orgo midterm 3 reactions
1/80
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
81 Terms
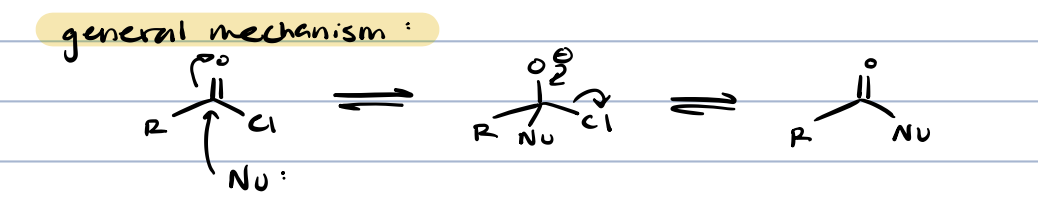
general mechanism for acid chloride reactions
acid chloride + HOR
esters
acid chloride + NH3
primary amide
acid chloride + NH2R
secondary amide
acid chloride + NHR2
tertiary amide
acid chloride + N3-
acyl azides
acid chloride + CN
acyl nitriles
acid chloride to anhydride
R'COOH/R'COO-
can acid chlorides do friedel crafts
yes only with aromatic compounds
acid chloride + metal hydride
primary alcohol with intermediate aldehyde
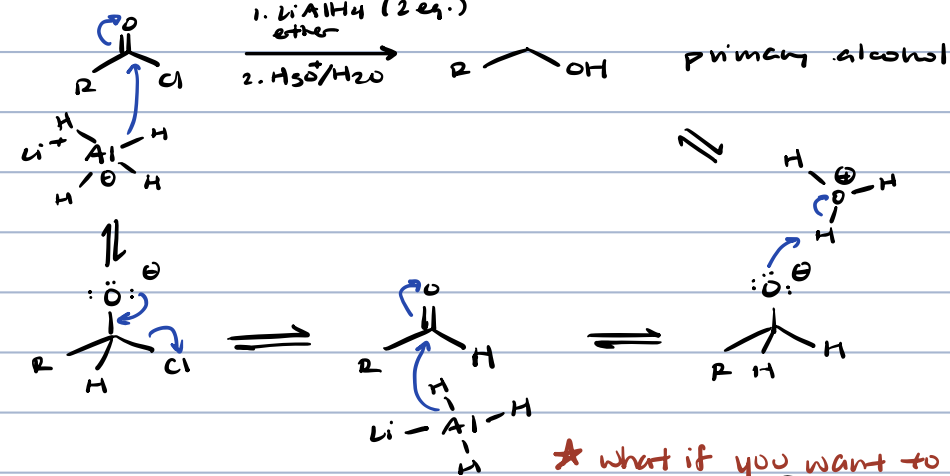
acid chloride to aldehyde
rosenmund reaction OR 1. LiAl[OC(CH3)3]3 2. H2O
![<p>rosenmund reaction OR 1. LiAl[OC(CH<sub>3</sub>)<sub>3</sub>]<sub>3</sub> 2. H<sub>2</sub>O</p>](https://assets.knowt.com/user-attachments/d86cd392-a736-4ea6-8b09-a36a27783588.png)
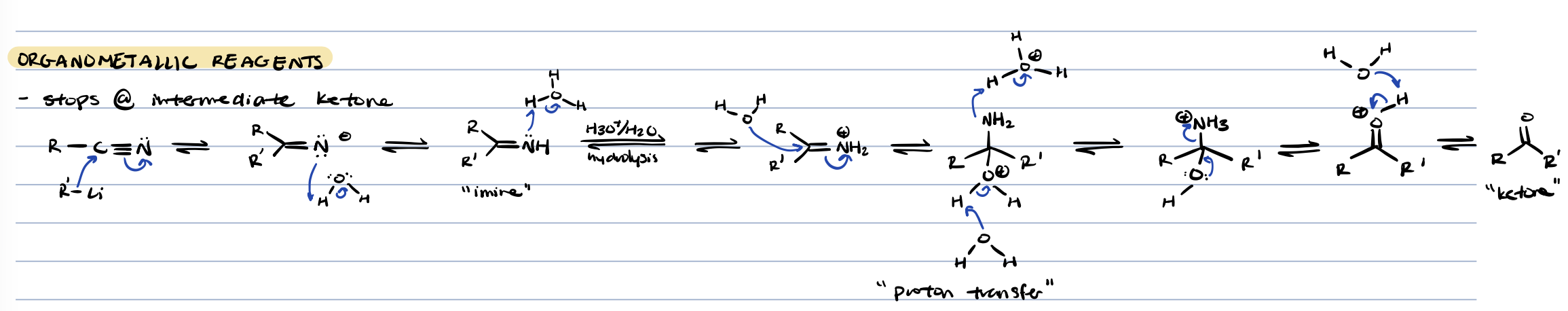
