a level inorganic
1/186
Earn XP
Description and Tags
USE THIS FOR NOTES !!! i cba to update other fcs from before combining
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
187 Terms
finish the eqn: acid + base →
acid + base → salt + water
give and explain the eqn for the reaction between sodium oxide and hydrochloric acid:
Na2O + 2HCl → 2NaCl + H2O
Na2O is basic - neutralises HCl (acid)
give and explain the eqn for the reaction between magnesium oxide and sulfuric acid:
MgO + H2SO4 → MgSO4 + H2O
MgO is basic - neutralises H2SO4 (acid)
give and explain the eqn for the reaction between silicon dioxide and sodium hydroxide:
SiO2 + 2NaOH → Na2SiO3 + H2O
SiO2 is acidic so neutralises NaOH (base)
give and explain the eqn for the reaction between phosphorous (V) oxide and sodium hydroxide:
P4O10 + 12NaOH → 4Na3PO4 + 6H2O
P4O10 is acidic so neutralises NaOH (base)
give and explain the eqn for the reaction between sulfur dioxide and sodium hydroxide:
SO2 + 2NaOH → Na2SO3 + H2O
SO2 is acidic so neutralises NaOH (base)
give and explain the eqn for the reaction between sulfur trioxide and sodium hydroxide:
SO3 + 2NaOH → Na2SO4 + H2O
SO3 is acidic so neutralises NaOH (base)
give and explain the eqn for the reaction between aluminium oxide and sulfuric acid:
Al2O3 + 3H2SO4 → Al2(SO4)3 + 3H2O
Al2O3 is amphoteric but in this case neutralises H2SO4 (acid)
give and explain the eqn for the reaction between aluminium oxide and sodium hydroxide:
Al2O3 + 2NaOH + 3H2O → 2NaAl(OH)4
Al2O3 is amphoteric but in this case neutralises NaOH (base)
how does Na react with water?
floats on and moves about surface of water
fizzes and melts to form a silvery ball
eventually disappears and solution remaining is colourless
give the eqn for the reaction of Na with water:
2Na (s) + 2H2O (l) → 2NaOH (aq) + H2 (g)
how does Mg react with water?
reacts v slowly w/ water
will produce a small vol of hydrogen when left in contact w/ water for several weeks :(
give the eqn for Mg reacting with water:
Mg (s) + 2H2O (l) → Mg(OH)2 (aq) + H2 (g)
how does Mg react with water vapour? give the observation:
reacts vigorously with water vapour at temps > 100 oC (373K) in absence of air
forms a white powder and bright/white light
give the eqn for Mg’s reaction with water vapour:
Mg (s) + H2O (g) → MgO (s) + H2 (g)
give the observation and the eqn for the reaction of sodium with oxygen:
yellow flame observed, white solid formed
4Na + O2 → 2Na2O
give the observation and the eqn for the reaction of magnesium with oxygen:
burns in air - white light observed and white powder/solid formed
2Mg + O2 → 2MgO
state and explain the difference in how aluminium powder and foil react with oxygen/water:
aluminium powder reacts readily
but aluminium foil does not react as easily - has protective oxide layer which prevents reaction with oxygen and water, ∴ preventing corrosion
give the eqn for the reaction of aluminium with oxygen:
4Al + 3O2 → 2Al2O3
give the explanation and eqn for the reaction of silicon with oxygen:
finely /ed silicon reacts with oxygen when heated, forming silicon dioxide
Si + O2 → SiO2
give the observation and eqn for phosphorous reacting in limited oxygen:
phosphorous burns vigorously with a white flame
P4 + 3O2 → P4O6
give the observation and eqn for phosphorous reacting in XS oxygen:
phosphorous burns vigorously with a white flame
P4 + 5O2 → P4O10
give the observation and eqn for sulfur’s initial reaction with oxygen:
burns in air - yellow solid sulfur melts to form a red liquid
this then burns with a blue flame, forming misty fumes of a choking, pungent gas
S + O2 → SO2
give the eqn for sulfur’s further reaction with oxygen and state the conditions:
2SO2 + O2 → 2SO3
more oxygen, vanadium (V) oxide catalyst, specific temp/pressure conditions
how do halogens react w/ oxygen?
form unstable oxides
do noble gases react with oxygen?
