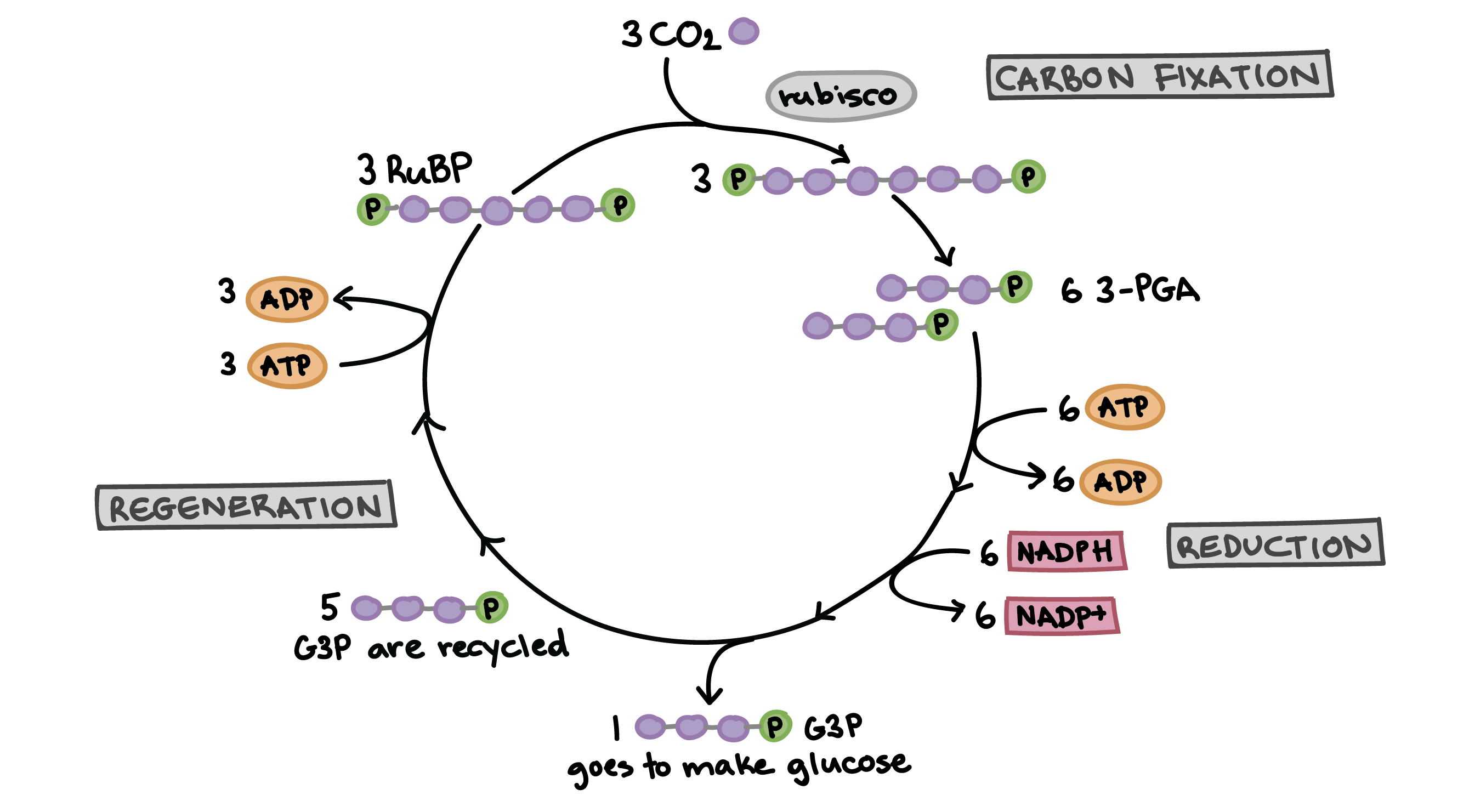
Carbon fixation steps explained in simpler terms
Fixation: CO2 is “fixed” into RuBP by Rubicose (enzyme) and makes 3-PG
Reduction: ATP and NADPH are used to reduced 3-PG to G3P. Some G3P is used to make sugars
Regeneration: G3P is used to regenerate RuBP to continue the cycle
Cellular Respiration
Process by which macromoleclues (namely carbonhydrates) are used to synthesize ATP using enzyme-meditated reactions
In aerobic respiration, Glycolysis and Krebs Cycle main objective is to produce NADH and FADH2, which are ¨reducing agents” ( a moleclue that reduces another moleclue by transferring an electron)
Glycolysis
Metabolic Pathway that produces 2 ATP and 2 NADH by converting glucose into pyruvate
Occurs in the cytosol of the mitochondria

However, to make ATP without oxygen (anerobic)
Fermentation can occur where production of ATP without oxygen, relying upon glycolysis, converting pyruvate into ethanol or lactic acid.
Krebs Cycle (Critic Acid Cycle)
Cycle of Reactions where pyruvate is used to reduce NAD+ to NADH and FAD to FADH2, while releasing CO2
Occurs in mitochondrial matrix
Electron Transport Chain
Establishes a proton gradient that ATP synthase uses to phosphorylate ADP + Pi into ATP in large amounts (oxidative phosphorylation)
NADH and FADH2 transfer…
NADH and FADH2 transfer electrons (oxidized) to the proteins on the inner mitochondrial membrane
While electrons go from protein to protein, they…
While electrons go from protein to protein, they pump protons to the inner mitochondrial membrane, establishing a gradient
The gradient is then used to by ATP synthase to phosphorylate ADP + Pi through chemiosis (oxidative phosphorylation)
What are the electrons accepted by?
The elctrons are accepted by oxygen, which binds with H+ ions to produce water moleclues.
Keeps the entire ETC chain running smoothly to produce ATP.