AQA GCSE Physics: Atomic Structure
1/89
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
90 Terms
What is the radius of an atom?
~1x10⁻¹⁰m (very small)
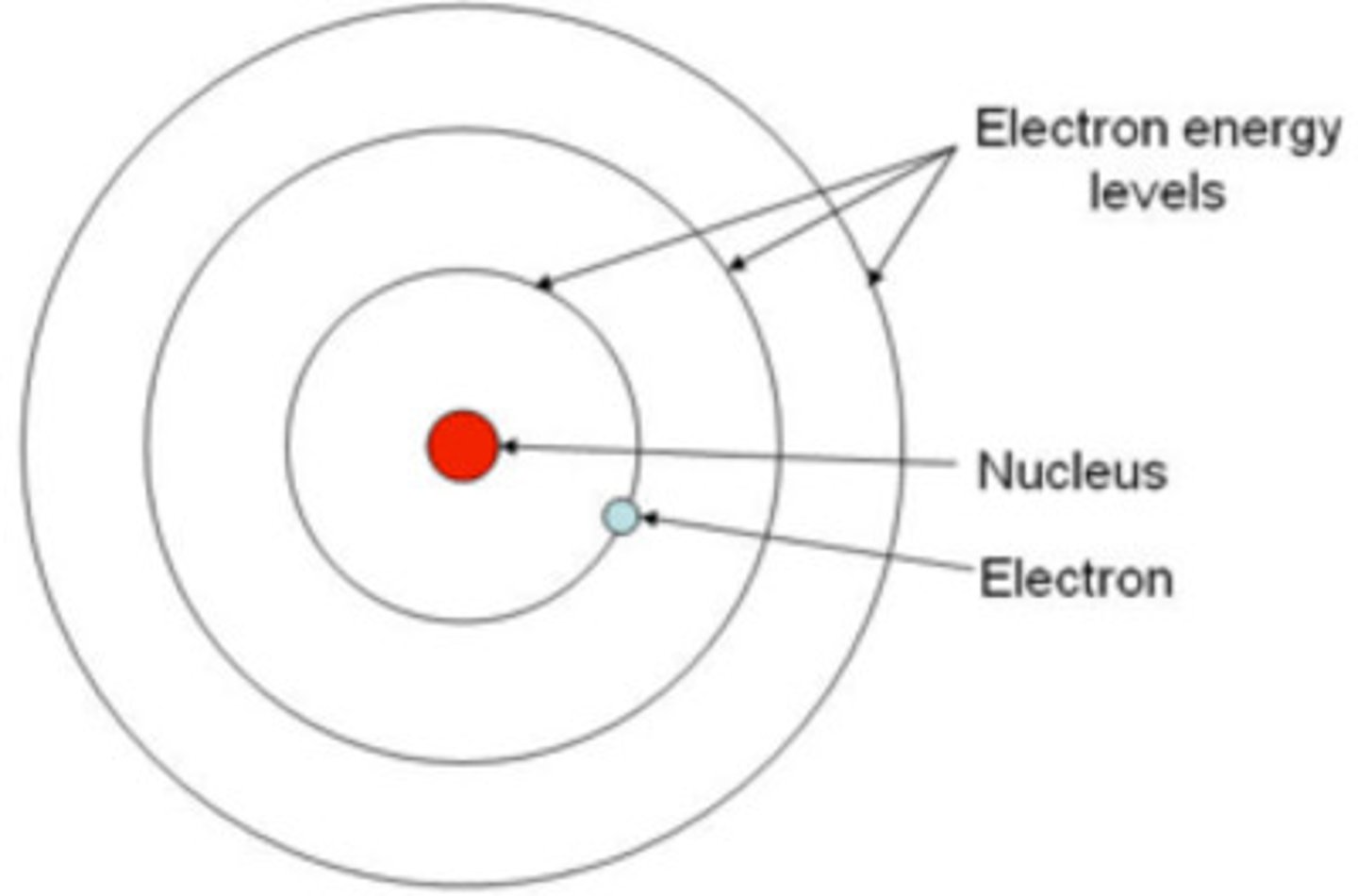
What is the basic structure of an atom?
- an overall positively charged nucleus (composed of both protons and neutrons)
- nucleus is surrounded by negatively charged electrons at different energy levels (different distances from the nucleus)
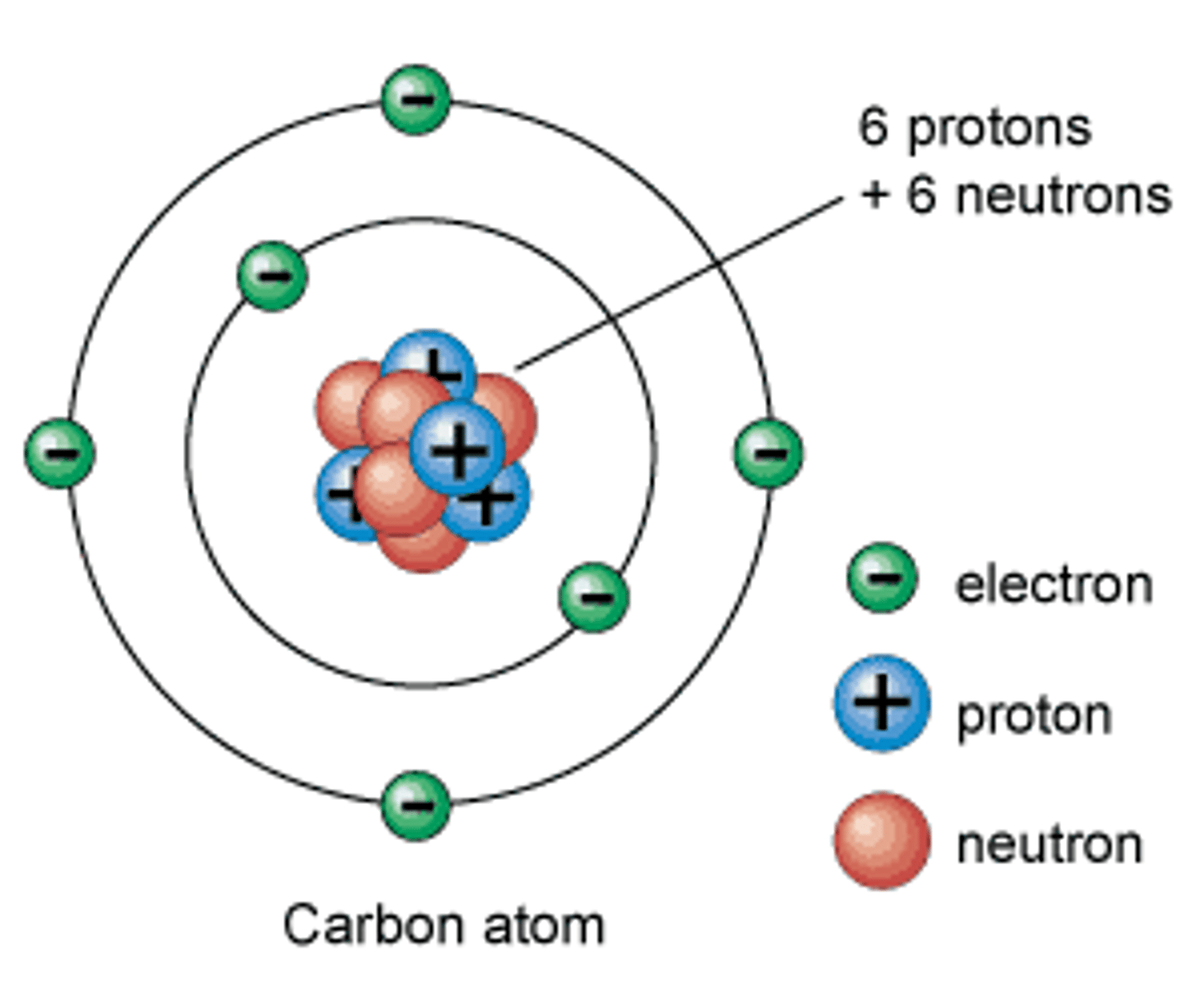
What is the radius of the nucleus in comparison to the radius of the atom?
