CHEM 2.5 - TRANSITION METALS
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
transition element
transition element = is a d-bloc element that can form at least one stable ion with a partially filled (incomplete) d subshell
for period 4 d block elements, scandium and zinc are not transition metal because they dont form a stable ion with a partial filled d subshell
chromium and copper are exceptions to the electron configuration
loose electrons from 4s first then 3d

scandium and zinc not a transition metal
transition metals have to have at least one stable ION with a partially filled d subshell
scandium forms a stable 3+ ion but it does not have a partially full d subshell. the d sub shell is empty for Sc3+
zinc forms a stable 2+ ion but it does not have a partially full d subshell. the d subshell is full for Zn2+

properties of transition metals
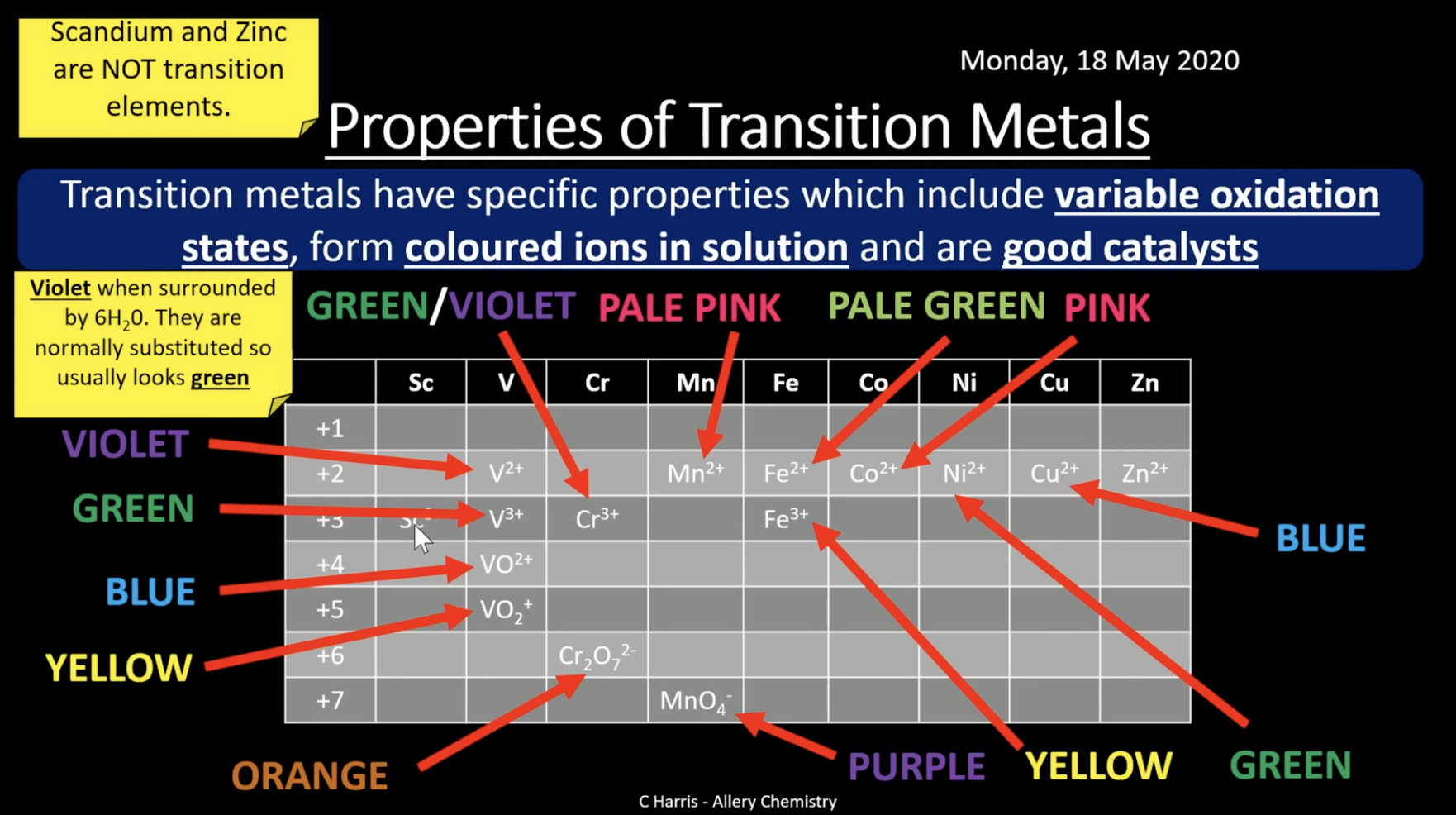
transition metals have specific properties which include variable oxidation states, form coloured ions in solution and are good catalysts
transition metals have variable oxidation states. this is because the electrons sit in 4s and 3d sub shells which are very close in energy. therefore electrons are gained and lost using a similar amount of energy when they form the ions.
they form many different ions
they form coloured ions in solution
colours of transition metals
these ions form coloured ions in solution (aqueous in water)
metal complexes
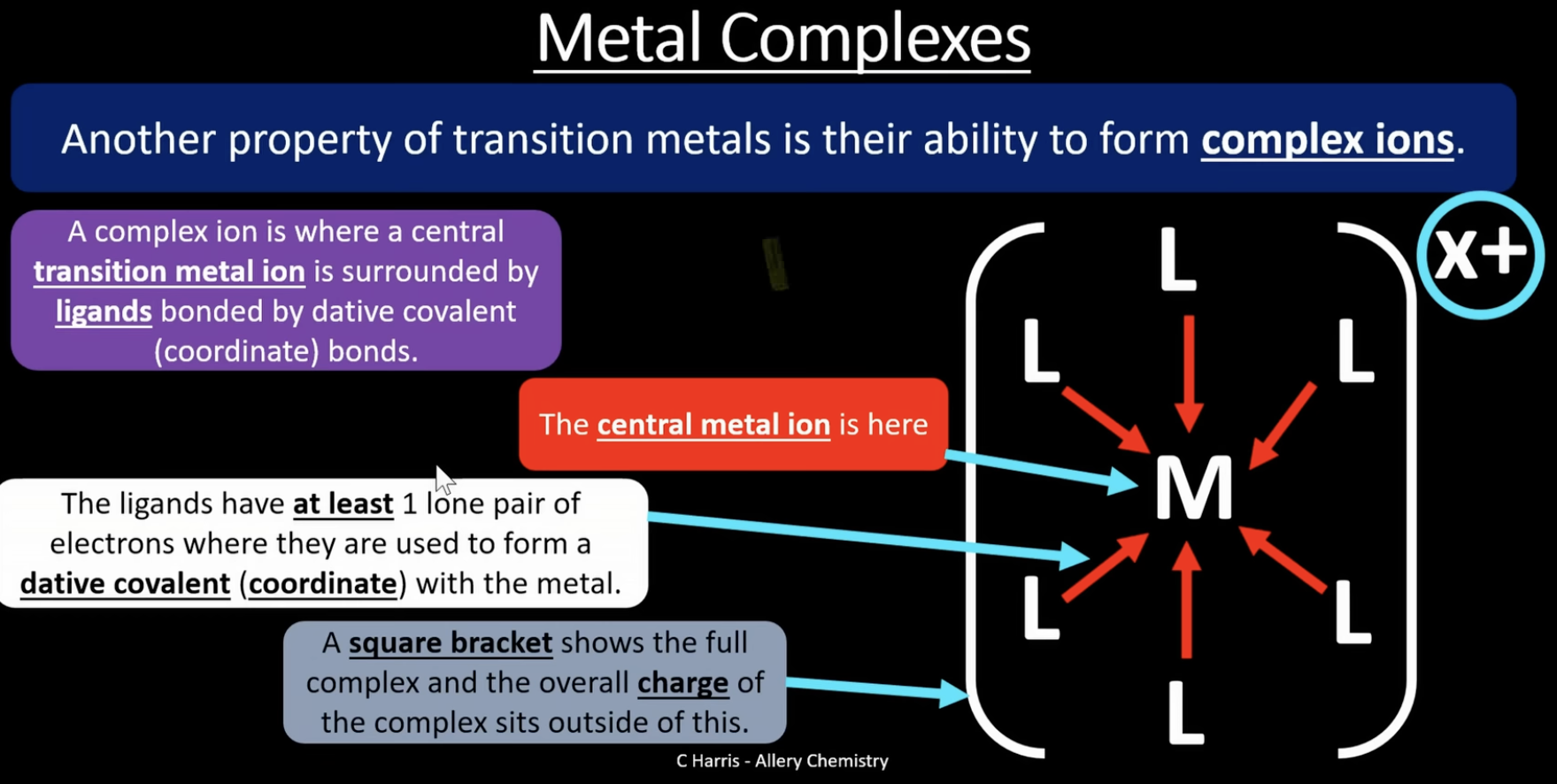
transition metals can form complex ions where a central transition metal ion is surrounded by ligands bonded by dative covalent/ coordinate bonds
ligand = a molecule or ion that forms a dative covalent bond with a transition metal by donating a pair of electrons
ligands have at least 1 lone pair of electrons which are used to form a dative covalent bond with the metal
types of ligands
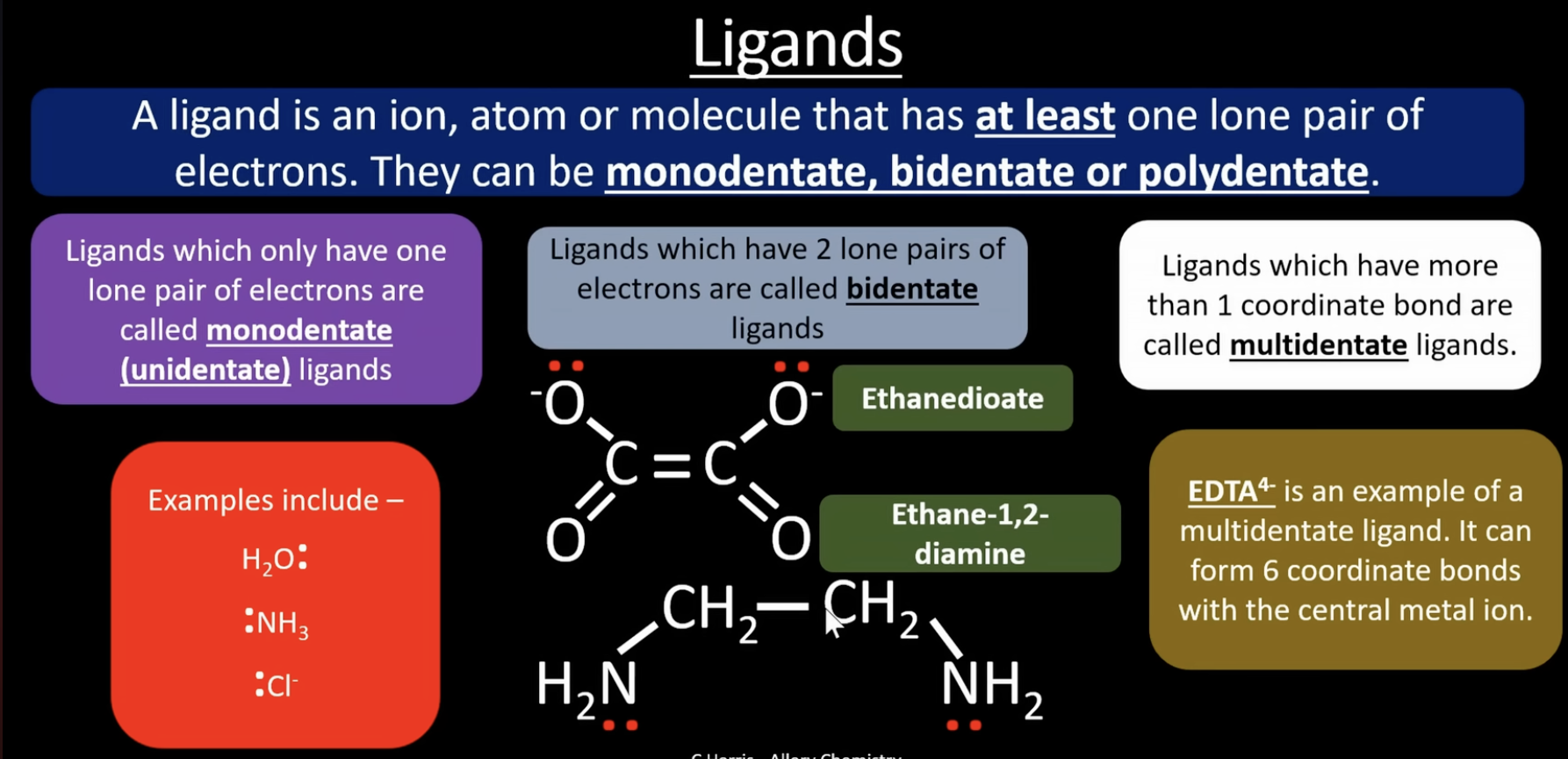
monodentate = a ligand that forms one coordinate bond to the metal ion.
-examples: H2O, NH3, Cl-
bidentate = a ligand that forms two coordinate bonds to the metal ion
-example: ethanedioate C2O42-, ethane-1,2-diamine C2H8N2
multidentate = a ligand that forms multiple coordinate bonds to the metal ion
-example: EDTA4- (forms 6 coordinate bonds)
complex shapes
the shape is dependant on the coordination number and the size of ligands
coordination number = the coordination number is the number of coordinate bonds in a complex, NOT the number of ligands
size of ligands = can fit a certain no of ligands around a metal ion depending on its size
-can fit 6 of H2O and NH3
-can fit 4 of Cl-
-can fit 3 of ethanedioate C2O42- and ethane-1,2-diamine C2H8N2