no - inert
describe and explain the overall trends in the mpts of the period 3 oxides from Na2O → Al2O3:
increase, decrease (respectively)
overall: high mpts as form giant ionic lattices - strong electrostatic forces of attraction require a large amount of E to overcome
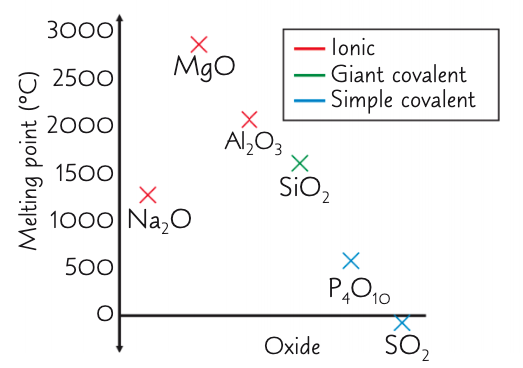
how and why does the mpt change from Na2O to MgO?
increase
Mg is smaller and has a bigger charge and so a greater charge density
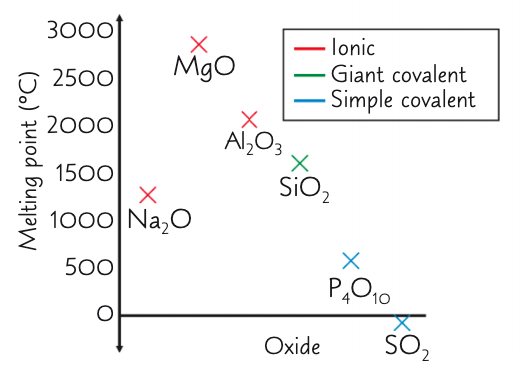
how and why does the mpt change from MgO to Al2O3?
decrease
smaller electronegativity diff between Al and O than Mg and O
so oxygen ions in Al2O3 don’t attract e- in the metal-oxygen bond as strongly as in MgO
so Al2O3 bonds are partially covalent
how and why does the mpt change when you reach SiO2?
decrease - but still higher compared to other non metal oxides
as has a giant covalent structure with strong covalent bonds which require a large amount of energy to break
describe and explain the overall trend in the mpts of the period 3 oxides from P4O10 → SO2:
decrease, decrease (respectively)
low mpts as are simple molecular structures - weak IMF: DPDP and VDWs which require little energy to overcome
how do Na2O and MgO react with water? refer to the ions and state the ionic eqn:
dissolve in water
O2- ions react with water to form OH-, forming alkaline solutions
O2- + H2O → 2OH-
give the eqn for Na2O reacting with water:
Na2O (s) + H2O (l) → 2NaOH (aq)
give the eqn for MgO reacting with water and state the additional observation:
MgO (s) + H2O (l) → Mg(OH)2 (aq) - bright white flame
are Na2O and MgO acidic, amphoteric or basic oxides? why?
basic:
react with acids (to form salts)
react w/ water to form bases
which 2 group 3 oxides are insoluble in water and why?
Al2O3 and SiO2
dessicants - absorb water into structure
is SiO2 basic, acidic, or amphoteric? what does this mean?
acidic:
reacts with bases (to form salts)
reacts w/ water to form acids
are sulfur oxides acidic, basic or amphoteric? what does this mean?
acidic:
reacts with bases (to form salts)
reacts w/ water to form acids
give the eqn for the reaction of phosphorous oxide with water:
P4O10 (s) + 6H2O (l) → 4H3PO4 (aq)
give the eqn for the reaction of sulphur dioxide with water and name the product:
SO2 (g) + H2O (l) → H2SO3 (aq) - sulphurous acid
give the eqn for the reaction of sulfur trioxide with water and name the product:
SO3 (g) + H2O (l) → H2SO4 (aq) - sulphuric acid
is silicon dioxide soluble in water? is it acidic, basic or amphoteric?
insoluble
acidic - reacts with bases
is aluminium oxide water soluble? is it acidic, basic or amphoteric?