acid chloride + organometallic reagents
tertiary alcohol (kinda)
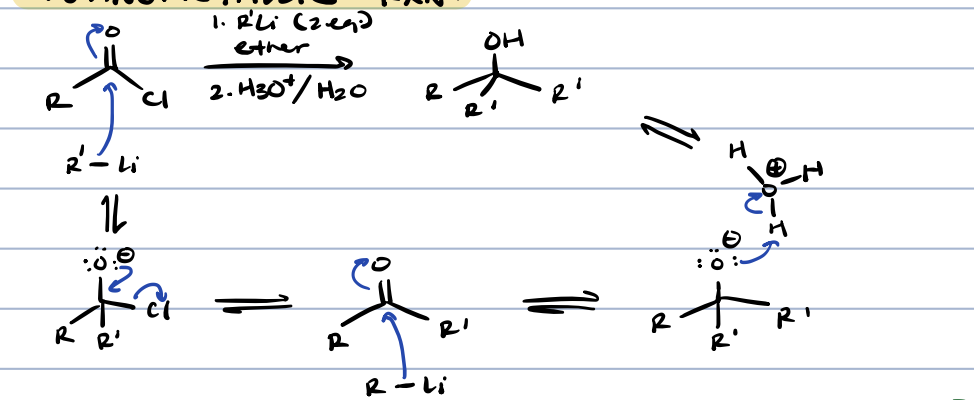
acid chloride to ketone
R2CuLi

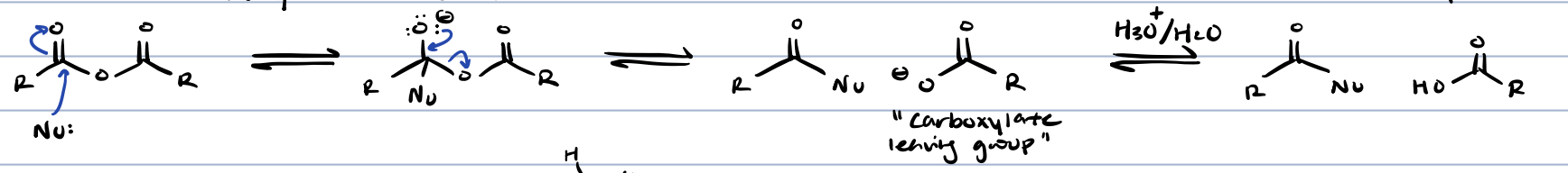
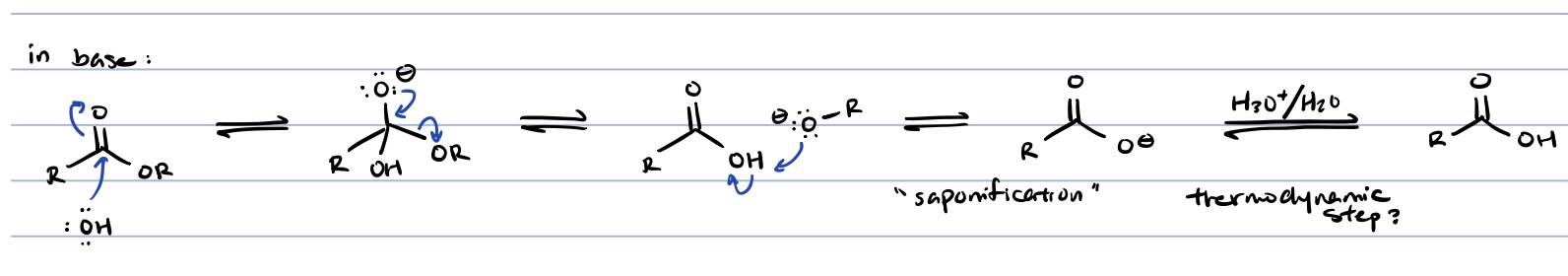
ester hydrolysis in acid
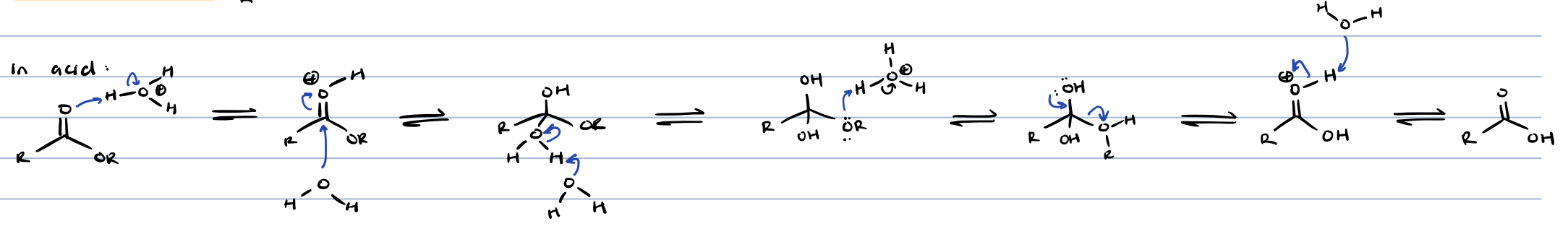
transesterification in acid
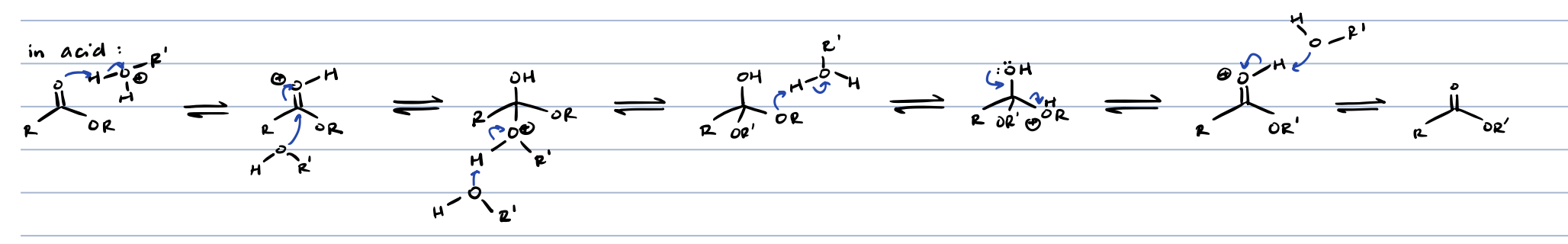
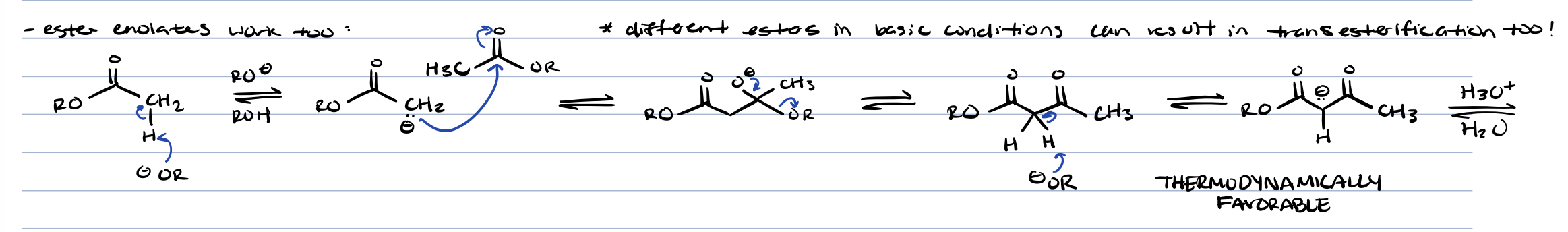
transesterification in base
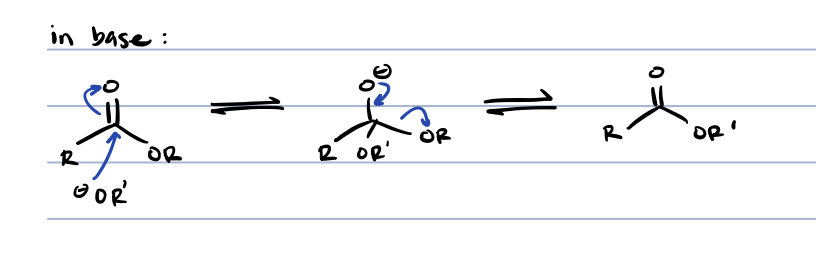
esters + organometallic reagents
tertiary alcohol (kinda), same as acid chloride + organometallic reagentse
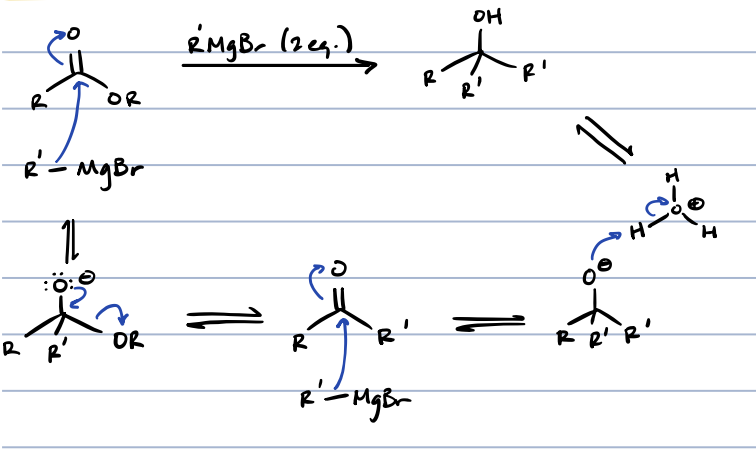
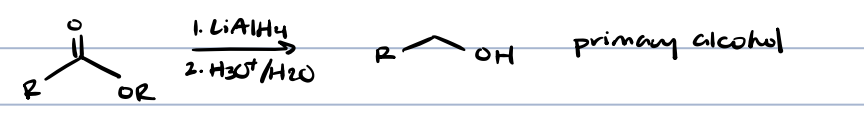
esters + metal hydrid
primary alcohol
ester + DIBAL-H
aldehyde
why does DIBAL-H make an intermediate aldehyde but metal hydrides make alcohol
DIBAL-H is more sterically hindered
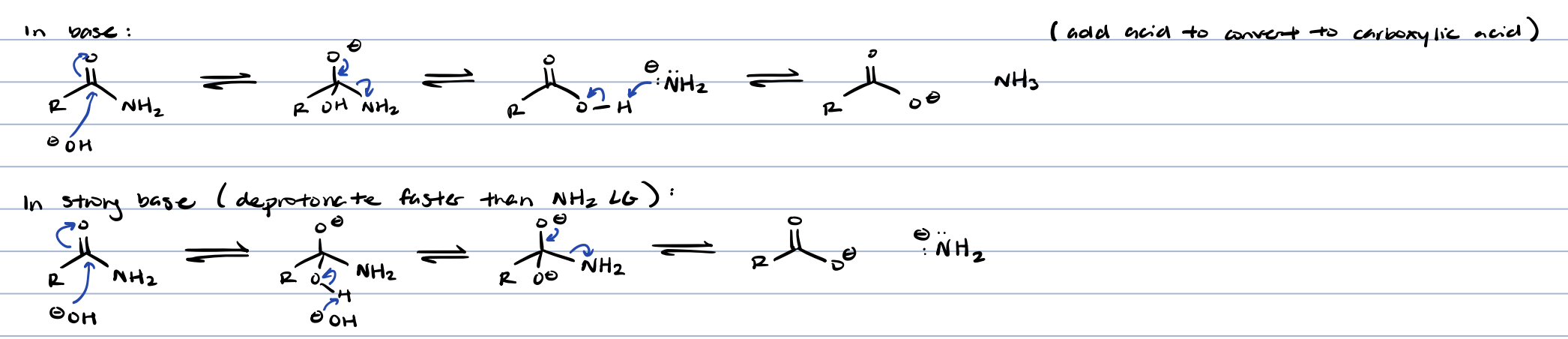
amide hydrolysis in base
to protonate, add acid
