Bio Lab practical questions
1/189
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
190 Terms
Lab basics
kilo (k) factor
base unit factor
centi factor
milli factor
micro factor
1000
1
0.01
0.001
0.000001
submultiple are
multiples are
smaller than the base
bigger than the base
Milli
a submultiple 1000 times smaller than the base unit
Micro-
a submultiple one million times smaller than the base unit
Centi-
a submultiple 100 times smaller than the base unit
Kilo-
a multiple 1000 times larger than the base unit
What is the total magnification of a microscope with 20x ocular lenses and a 30x objective lens
600
What happens to the field diameter as magnification increases?
The field diameter gets smaller.
(the less of the specimen you can see)
You know that the field diameter of a scope using a 10x objective lens is 20 mm. What is the field diameter of a 40x lens in mm? Do not include units in your answer
5
Field diameter= FD1 x Mag1 = FD2 x Mag2
Due to the short working distance, the high magnification objective lens is the one most likely to contact a slide, potentially breaking the slide and damaging the lens
True
Which objective lens should be used to initially get a specimen in focus under the microscope?
4x (lowest one)
Why should you only use the fine focus when trying to put a specimen in sharp focus when using the 40x objective lens?
because the working distance is too small, and the coarse focus moves the stage too quickly.

Name
A = Flask
B = Graduated cylinder
C = Beaker
D = Test Tube
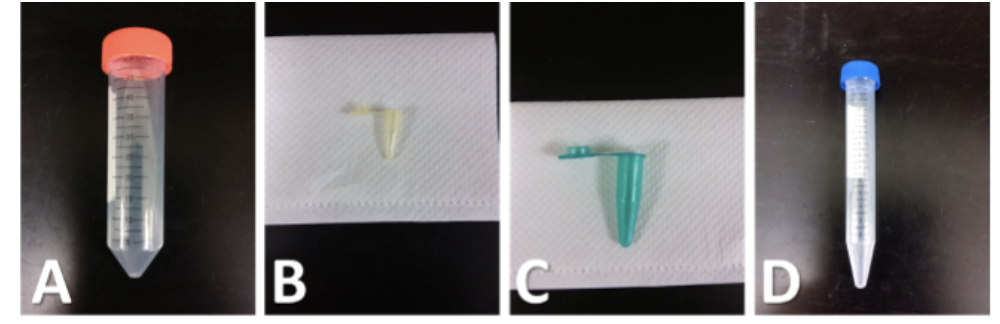
Name
A = Centrifuge tube (50mL)
B = PCR tube
C = microcentrifuge tube
D = centrifuge tube (15mL)
Total mag formula
objective mag x ocular mag = total mag
As magnification increases, field diameter becomes _______.
smaller
How many mL are in 4.6 L?
big to small = mulit
4600
How many mg are in 63.6 g
big to small = multi
63600 (x1000)
Convert 899 µg to mg
small to big = divide
0.899 (/1000)
Convert 62 µL to mL
small to big (divide)
0.062 (/1000)
Convert 81 L to mL
big to small (mulit)
81000 (x1000)
Convert 6 L to µL
L to mL to uL
big to small (mulit)
6000000 (x1000ml x1000 ul)

Name
A = beaker
B = graduated cylinder
C = erlenmeyer flask
D = test tube
microgram
ug
small to large = dividing so you move deci to ___
large to small = multiplying so you move deci to ___
left
right
nm to mm
divide by 10^6 (6 becomes pos because moved to denomonator)
m to um
multiply (10^6 6 stays pos because you are multiplying which is in the numorator)
kg to cg
multiply (times by 10² stays pos 2 bc multiplying happens in numorator)
unknown field diameter
(known FD) x (initial FD) / final Mag
Molarity
mol/L
MW
g/mol
initial con. x volume initial = final con. x volume final
C1 x V1 = C2 = V2
in this formula units MUST MATCH
make 5ml of 10 mM NaCl from 50 um stock
1.0 uL of 50 um
mL to uL
x 1000
can a hypothesis be phrased as a question
no
Independent variable vs Dependent variable
IV : the thing you manipulate or change
DV; what changes bc of the IV
What button is used to subtract the mass of the weighing vessel from the display on a digital scale?
