OCHEM NAGORSKI EXAM 4 REAGENTS
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
How do i determine E/Z?
E is trans, Z is cis!
How to find degrees of unsaturation:
Determine # of C’s (nC), Halogens(nX), and Nitrogens(nN).
The number of Hydrogens (Saturated) would be
= 2nC + 2 -nX + nN
Then divide (Hsat-Hactual)/2 = Degrees of unsaturation
If you see a double bond → H2, and PdC/PtO2/Ni/Ra-Ni
Catalytic hydrogenation: adds a Hydrogen across a O=C Bond or C=C double bond,
is stereospecific, so both hydrogens will go to one face of the bonds. either cis wedge or cis dash.
If you see a double bond → HX
Addition of the halogen, will go to the most substituted side. (Markovinikov addition)
Double bond → HX over H2O2 and light/heat
Anti-markovinikov Addition, where the halogen is added to the LEAST substituted end.
Double bond → just H20 over acid
acid removes its proton, then H+ attacks the hydrogen on the double bond, then H2O is added to the carbocation, then you have a hydronium ion, which then loses a proton.
Joins in a markovinikov fashion, where the OH joins the most substituted end.
Double bond → H2SO4 then → H2O
Addition of a HSO4 LG, which is a great LG, can be worked up with H2O and make an alcohol.
Double bond → BH3 or THF → H2O2/H2O
Anti-markovinikov reaction where the H2O2 leaves an OH on the LEAST subtituted side
if you see a CH-CH bond with a Halogen and an R group on both C’s → Base (2 parts NaNH2/NH3 liquid) then → H2O
Creates a C-C triple bond, as one halogen leaves, makes a double bond, then the very strong base NaNH2 will take the proton, then the other halogen leaves, and the second NaNH2 takes that proton, leaving you with a c-c triple bond.
What if you se a c-c bond with a halogen on each end, but → a weaker base like NaOCH3 over HOCH3?
This will stop at the formation of a c=c rather than a c-c triple bond.
IF i start with a c-c triple bond → NaNH2 over NH3 liquid?
This will deprotonate one side of the c-c triple bond, leaving a carbocation intermediary which can be substituted by a second step.
If I see a C-C triple bond → H2 over PtO2 (Adams catalyst), Pd-C, Ra-Ni with a solvent like methanol, acetic acid, or ethyl acetate.
this will cause the C-C triple bond to be hydrogenated, making it a c-c single bonded alkane.
What if a see a c-c triple bond → H2/Lindlar’s Catalyst?
This stops at the alkene, or c=c bond. The hydrogen are added in a cis fashion, meaning any substituents are also in the Cis
c-c triple bond → Na*, NH3 Liquid (Sodium radical) then → H2O
This stops at a c=c double bond, but in a trans fashion in the final product
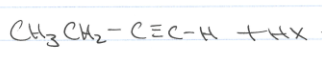
What if I see a c-c triple bond → 1HX?
Ex, Ch3CH2C-CH where C-CH is a triple bond
The halogen will attatch to the most substituted end of the c-c triple bond, ie Ch3-CH2-X-C=CH2.
both cis and trans products will be observed.
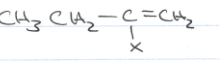
What if I see a c-c triple bond reacting with excess HX?
the halogen will substitute to the same side of the c-c triple bond twice!
What if I see a c-c triple bond → H+ with H2O solvent?
one of the carbons on the c-c triple bond will become a ketone alpha carbon (C=O with two R groups on the C), and the other becomes a CH2.
What if I see a terminal c-c triple bond, → H2O/H+ over HgSO4?
Then a R-C=O-CH3 will form
what if I see an internal c-c triple bond →H2O/H+ over HgSO4?
you get equal parts of a =O on each of the carbons across the triple bond

what if I see R-C-=-C-R’ triple bond → 1) Ozone 2)H2O?
The ozone will cleave the triple bond which then creates R-COOH (Carboxcylic Acid) or COOH-C-R’, where it clips one side of the triple bond off and replaces it with a COOH group.
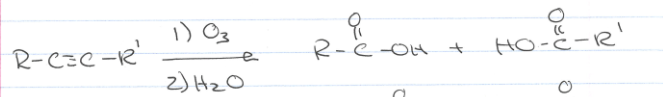
What if I see a Primary Haloalkane → H2O ?
The Halide leaves and then forms an alcohol.
what if I have a strong leaving group on a secondary carbon with chirality → H2O
The leaving group is replaced by OH, and you see a racemixation of each.
which is the most oxidized; CO2, COOH, H2C=O, CH3OH, CH4?
CO2 is the most reduced.
What if I see a O=C double bond → H2 over a catalyst (PtO2, Pd-C, or Ni-C) ?
you have an addition of H on the Carbon, leading to a C-OH bond
What if I see a Ketone and Aldehyde → 1) NaBH4/EtOH 2)H+/H2O ?
Fully reduces ketones and aldehydes to an alcohol, but cannot reduce COOH groups.
What if I see a ketone, Aldehyde, or Carboxcylic Acid → LiAlH4/EtOEt then H+/H2O
It will full reduce them to an alcohol
What if i want to oxidize a molecule?
Use a chromium reagent, such as K2Cr2O7, Na2Cr2O7, or CrO3
What if i see a secondary alcohol (2R-CH-OH) → Na2Cr2O7/H2SO4/H2O
This will oxidize the secondary alcohol into a O=C
What if I see a primary alcohol (R-CH2-OH)→ K2Cr2O7 over H2SO4/H2O?
The OH will oxidize to a COOH.
What if I see a primary alcohol → PCC?
This will cause the oxidation to stop at a R-CH=O (aldehyde) rather than all the way to a COOH.
What will KMnO4 do?
Very strong oxidizer, similar to chromium reagents.
2* alcohols → Ketones
1* alcohols → COOH