<1/10000 of the radius of an atom
Where is most of the mass inside an atom?
- most of the mass of an atomis concentrated in the nucleus
What is the charge of protons, neutrons, and electrons?
protons (+) positive
neutrons (0) neutral
electrons (-) negative
How does the energy change in energy levels?
- energy levels which are further from the nucleus are at a higher energy than those which are closer to the nucleus
Why might the electron arrangements change (why might the electrons change which energy level they are orbiting the nucleus from)?
- if the atom absorbs of electromagnetic radiation, an electron can move to a higher energy level (move further away from the nucleus)
- when the atom emits electromagnetic radiation, the electron returns back to the lower energy level (move closer to the nucleus)
Why do atoms have no overall charge?
- in an atom, the number of negative electrons is equal to the number of positive protons => charges cancel out
What is the atomic number of an atom?
- the number of protons in an atom of an element
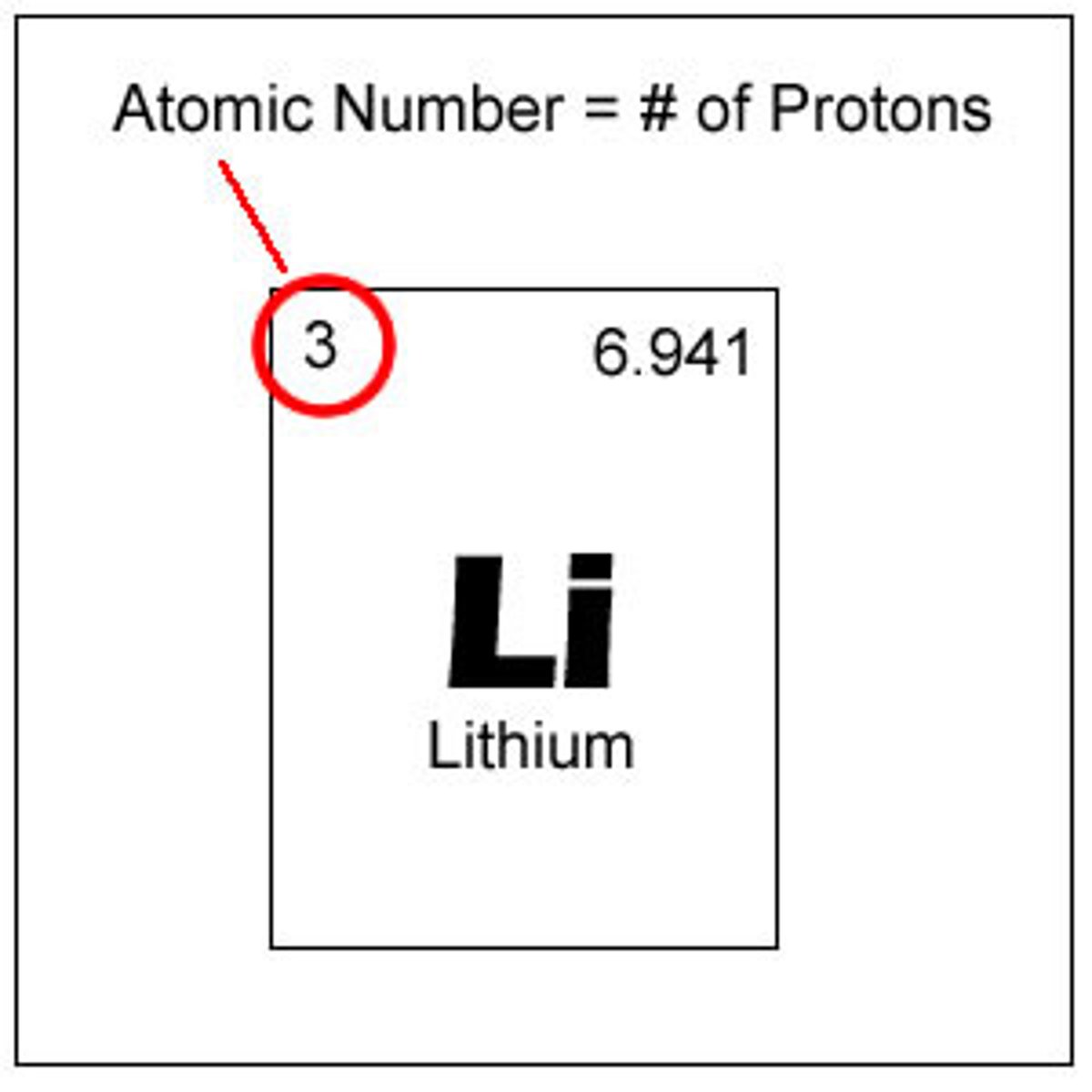
What is the 'mass number' of an atom?
- the total number of protons and neutrons in an atom

What are isotopes?
- isotopes are atoms of the same element that have different numbers of neutrons
What is an ion?
- a charged particle formed when an atom, or number of atoms, loses/gains electrons
How are the 2 types of ions formed?
- a positive ion is formed when an atom loses electrons
- a negative ion is formed when an atom gains electrons
Why may a scientific model be changed or replaced?
new experimental evidence
What did the Ancient Greeks believe about atoms?
- before the discovery of the electron, the Greeks thought atoms were TINY SPHERES THAT COULD NOT BE DIVIDED
What was discovered in 1897?
What did this discovery prove wrong?
What model was made from this discovery?
- scientists discovered that atoms contain tiny negative particles called electrons
=> showed that atoms are not spheres that cannot be divided (they have an internal structure)
=> plum pudding model
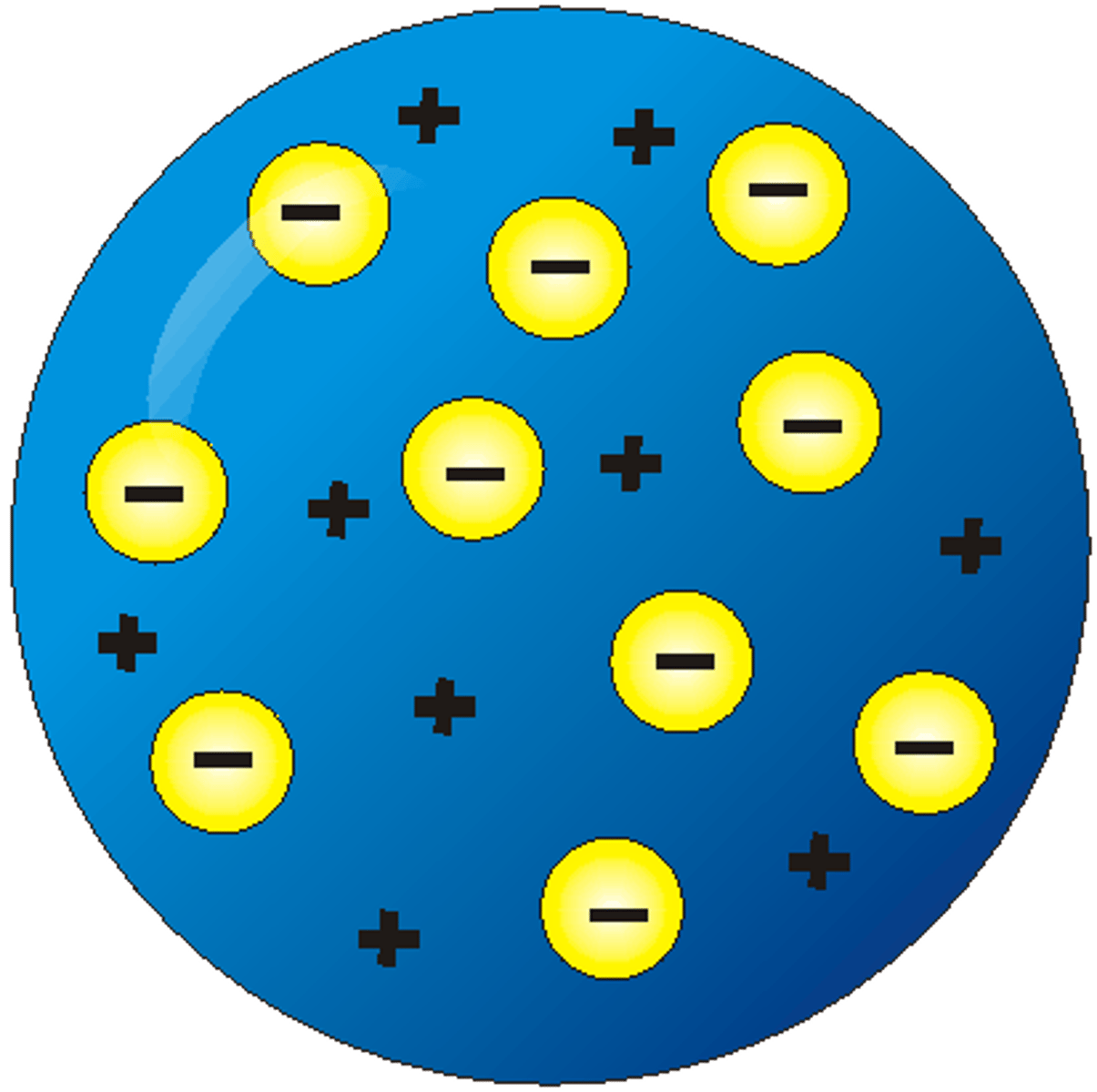
What did the plum pudding model suggest about atoms?