Tare
solute vs solvent
Solute: the substance being dissolved in a solution
Solvent: the liquid in greater quality (where the solute is being dissolved in)
How many grams of KOH are needed to make 1 liter of a 1 M KOH solution? FW KOH = 56.11 g/mol.
1
How many moles are in 1 L of a 2 M KOH solution? (FW KOH = 56.11 g/mol
2
How many moles are in 0.1 L of a 2 M KOH solution? (FW KOH = 56.11 g/mol)
0.2
How many grams of CaCO3 are needed to prepare 2 L of a 6 M CaCO3 solution (FW of CaCO3 = 100.09 g/mol). Do not include units in your answer. Report your answer to the nearest two decimal places
1201.08
How many grams of NaCl are needed to prepare 1200 mL of a 2 M NaCl solution (FW of NaCl = 58.44 g/mol). Do not include units in your answer. Report your answer to the nearest two decimal places
140.26
Which is the best instrument to move 4 mL of water?
5mL serological pipette
best instrument for
100uL
300uL
1.5mL
7mL
P200 micropipette
P1000 micropipette
2mL serological pipette
10mL serological pipette
What is the purpose of the "first stop" when using a micropipette?
This is the appropriate starting position for drawing liquid up into the pipette
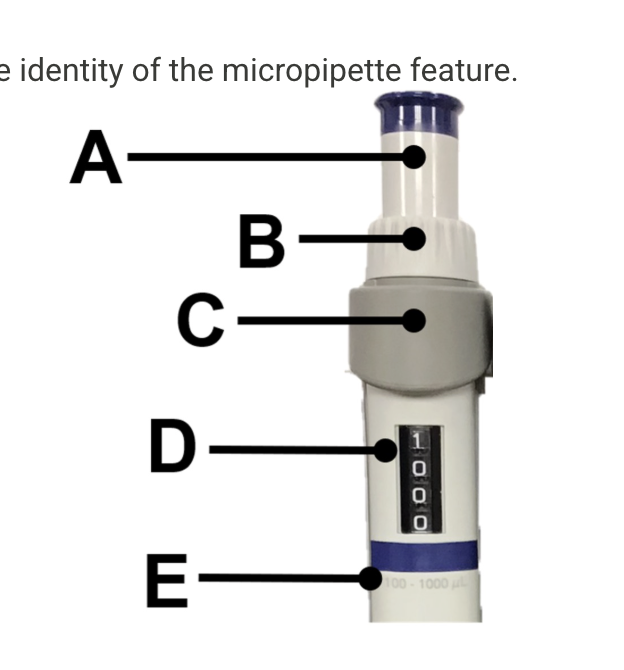
Name
A) plunger
B) volume Adjustment dial
C) Tip ejector button
D) volume display
E) volume range
How many grams of NaCl are needed to prepare 40 mL of a 2 M NaCl solution? (FW NaCl = 58.44 g/mol)
4.675
What's the difference between the tare and zero function on the balance? Describe a situation in which it would be beneficial to use the tare function over the zero function.
The zero button is used when there is nothing on the scale and you just want to calibrate the scale. However, the tare button is used to cancel out the weight of the object that is on the scale. It would be beneficial to use the tare button over the zero button if you are weighing a new mass in a beaker that already has liquid in it.