insoluble
amphoteric - reacts with both acids and bases
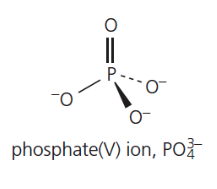
give the formula and structure of the phosphate (V) ion:
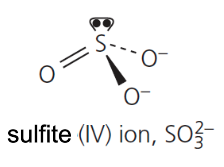
give the formula and structure of the sulfite (IV) ion:
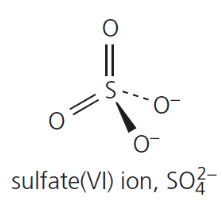
give the formula and structure of the sulfate (VI) ion:
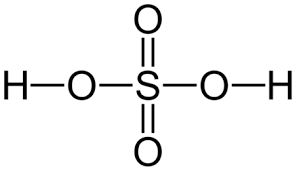
give the formula and structure of sulfuric acid:
H2SO4
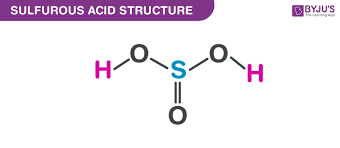
give the formula and structure of sulfuruous acid:
H2SO3
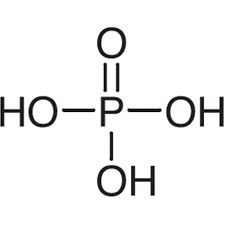
give the formula and structure of phosphoric acid:
H3PO4
give the formula of a hydroxide of an element in period 3 used in medicine:
Mg(OH)2
why are transition metals good catalysts?
have variable oxidation states
so are able to form a range of compounds by gaining/losing e- w/in their d orbitals
in which process does vanadium act as a catalyst? give the eqns for this process:
Contact Process - industrial process for making H2SO4 :
S + O2 → SO2
SO2 + ½ O2 → (V2O5 catalyst) SO3
SO3 + H2O → H2SO4
give and explain pair of eqns for the use of V2O5 as a catalyst in the Contact Process:
V2O5 oxidises SO2 to SO3 and is itself reduced to V2O4:
V2O5 + SO2 → V2O4 + SO3
the reduced catalyst is then oxidised back to its original state:
V2O4 + ½ O2 → V2O5
what is a heterogeneous catalyst?
catalyst which exists in a different phase from the reactants
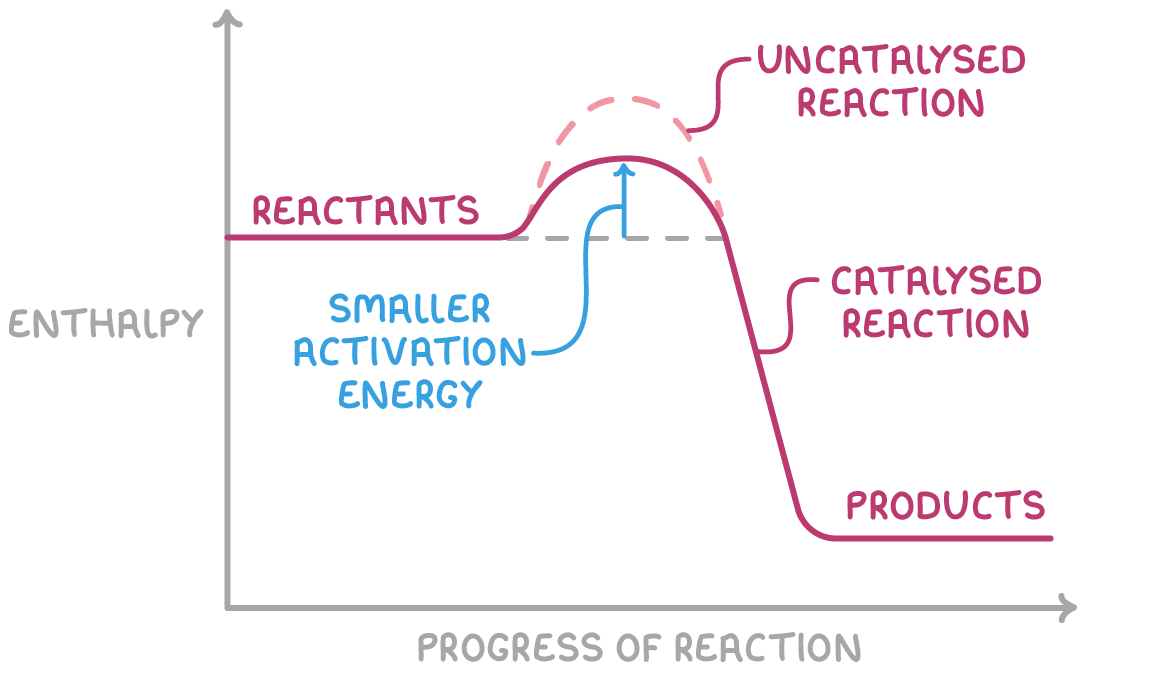
explain how a heterogenous catalyst works:
reactants adsorb onto active sites on the surface of the heterogenous catalyst
bonds weaken/reaction takes place
products desorb from surface
give 2 egs of heterogenous catalysts and their eqns:
Fe in the Haber process:
N2 (g) + 3H2 (g) → (Fe (s) catalyst) 2NH3 (g)
V2O5 in the Contact process:
SO2 (g) + ½ O2 (g) → (V2O5 (s) catalyst) SO3 (g)
how can we minimise the cost of a reaction using a heterogenous catalyst?
