Module #8 (Ectodermal Derivatives 2)
1/94
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
95 Terms
Most important part relates to the
stage that is very critical for a correct closing of the tube
What do you got here?
Area of the Ectoderm
Neural tube formation start
Notochord that release signals (sonic Hh)
Presence of adhesion molecules, what the most important in terms of Neurulation?
Neurulation requires specific cadherin adhesion molecules for patterning
Cadherin (calcium-dependent adhesion molecules)
Transmembrane proteins → that will connect two cells
What are the three primary structural domains of a cadherin molecule?
Cadherins consist of an extracellular domain (containing adhesive recognition and calcium-binding sites), a transmembrane domain (which spans the cell membrane), and an intracellular domain (containing phosphorylation sites).
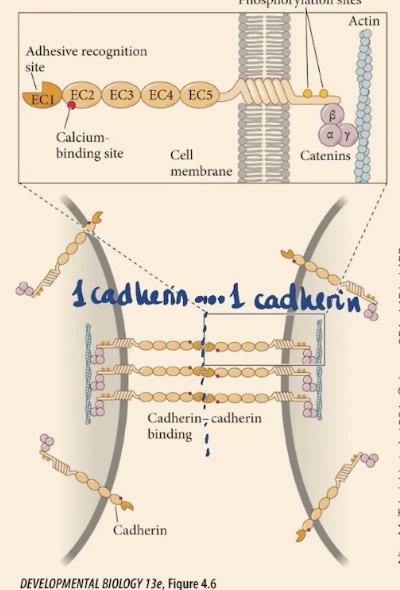
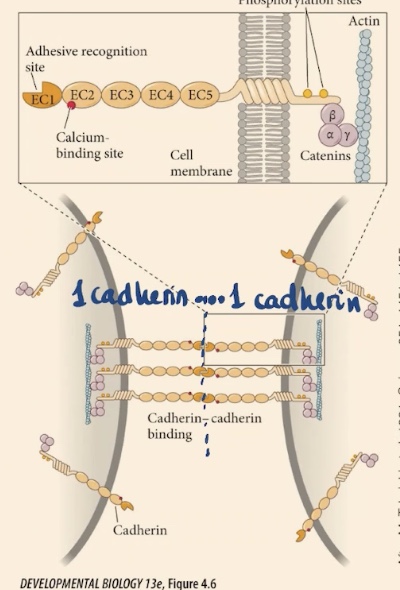
Cadherin’s intracellular domain is going to anchor the membrane to . There are also , on each of these domains that are repetitive domains. At one of the extremes they have this _.
Actin cytoskeleton, calcium binding sites, adhesive recognition site → they are going to recognize the same kinds of cadherin, that is going to attach one cell to the other
Different flavors of Cadherins, the most important are
E-cadherins → because they are found in the early embryo and also in the epidermis, the epithelium later on
Other types of Cadherins
N-cadherin → expressed in neural tissue (relevant to us)
P-cadherin → expressed in placenta
R-cadherin → expressed in the retina
What if you were to perform an In-Situ hybridization of E-cadherins (green)?
Will be expressed in the surface Ectoderm, but in the Neural plate remember that it is receiving signals from the notochord → so it is going to stop expressing E-cadherin and is going to express N-cadherin (red)
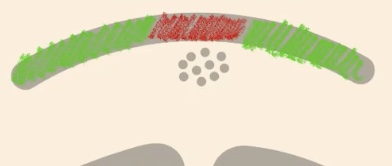
How does cadherin expression affect cell attraction?
Cells expressing E-cadherin have a much higher attraction (affinity) to other cells expressing that same kind of cadherin. This causes cells with like-cadherins to stick together and separate from cells expressing different types.
During neurulation, which tissues express E-cadherin vs. N-cadherin?
E-cadherin: Expressed by the surface ectoderm (prospective skin).
N-cadherin: Expressed by the neural plate/neural tube cells.
Why does the neural tube separate from the surface ectoderm?
Because the cells feel more attracted to those expressing their same kind of cadherin, they stop being attached to cells with different cadherins. This allows the tube to close and separate from the surface.
How does neural tube closure/fusion occur across the embryo?
This process of closure and fusion happens at different positions along the embryo rather than all at once.
How many initial closure points are there in human and mouse embryos?
There are three initial points (Closure 1, 2, and 3) where the neural tube first makes contact and begins to fuse.
What happens after the initial closure points (1, 2, and 3) are established?
Once these areas are closed, a "zipper" effect occurs. The neural tube continues to close by extending from these points in either an anterior or posterior direction.
In what direction does the neural tube close during the "zippering" phase?
The neural tube closes in the direction of the specific closure point, moving toward the anterior or posterior poles to complete the tube.
What are the open gaps in the neural tube called before closure is complete?
These areas are called neuropores. They are the normal, temporary regions of the neural tube that have not yet fused.
Anterior neuropore
located between two of the most anterior sides of the closure
How does the huge posterior neuropore close?
it closes progressively starting from the cervical point and moving toward the tail of the animal.
Which two other specific gaps (neuropores) are mentioned in the closure process?
The hindbrain neuropore and a huge posterior neuropore.
How is neural tube closure regulated, and what is the impact of a failure in this process?
It is highly regulated. A failure in the closure "fix" results in tremendous, really negative consequences for the embryo.