amide hydrolysis in acid
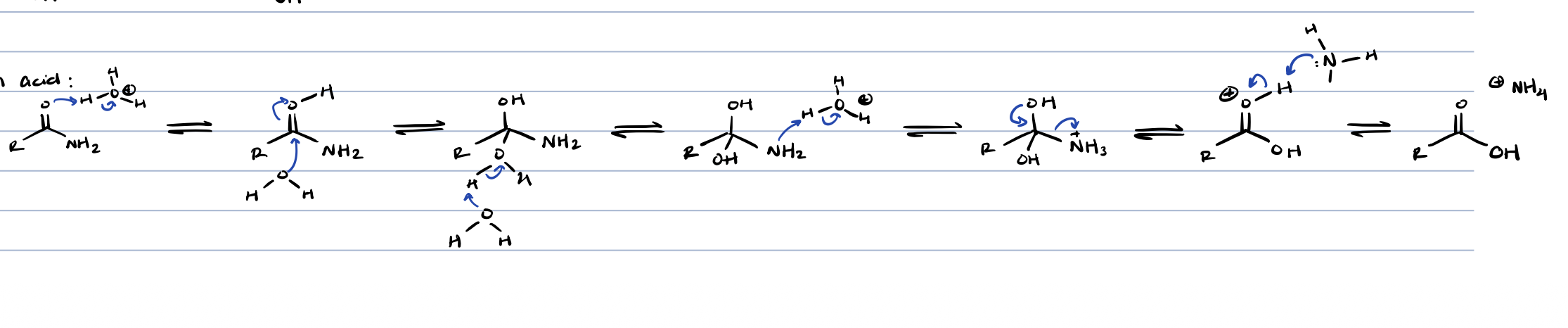
amide + metal hydride
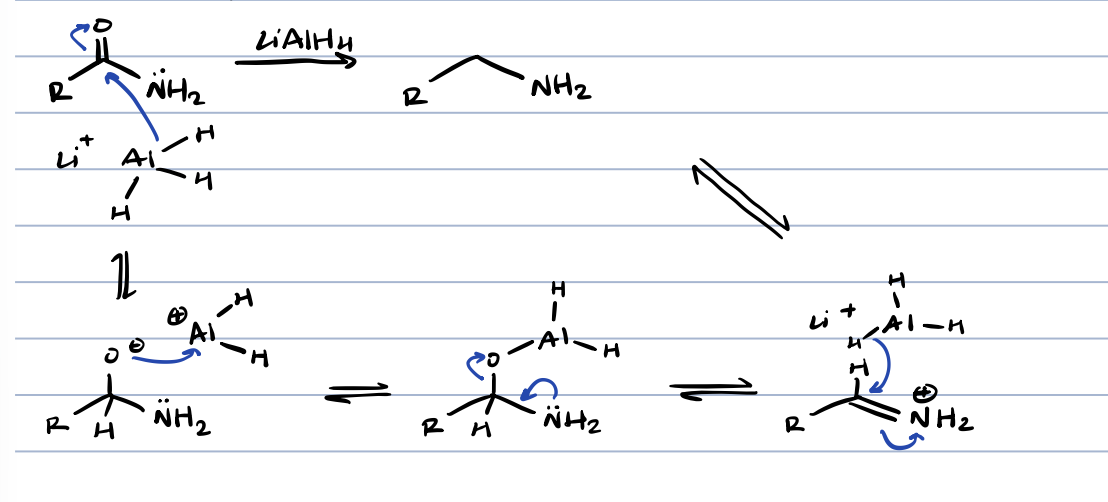
nitrile hydrolysis in acid
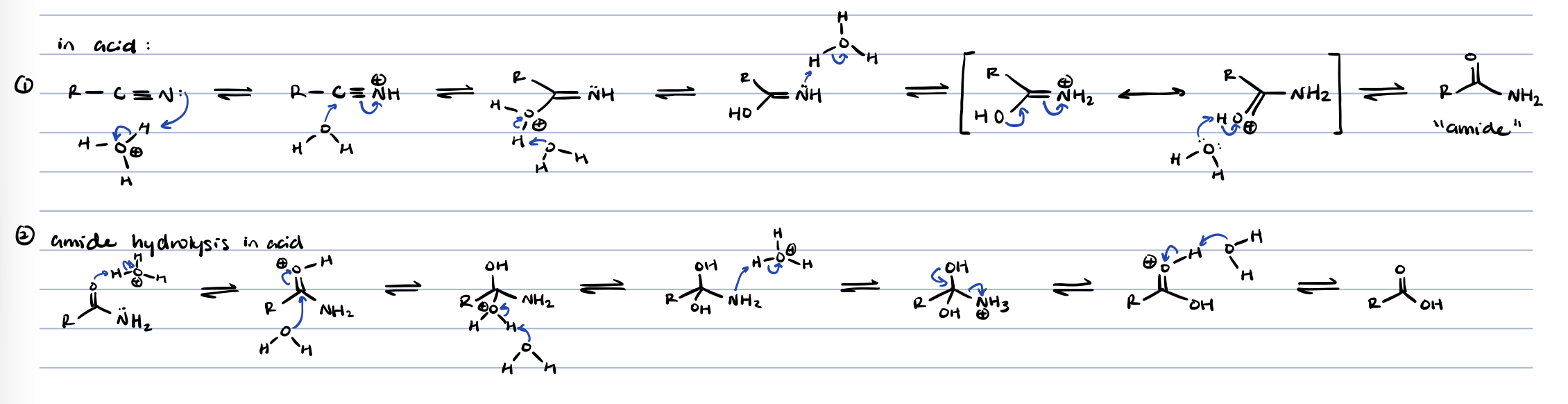
nitrile hydrolysis in base
which is better: nitrile hydrolysis in base or acid
acid, base creates side effects
nitrile + organometallic reagents
ketone
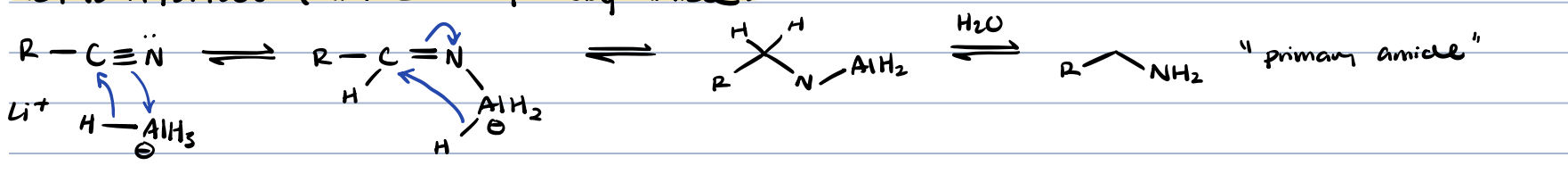
nitrile + metal hydride
primary amide
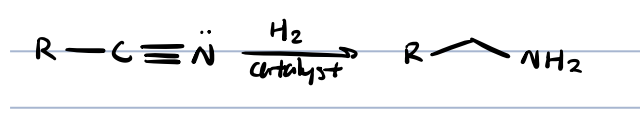
nitrile + catalytic hydrogenation
primary amide
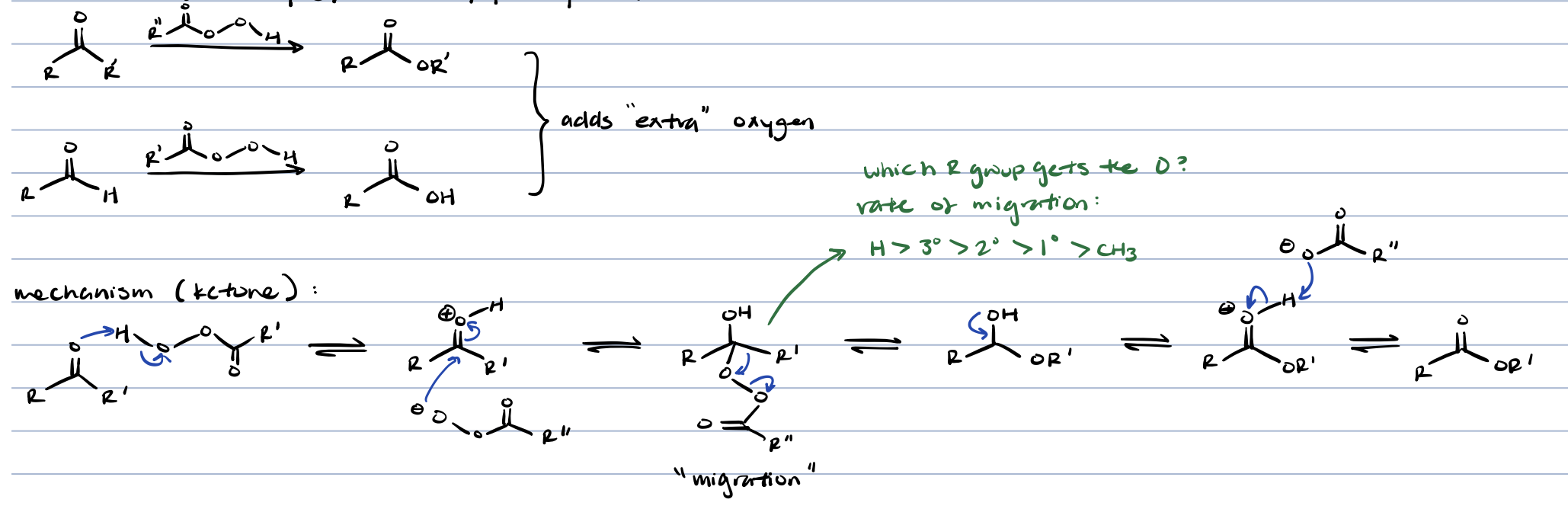
Baeyer-Villiger reaction (aldehyde/ketone + peroxy acid)
aldehyde → carboxylic acid
ketone → ester
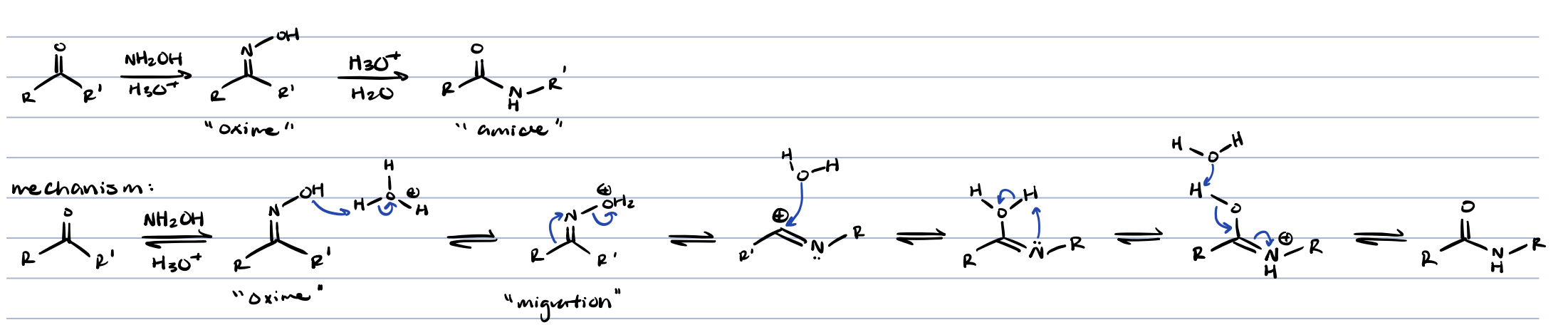
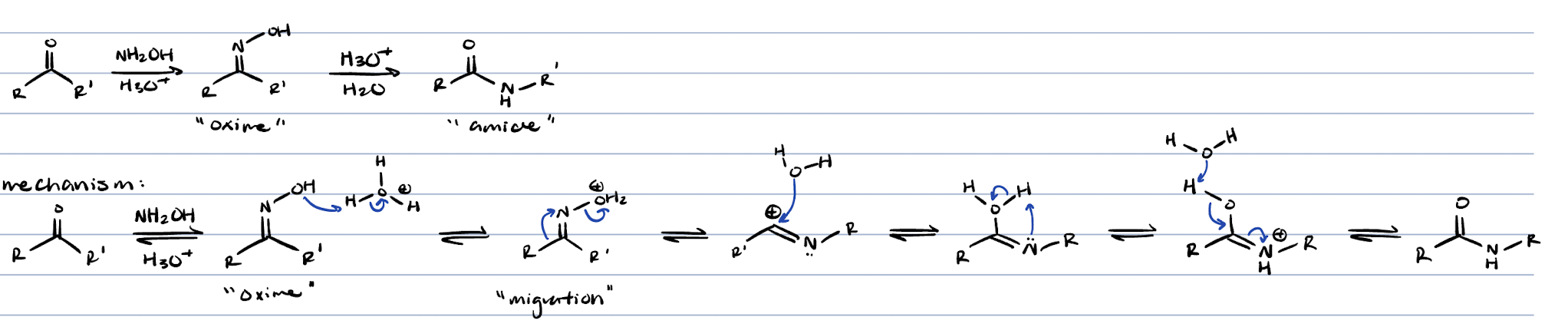
beckmann rearrangement (ketone + NH2OH and H3O+)
amide
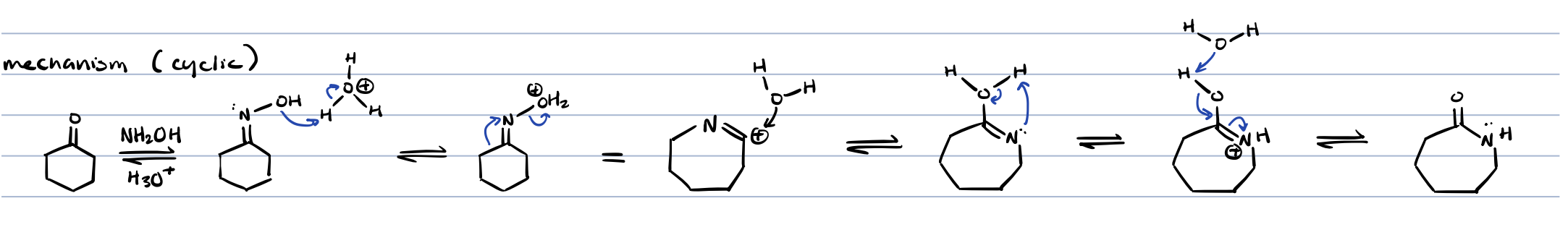
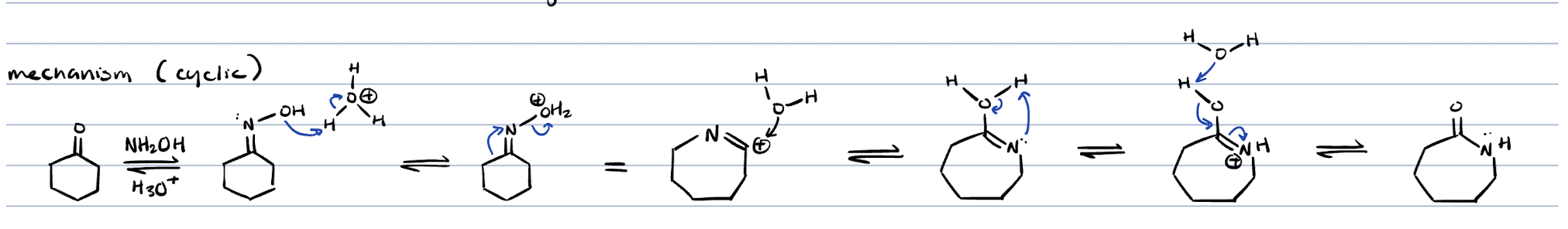
Beckmann rearrangement for cyclic compounds