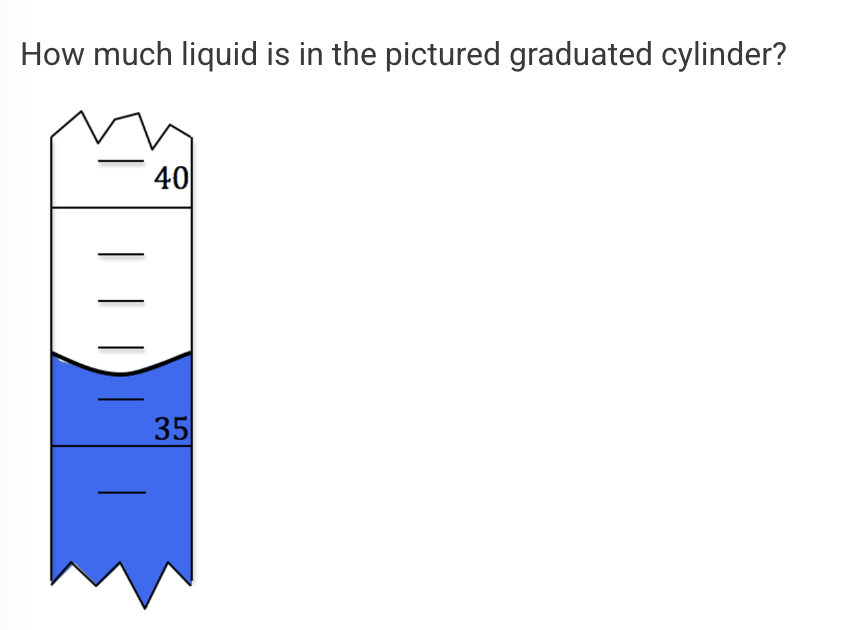
How much
36.5
Density formula
D = Mass/Volume
Chemistry
What must you make sure to always do when moving the probe between solutions
rinse the bottom third of probe w/ dH2O
How many grams of sucrose (FW = 342.3 g/mol) are needed to make 40 mL of a 0.2 M sucrose solution?
2.7384
How many mL of 1 mM BSA stock solution do you need to make 40 mL of an 0.2 mM BSA working solution
8
How can you tell that disodium phosphate (Na2HPO4) is the conjugate base and monosodium phosphate (NaH2PO4) is the weak acid
Disodium phosphate has one less hydrogen atom than monosodium phosphate
How many mL of the 100 mM NaH2PO4 stock solution will you need to make 40 mL of a 1.8 mM NaH2PO4 working solution
0.72
V= mL x mM / mM
Biological molecules
Glucose =
lactose =
starch =
maltose =
galactose =
glycogen =
glucose =
cellulose =
sucrose =
monosaccaride
disaccharide
polysaccharide
disaccharide
monosaccharide
polysaccharide
monosaccharide
polysaccharide
disaccharide
Enzymes
Both catalysts and enzymes work by lowering the activation energy needed to start a reaction.
true
A biological molecule that increases the rate of a reaction is most likely...
an enzyme
enzymes are ____
proteins
an enzyme is not used up in the reaction it catalyzes
true
This part of the enzyme binds to the substrate.
active site
True/False: A protein that has lost its three-dimensional structure due to low pH would still function as an enzyme.
False
A phosphatase enzyme will...
remove a phosphate group from a molecule.
Hue reflected is the one ____
seen by use
lipids =
nucleic acids =
proteins =
starch =
lipase
nuclease
pepsin
amylase
amylase produces
maltose
Lipase produces which product(s)?
glycerol and fatty acids
Pepsin produces which product(s)?
amino acids and peptides
How many essential amino acids are there?
9
Salivary amylase is an enzyme that begins breaking down carbohydrates while the food is still in the mouth. Once the food passes into the stomach, what happens to the salivary amylase?
It is inactivated by the acidic conditions of the stomach.
Which biological macromolecule(s) is/are chemically digested in the duodenum?