maximise SA so increase no. of molecules that can react at the same time
this can be achieved by using powder/small pellets/support medium
give and explain an example of a support medium and provide the eqn:
catalytic converters contain a ceramic lattice coating in a thin layer of Rh/Pt/Pd
2CO (g) + 2NO (g) → (Rh (s) catalyst) 2CO2 (g) + N2 (g)
what is catalyst poisoning? what effects does this have?
catalysts can be poisoned when impurities adsorb onto the active sites of the catalyst, blocking them
this decreases the efficiency of catalysis, increasing costs
give 2 examples of catalyst poisoning:
lead can coat the inner surface of a catalytic converter
sulfur can poison the active sites on the iron catalyst in the Haber process, forming iron sulfide (sulfur found in fossil fuels used to produce H2)
what is a homogeneous catalyst?
catalyst which exists in the same phase as the reactants (typically liquids/solutions)
how do homogeneous catalysts work?
work by forming an intermediate species which then reacts to form the products
the Ea needed to form the intermediate species is lower than that needed to make the products directly
give an example of a homogeneous catalyst and give the eqn for the reaction it catalyses:
Fe2+ ions catalyse the reaction between peronodisulfate ions and iodide ions:
S2O82- (aq) + 2I-(aq) → (Fe2+ (aq) catalyst) I2 (aq) + 2SO42- (aq)
why is the reaction between peronodisulfate ions and iodide ions slow without a catalyst?
-ve charges repel so high Ea
give a pair of eqns to show how Fe2+ acts as a catalyst in the reaction between peronodisulfate ions and iodide ions:
S2O82- (aq) + 2Fe2+ (aq) → 2Fe3+ (aq) + 2SO42- (aq)
2Fe3+ (aq) + 2I- (aq) → I2 (aq) + 2Fe2+ (aq)
other than having variable oxidation states, explain why Fe2+ ions are good catalysts for the reaction between peronodisulfate ions and iodide ions:
+ve ions attract -ve ions in catalysed process
give an example of a homogeneous catalyst which undergoes autocatalysis and give the overall eqn for the reaction it catalyses:
Mn2+ ions autocatalyse the reaction between C2O42- and MnO4- :
Mn2+ is a product of the reaction and acts as a catalyst
∴ as the reaction progresses, amount of product increases
2MnO4- (aq) + 16H+ (aq) + 5C2O42- (aq) → 2Mn2+ (aq) + 8H2O (l) + 10CO2 (g)
give a pair of ionic eqns to show how Mn2+ ions act as a catalyst in the reaction between C2O42- and MnO4- :
4Mn2+ (aq) + MnO4- (aq) + 8H+ (aq) → 5Mn3+ (aq) + 4H2O (l) (Mn2+ oxidised back to Mn3+ by MnO4-)
2Mn3+ (aq) + C2O42- (aq) → 2Mn2+ (aq) + 2CO2 (g) (Mn3+ reduced to Mn2+ by C2O42-)
the reaction between C2O42- and MnO4- is very slow at first - explain why the reaction is initially slow (3)
no catalyst at start
2 -vely charged ions repel
Ea for reaction high
why does Mn2+ lower the Ea of the reaction between C2O42- and MnO4- ?