For nitriles, how can you predict stereochem of product
the R group anti to the OH is the group that migrates
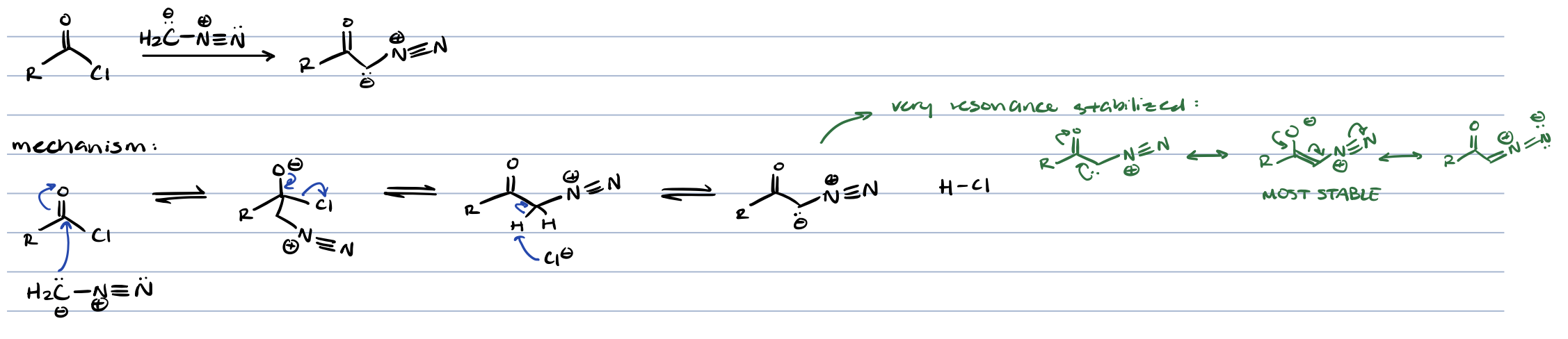
wolff rearrangement (acid chloride + CH3N2)
diazo ketone
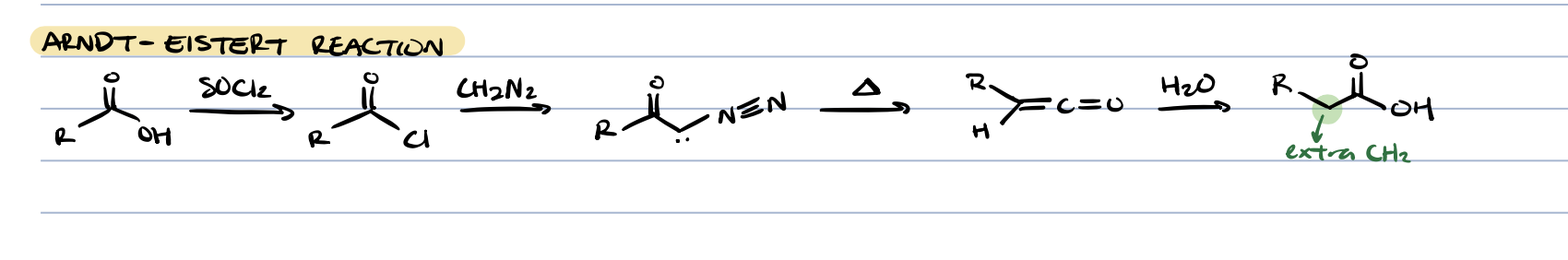
Arndt-Eistert reaction (ketone + CH2N2 + heat)
adds extra CH2 on R group of starting carboxylic acid
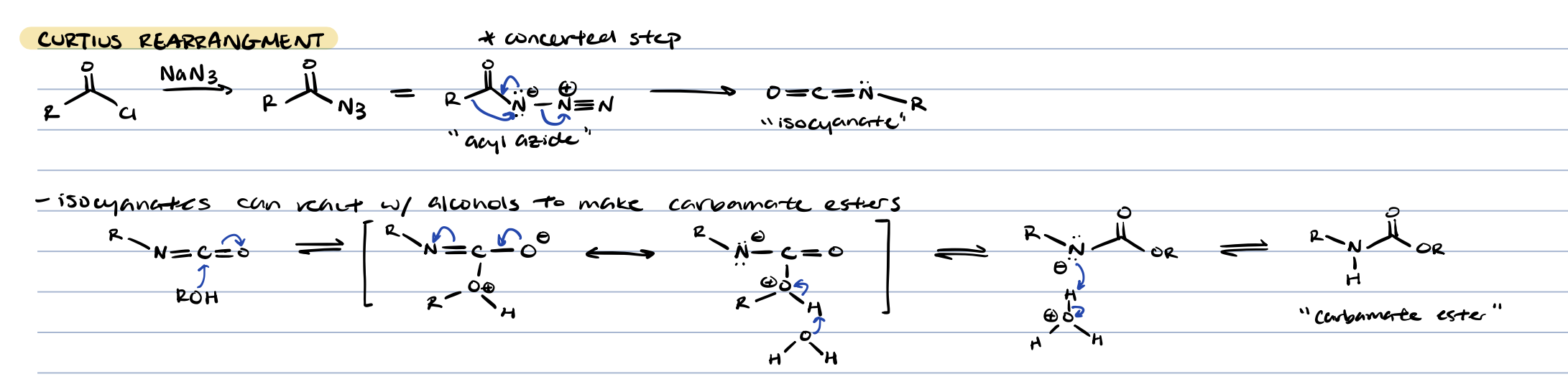
curtius rearrangement (acid chloride + NaN3)
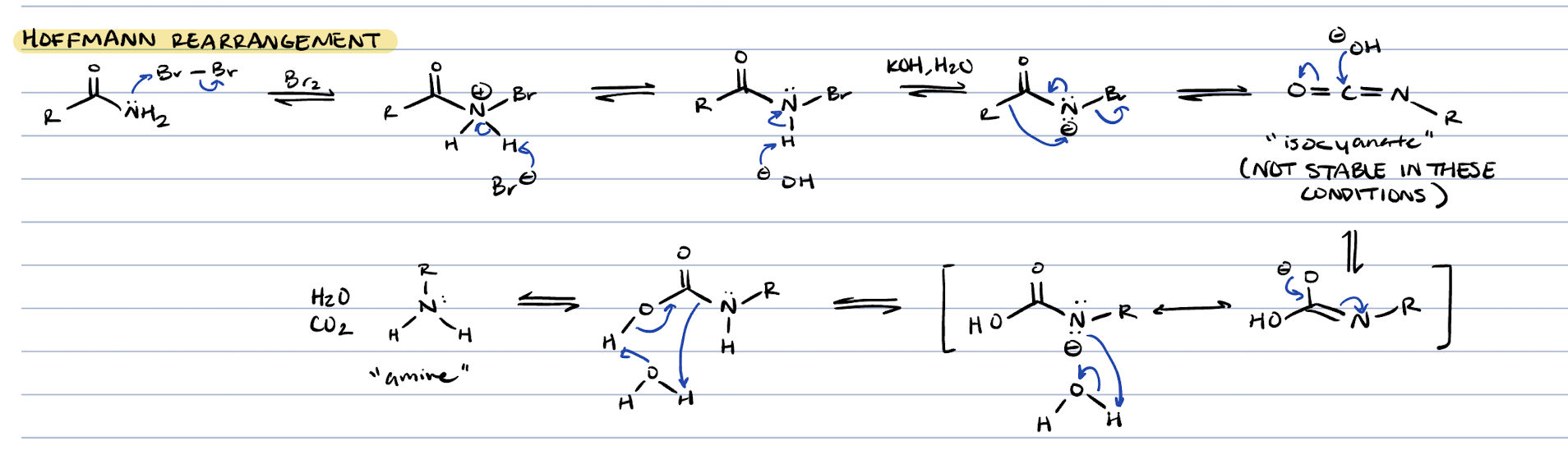
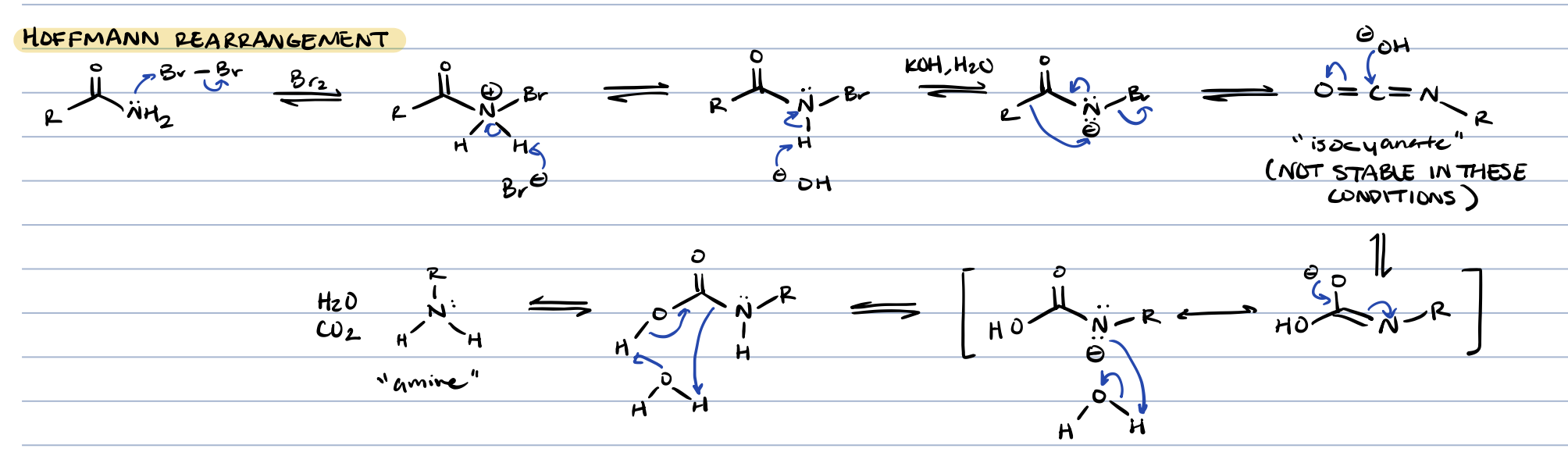
hoffmann rearrangement (amide + Br2, KOH, H2O)
amine, CO2, H2O
why is removal of an ⍺ hydrogen favored over further hydrogens
resonance stabilized
stabilization from C=O dipole (electrostatically stabilized)
what are the two nucleophiles for carbonyl chemistry at ⍺ position
enol or enolate
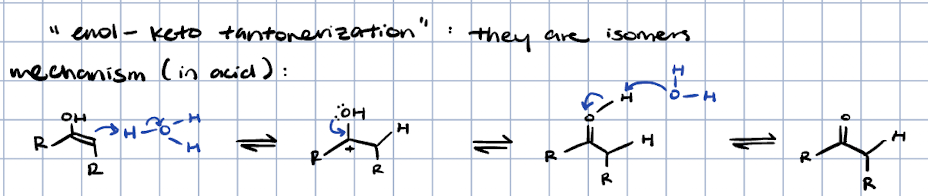
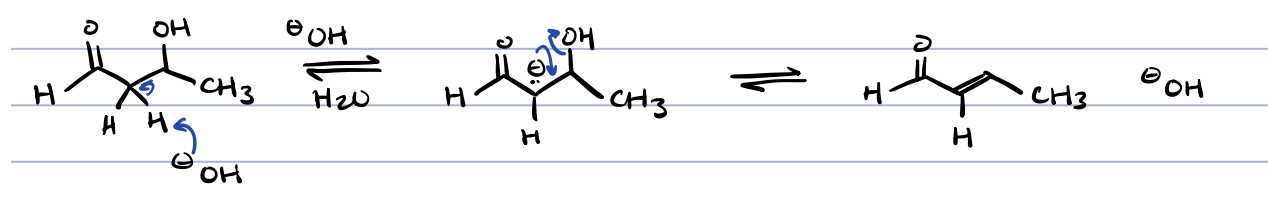