- suggested that the atom is a ball of positive charge with negative electrons embedded in it
Why did scientists use gold as the foil in the alpha-scattering experiment?
- gold could be hammered into a very thin piece (just a few atoms thick)
What 3 results happened when alpha particles were fired at the gold foil in the alpha-scattering experiment?
A. Most of the alpha particles went straight through without changing direction
B. Some alpha particles were deflected (changed direction) as they passed through
C. Some alpha particles bounced straight off
What 3 conclusions did the alpha-scattering experiment lead to?
A. Atoms are mainly empty space (plum pudding model had to be wrong)
B. Centre of an atom must have a positive charge that repelled the positive alpha particles
C. Mass of the atom must be concentrated in the centre (now called the nucleus)
After the alpha-scattering experiment, what new model replaced the plum pudding model?
the nuclear model v1
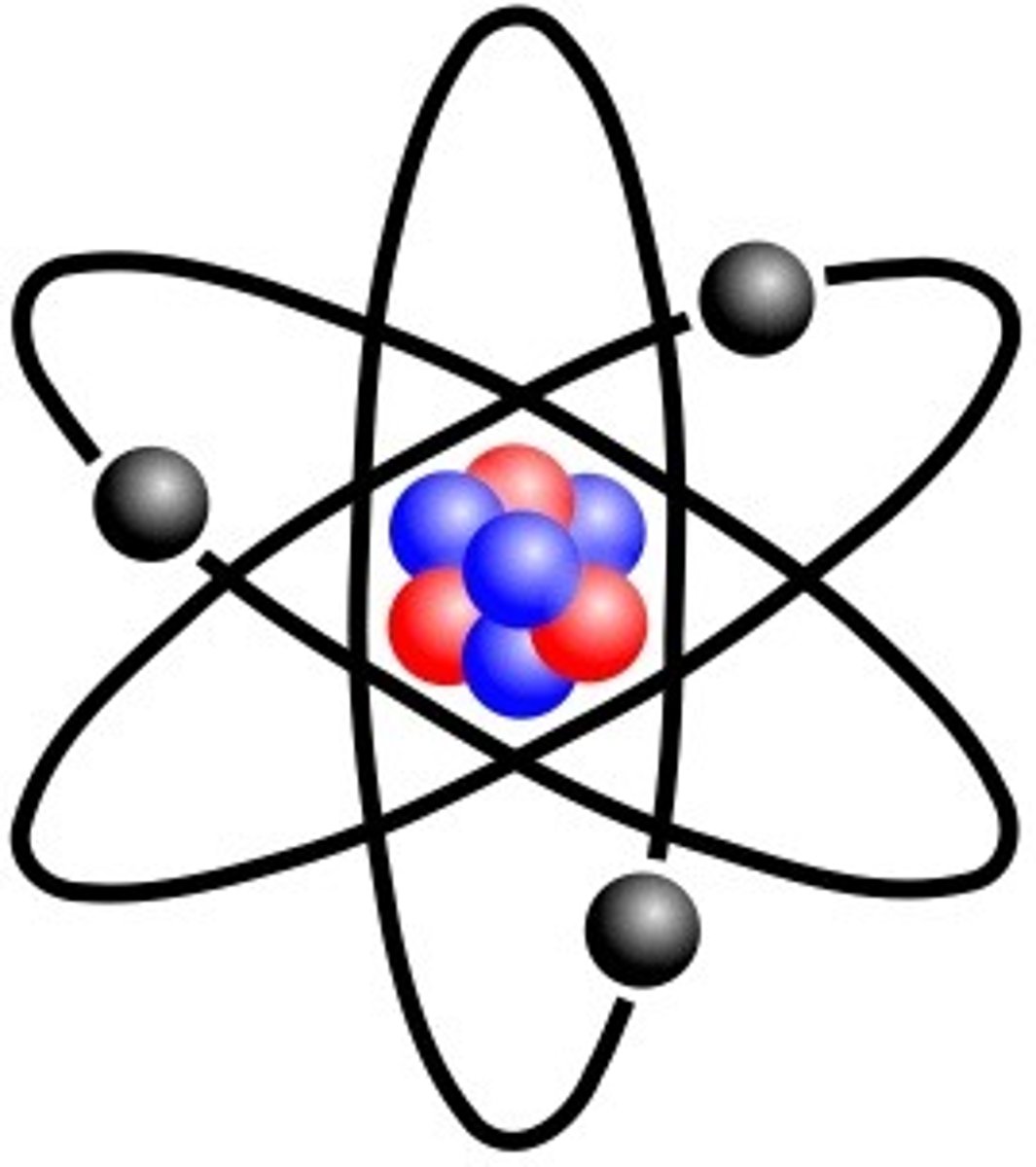
How did Niels Bohr adapt the nuclear model?
- he suggested that electrons orbit the nucleus at specific distances (now called energy levels/shells)
- his theoretical calculations agreed with experimental observations by other scientists
What was discovered to cause the positive charge of any nucleus several years after Bohr adapted the nuclear model?
- later experiments led to the idea that the positive charge of any nucleus could be subdivided into a whole number of smaller particles, each particle having the same amount of positive charge
- protons
How did James Chadwick's experimental work on the atom lead to a better understanding of isotopes? (Exam Q)
- Chadwick provided the evidence to show the existence of neutrons
- isotopes have the same number of protons but with different numbers of neutrons
What is radioactive decay?
- some atomic nuclei are unstable (eg. certain isotopes)
- the nucleus gives out radiation as it changes to become more stable
- radioactive decay is a random process => we cannot predict when it will happen
What is 'activity' in terms of nuclear radiation?
What is activity measured in?
- activity is the rate at which a source of unstable nuclei decays
- measured in becquerel (Bq)
(1Bq = 1 decay per second)
What is 'count-rate' in terms of nuclear radiation?