naming complex shapes
expressing the shapes [Co(H2O)6]2+
for single atom ligands they dont need to be written with extra brackets around them
octahedral shapes = complexes with a coordination number of 6.l bond angles is 90
tetrahedral shapes = complexes with a coordination number of 4. bond angle is 109.5
square planar shapes = also complexes with a coordination number of 4. bond angle is 90. only example you need to know is cisplatin [Pt(NH3)2Cl2] which is an anti cancer drug. memorise the shape and expression
linear shapes = complexes with a coordination number of 2. bond angle is 180. example you need to know is tollens reagent [Ag(NH3)2]+. memorise the shape and expression
![<p>expressing the shapes <span>[Co(H<sub>2</sub>O)<sub>6</sub>]<sup>2+</sup></span></p><p><span>for single atom ligands they dont need to be written with extra brackets around them</span></p><p><strong>octahedral shapes</strong> = complexes with a coordination number of 6.l bond angles is 90 </p><p><strong>tetrahedral shapes</strong> = complexes with a coordination number of 4. bond angle is 109.5 </p><p><strong>square planar shapes</strong> = also complexes with a coordination number of 4. bond angle is 90. only example you need to know is cisplatin <span>[Pt(NH<sub>3</sub>)<sub>2</sub>Cl<sub>2</sub>] </span>which is an anti cancer drug. memorise the shape and expression</p><p><strong>linear shapes</strong> = complexes with a coordination number of 2. bond angle is 180. example you need to know is tollens reagent [Ag(NH<sub>3</sub>)<sub>2</sub>]<sup>+</sup>. memorise the shape and expression</p><p></p>](https://assets.knowt.com/user-attachments/aba3d245-f1dd-4a28-9d42-472547f4c046.png)
calculating oxidation state of the metal of complex shapes
oxidation state of metal = total oxidation state of complex shape - total oxidation states of ligands