keto-enol tautomerization in acid
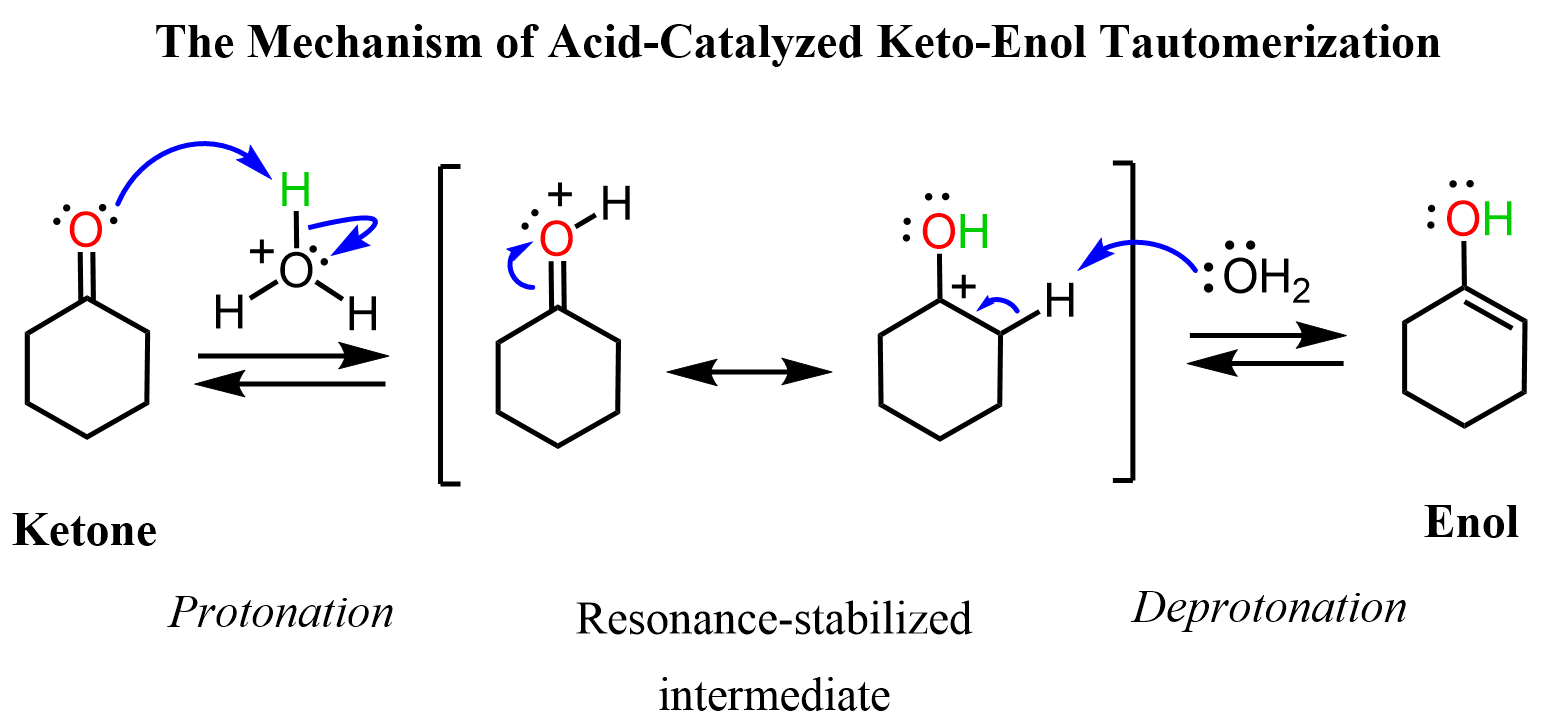
keto-enol tautomerization in base
first create an enolate
why are products of carbonyl chem at ⍺ position racemic
enols and enolates are planar, they can be attacked from both sides
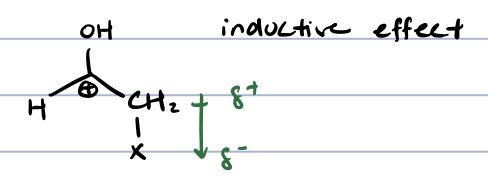
halogenation in ⍺ position for ketones and aldehydes
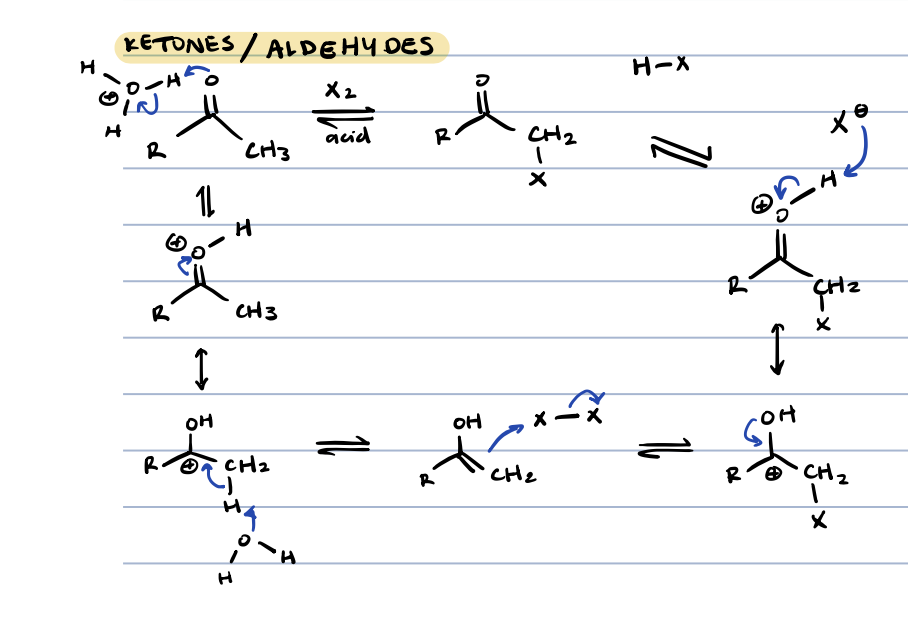
why doesn’t halogenation in acidic conditions continue like deuterium and replace all the ⍺ hydrogens
because adding a halogen would withdraw electrons at ⍺ position, disfavoring formation of enol
halogenation in ⍺ position for ketones/aldehydes in base
enolate is produced thus all ⍺ hydrogens are replaced
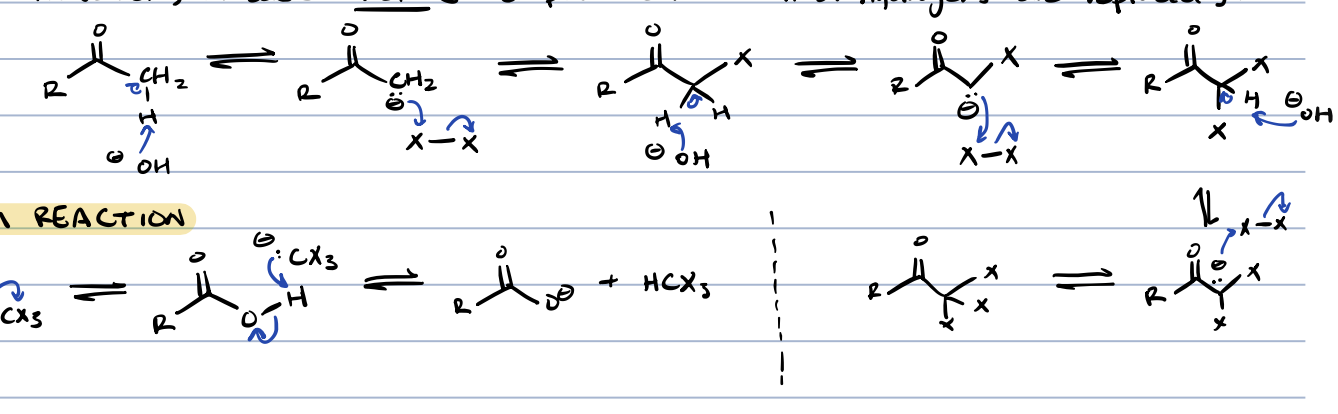
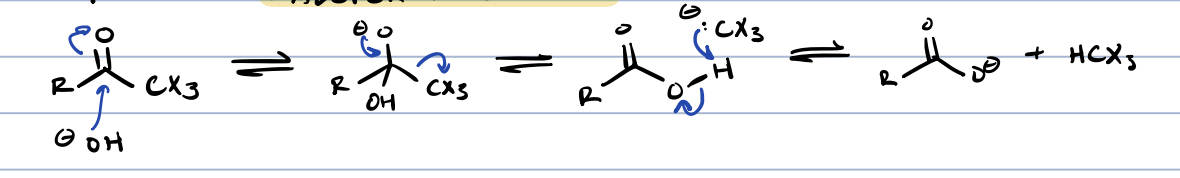
haloform reaction (methyl ketone + base)
product of halogenation at ⍺ position in base is further reacted with base