- the number of decays recorded each second by a detector
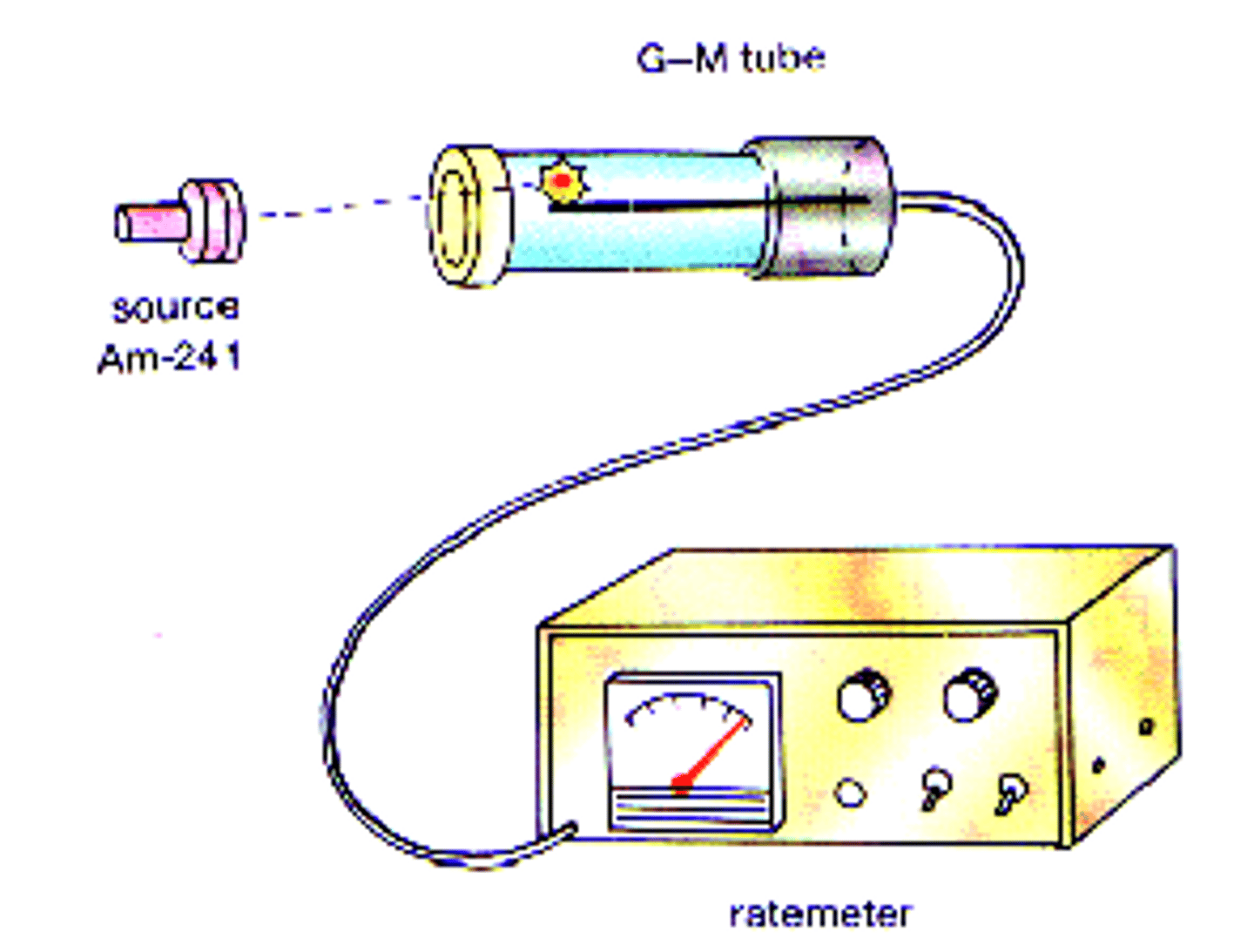
What instrument can be used to detect and measure the count-rate?
Geiger-Muller tube
What are the 4 types of nuclear radiation that can be emitted by an unstable nucleus? What are their symbols?
- alpha particle (α)
- beta particle (β)
- gamma ray (γ)
- neutron (n)
What is an alpha particle, in terms of nuclear radiation?
- this consists of two neutrons and two protons
- it is the same as a helium nucleus
What is a beta particle, in terms of nuclear radiation?
- a high speed electron ejected from the nucleus
- a beta particle is formed inside the nucleus when a neutron turns into a proton and an electron
What is a gamma ray, in terms of nuclear radiation?
- a type of electromagnetic radiation from the nucleus
What is a neutron, in terms of nuclear radiation?
- when a neutron is ejected out of the nucleus
How do alpha particles, beta particles, and gamma rays differ with their range in air?
- alpha particles are large
- they can travel ~5cm in air before they collide with air particles and stop
- beta particles can reach ~15cm in air before stopping
- gamma radiation can travel several metres in air before stopping
How do alpha particles, beta particles, and gamma rays differ with their penetrating power?
- alpha particles is stopped by a single sheet of paper (relatively easy to stop)
- beta particles is stopped by a few millimetres of aluminium
- gamma radiation is stopped by several centimetres of lead
What is 'ionising power'?
- the ability to lose electrons and form ions when radiaiton collides with atoms
How do alpha particles, beta particles, and gamma rays differ with their ionising power?
- alpha particles are very strongly ionising (they can produce a lot of ions when they collide with a material)
- beta particles are quite strongly ionising (not as ionising as alpha particles)
- gamma radiation is weakly ionising
What does a nuclear equation show?
- a nuclear equation shows what is produced when a radioactive nucleus decays
What may be caused by the emission of the different types of nuclear radiation?
a change in the mass and/or the charge of the nucleus
How can we represent an alpha particle in a nuclear equation?
- the symbol for helium
- 2 protons and 2 neutrons (mass number is 4 and atomic number is 2)
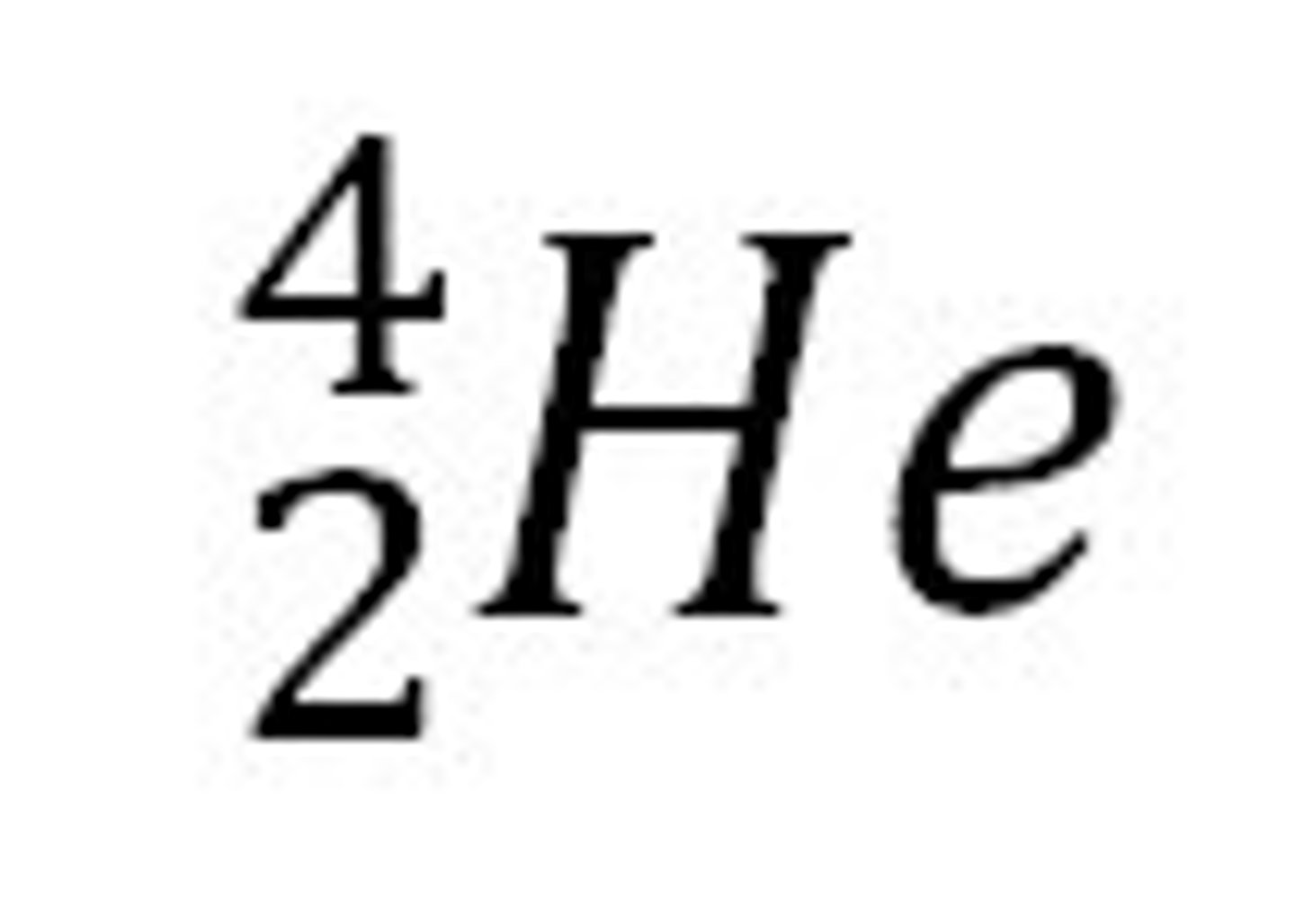
What happens during alpha decay?