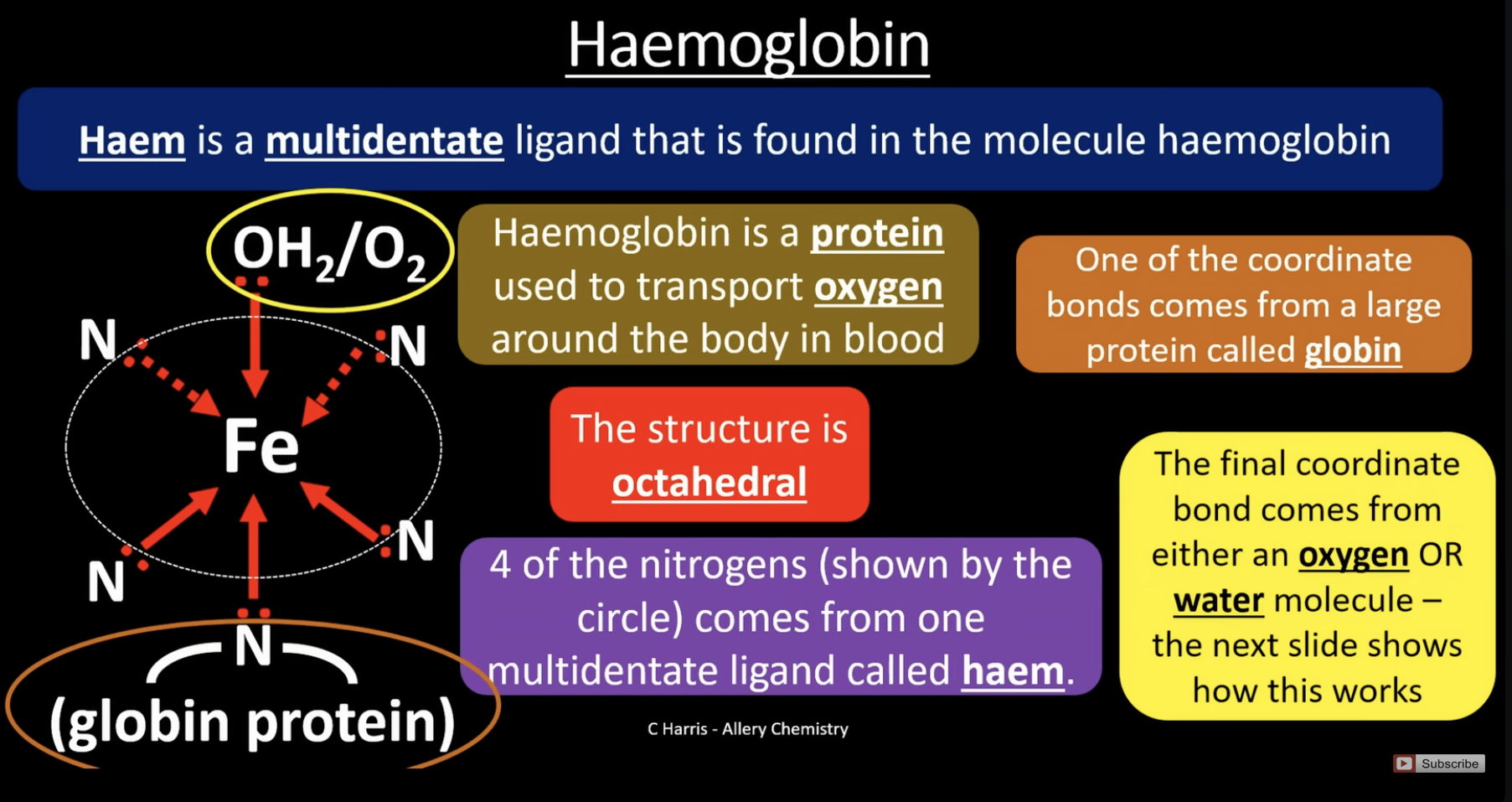
haemoglobin
is an octahedral shape
haem is a multi dentate ligand found in haemoglobin, the 4 nitogens bound to Fe come from the haem group (the circle). the 5th nitrogen comes from the globin protein
in the lungs when oxygen concentration is high, oxygen substitutes the water ligand and is transported around the body, forming oxyhaemoglobin. the oxygen is removed where its needed in the body and is replaced by water again. the haemoglobin returns to the lungs and process repeats
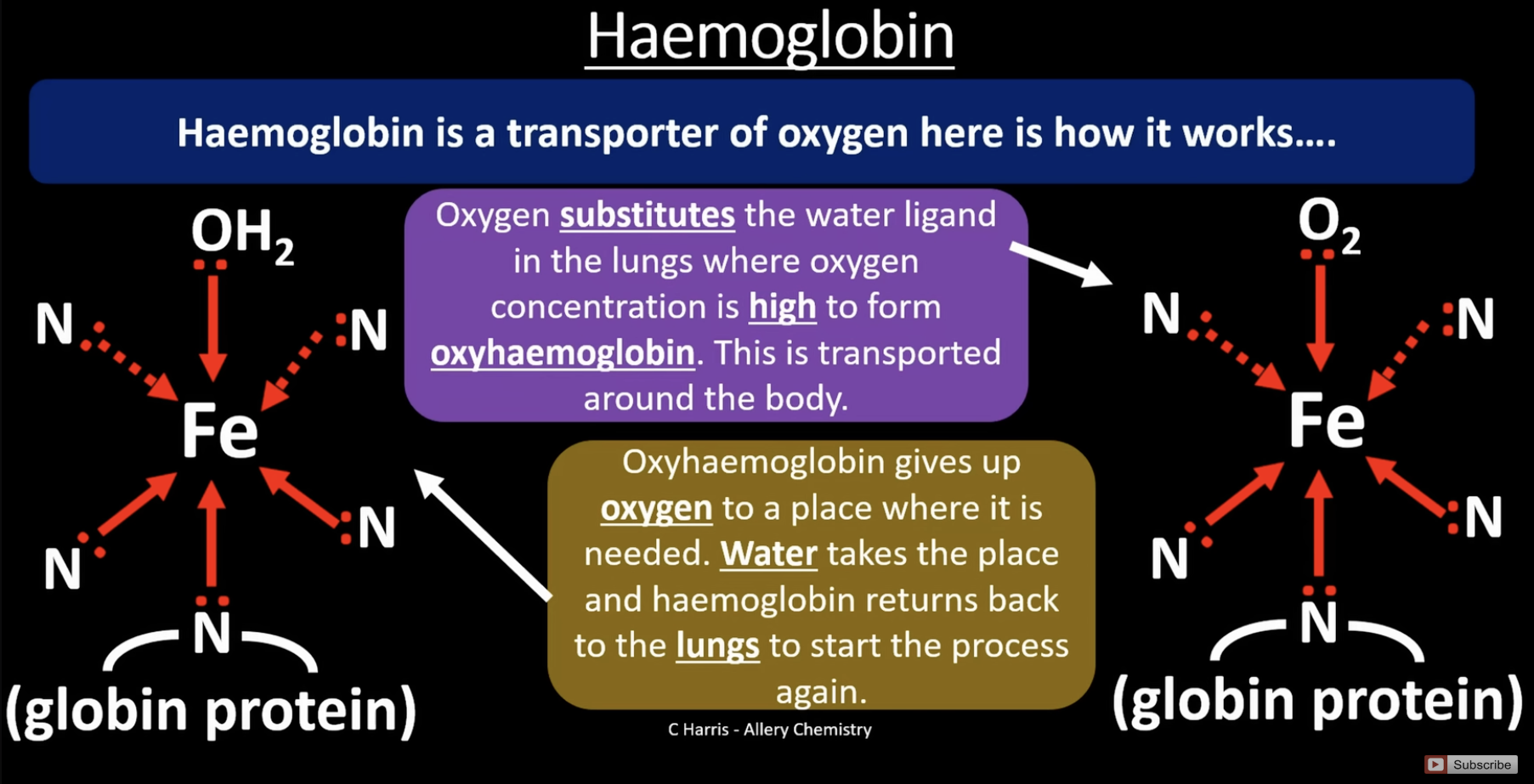
haemoglobin and carbon monoxide
carbon monoxide is dangerous because it can replace the water ligand in haemoglobin and the bond it forms is very strong so it cannot be replaced by water or oxygen. this leads to a lack of transport of oxygen around the body

