Lecture 17: amine and nucleotide metabolism
1/165
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
166 Terms
Utilizing amino acids in the cell:repurposing _____/____ backbones for specialized functions
carbon/nitrogen
Repurposing carbon/nitrogen backbones of amino acids to make
heme, biological amines, nucleotides
Specialized nitrogen molecules require ____
repurposing
Problem: with nitrogenous rings for light absorption, building them is
expensive
Solution to the expensive process of building nitrogenous rings
utilize simple nitrogen-containing compounds to build repeated structures
Nitrogenous ring synthesis: step 1: Build ___
a pyrolle from metabolic intermediates
Nitrogenous ring synthesis: step 2: Link 4 together to form a ___ ____
linear tetrapyrrole
Nitrogenous ring synthesis: step 3: Close the ring to form ____
porphyrin
Nitrogenous ring synthesis: step 4 Load specific ___ ___ to the center of the porphyrin ring
metal ion
Porphyrins
strongly absorb light in the visible spectrum
Porphyra geek word for
purple
Most abundant porphyrin in vertebrates
heme
Tetrapyrroles are derived from ___ and ___
glycine and succinyl-CoA
Aminolevulinate
dimerized then tetramerized to form a tetrapyrrole
Defects of enzymes in the biosynthetic pathway lead to ___
buildup of precursors
Porphyria
diseases linked to toxic intermediate building in erythrocytes, body fluids, liver (lacking one of 8 enzymes to build heme)
Symptoms of porphyria include
abnormal sensitivity to light, anemia (low iron in blood), receding gums, fluorescent teeth, dark red urine
Degradation of heme (black/purple) yields ___ and ___
biliverdin (green) and bilirubin (yellow)
when heme is broken down what happens to Fe2+
recycled
Degradation of heme is the only reaction in the human body producing ___
CO
Biliverdin and bilirubin removed as
waste
Bilirubin characteristics
highly non-polar, pyrrole rings are very stable (have to invest too much to recapture stored energy)
Bilirubin characteristics: pyrrole rings
very stable
Bilirubin
potent antioxidant in the blood
Buildup of bilirubin causes ___
yellowing of the eyes and skin (jaundice)
bilirubin is not
soluble
Attach ___ ____ _____ to make bilirubin more soluble
two glucuronic acids
To breakdown bilirubin in bile: gut microbiome digests to ___
stercobilin
Bilirubin breakdown: most stercoblinin (90%) removed in ___
feces (red-brown color)
Bilirubin breakdown: remaining 10% oxidized to ___
urobilin in urine (yellow color)
Amino acids are direct precursors to ___
biological amines
Biological amines
generated by decarboxylation of amino acids
Biological amines function
hormones and/or neurotransmitters
examples of biological amines
dopamine, norepinephrine, epinephrine, histamine, serotonin, GABA
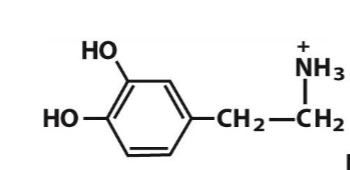
dopamine
norepinephrine
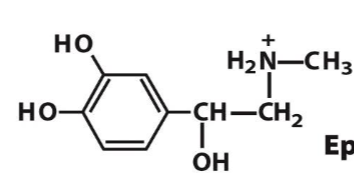
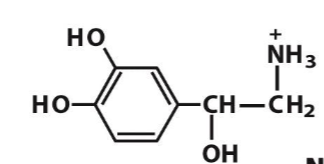
epinephrine
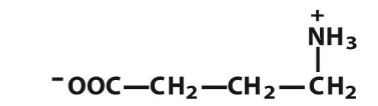
GABA
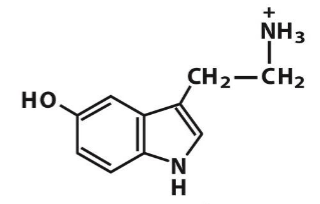
serotonin
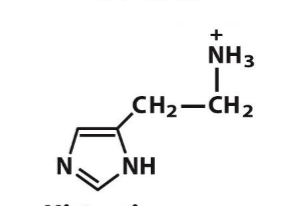
Histamine
PLP (Vitamin B6)
cofactor for transamination and decarboxylation
PLP acts as ___
electron sink
Different enzymes use PLP to attack ___
different functional groups
Tyrosine is a hub for
catecholamines (for making neurotransmitters)