- alpha decay causes both the mass and the charge of the nucleus to decrease
- mass number decreases by 4
- atomic number decreases by 2
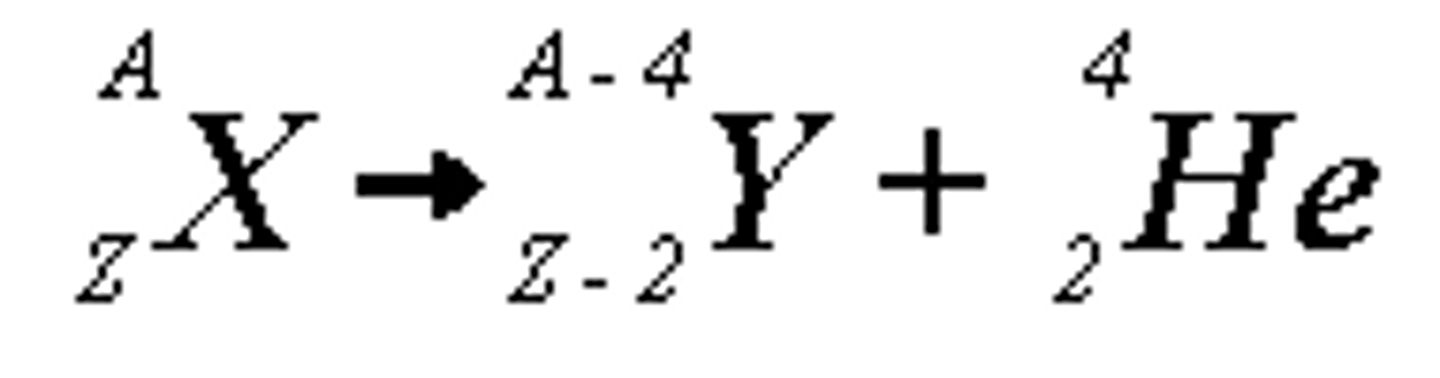
How can we represent a beta particle in a nuclear equation?
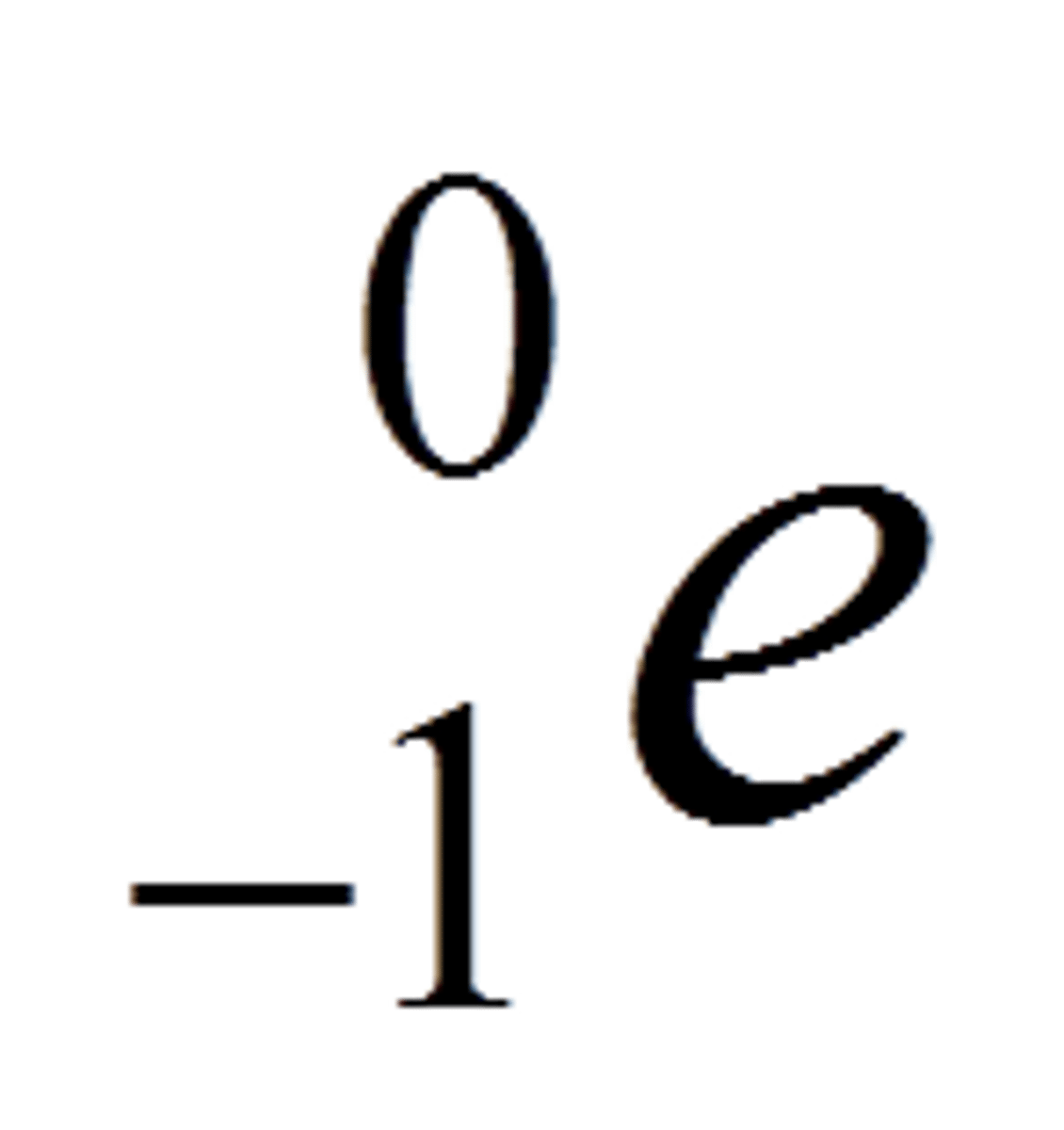
What happens during beta decay?