Ea lowered because oppositely charged ions attract
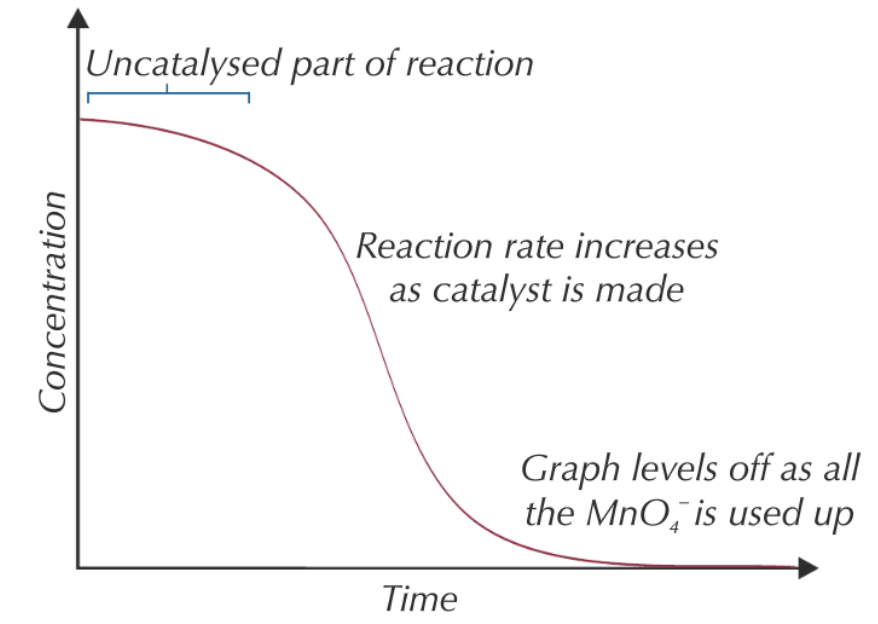
give and explain the shape of a conc-time graph of MnO4-:
initially gradient shallow as RoR low as reaction is uncatalysed as too few Mn2+ ions made ∴ high Ea
RoR increases w/ time as more of autocatalyst is made
graph begins to level off as MnO4- used up
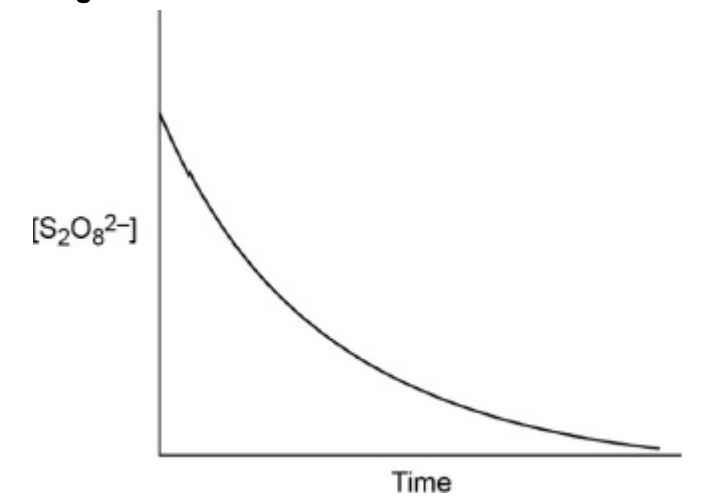
give and explain the shape of a conc-time graph of S2O82- in the reaction between S2O82- and I-:
reaction slow because 2 -ve ions repel
Fe2+ attracts S2O82- so lowers Ea
Fe2+ oxidised to Fe3+
a student determines the conc of ethanedioate ions in an acidified soln by titration w/ potassium managanate (VII) soln.