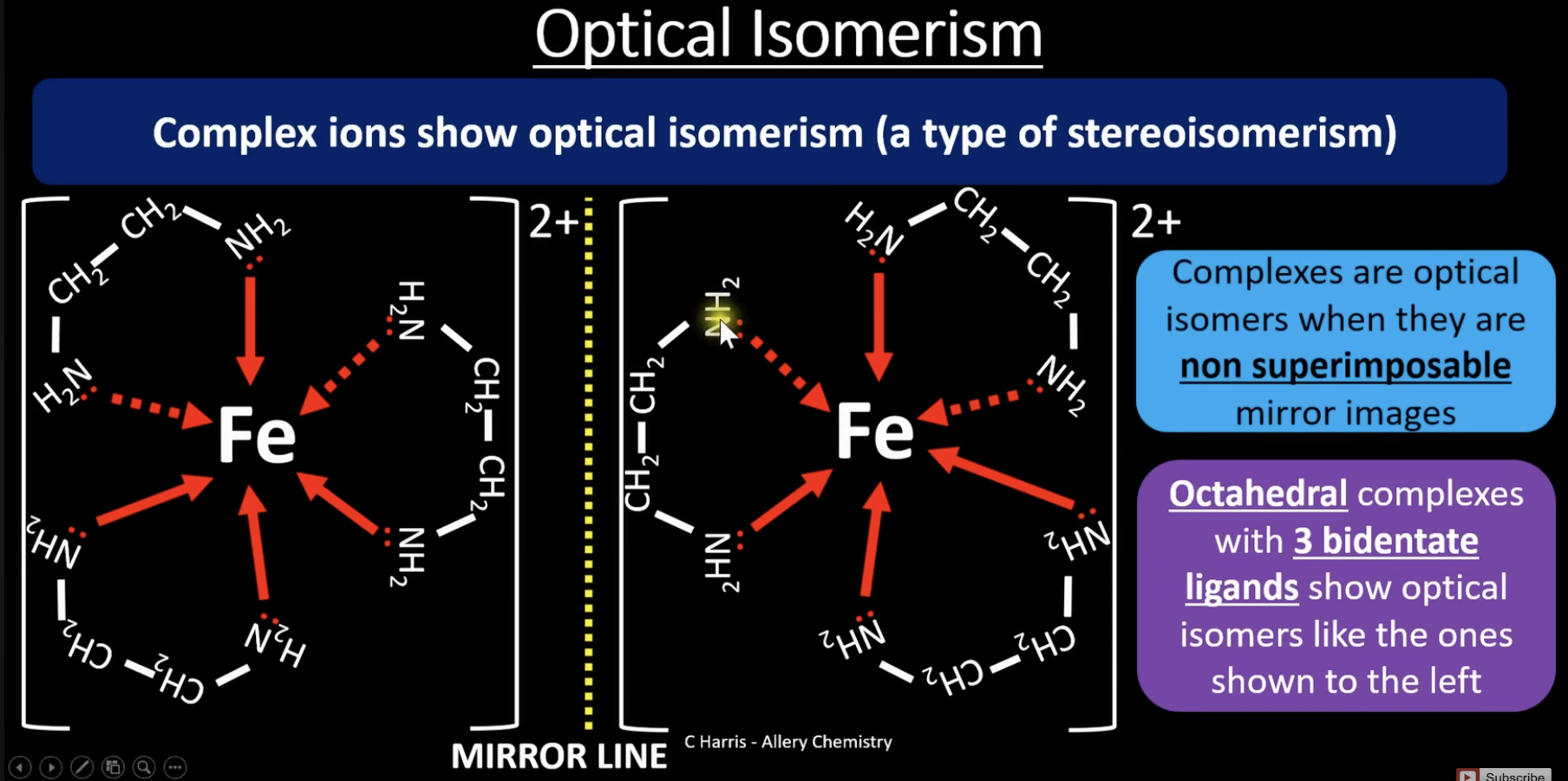
optical isomerism
optical isomerism = a type of stereoisomerism where molecules have the same structural formula but are non-superimposable mirror images of each other
non superimposable = they cannot be placed exactly on top of each other so that everything matches perfectly
octahedral shapes with 3 bidentate ligands show optical isomerism (image)
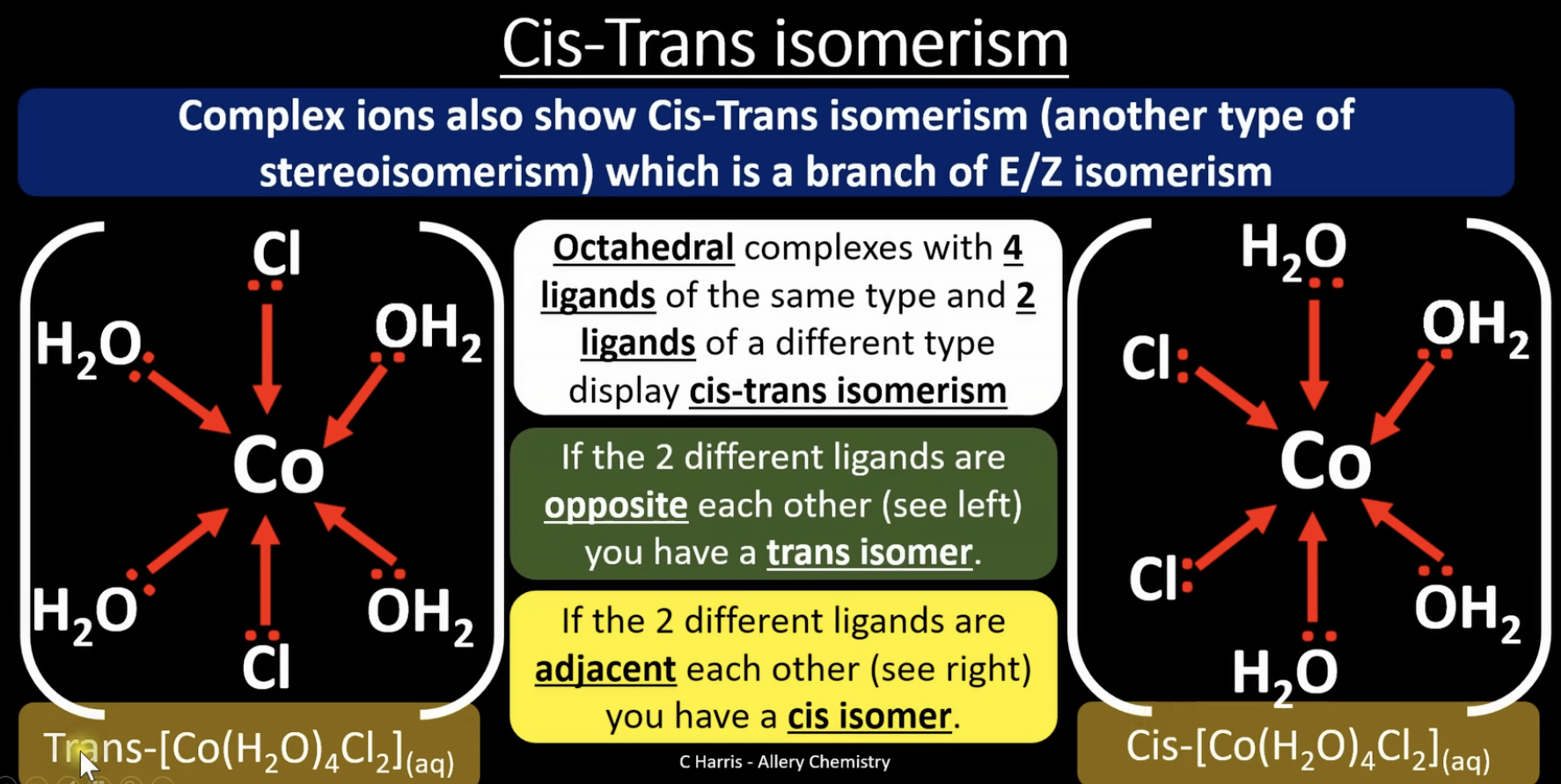
cis trans isomerism
cis-trans isomerism = molecules have the same structural formula but different arrangement in space around a rigid bond/structure
octahedral complexes with 4 of the same ligands and 2 other different ligands display cis-trans isomerism
if the 2 different ligands are opposite each other you have a trans isomer
if the 2 different ligands are adjacent each other you have a cis isomer
square planar complexes with 2 of the same ligand and 2 other different ligands display cis-trans isomerism
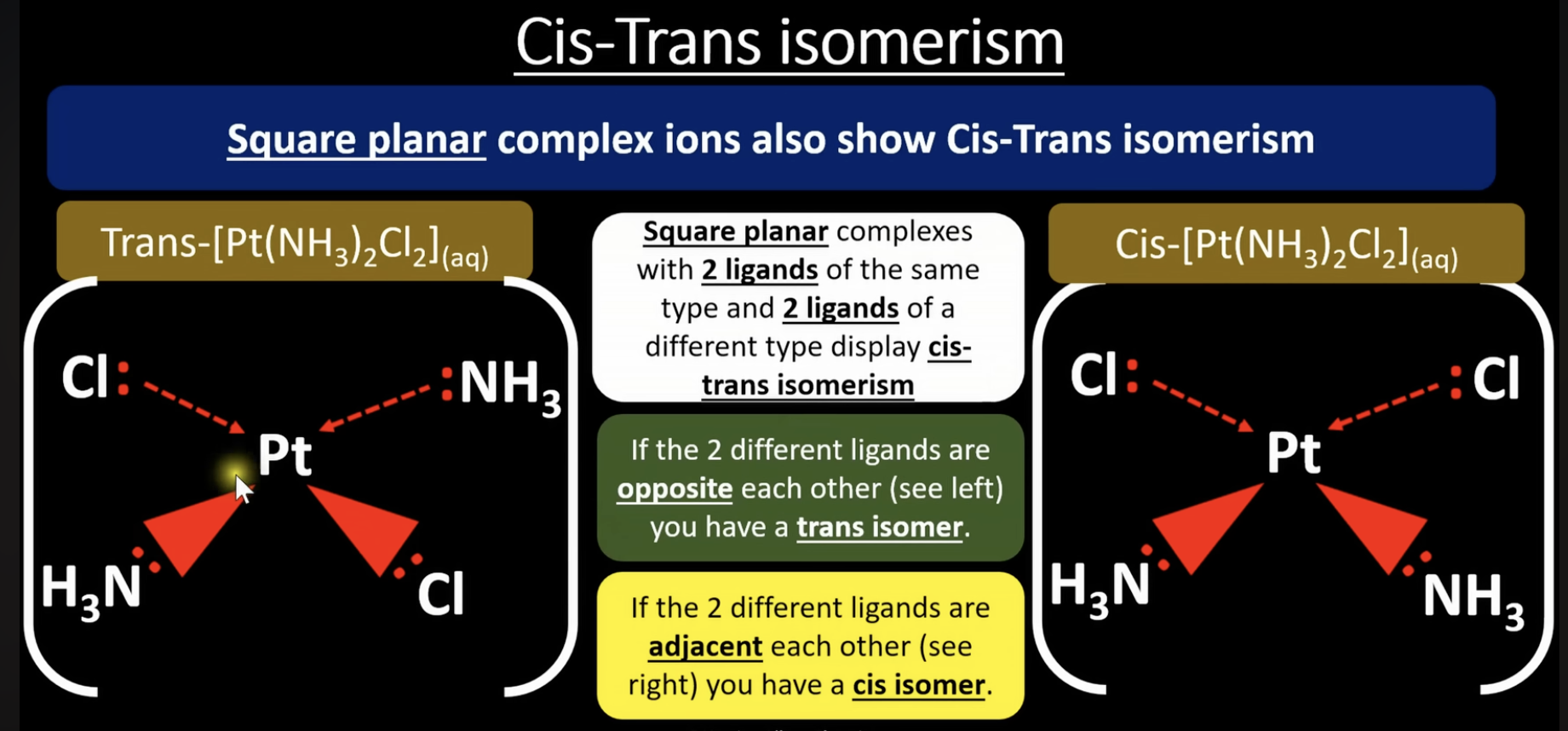
cis trans isomerism 2
square planar complexes with 2 of the same ligand and 2 other different ligands display cis-trans isomerism
if the 2 different ligands are opposite each other you have a trans isomer
if the 2 different ligands are adjacent each other you have a cis isomer
example: cisplatin cures cancer, transplatin cannot
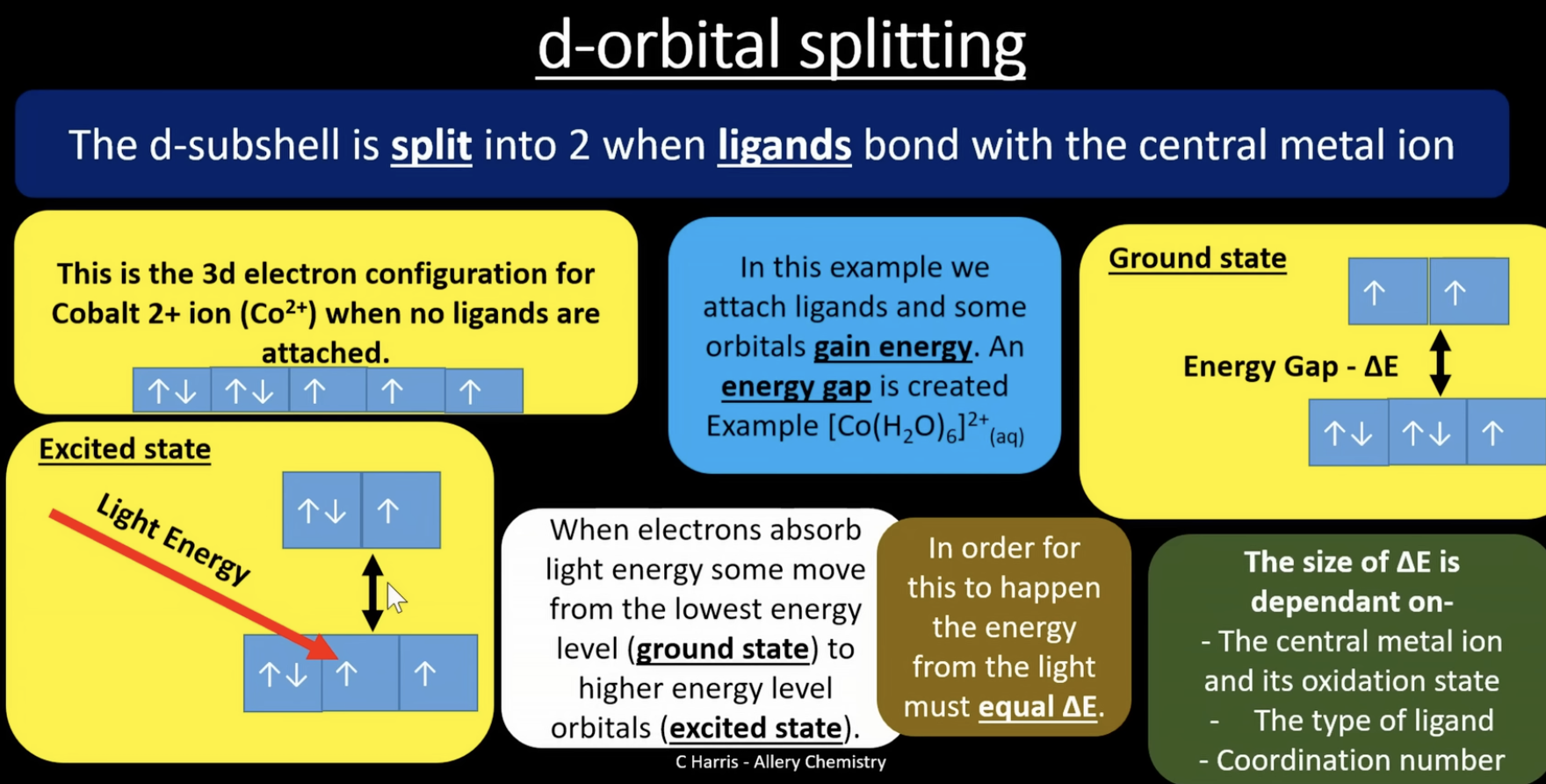
d-orbital splitting
when a ligand bonds onto the central atom, the d-subshell splits into 2. when the ligands attach some orbitals gain energy, creating an energy gap
when electrons absorb light energy some move from the lowest energy level (ground state) to a higher energy level orbital (excited state). for this to happen, the light energy must equal ^E
the size of ^E is dependant on: (calculation in next flashcard)
-the central metal ion and its oxidation state
-the type of ligand
-coordination number
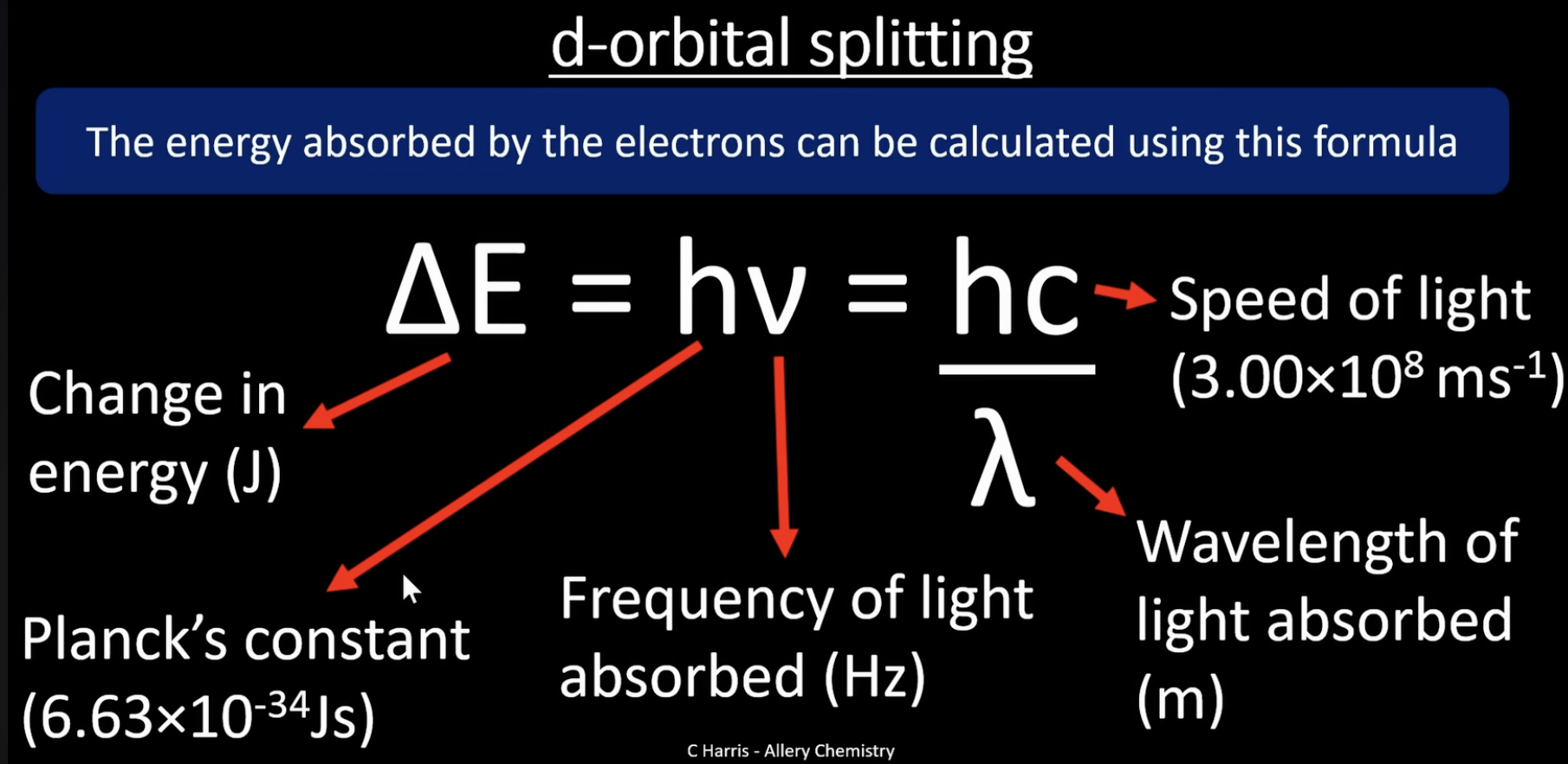
calculating ^E = change in energy
this is the formula for the energy absorbed by electrons
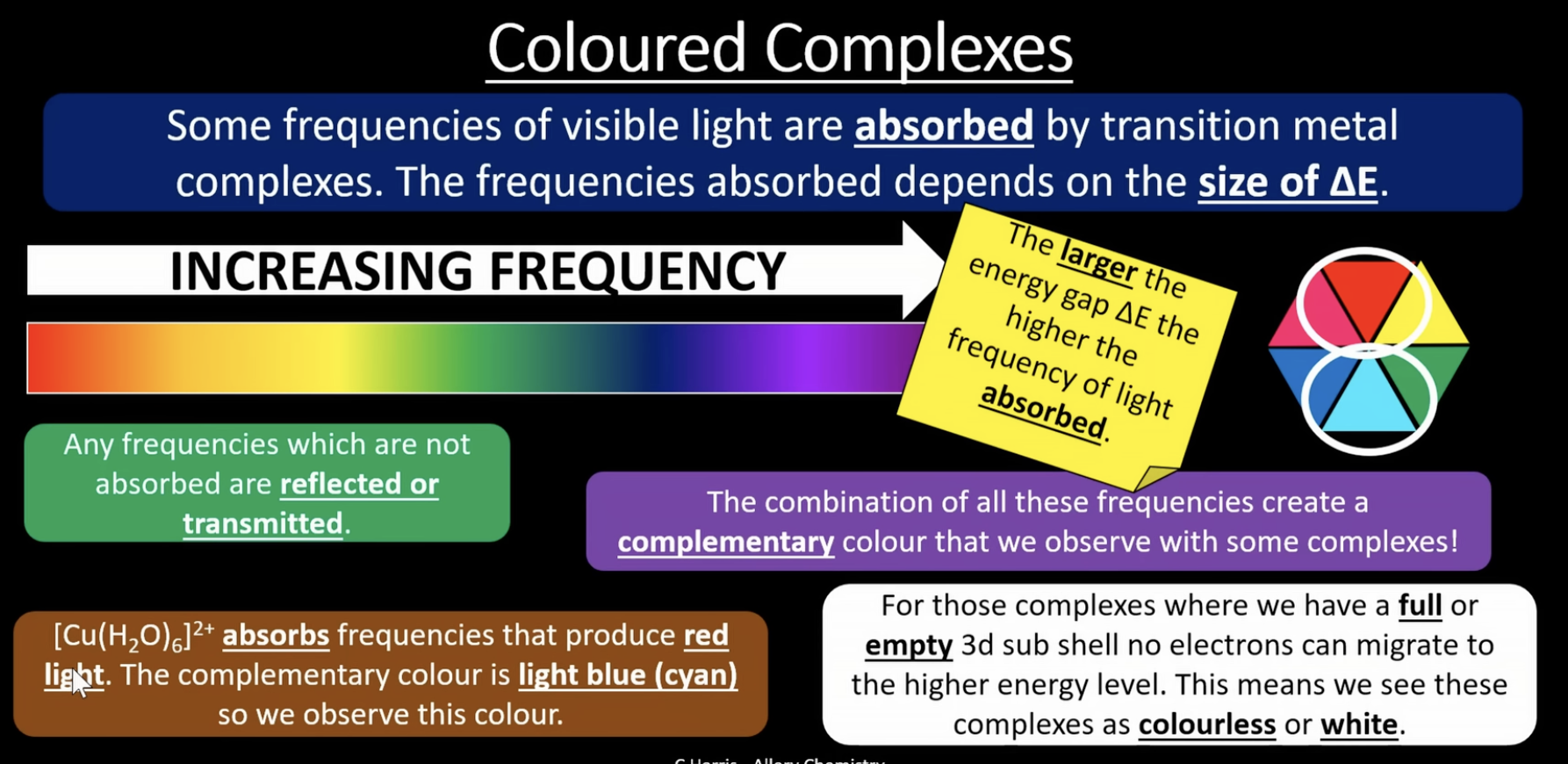
coloured complexes
the larger the change in energy ^E, the higher the frequency of light absorbed
any frequencies which are not absorbed are reflected or transmitted
the colour observed is the complementary colour to the colour/frequency of light absorbed
transition metal complexes are coloured because ligands cause the d orbitals to split into different energy levels. when the electrons absorb visible light energy and get excited, a colour change occurs. this explains why Sc and Zn are not transition metals and do not produce this colour change
colour is dependant on:
-the type of ligand
-the shape of the ligand
-the oxidation state of the central metal ion
ligand substitution
ligand substitution can change:
-colour
-coordination number
-shape
colour changes because different ligands change the d-orbital splitting (ΔE)
memorise the second one for Cu