Dopamine is a
neurotransmitter
neurotransmitter
chemcial signal that facilitates neuronal communication
Parkinson’s patients lose ___
dopamine-producing neurons
used to treat Parkinson symptoms
L-DOPA
Tryptophan and glutamate yield ___
distinct neurological signals
Serotonin and GABA
neurotransmitters
Histidine becomes ___ during immune response
histamine
Histamine
triggers vasodilation and allergic responses
Antihistamines
block histamine receptors
Seterotin and GABA are also
neurotransmitters
first step in polyamine synthesis
decarboxylation
Long, positively charged molecules that stabilize the negative backbone of DNA and RNA
Polyamines
Polyamines stablize the ___ ___ of DNA and RNA
negative backbone
DFMO inhibits ___
ornithine decarboxylase
Take Home: amino acids are not just for protein synthesis or energy; their chemical structures serve as the backbone for
communication and oxygen-transport networks
Nucleotide monophosphates
monomers of nucleic acids
ATP is an example of a
nucleotide
Nucleotide
nitrogenous base, pentose sugar, phosphate group
Nucleoside
nitrogenous base and pentose sugar (NO PHOSPHATE GROUP)
Alphabet for biological information includes
five nitrogenous bases
Five nitrogenous bases
adenine, guanine, cytosine, thymine, uracil
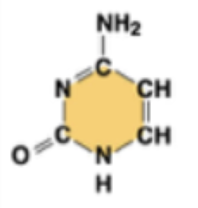
cytosine (C)
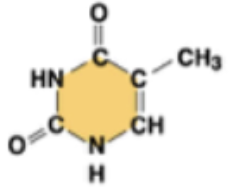
thymine (T, in DNA)
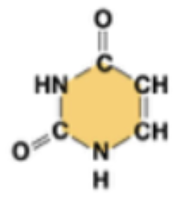
uracil (U, in RNA)
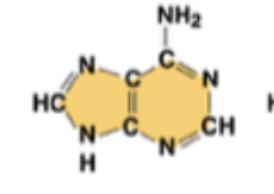
adenine (A)
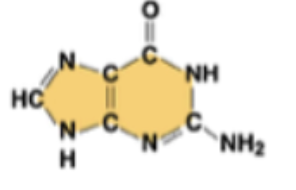
guanine (G)
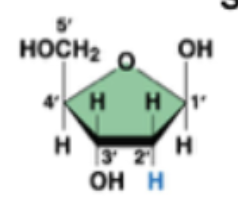
deoxyribose (in DNA)
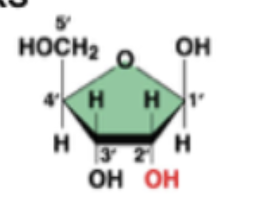
ribose (in RNA)
PRPP
donor of ribose-phosphate unit
PRPP formed from ___
ribose-5-phosphate and ATP
Ribose-5-phosphate must be activated at
C1
Sugar First: bases are built stepwise onto PRPP
purine
Base First: precursor base synthesized then added to PRPP
pyrimidine
Purine biosynthesis ___ first
Sugar
purine is made up of
co2, aspartate, formate, glutamine, formate, glycine
Purine ring synthesized onto ___
ribose
Purine ring derived from ___
amino acids, N10-Formyl-THF, CO2
Synthesis of inosinate costs ___
5 ATP
___ and ___ are synthesized from IMP
AMP and GMP
Inosinate (IMP) is a ___ point
branch point
AMP requires ___
GTP hydrolysis
GMP requires ___
ATP hydrolysis
Relying on the opposite NTP for energy naturally
balances the pools
purine biosynthesis: inhibit the commitment step of the common pathway
IMP, AMP, GMP
___and ___ inhibit the entry point enzymes for their specific synthesis from IMP
AMP and GMP
Purine production remains ___so the pools are relatively equal for DNA and RNA synthesis
balanced
Uracil is the ___ _____ (point)
entry point
uracil: entry point modified to form ______ or _______ (___)
cytosine or thymine (DNA)
Pyrimidine biosynthesis ___ first
Base
Pyrimidine ring synthesized as ___ ____
base precursor
Pyrimidine ring derived from ___
carbamoyl phosphate and aspartate
Carbamoyl phosphate synthetase I (CPS I) is in the
mitochondrial matrix
Carbamoyl phosphate synthetase II (CPS II) found in
cytosol
CPS II: ____ never leaves the enzyme
ammonia
CPS II structure
a large channel links active sites
pyrimidine biosynthesis: what is the precursor base
orotate