carbohydrates, lipids, and proteins
proper way to load a centrifuge tube into the rotor
hinges out
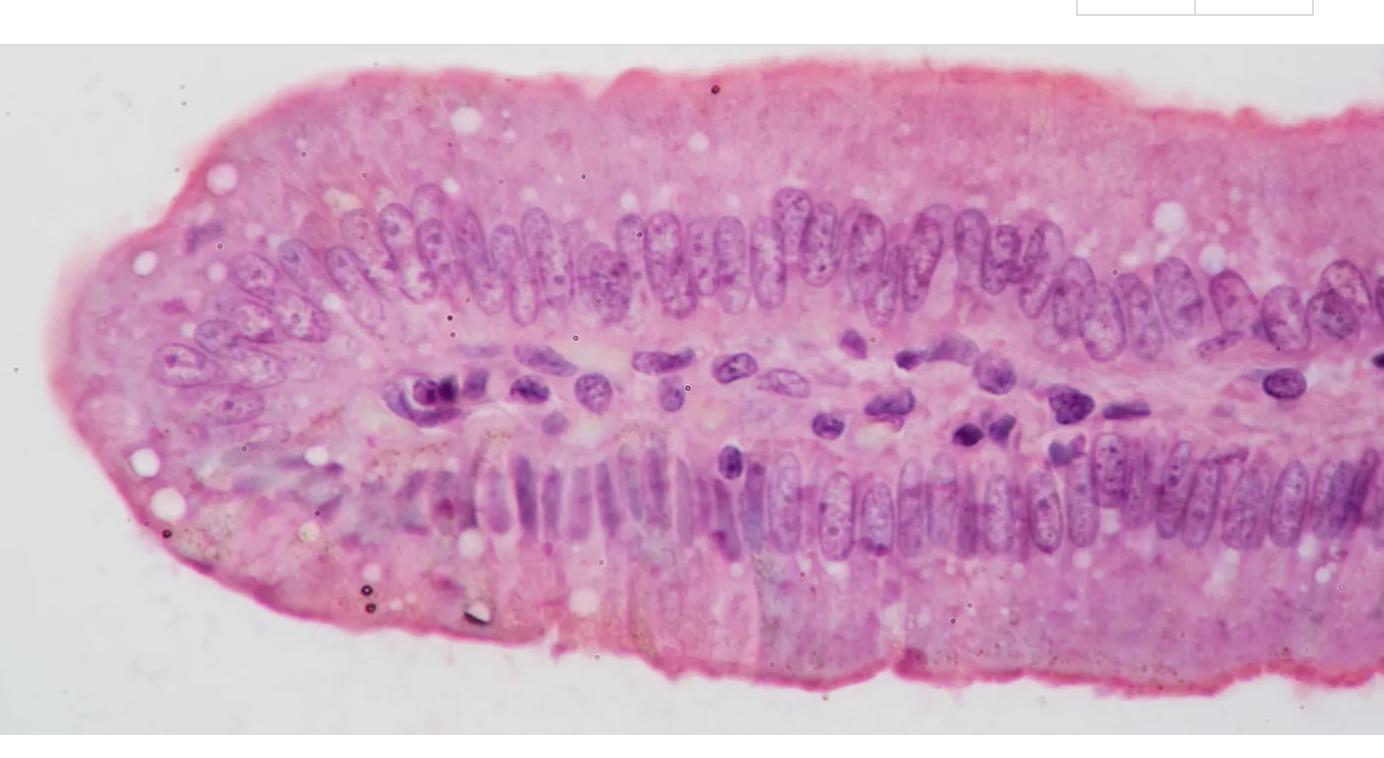
What type of tissue
simple columnar epithelium
