Crystalline Solids
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
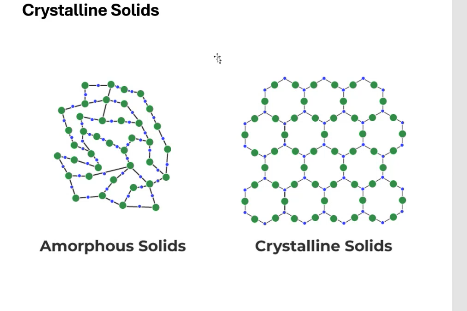
Crystalline solids have ____ ordering.
periodic
Amorphous solids lack ____.
periodic ordering
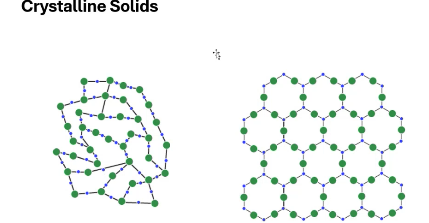
which is the crystalline solid and which is the amorphous solid
a unit cell is ____ in all directions
repeated
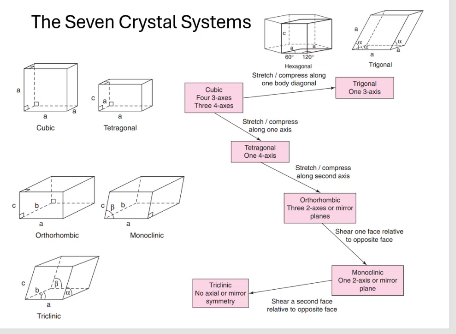
what are the seven crystal systems?
cubic, tetragonal, orthorhombic, monoclinic, triclinic, trigonal, hexagonal
Seven crystal systems flow chart
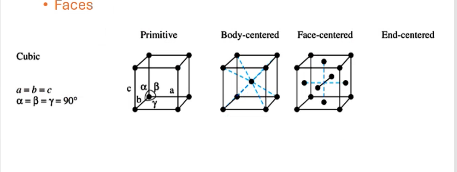
For cubic solids, what are the allowed symmetries, what are the unit cell shapes?
primitive, body-centered, face-centered
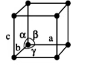
For tetragonal solids, what are the allowed symmetries, what are the unit cell shapes?
primitive, body-centered
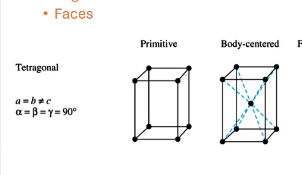
what symmetry does this structure have?
face-centered, atoms occupy center of each face of the structure
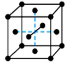
what symmetry does this structure have?
body-centered, atoms on corners and atom directly in center
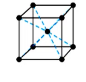
what symmetry does this structure have?
primitive, only atoms on corners
To be body-centered, all atoms MUST _____ on the structure.
be the same
cubic symmetry cannot have ____ symmetry
end-centered
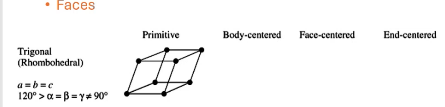
For Trigonal (rhombohedral) solids, what are the allowed symmetries, what are the unit cell shapes?
can only have primitive,
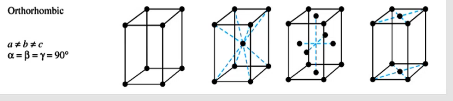
For orthorhombic solids, what are the allowed symmetries, what are the unit cell shapes
primitive, body-centered, face-centered, end-centered
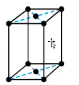
What type of symmetry does this solid have
end-centered, atoms on corners and one in center of two faces
For monoclinic solids, what are the allowed symmetries, what are the unit cell shapes
primitive and End-centered
every structure can have a ____ structure
triclinic
what is the lowest symmetry crystal system?
triclinic
Cubic: Four __ axes, three __ axes
3, 4
tetragonal: one __ axis
4
to go from cubic to tetragonal:
stretch/compress along one axis
to go from tetragonal to orthorhombic:
stretch/compress along second axis
to go from orthorhombic to monoclinic:
shear one face relative to opposite facet
o go from monoclinic to triclinic:
shear a second face relative to opposite face
Orthorhombic: three __ axes or ____
2, mirror planes
Monoclinic: one __ axis or __
2, mirror plane
Monoclinic: one __ axis or __
2, mirror plane
Triclinic: no __ or ____ symmetry
axial, mirror
To go from cubic to trigonal:
stretch/ compress along one body diagonal
trigonal: one __ axis
3
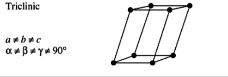
triclinic solids have what symmetry, what are the unit cell shapes
primitive
corner site, face site, edge site
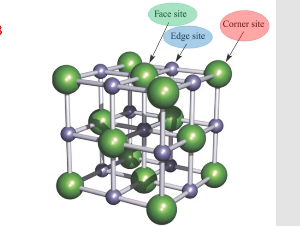
how much do the corner atoms, edge atoms, and face atoms account for?