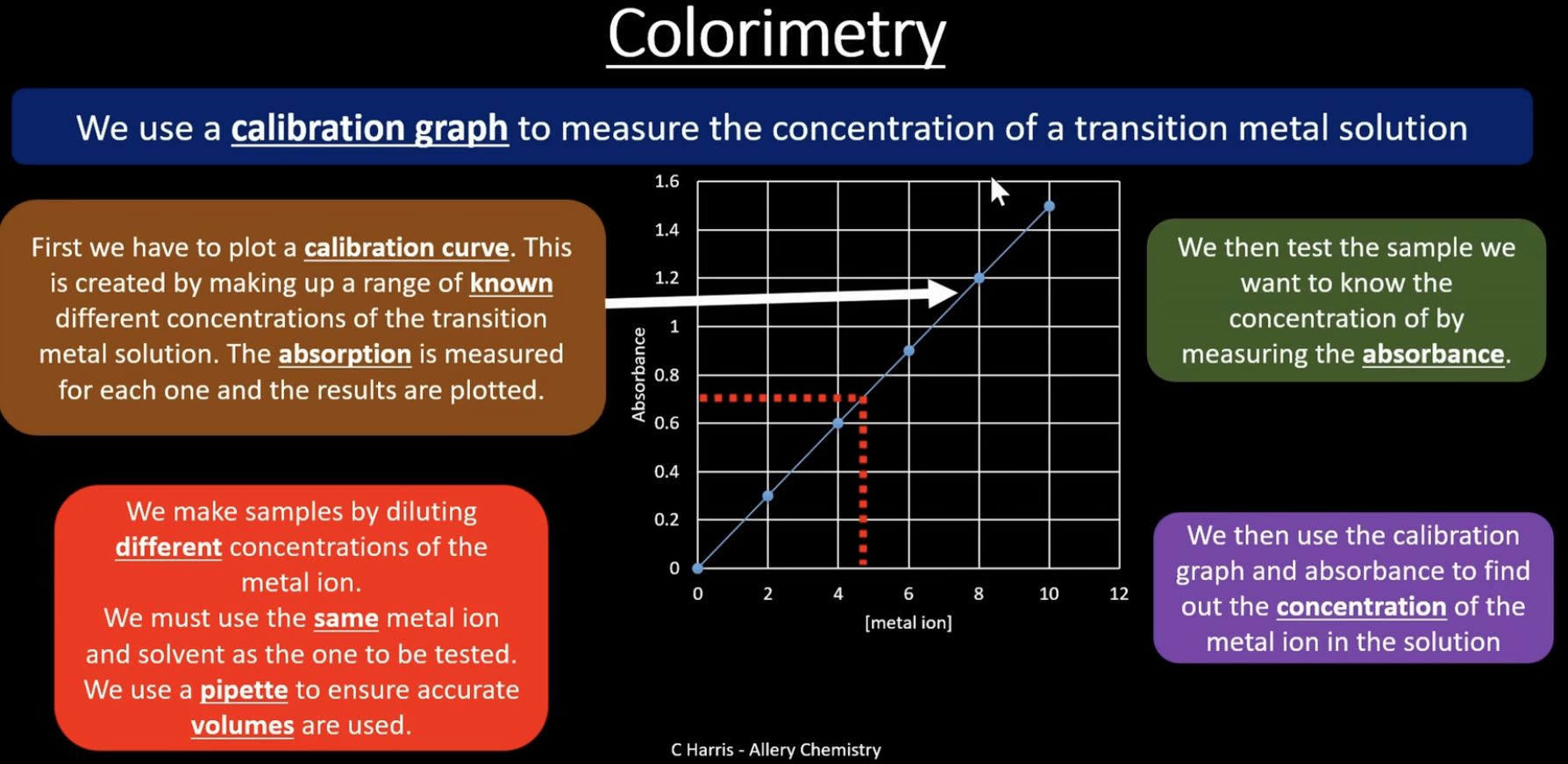
colorimetry
monochromatic light = light of a single wavelength/ frequency, so one colour of light only
the calorimeter must be set to zero. to do this we measure the absorbance of a blank sample, normally the solvent you are using to dissolve your transition metal ion (water). white light is filtered into a narrow range of frequencies to produce monochromatic light (single colour). the sample is held in a vessel called a cuvette. the monochromatic light passes through the sample and some the light is absorbed. light not absorbed travels to the detector. the detector will measure the level of absorbance by comparing it to absorbance in the blank sample
more concentrated sample = more light absorbed = darker colour