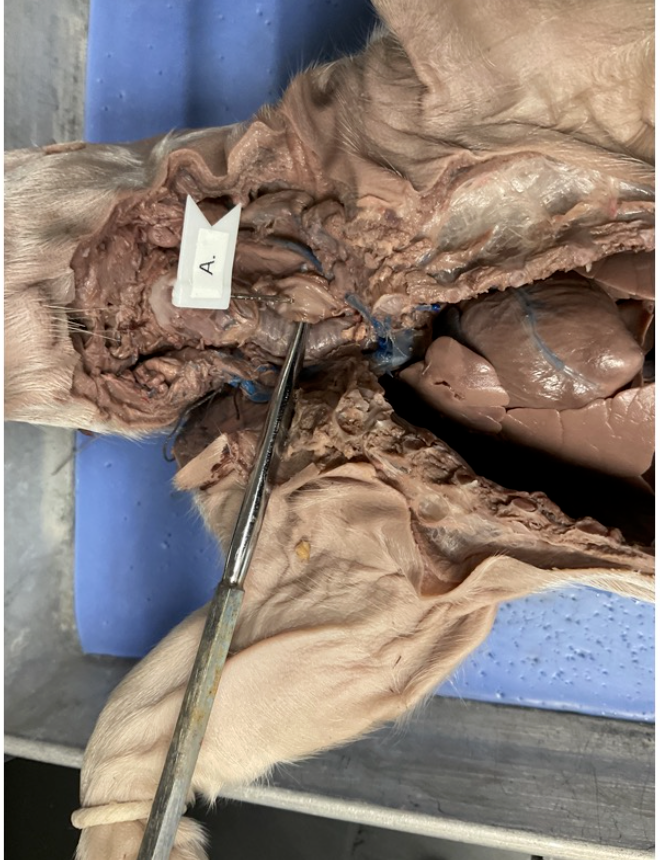
esophagus
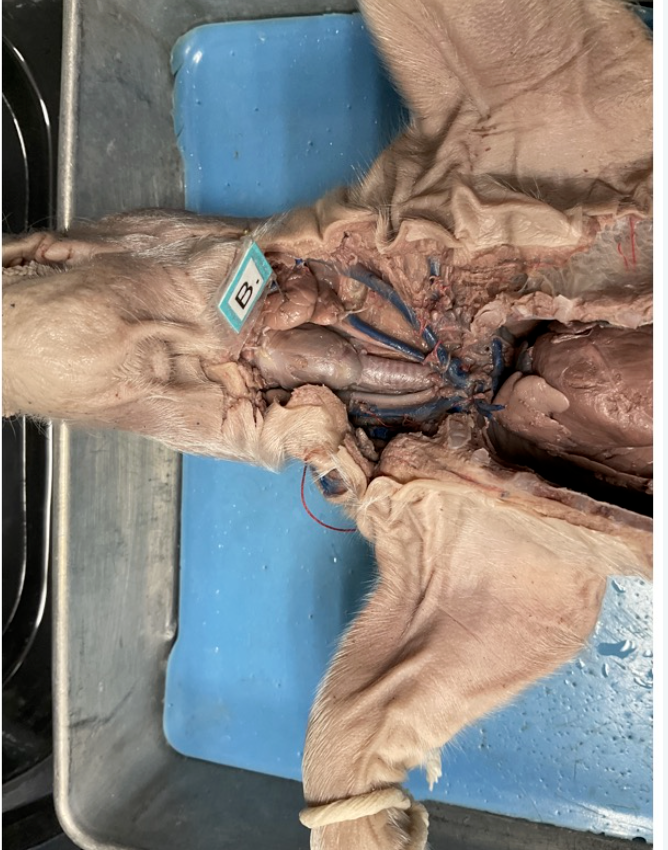
salivary gland
cecum
stomach

duodenum
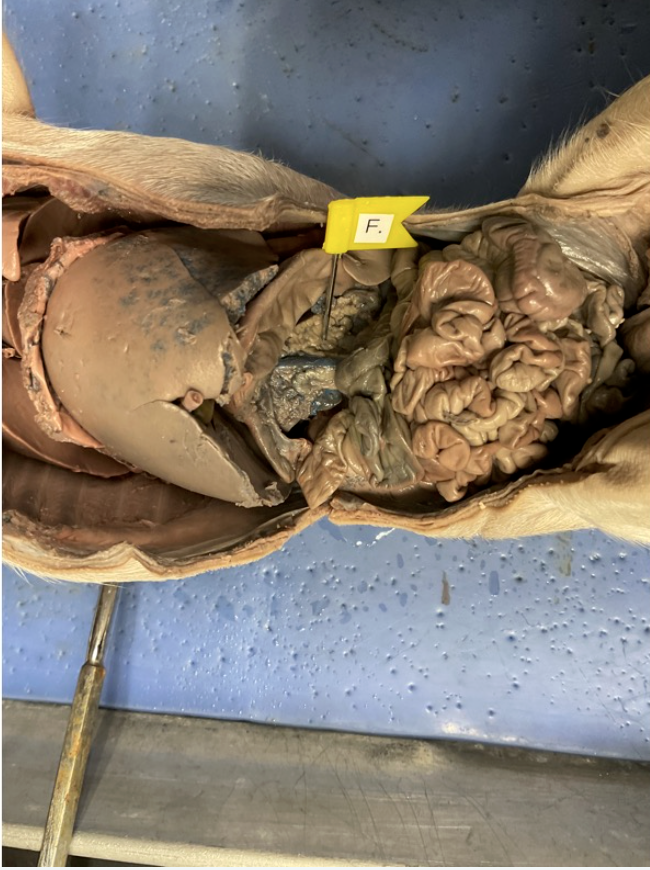
pancrease