corners: 1/8
edges: ¼
faces: ½
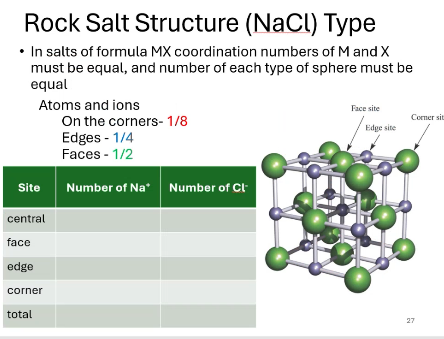
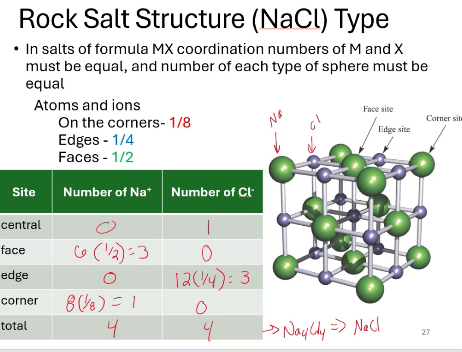
what is the chemical formula for this salt structure?
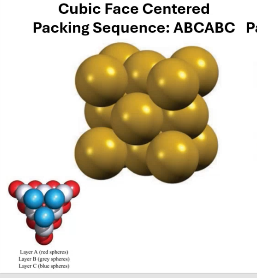
____ of spheres is most efficient and most common in nature.
close-packing
Cubic Face Centered packing sequence:
ABCABC
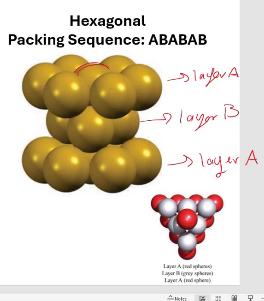
Hexagonal packing sequence:
ABABAB
interstitial holes:
hexagonal and cubic close-packing
interstitial holes form ____ and ___ shaped holes
octahedral and tetrahedral
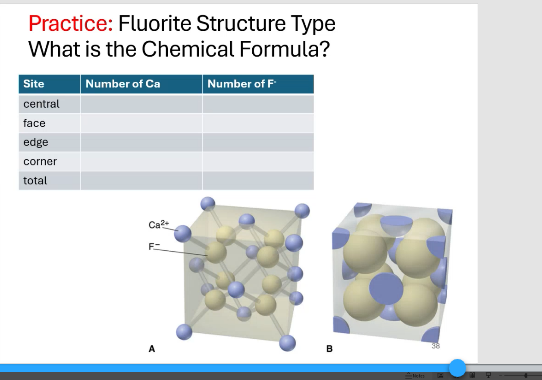
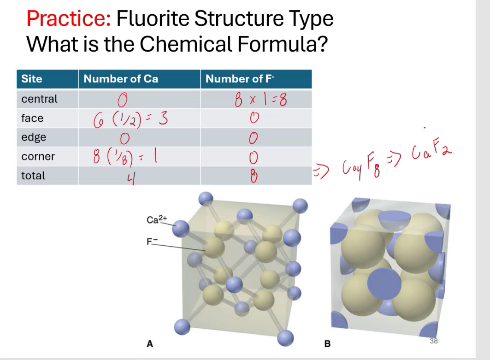
fluorite chemical structure
MX2
antifluorite chemical structure
M2X
anti fluorite structure reverses position of
metal and fluorine
Zinc blends have a cation:anion ratio of?
1:1
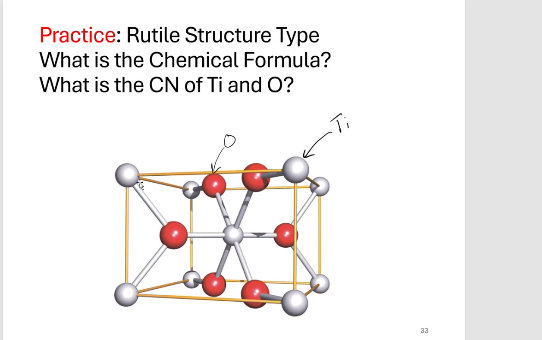
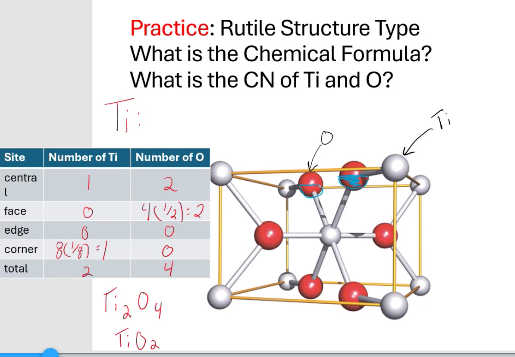
CN for Ti = 6 , CN for O = 3
If Ti occupies the center of a cubic unit cell and Ba occupies the corners and O each face, what is the empirical formula of the compound?