- neutron changes to a proton => atomic number increases by 1
- mass of the nucleus (mass number) does not change as total number of protons and neutrons stays the same (lose 1 neutron but gain 1 proton so cancels out)
- charge of the nucleus does increase
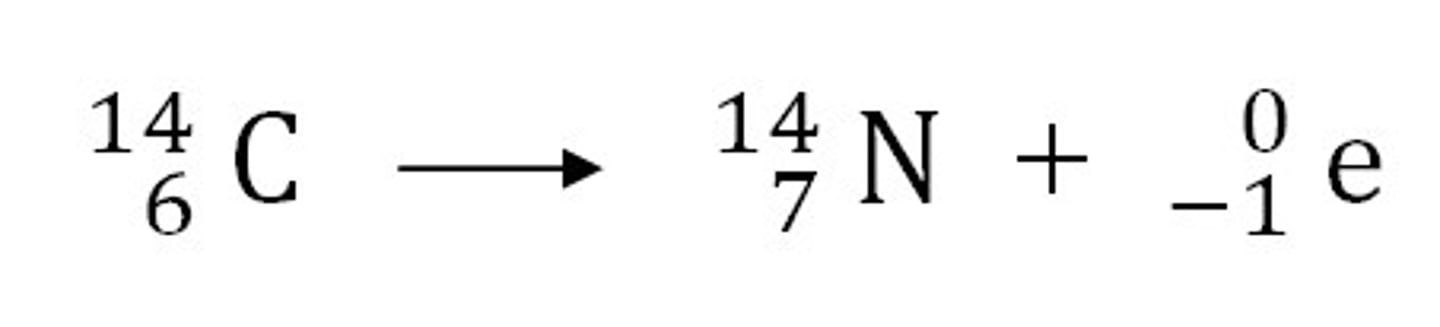
What is the nuclear equation for the alpha decay of a radium atom?
→ alpha particle + radon
- atom number + 2
- mass number + 4
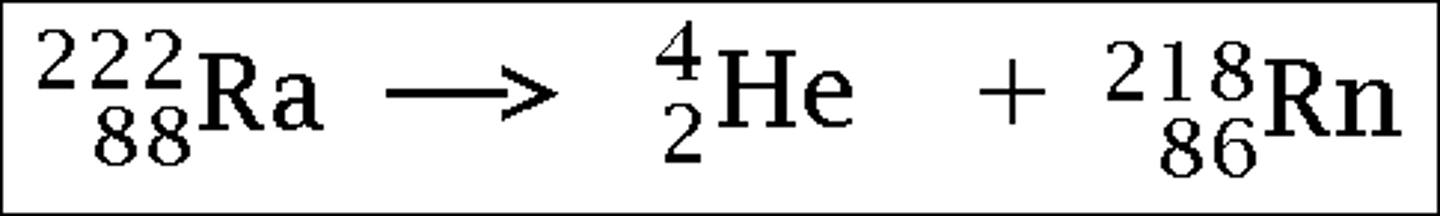
What is the nuclear equation for the beta decay of an iodine atom?
- mass number does not change
- but atomic number + 1
→ beta particle + xenon
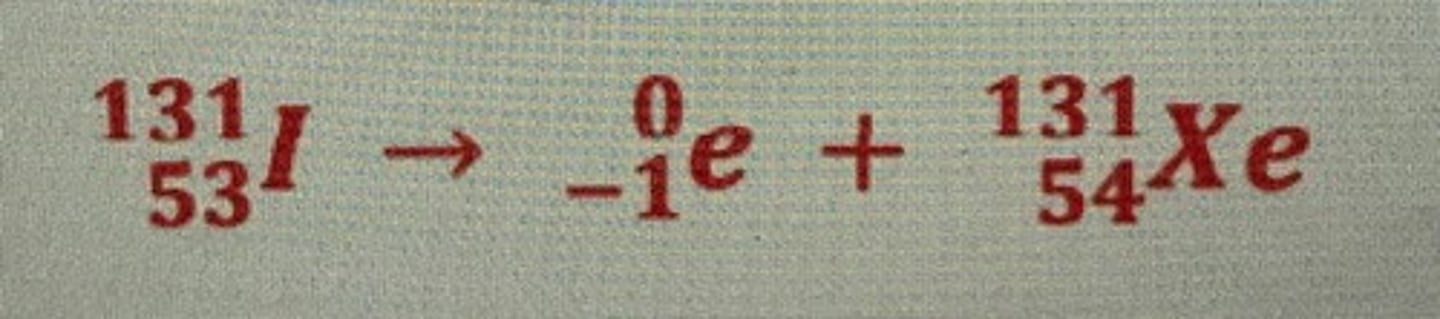
What is different about gamma decay compared to both alpha and beta decay?
- the emission of a gamma ray does not cause the mass or the charge of the nucleus to change
(neither mass number nor atomic number change at all)
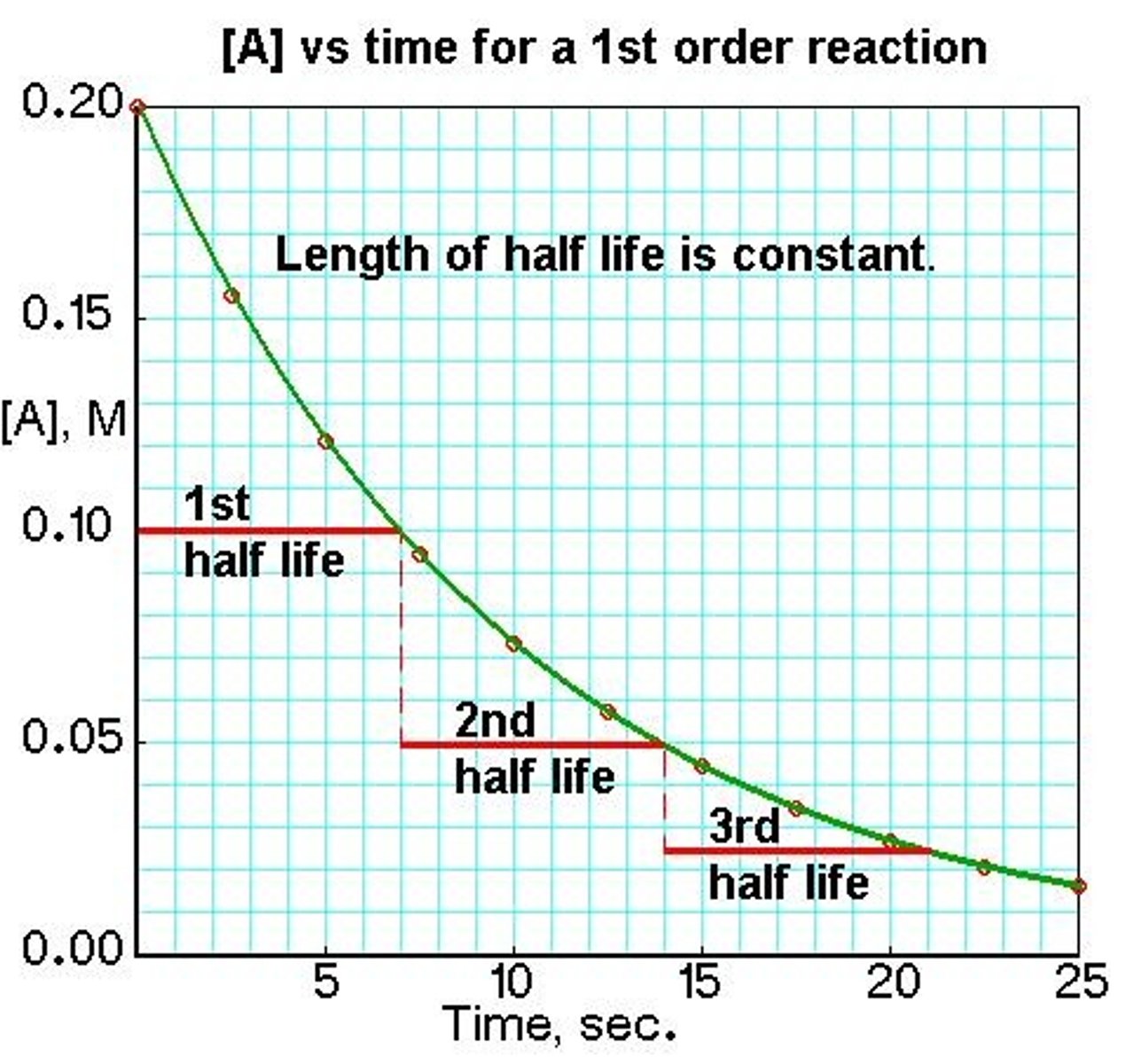
What is the 'half-life' of a radioactive isotope?
What is the second definition of half-life, in terms of the count-rate or activity?