calibration curve
plotting concentrations of known solution to work out the concentration of an unknown
more ligand substitution
memorise the second one
is in excess ammonia
ligands of a similar size are being exchanged
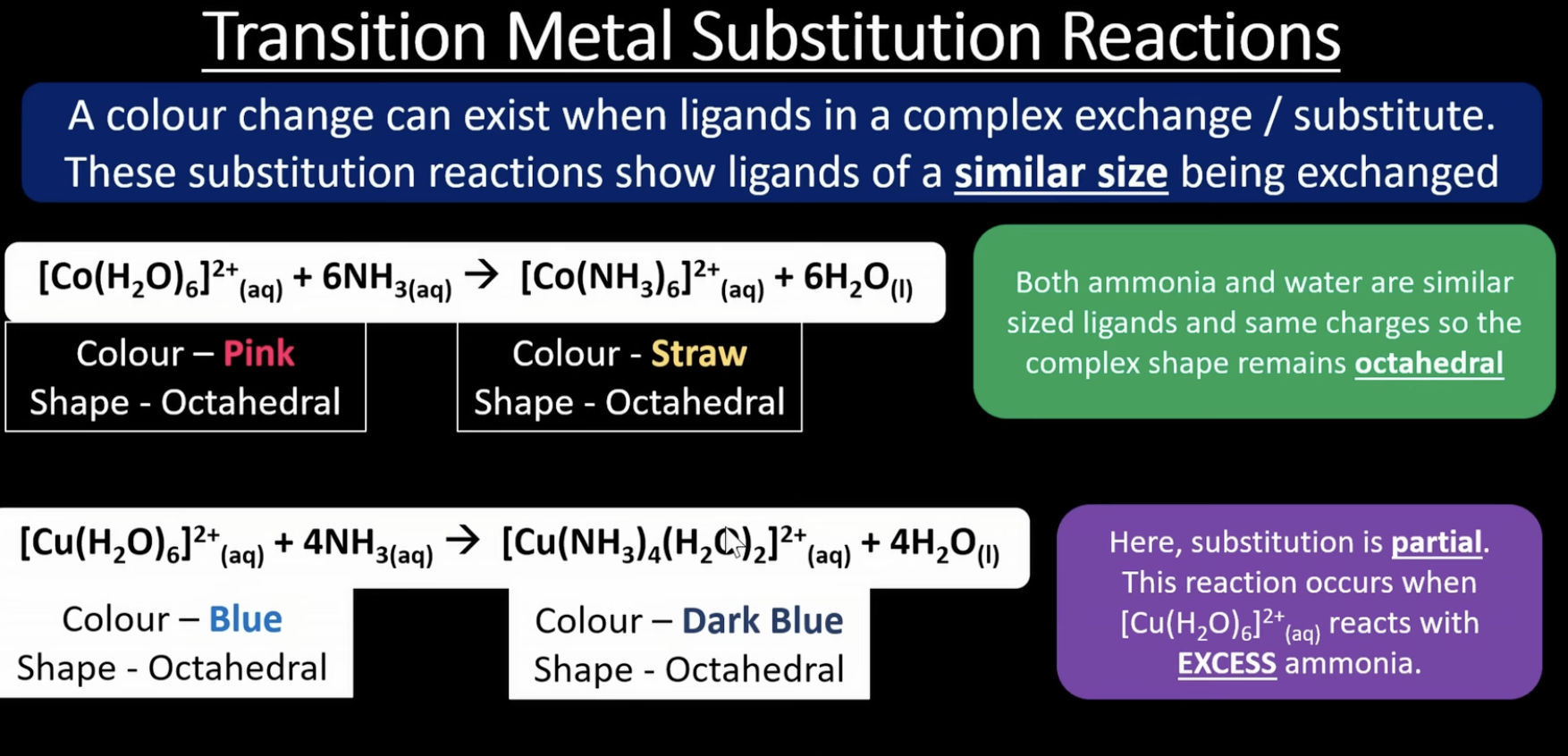
more ligand substitution 2
larger ligans replacing smaller ones