2MnO4- + 5C2O42- + 16H+ → 2Mn2+ + 10CO2 + 8H2O
the mixture is warmed before the addition of potassium manganate (VII) soln because the reaction is slow at first, when more potassium manganate (VII) is added, the mixture goes colourless quickly due to the presence of an autocatalyst.
explain the meaning of the term autocatalyst and explain, using eqns where appropriate, why the reaction is slow at first and then goes quickly (6)
autocatalyst - product of reaction catalyses the reaction
slow: -ve ions repel
high Ea
attraction between oppositiely charged ions
4Mn2+ + MnO4- + 8H+ → 5Mn3+ + 4H2O
2Mn3+ + C2O42- → 2Mn2+ + 2CO2
explain why complexes formed from TM ions are coloured (3)
d orbitals have different e/d orbital Es are split
e- absorb some wavelengths of visible light
to excite e- in d orbitals
remaining/complementary wavelengths of light reflected to give colour seen
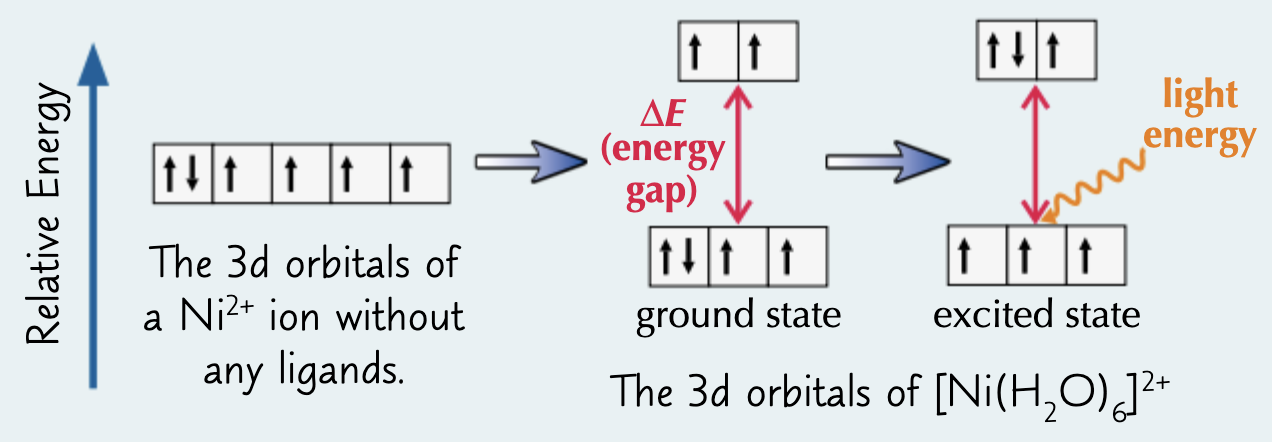
state the origin of the colour of TM complexes (1)
e- excited in d shell
E in visible range
give the eqn for calculating ΔE and state what each symbol corresponds to:
ΔE = h ν = hc / λ, where:
ΔE: energy absorbed by e- from ground to excited state / J
h: Planck’s constant (6.63 × 10-34 J)
v (Greek letter nu): freq of light absorbed / Hz
c: speed of light = (2.998 × 108 ms-1)
λ: wavelength of light absorbed / m
give 3 factors which might affect the colour of a transition metal complex:
change in oxidation state
change in coordination number
change in ligand
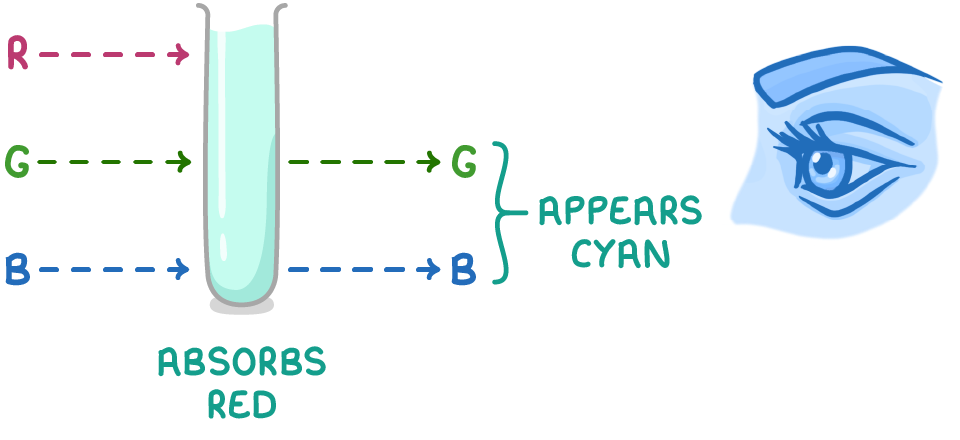
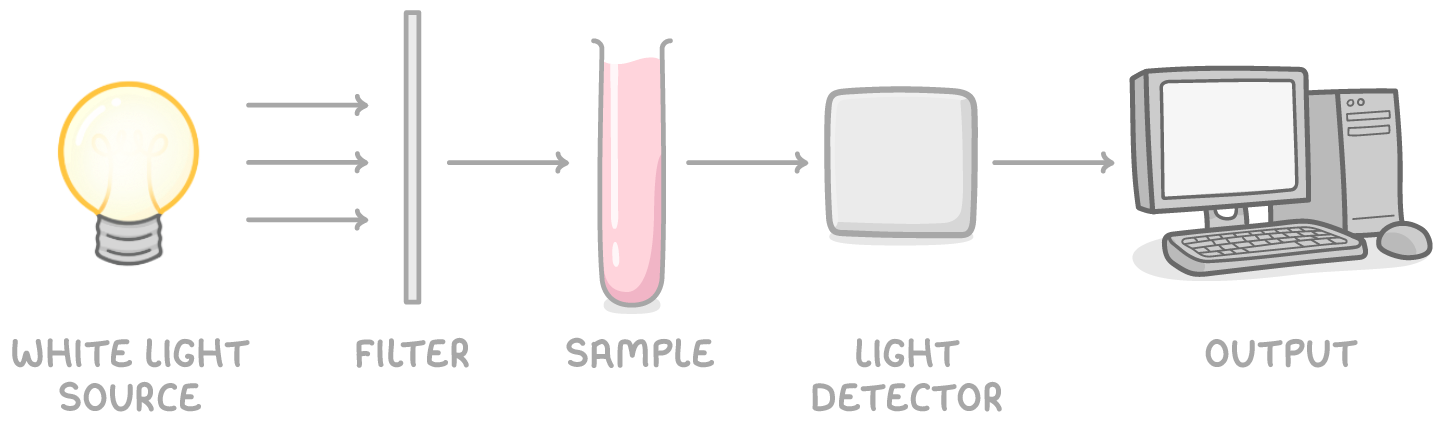
how can we use colorimetry to determine the concentration of transition metal ions in a solution of unknown concentration?
add an appropriate ligand to intensity colour