halogenation of carboxylic acids at ⍺ position
⍺-bromo acid bromide
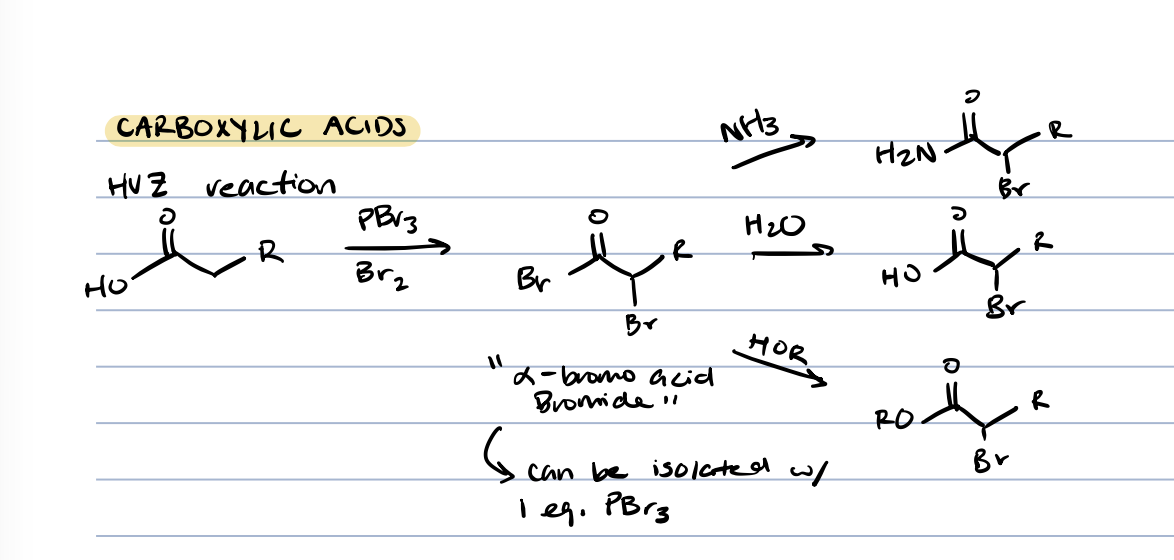
halogenation of carboxylic acids at ⍺ position (catalytic PBr3)
adding strong acid reacts with the ⍺-bromo acid bromide
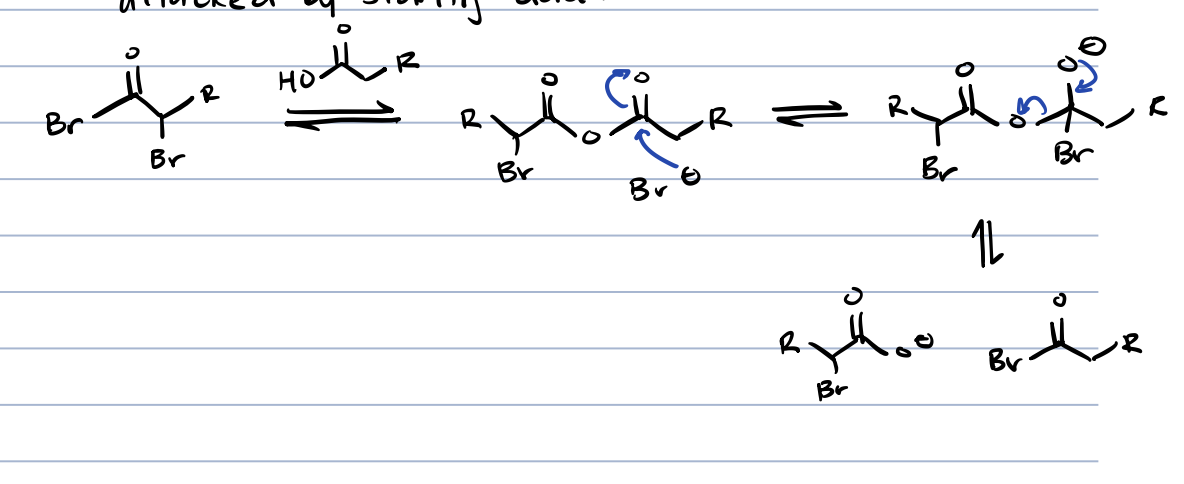
what is ⍺-bromo acid bromide similar to
acid chloride (bromide can be replaced to make amide, carboxylic acid, ester)
halogenation of esters in ⍺ position
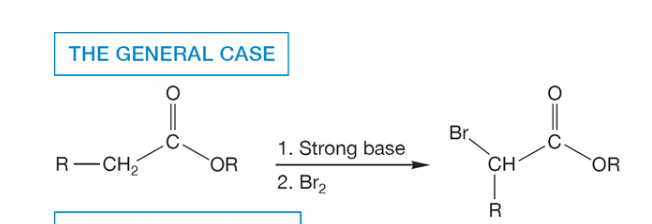
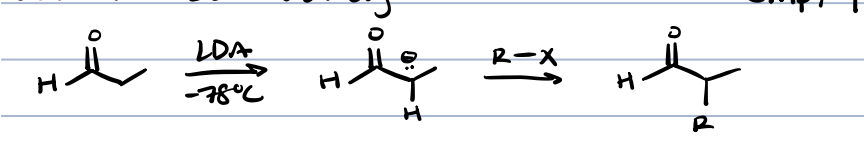
alkylation at ⍺ position for ketones and aldehydes
alkylation for carboxylic acids
requires 2 eq of strong base to remove both carboxyl hydrogen and ⍺ hydrogen
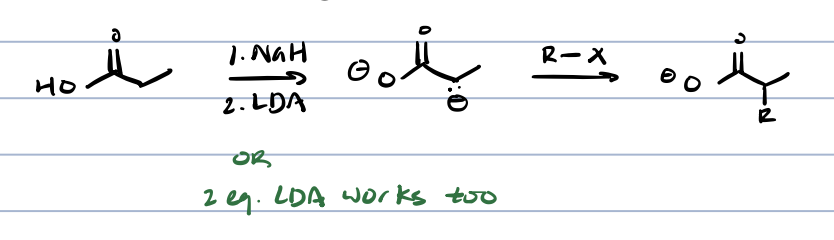
alkylation of ß-dicarbonyl compounds

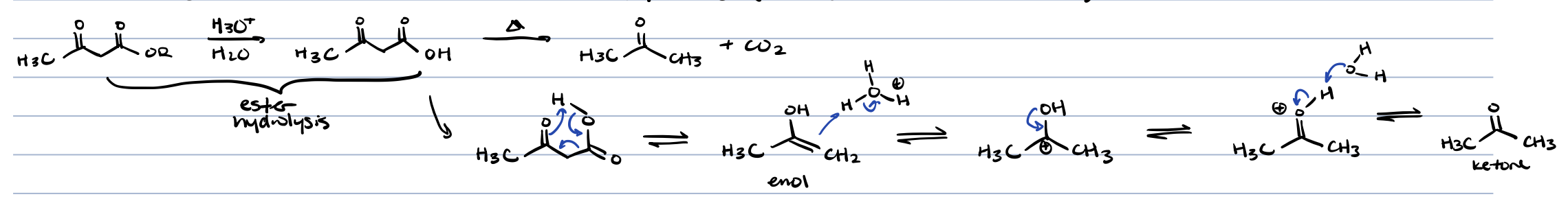
hydrolysis and decarboxylation of ß-dicarbonyl compounds
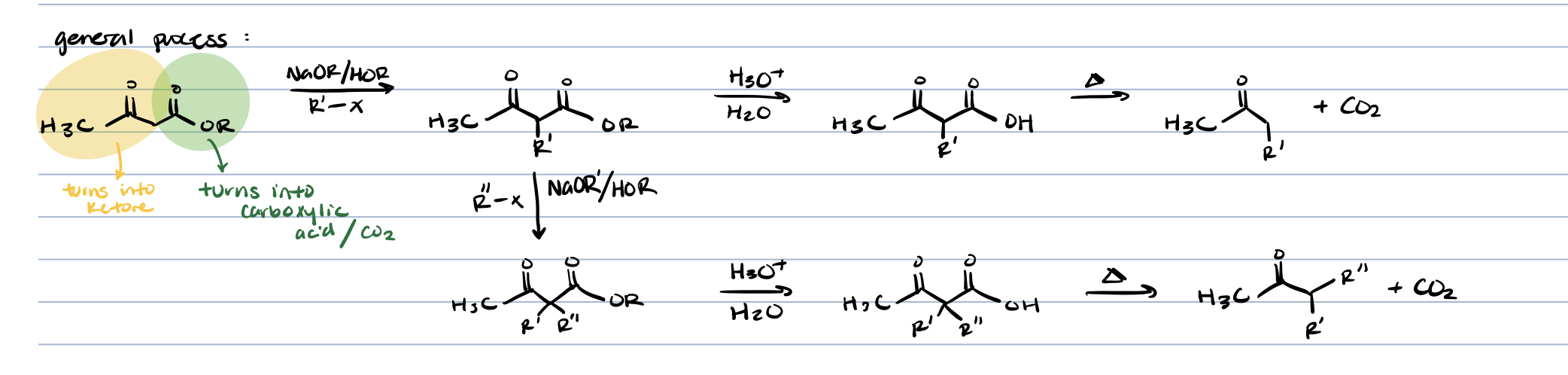
ß-keto ester synthesis