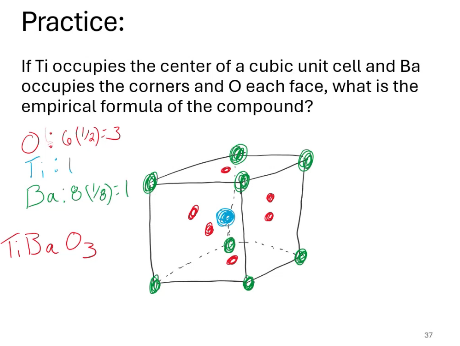
General formula for perovskites:
ABO3
What is lattice energy
How much energy it takes to form/break down into gaseous ions
most useful properties of solids are related to:
___ vibrations of atoms or ions
the presence of _____
the existence of ____
thermal vibrations, impurities, defects
Examples of defects:
simple ____
unexpected occupation of ____ sites
incorporation of ____: atoms or ions
Various ____ imperfections
vacancies, interstitial sites, impurities, lattice
Schottky Defect:
the difference in size between cation and anion is ___.
Both anion and cation ___ the solid crystal.
Atoms ____ leave the crystal.
There is formation of two ____.
The density of the solid ____.
small, leave, permanently, vacancies, decreases
Frenkel Defect:
Contains ionic crystals where the ___ is larger than the ____.
Usually the ___ ion cation leaves its original lattice structure.
Atoms ___ within the solid crystal.
only ___ vacancy is created.
____ remains the same.
anion, cation
smaller, one, density
In solids, atomic orbitals overlap —> energy levels spread into ___
bands
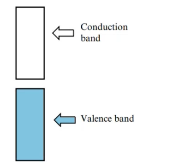
Valence band
usually filled with electrons
conduction band
higher energy states, may be empty or partially filled
Band gap:
energy separation between valence and conduction bands
a charge carrier in a semiconductor, is either a _____ or an electron that is able to ___ electricity.
positive hole, conduct
The valence bands participate in ___ and ____
bonding, magnetism
The ___ band is the empty ____
conduction, LUMO
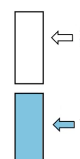
Which is the conduction band, which is the valence band
label the three conductors
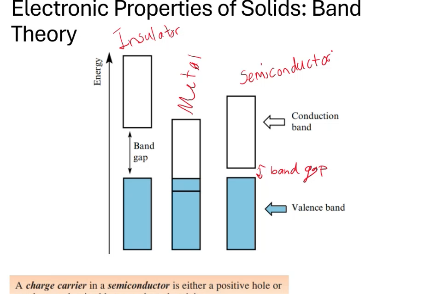
This is an ____
band gap is so big, energy jump is unable to be made
fermi level is the ?
measure of the energy of the least tightly held electrons within a solid
The electrical conductivity of a ____ decreases with temperature; that of a ______ increases with temperature.
metal, semiconductor
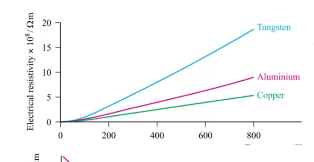
this graph is for ___ and ____
metals and semi conductors
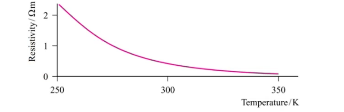
this graph is for ____
insulators
a dopant is an ____ introduced into a semiconductor in minute amounts to enhance its _____.
impurity electrical conductivity
____ contain dopants
extrinsic semiconductors
p-type semiconductor:
Group ____ element is added as a doping element
Trivalent impurity like __, __, __, etc. are added, NOT B
The fermi level is between acceptor energy level and the valence band, closer to the ____ band than the ____ band.
III
Al, Ga, In
valance, conduction
N-type semiconductor:
Group __ element is added as a doping element
Pentavalent impurity like __ , __, __, __etc. are added NOT N
Fermi Level is between donor energy level and the conduction band, appears closer to the ___ band than the ___ band
V
P, As, Sb, Bi
conduction, valence
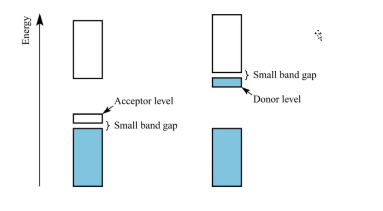
what are these types of conductors?
The semiconductor on the left is a p-type semiconductor, the fermi level is closer to the valence band than the conduction band
The semiconductor on the right is an n-type semiconductor, the fermi level is closer to the conduction band than the valence band