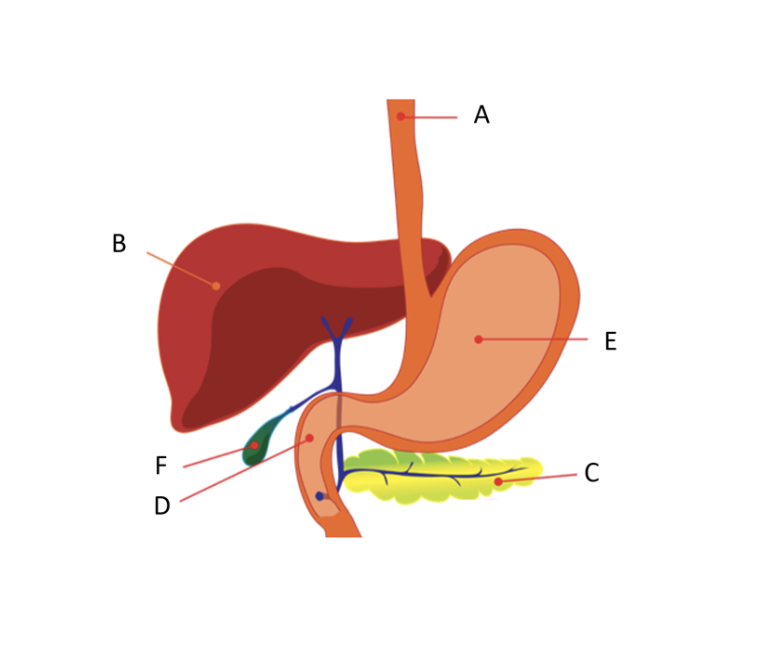
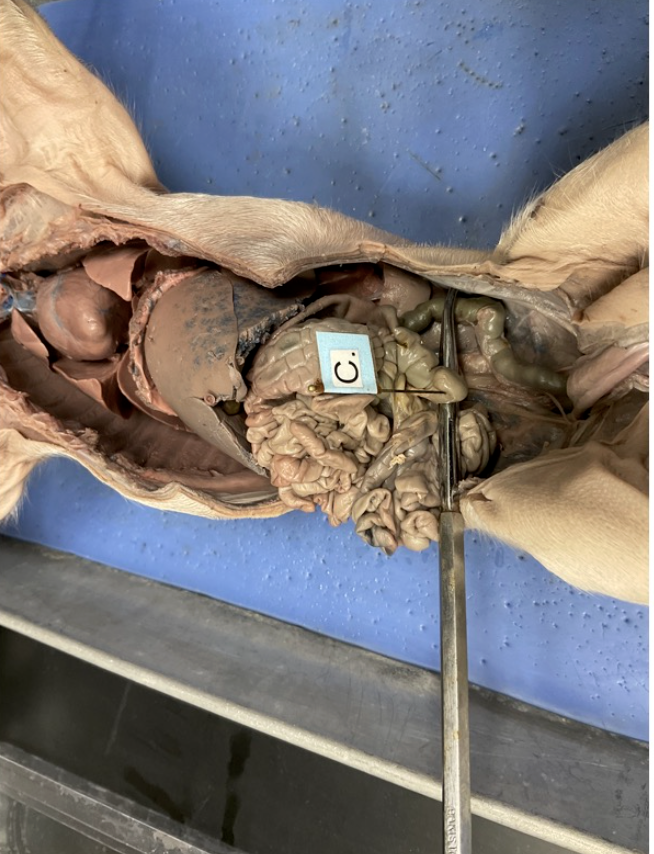
Name
A) esophagus
B) liver
C) pancreas
D) duodenum
E) stomach
F) gallbladder
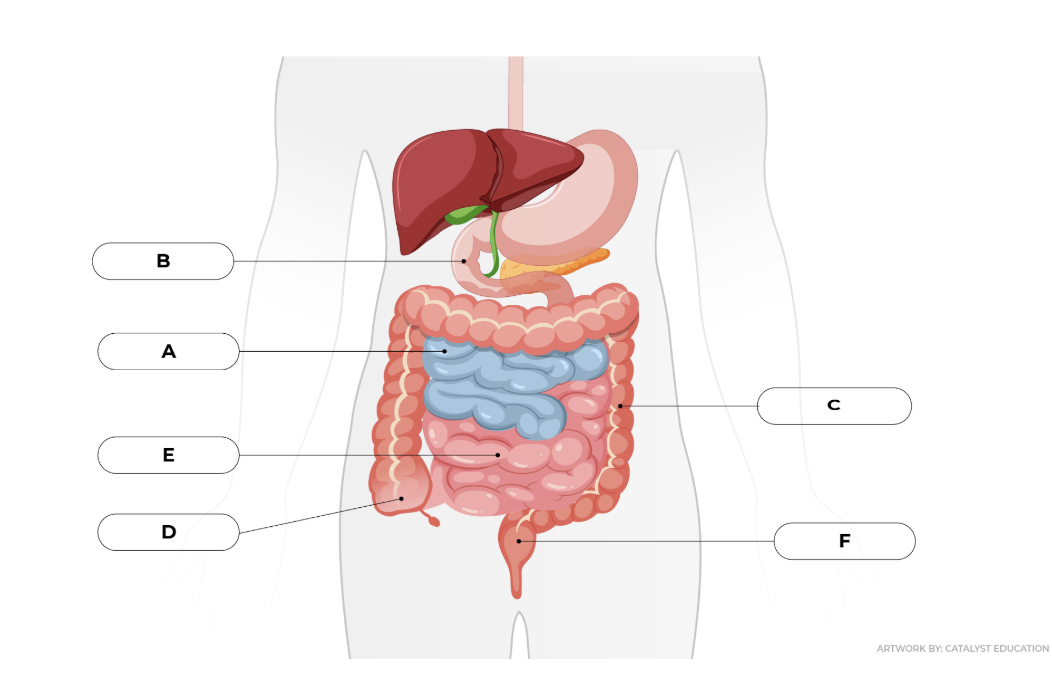