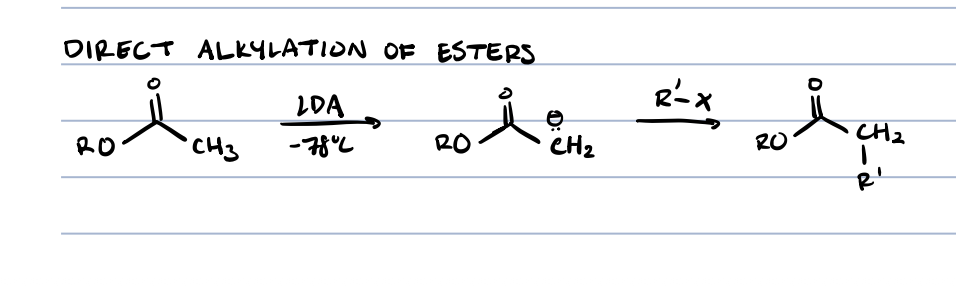
direct alkylation of esters
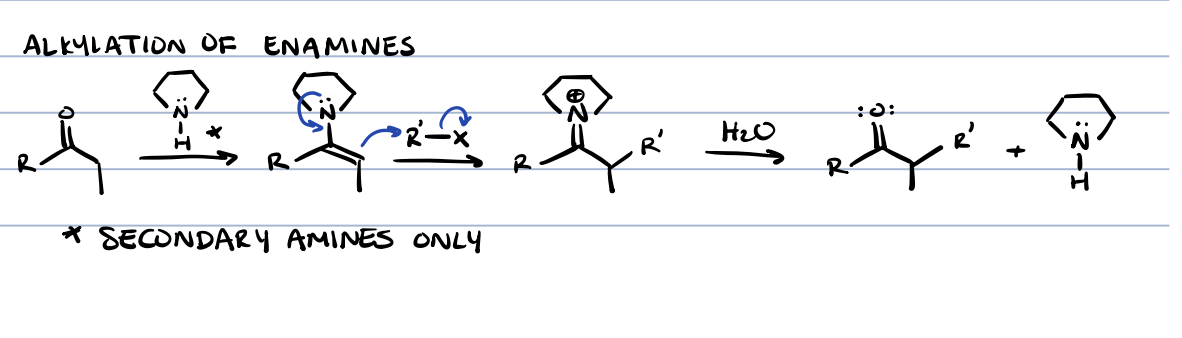
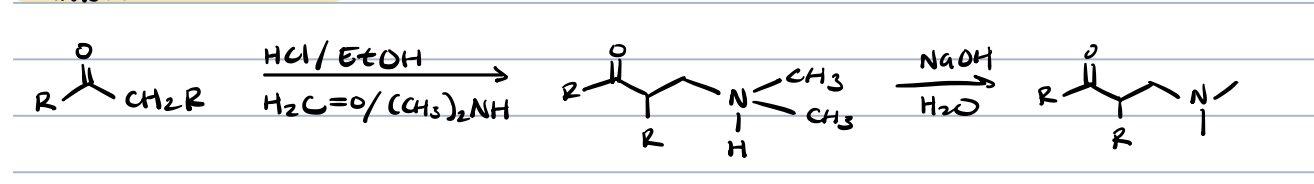
alkylation of amines
secondary amines only
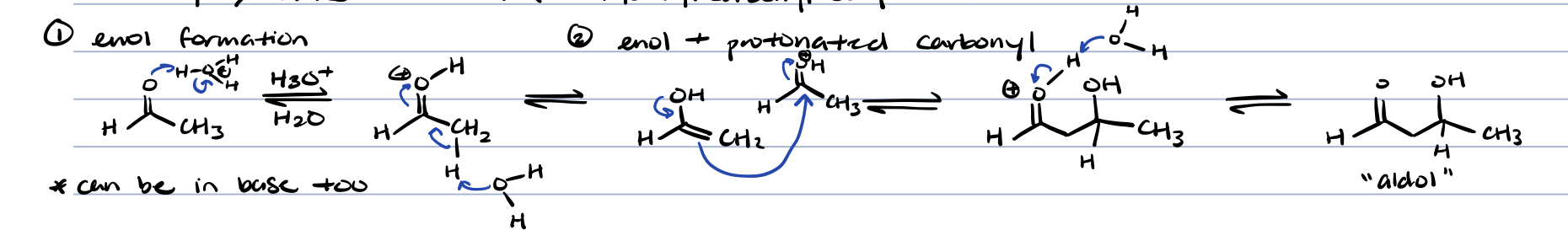
adol condensation of aldehydes/ketones in acid
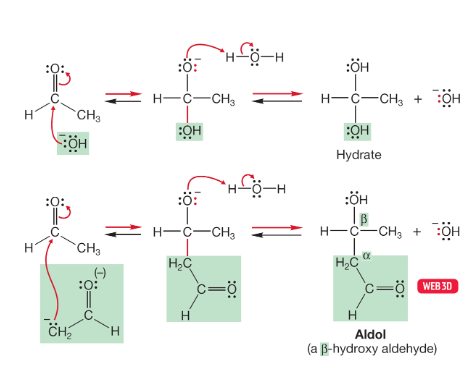
aldol condensation of aldehydes/ketones in base
aldol condensation to unsaturated aldehyde/ketone (in acid)
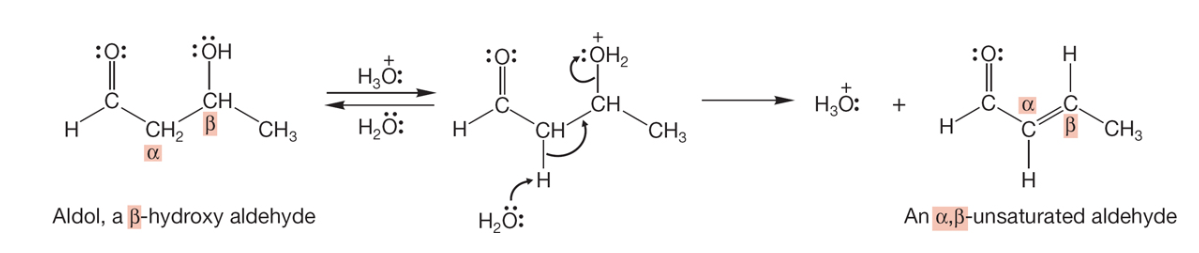
aldol condensation to unsaturated aldehyde/ketone (in base)
knoevenagel condensation (ketone/aldehyde + base)
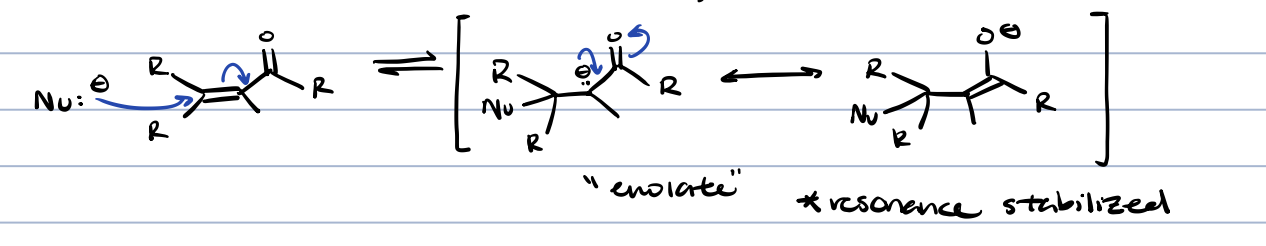
⍺, ß-unsaturation diester (anion stabilizing groups can be carboxylic acids, carbonyls, etc)
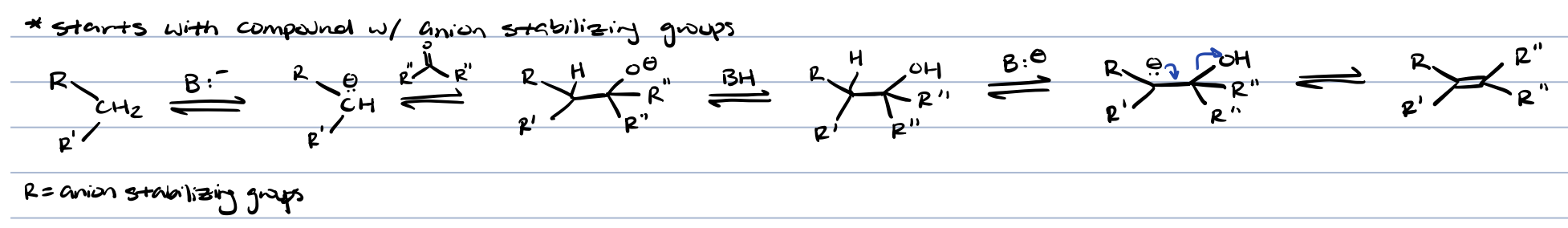
michael reaction (⍺, ß-unsaturated carbonyl compounds)
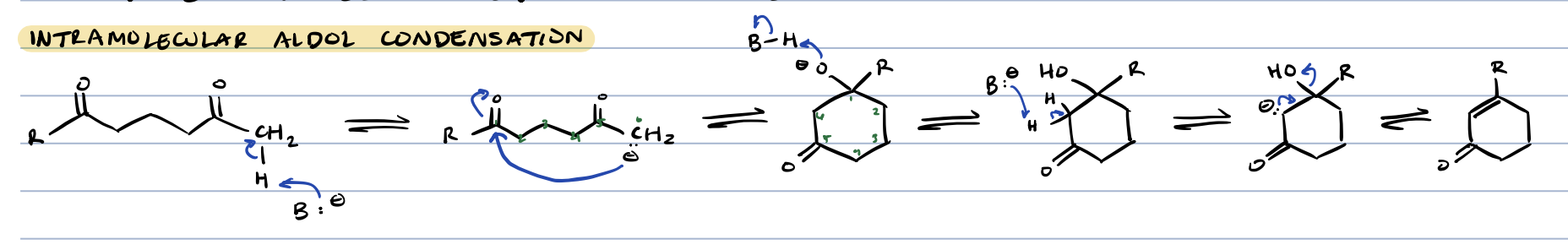
intramolecular aldol condensation
crossed aldol condensation