- the time it takes for the number of nuclei of the isotope in a sample to halve
- the half-life is alsothe time it takes for the count rate (or activity) from a sample containing the isotope to fall to half its initial level
Why do scientists use half-life?
- radioactive decay is a completely random process => scientists cannot predict when any individual nucleus will decay
=> instead, scientists determine the time it takes for half the original nuclei to decay (half-life)
From a graph, how canwe find the half-life of a radioactive isotope?
- half the number of initial nuclei and then find the corresponding time value
What is 'radioactive contamination'?
- the unwanted prescence of materials containing radioactive atoms on other materials
What is the hazard from radioactive contamination caused by?
due to the decay of the contaminating atoms
What factor affects the level of hazard from radioactive contamination?
the type of radiation
What can easily stop alpha radiation in humans?
- alpha radiation (low penetration power) can be stopped by dead cells on the skin surface
When is alpha radiation dangerous?
- alpha emitters can be dangerous if you inhale them (eg. from dust) or swallow them (eg. from contaminated food)
Why is gamma radiation less hazardous than alpha or beta radiation?
- weakly ionising
- strong penetrating power => likely to just pass straight through the body
Why is peer review important in exploring the effects of radiation on humans?
- it is important that studies are published and then shared with other scientists to check validity and prevent falsified work from being accepted
What is 'irradiation'?
- the process of exposing an object to nuclear radiation
What is the main use of irradiaiton?
- sterilisation of medical equipment (killing any bacteria) as sometimes, the object cannot be heated
- the irradiated object itself does not become radioactive (object does not come into contact with the radioactive isotope but only with the radiation)
Why is the object to be sterilised usually wrapped in plastic?
to prevent bacteria from entering
Why is a lead shield used in irradiation?
to protect workers from the gamma radiation from the radioactive isotope
What is a risk of working with ionising radiation?
- ionising radiation can increase the risk of cancer => people who work with radioactive isotopes have to take precautions
What are some precautions a worker must take to reduce exposure to ionising radiation?
- using a shield to stop the radiation (eg. gloves can protect against alpha radiation, which has very low penetrating power; beta and gamma radiation can be reduced by using a lead apron)
- using a radiation monitor to measure how much radiation has been received (if too much has been received, the person can be stopped from working with radioactive isotopes)
Why would a Geiger-Muller tube still give a reading if the radioactive isotope is taken away?
- there is background radiation around us all the time
What are 2 natural sources of background radiation?
- certain rocks are radioactive (eg. granite)
- cosmic rays (eg. from supernovae) which are very high energy particles that travel through space and crash into the Earth's atmosphere
What are 2 man-made sources of background radiation?
- fallout from nuclear weapons testing (radioactive particles that are carried into the atmosphere)
- nuclear accidents (radioactive isotopes can be released from accidents at nuclear power stations)
What factors can affect a person's exposure to background radiation?
- location (eg. people living in areas with large amounts of granite, such as Cornwall or N. Scotland will have a higher exposure to background radiation)
- occupation (eg. cabin crew and airline pilots may be exposed to higher levels of cosmic radiation)
What is radiation dose measured in?
sieverts (Sv)
1000mSv = 1Sv
What are the two main uses of nuclear radiation in medicine?
- exploration of internal organs
- control or destruction of unwanted tissue
Suggest how Dr Eshan could use nuclear radiation to explore the faulty thyroid gland in a patient?
- the thyroid gland works by absorbing iodine and using it to make hormones
- the patient is given a solution of radioactive iodine to drink
- radioactive iodine emits gamma radiation which passes out of the body (high penetrating power)
=> this gamma radiation can be detected
- if the scan shows that the thyroid gland has absorbed too much or too little iodine, then Dr Eshan can use this to diagnose the patient's condition
Why might Dr Eshan use a radioactive tracer to take bone scans in a patient?
- to visualise damage caused by arthritis
- or to detect tumours
What are some issues Dr Eshan must consider when using a radioactive tracer?
- the tracer must emit radiation that can pass out of the body and be detected (not alpha radiation but beta or gamma radiation)
- the tracer must not be strongly ionising to minimise unwanted damage to body tissue (gamma emitters are god tracers as they are weakly ionising)
- the tracer must not decay into another radioactive isotope
- the tracer must have a short half-life so it is not present in the body for a long period of time
Why would a radioactive tracer with a long half-life be unsuitable for exploring internal organs?
- they continue to decay in the body for a long time => cause significant damage to body tissues
What is radiotherapy?
- the treatment and destruction of disease, especially cancer, using ionising radiation
What type of radiation would be usually used to destroy cancers? Why would this type of radiation be suitable?
gamma radiation
- weakly ionising (minimal damage to body tissue)
- high penetrating power (can destroy tumours)
What is one problem with radiotherapy to control or destroy unwanted tissue?
- healthy tissue may also be damaged
What is the main benefit of having the source of radiation inside the body in controlling or destroying unwanted tissue?
- the radiation is targeted very precisely to the tumour => less damage to health tissue
What is 'nuclear fission'?
- the splitting of a large and unstable nucleus (eg. uranium or plutonium)
What must usually happen in order for nuclear fission to occur?
- spontaneous fission is rare
- the unstable nucleus must first absorb a neutron, which then triggers the nucleus to split
What is produced when a nucleus undergoes fission?
- two smaller nuclei, roughly equal in size (daughter nuclei)
- two or three neutrons
- gamma rays
- energy is released by the fission reaction (all of the fission products have kinetic energy)
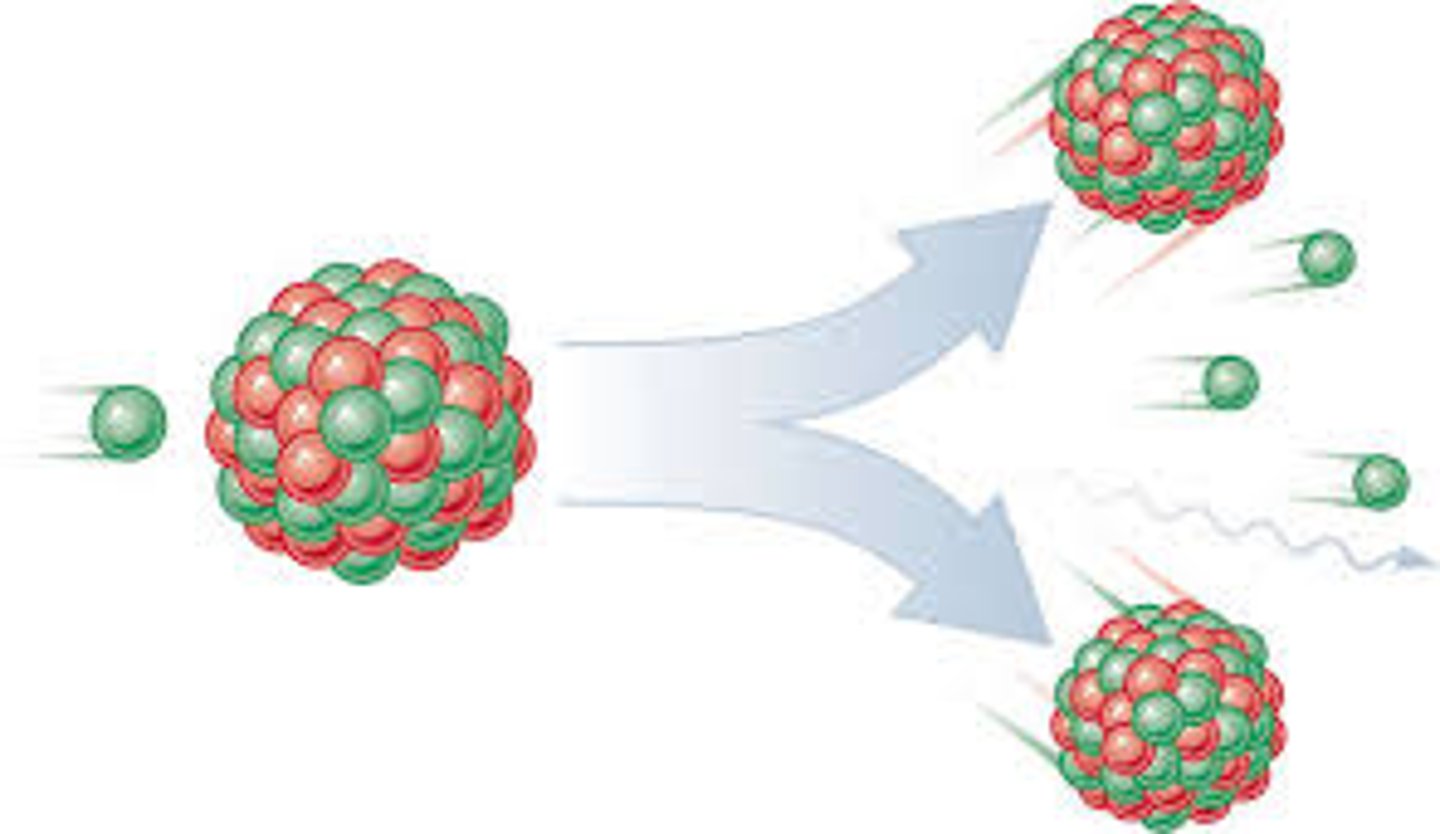
Describe the process of nuclear fission. (4 marks) (Exam Q)
- a neutron is absorbed by a large nucleus
- the nucleus splits into two smaller nuclei
- releasing energy and gamma rays
- and two or three neutrons
What does it mean if the neutrons may go on to start a chain reaction?