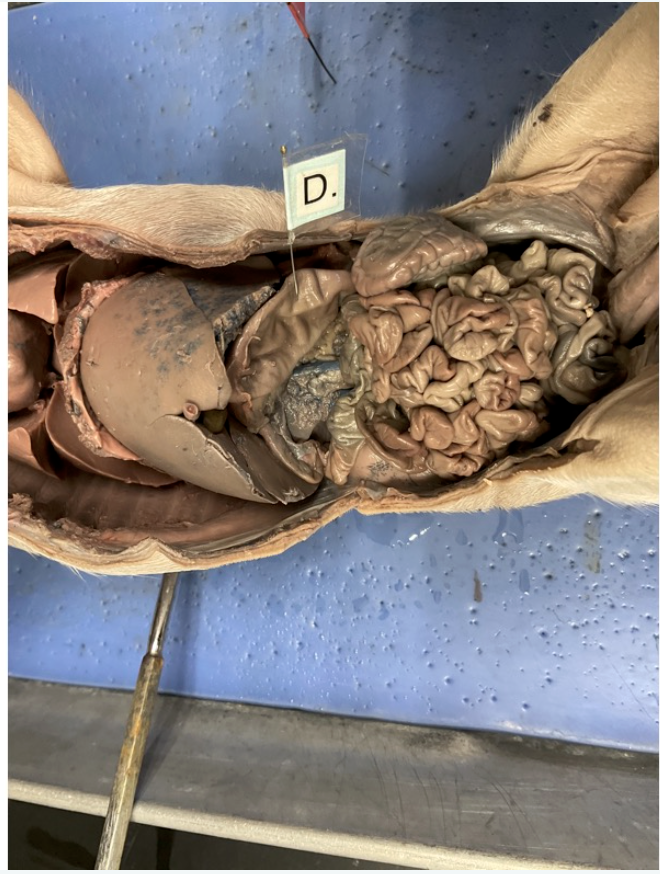
A) jejunum
B) duodenum
C) descending colon
D) cecum
E) ileum
F) rectum
Membranes
True or False: Osmosis is a an active process that requires ATP.