make up solutions of some known concentrations and measure absorbance
plot calibration curve of of absorbance v conc
measure absorbance of coloured complex and find conc from graph
what is a transition metal?
a metal that can form one or more stable ions w/ an incomplete d sub level
by definition, which 2 elements in the d block of the periodic table are not transition metals?
Sc
Zn
when figuring out e- configurations of the transition metals, in which order do we fill the orbitals? what does this mean for the ionisation of transition metals?
4s before 3d
so when transition metals become ions, they lose 4s e- before 3d e-
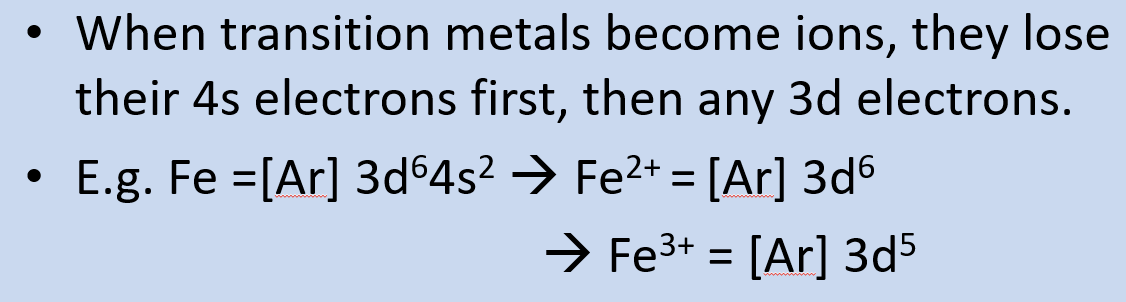
give and explain the two exceptions to the electron configuration rule:
Cr - [Ar] 4s1 3d5
Cu - [Ar] 4s1 3d10
as a half or fully filled subshell is more stable than a partially filled one
give 2 physical properties of transition metals:
high densities
high mpts/bpts
what is a physical property?
a characteristic of a substance that can be observed or measured without changing its chemical composition
give 4 chemical properties of transition metals:
form coloured ions
very good catalysts
have variable oxidation states
form complex ions
what is a chemical property?
properties which can only be observed or measured during a chemical reaction
give a transition metal with a variable oxidation state and explain why it has this:
Fe2+ and Fe3+ as the 4s and 3d orbitals are very similar in energy so different nos. of e- can be lost/gained
suggest why the oxidation stat eof chromium is zero in Cr(PF3)6 (1)
PF3 is neutral and whole complex is neutral
what is an enantiomer?
isomers which are non-superimposable mirror images of each other
what is a chiral carbon?