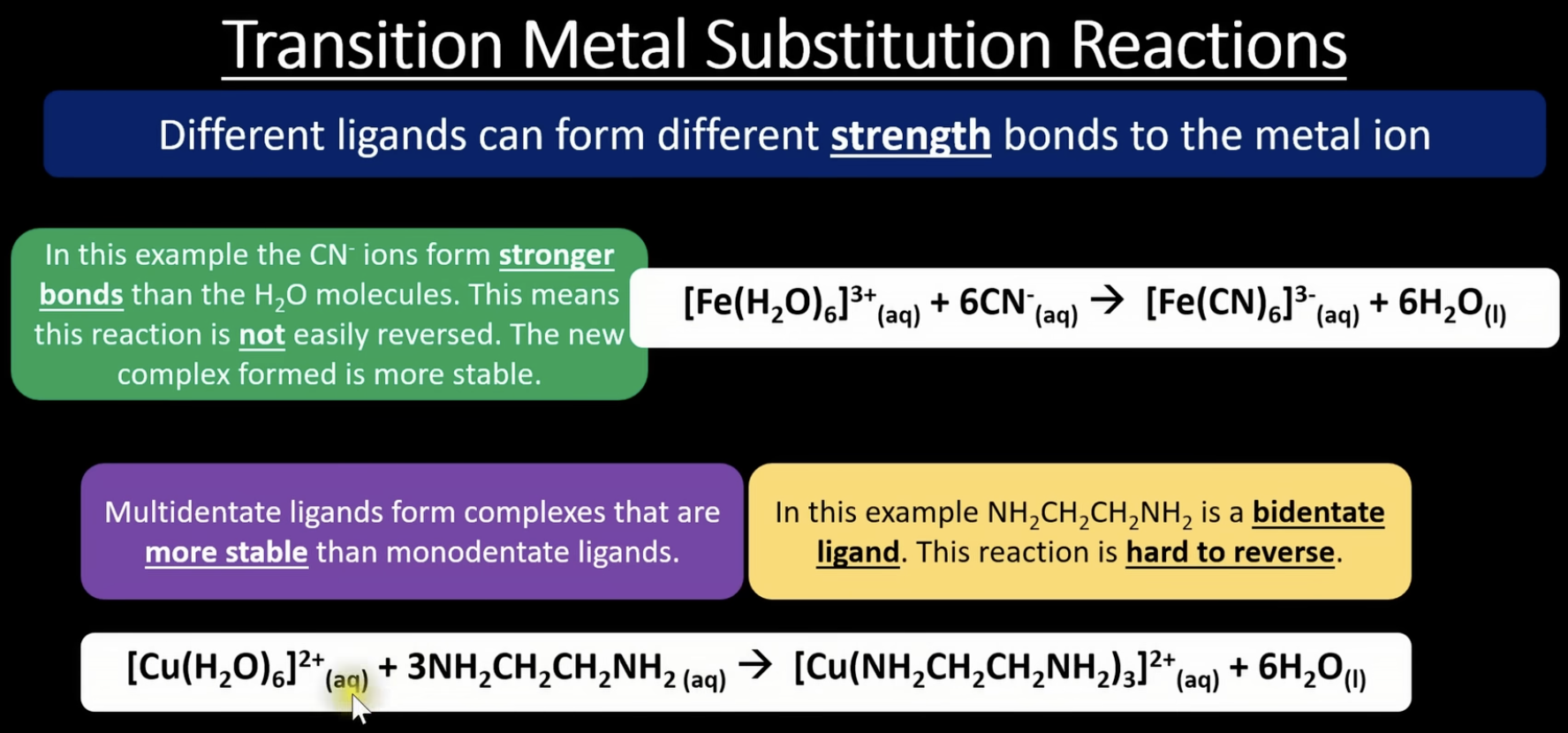
more ligand substitution 3
when ligand substitution occurs, some ligand-metal bonds are stronger than other, which means its harder to reverse some reactions
multi dentate ligands form more stable complexes than monodentate
entropy in ligand substitution
increased entropy helps favour formation of a more stable complex
often the energy needed to break the bonds is similar as the energy released when new ones are formed, so the enthalpy change (AH) is small like the reaction in the image

entropy in ligand substitution 2
chelate effect = multidentate ligands form more stable complexes than equivalent monodentate ligands. this is because of an increase in entropy
when we substitute monodentate ligand with bidentate or multidentate ligand we create a solution with more particles in it = increase in entropy. reactions that have an increase in entropy are more likely to happen. it's difficult to reverse these reactions as this would mean a decrease in entropy
multidentate ligands bond to the metal ion and replace several smaller ligands already attached, this releases more free molecules into the solution = entropy

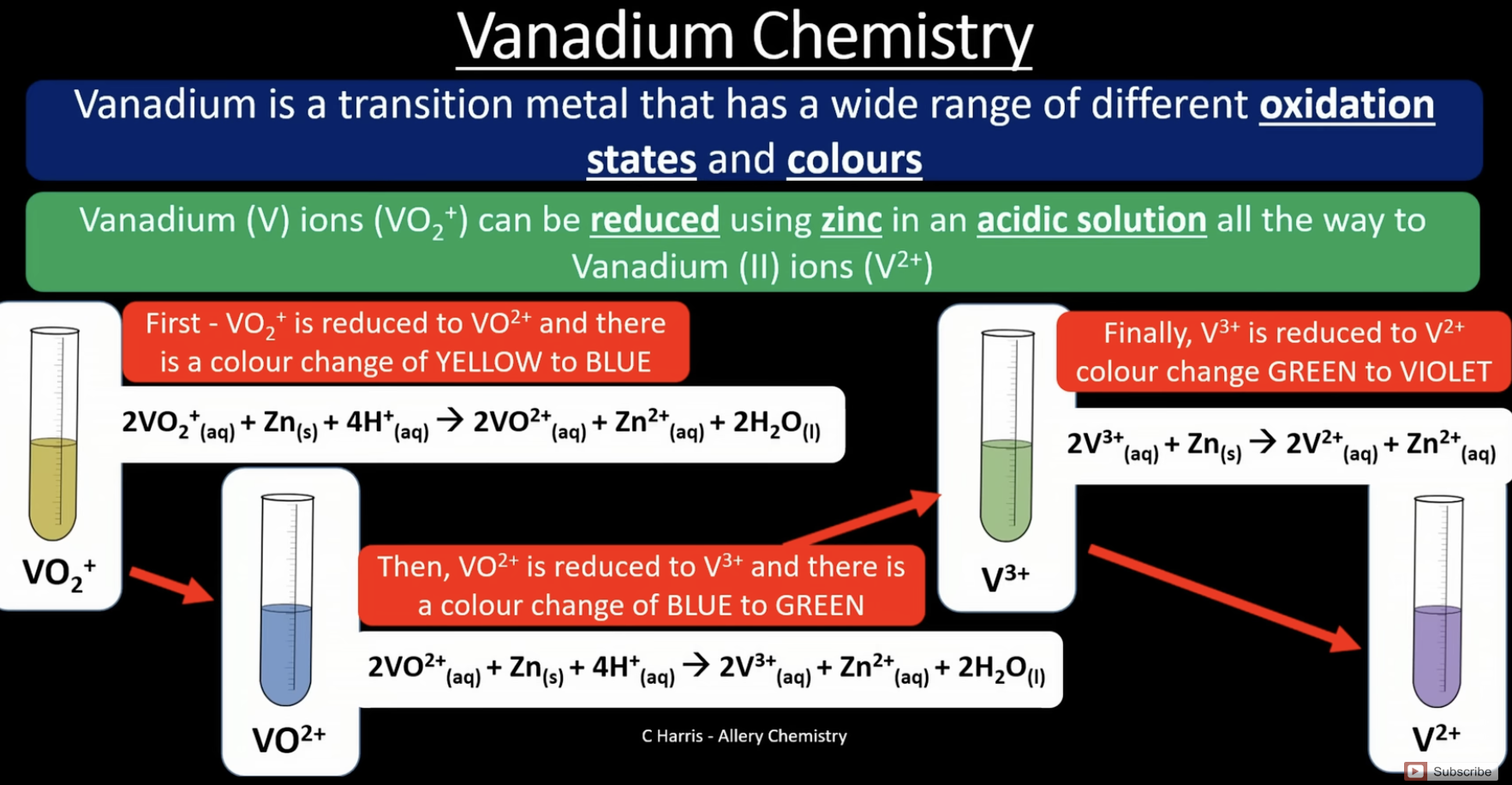
vanadium
has a wide range of oxidation states and colours
when transition metals change oxidation state a redox reaction occurs
memorise those colours