F
Osmosis
the diffusion of water across a membrane.
The osmolarity of a cell is 0.3 Osm. Which solution of NaCl is isosmotic to the cell?
0.15
A cell is placed into a solution of pure water. What is the tonicity of the water solution?
hypotonic
Placing an animal cell into a hypertonic solution will result in...
crenation
A synthetic "cell" is made out of semi-permeable dialysis tubing, and it contains a 10% sucrose solution. It is placed into a beaker containing a 20% sucrose solution. The solution in the beaker is...
hypertonic
Imagine another scenario. This time, a cell is placed in a solution of NaCl that is more concentrated than the cytosol inside the cell. NaCl and its ions are not able to cross the cell membrane. What will happen?
Water will flow out of the cell.
How many grams of maltose do you need to make 550 mL of a 13 % w/v solution?
71.5
How many mL of acetone do you need to make 450 mL of a 8% v/v solution?
36
How many grams of NaOH do you need to make 750 mL of a 8% w/v solution?
60
How many mL of ethanol do you need to make 150 mL of a 65% v/v solution?
97.5
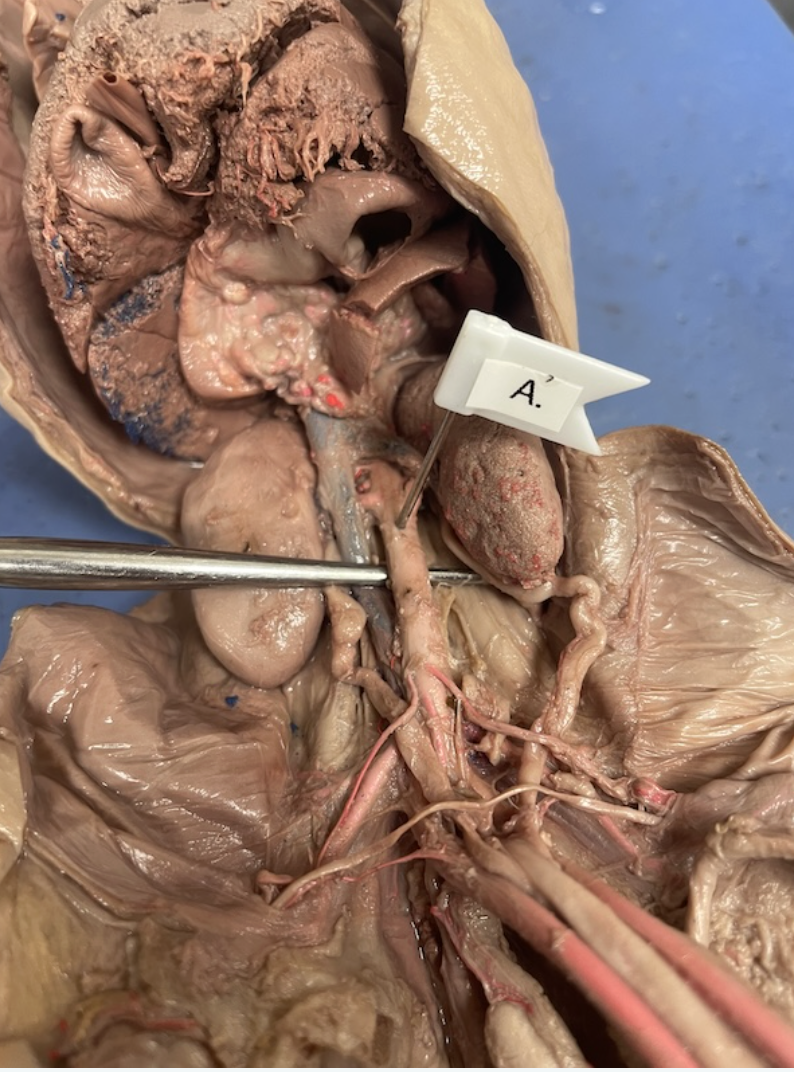
abdominal aorta
inferior vena cava