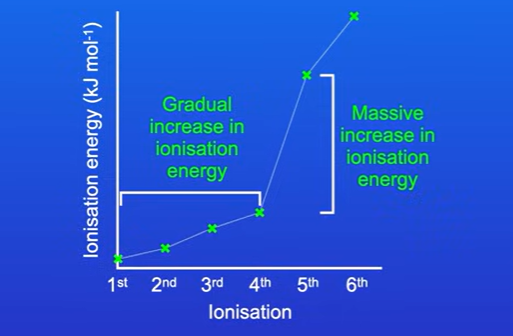
Successive ionisation energies
1/5
Earn XP
Description and Tags
https://www.youtube.com/watch?v=eBLJLD2D5ao
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
Key idea about electrons
Electrons in an atom are attracted to the positive protons in the nucleus, the greater the attraction between the outer electrons and the nucleus, the greater the ionisation energy
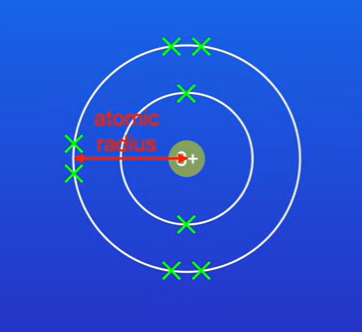
What is 1st factor affecting ionisation energies.
The distance between the nucleus and the outermost electrons. (atomic radius).
As atomic radius increases, the force of attraction between the positive nucleus and the outer electrons decreases.
2nd factor affecting ionisation energy
The charge on the nucleus.
Electrons are attracted to positively charged protons in nucleus, greater number of protons means there is a greater force of attraction between the outer electrons and the nucleus
3rd factor affecting ionisation energy.
Shielding.
Electrons in outer shell are repelled by electrons in inner shells, This shielding effect reduces the attraction between the outer electrons and the nucleus.
What can we see about the successive ionisation energy graph of oxygen,
We see a gradual increase in ionisation energy as we remove fiirst six electrons, this is because each time we remove an outer electron, the remaining electrons in the outer shell are pulled slightly closer to the nucleus meaning there is greater attraction between the outer electrons and the nucleus, causes ionisation energy to gradually increase.
Also massive increase in ionisation energy as we remove seventh electron, this can be explained by looking at electrons in oxygen atom, the first six electrons are all found in the second electron shell, once these electrons are removed, the seventh electron removed from first electron shell. Compared to the second electron shell, the first electron shell is closer to nucleus, and electrons in first shell experience much less shielding, so greater attraction to nucleus.
How do we identify elements using ionisation energy data.
First six ionisation energies for element in period 3 shown.
First must work out number of electrons in outer shell, we can see ionisation energy gradually increases upto 4th ionisation, and massive increase in ionisation 5. Tells us that theres 4 electrons in outer shell.5th electron must have been removed from internal shell.
Element must be in group 4. element in period 3 group 4 is sillicon.