vanadium reactions - reduction using zinc
VO2+ (+5) to V2+(+2)
VO₂⁺ → VO²⁺ → V³⁺ → V²⁺
yellow → blue → green → violet
+5 → +4 → +3 → +2
redox potentials
redox potentials tell us how easily an ion is reduced which is the same as electrode potentials. the least stable ions have the largest redox potential and are more likely to be reduced
must have:
-temperature at 298K
-pressure at 100kPa
-concentrations of ions at 1 moldm-3
there may be a difference in redox potential to the standard values seen in a data book. it is dependent on the environment the ions are in

what affects redox potentials
ligands:
the standard electrode potentials is always measured in aqueous solutions. the metal ion is surrounded by water molecules. however ligands other than water can form stronger bonds to the metal ions with different oxidation states. this means the redox potential can be higher or lower than the standard value
pH:
the pH of the solution affects the size of redox potentials with some reactions. the more acidic the solution the larger the electrode potential. this means the ion is more easily reduced. pH affects redox potentials because H⁺ ions are involved in some redox equilibria. changing the H⁺ concentration shifts the equilibrium and changes the electrode potential

tollens reagent
[Ag(NH3)2]+
formed by ligand substitution
Ag⁺ is the metal ion
NH₃ ligands replace water ligands
redox titrations
redox titrations are used to work out the titration of a reducing or oxidising agent
have an oxidising/reducing agent in burette with a known concentration. have your reducing/ oxidising agent solution with an unknown concentration but known volume in conical flask. add excess dilute sulfuric acid into flask too, to ensure you have sufficient H+ ions to allow the reduction of the oxidising agent. add the ions in the burette to the conical flask until you see the faint colour appear = end point. add drop by drop. record your results to 2 decimal places and repeat until you get 2 results that are concordant (within 0.10cm of each eachother)
make sure in titration to record values with 2dp

types of catalyst
heterogenous:
a catalyst that is in a different phase/ state from the reactants
example: haber process
N2(g) + 3H2(g) > 2NH3(g). a solid iron catalyst is used for this reaction which is a different state the gaseous reactants
Increasing the surface area of the heterogeneous catalyst will increase the rate of reaction. More particles can react with the catalyst at the same time.
homogenous:
a catalyst that is in the same phase as the reactants
generally homogeneous catalysts are aqueous in aqueous reactants. for example using sulphuric acid to make an ester.
homogeneous catalysts forms intermediate species by reactants combining with the catalyst. the catalyst is reformed by the end

the contact process
the contact process uses vanadium (V) to make sulfuric acid. in the manufacture of sulfuric acid we use V2O5 (Vanadium V) as a catalyst. it's used to catalyse SO2 to SO3. this is a heterogeneous catalyst
first V2O5 oxidises SO2 to SO3 and is itself reduced to V2O4:
V2O5 + SO2 → V2O4 + SO3
second V2O4 is oxidised by oxygen in the air to reform the catalyst V2O5:
V2O4 + ½ O2 → V2O5

poisoned heterogenous catalysts
impurities can bind to the surface of a catalyst and block the active sites for reactants. when an impurity blocks a site we call this poisoning. catalytic poisoning reduces the surface area of the catalyst for the reactants bind to. this slows down the reaction
a poisoned catalyst means
-less product is made
-the catalyst needs to be replaced or cleaned more often
-increased cost of the chemical
in the harber process the hydrogen is made from methane which contains sulfur impurities, which can form iron sulfide and block the iron catalyst less effective

homogenous catalyst energy diagram
has two peaks for activation energy because the reaction occurs in more than one step. the catalyst reacts with the reactants to form an intermediate. each step has its own activation energy
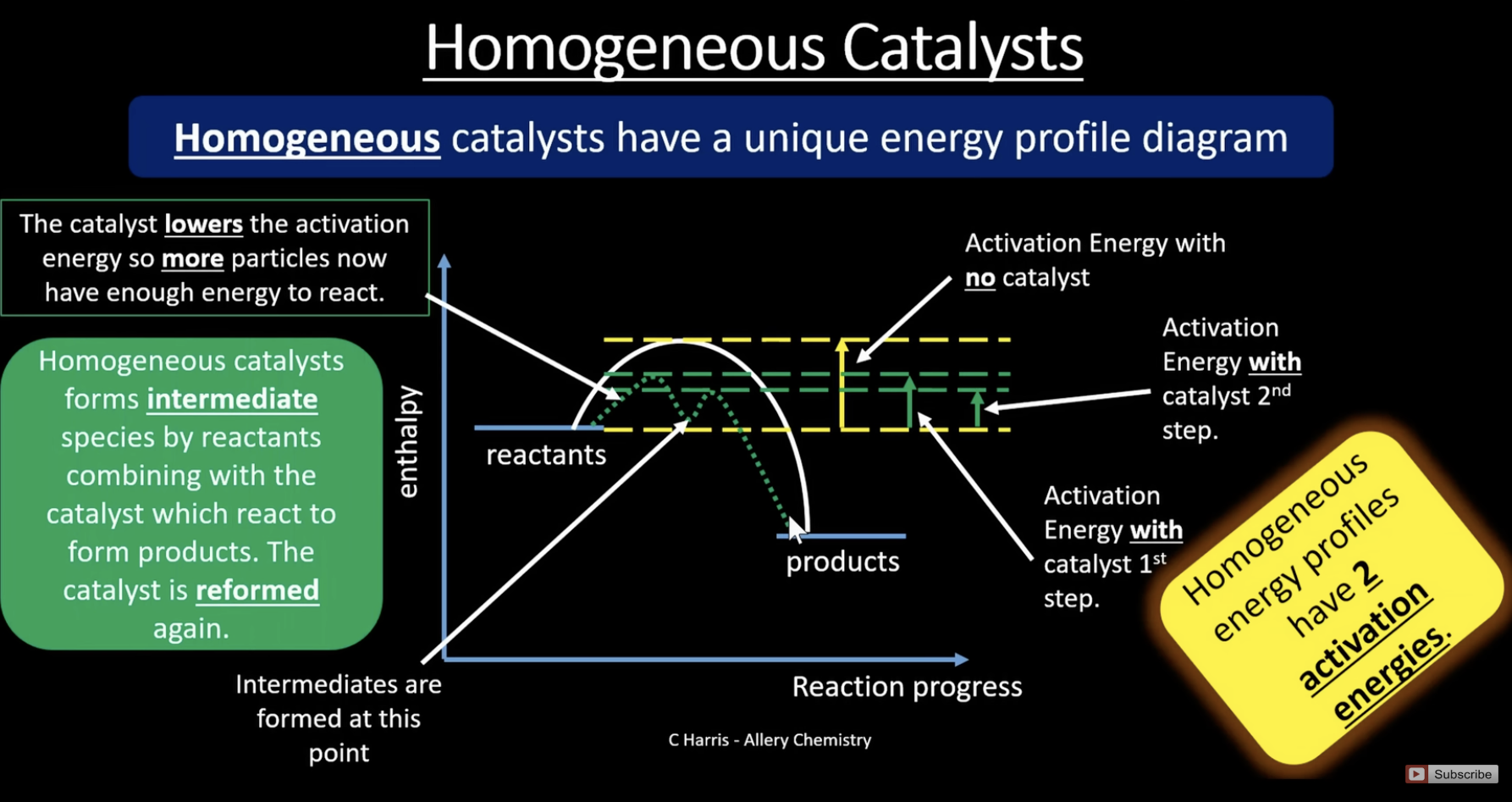
example of homogenous catalyst pathway
the original reaction is very slow because it involves 2 negative ions reacting and they repel, so Fe2+ is used as a homogenous catalyst to react peroxodisulphate S2O82- and iodide ions. Fe2+ is oxidised to Fe3+, which is the intermediate formed and S2O82- is reduced to SO42-
Fe3+ then reacts with iodine ions in the second step to reform the catalyst

autocatalysis
autocatalysis is another form of homogeneous catalysis where the product catalyses the reaction/ it catalyses itself
an example of autocatalysis is where Mn2+ is the catalyst in the reaction between C2O42- and MnO4- . Mn2+ is a product and a catalyst
the original reaction is very slow because it involves 2 negative ions reacting and they repel. Mn2+ catalyses the conversion of MnO4- to Mn3+. then Mn3+ reacts with C2O42- and Mn2+ are reformed