C atom with 4 different attached groups
how does optical isomerism exist in transition metal complexes?
exists in octahedral complexes when the ion has 3 bidentate ligands bonded to it
these can ∴ form a pair of enantiomers
give an example of a transition metal complex displaying optical isomerism:
[Cu(en)3]2+
![<p>[Cu(en)<sub>3</sub>]<sup>2+</sup></p>](https://knowt-user-attachments.s3.amazonaws.com/d8fbc9f2-9512-4568-9ec7-8713bc8a957e.png)
what is cis-trans isomerism?
form of steroisomerism where identical groups can be positioned either adjacent (cis) or opposite (trans) each other
where can cis-trans isomerism be seen in transition metal complexes?
square planar complexes that have 2 pairs of ligands
octahedral complexes - 2 of one ligand, 4 of another
give a complex that displays cis-trans isomerism and name the two isomers:
PtCl2(NH3)2:
cis isomer = cisplatin
trans isomer = transplatin
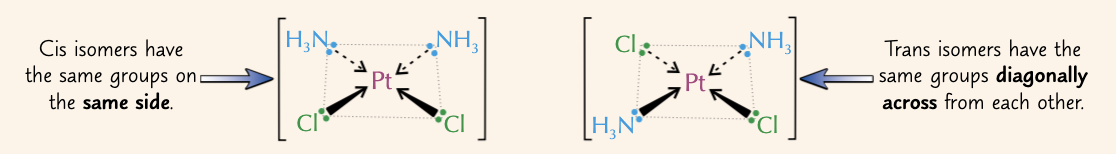
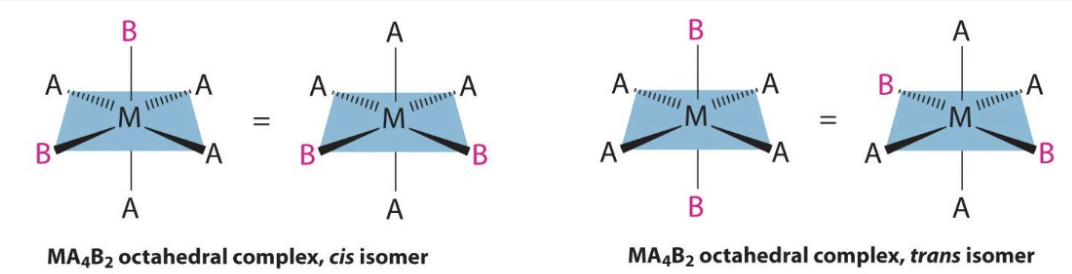
how do we distinguish between cis and trans isomers in octahedral complexes?
if the 2 ligands which are the same are opposite each other and so have an angle of 180o between them = trans
if the 2 ligands which are the same next to each other and so have an angle of 90o between them = cis
what are redox titrations used for?
to find the concentration of an oxidising/reducing agent in solution (i.e. how much oxidising agent is needed to exactly react with a quantity of reducing agent)
why do you not need an additional indicator for a redox titration? give an example:
redox titrations are self indicating
purple potassium manganate (VII) is reduced to pale pink manganese (II) ions
give the ½ eqn for the reduction of potassium manganate (VII) → manganese (II) ions:
MnO4- + 8H+ + 5e- → Mn2+ + 4H2O
give the method for a typical redox titration:
measure out the quantity of reducing agent using a pipette (either Fe2+ or C2O42- ions)
add dilute sulfuric acid so it is in excess
add the oxidising agent to the burette (usually potassium manganate)
add the oxidising agent from the burette until the solution just changes colour - to the colour of the oxidising agent (in this reaction it would be colourless to pale pink)
repeat to get concordant results
calculate the concentration of the reducing agent