- the neutrons produced by nuclear fission can now be absorbed by more nuclei (eg. the nucleus of an isotope of uranium) => triggers fission again (nuclear fission needs the absorption of neutrons)
- these isotopes undergo fission and produce more neutrons => nuclear fission repeats in another uranium isotope again and again (a chain reaction)
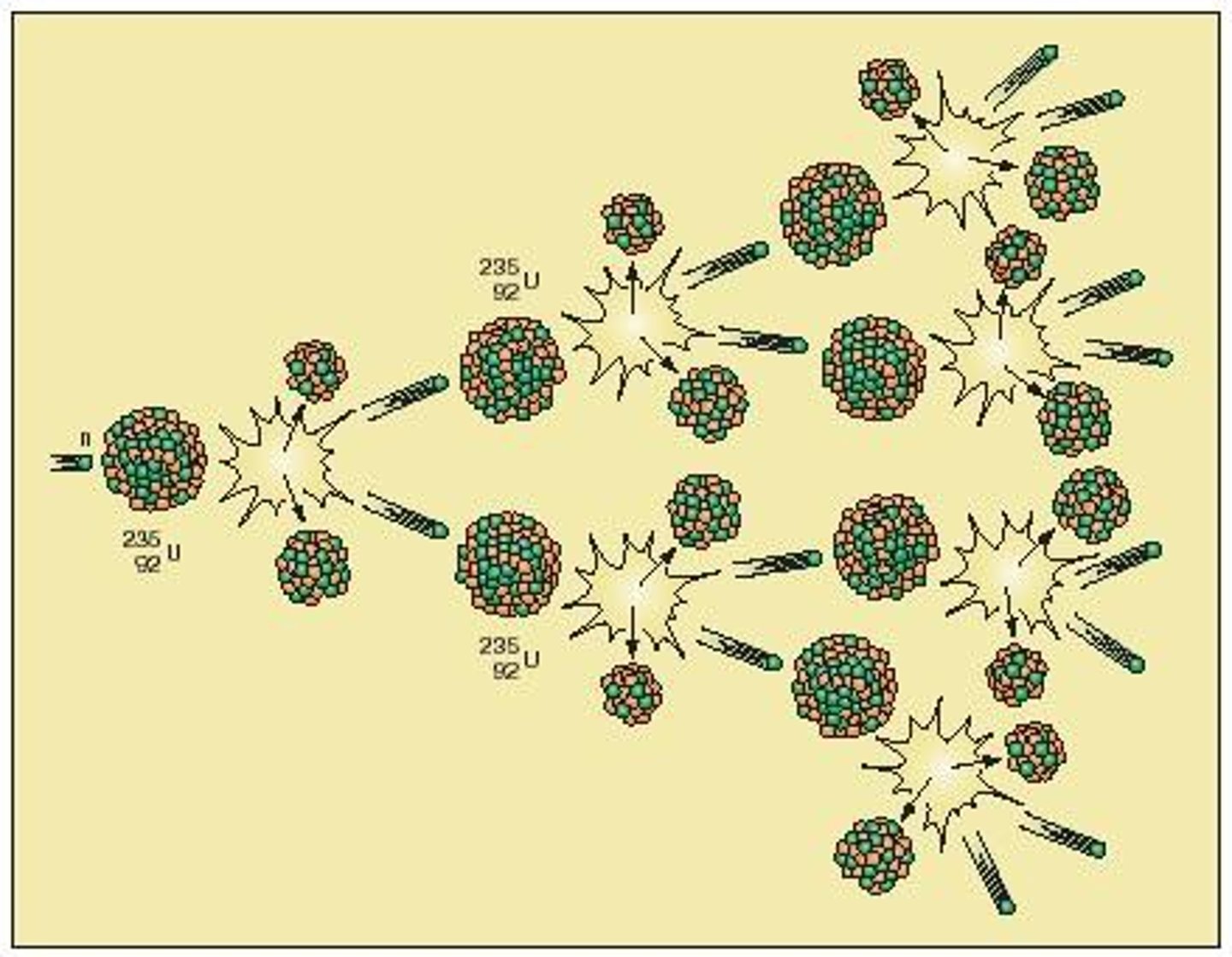
What can a controlled nuclear fission chain reaction be used for?
- to release energy in a nuclear factory
What is an explosion in a nuclear weapon caused by?
an uncontrolled nuclear fission chain reaction

What is 'nuclear fusion'?
- the joining of two light nuclei (eg. hydrogen) to form a heavier nucleus
- some of the mass of the nuclei may be converted into energy, which is released as radiation
A teacher used a Geiger-Muller tube and counter to measure the number of counts in 60 seconds for a radioactive rock.
The counter recorded 819 counts in 60 seconds. The background radiation count rate was 0.30 counts per second.
Calculate the count rate for the rock. (Exam Q)
- count rate = 819/60
- count rate = 13.65
- corrected count rate = 13.35 per second
A householder is worried about the radiation emitted by the granite worktop in his kitchen.
1 kg of granite has an activity of 1250 Bq. The kitchen worktop has a mass of 180 kg.
Calculate the activity of the kitchen worktop in Bq. (Exam Q)
- activity = 1250 × 180
- activity = 225 000Bq
Explain the ideal properties of a radioactive source for use in medical diagnosis. (Exam Q)
• short half-life or half-life of a few hours
• short half-life means less damage to cells / tissues / organs / body
• low ionising power
• low ionising power means less damage to cells / tissues /
organs / body
• highly penetrating
• highly penetrating means it can be detected outside the body
• emits gamma radiation
Describe the process of nuclear fission. (Exam Q)
- a neutron is absorbed by a large nucleus
- the nucleus splits into two smaller nuclei
- releasing energy and gamma rays
- and two / three neutrons
Beta decay does not cause the mass number of an atom to change. Explain why not. (Exam Q, only 2mks)
- beta decay is the emission of an electron from the nucleus when a neutrons decays into a proton and neutron
- this means that the number of neutrons decreases by one
- but the number of protons increases by one
- the mass number is the protons + the neutrons
- so there is no change to the mass number as the decrease and increase is equal
A student models the random nature of radioactive decay using 100 dice. He rolls the dice and removes any that land with the number 6 facing upwards. He rolls the remaining dice again. The student repeats this process a number of times.
Give two reasons why this is a good model for the random nature of radioactive decay. (Exam Q)
- cannot predict WHICH dice / atom will 'decay'
- cannot predict WHEN a dice / atom will 'decay'