two different carbonyl compounds (4 possible products)
claisen condensation with ketones
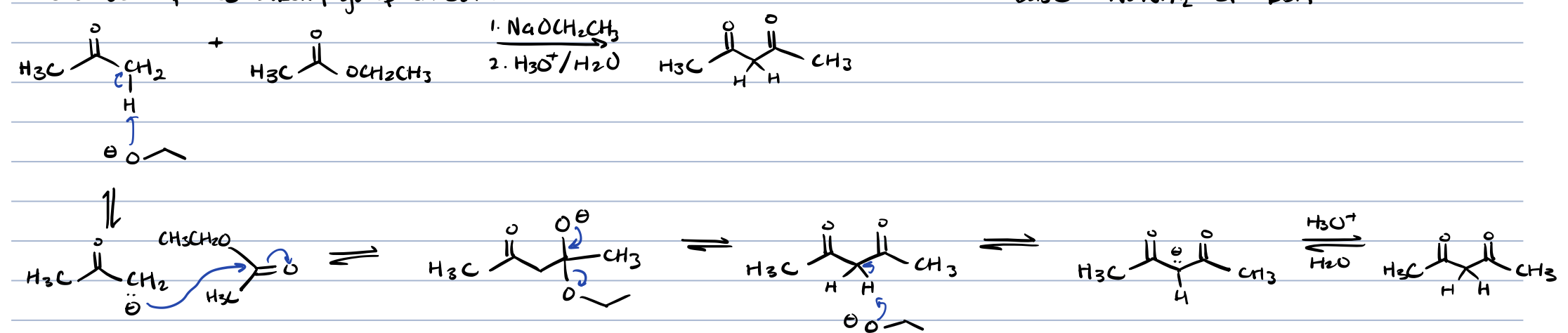
claisen condensation with 2 esters
results in claisen product and transesterification product
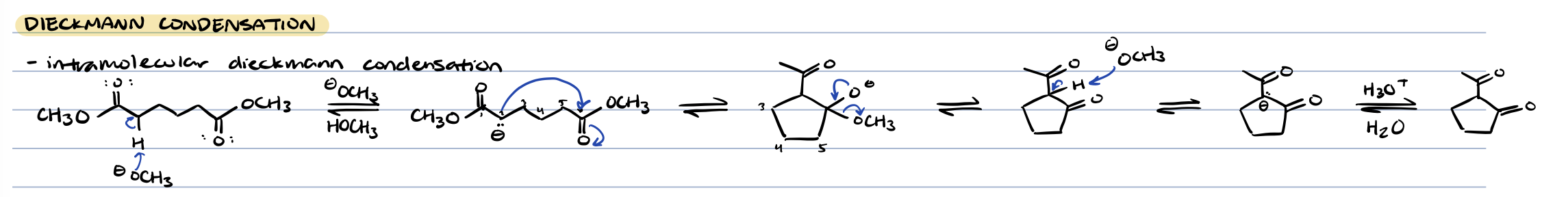
dieckmann condensation (intramolecular claisen condensation)
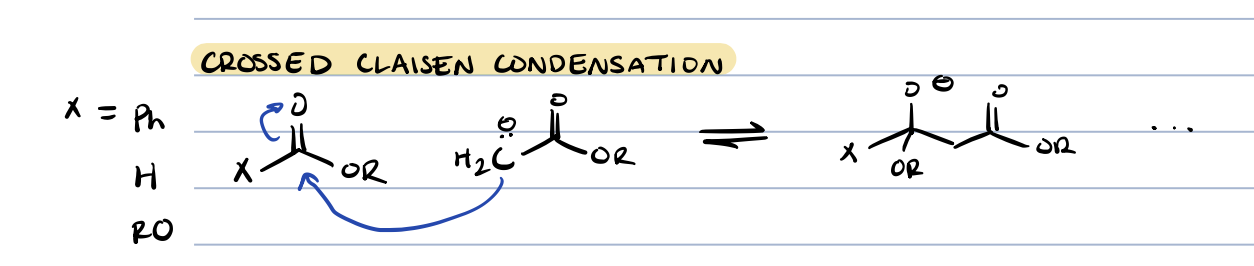
crossed claisen condensation
robinson annulation (michael addition + aldol condensation)
makes six membered ring
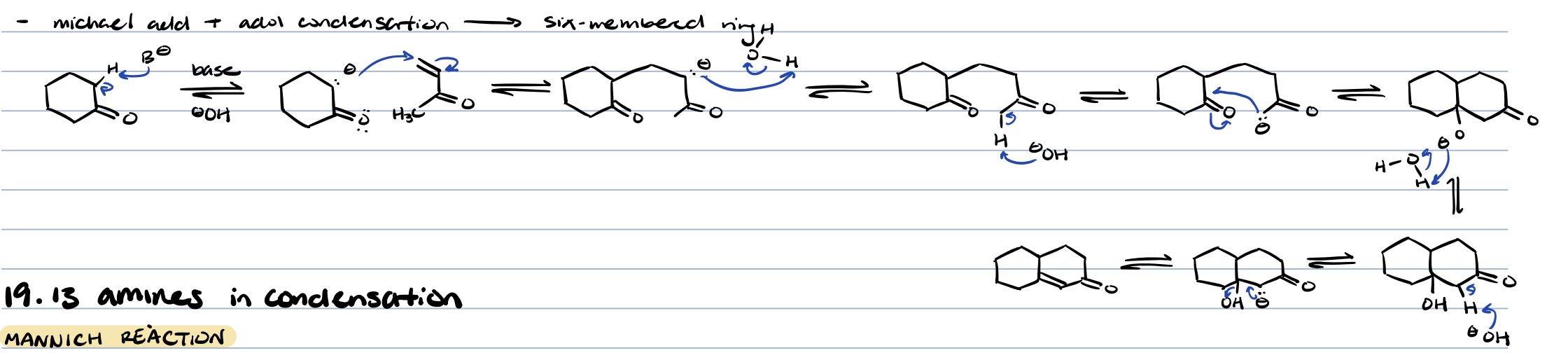
mannich reaction
beckmann rearrangement (ketone → amide)
beckmann rearrangement (cyclic)
wolff rearrangement (acid chloride → diaz ketone)
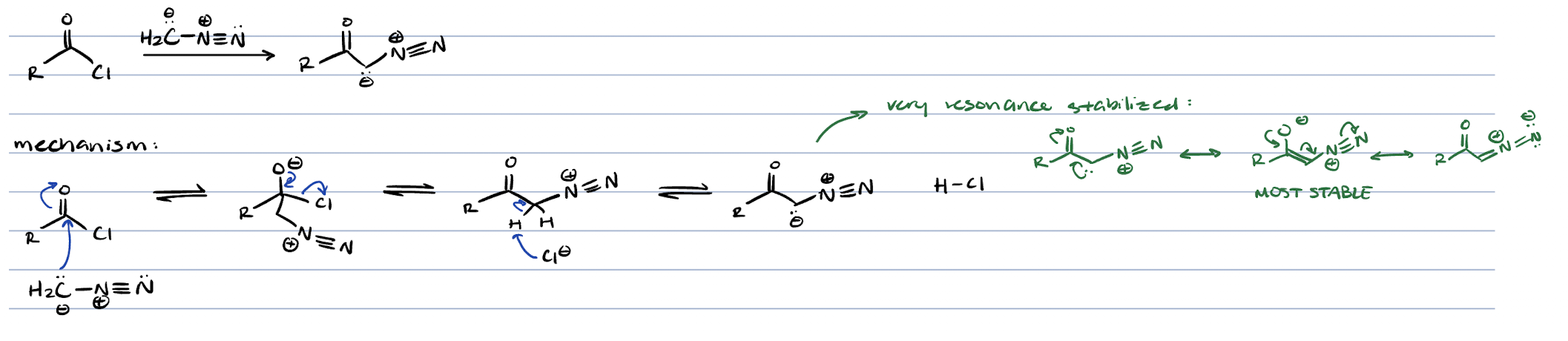
diazo ketone exposed to light
carbene
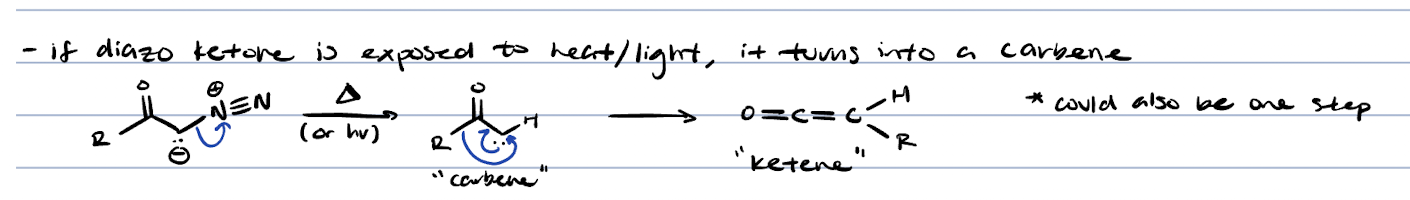
arndt-eistert reaction (extra CH2)
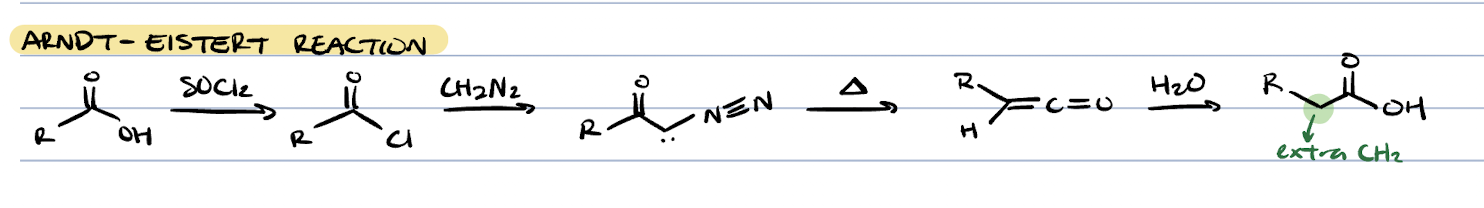
curtius rearrangement
hoffmann rearrangement (amide→amine)