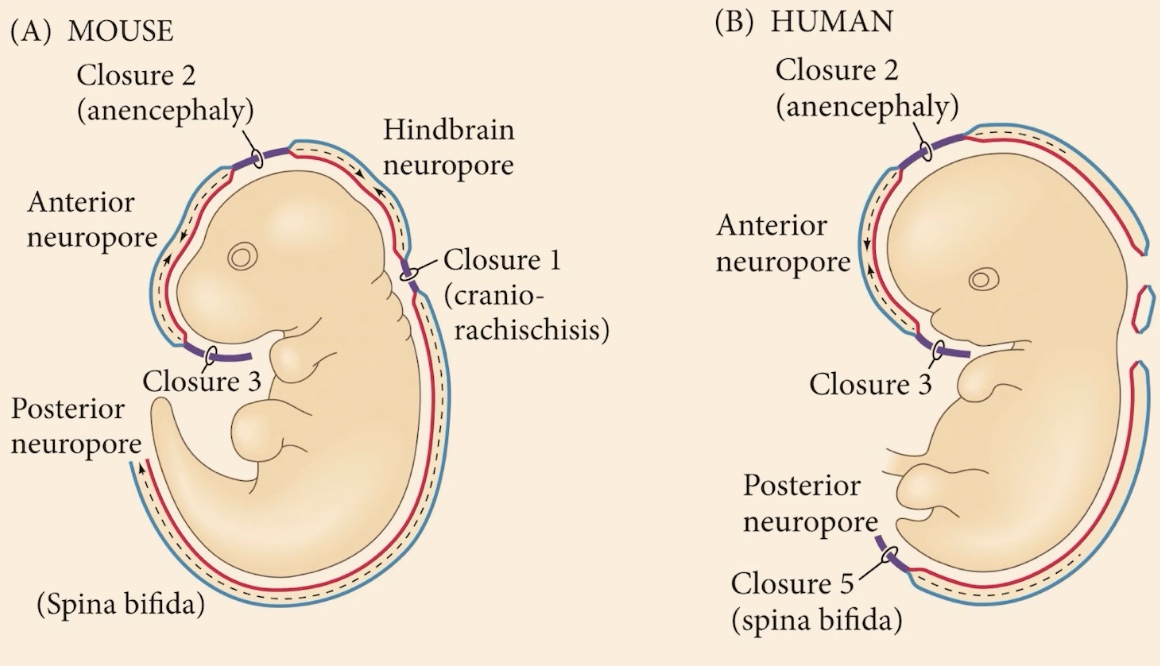
What occurs if the anterior neuropore fails to close between Closure 2 and 3?
The embryo develops anencephaly. The neural tube remains open, and the brain develops externally, exposed to the improper environment of the amniotic fluid.
What is the ultimate fate of the brain in anencephaly?
Due to exposure to amniotic fluid, the brain eventually degenerates, leaving the embryo without a brain.
What is the phenotype for a complete or partial failure to close the posterior side?
Spina bifida. Unlike anencephaly, this syndrome is compatible with life, and many humans live with it.
What are the physical consequences of spina bifida?
Depending on the specific location of the closure failure, it can result in paralysis or similar issues toward the posterior side of the body.
Some syndromes if you have an opening, a failure or closing of either the hindbrain neuropore or the most posterior side, those
are completely incompatible with life
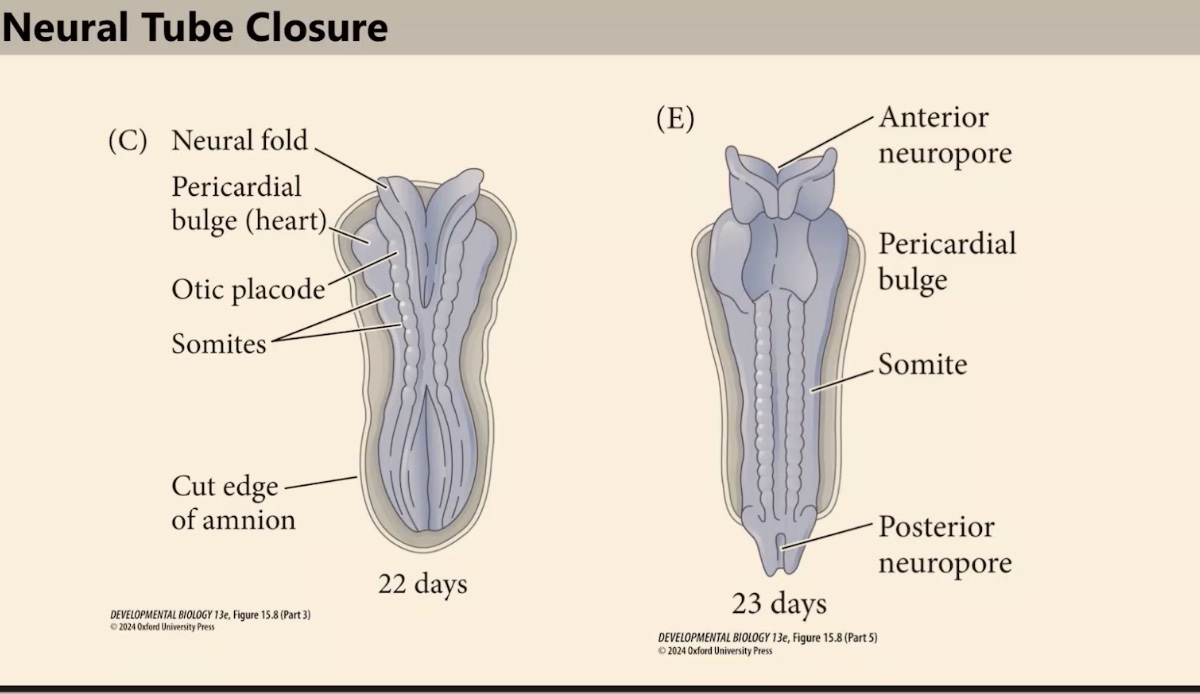
What are we seeing?
Closure begins in the middle of the embryo and proceeds through a zipping process. This closes the tube progressively, with the posterior neuropore closing from the cervical point toward the tail.
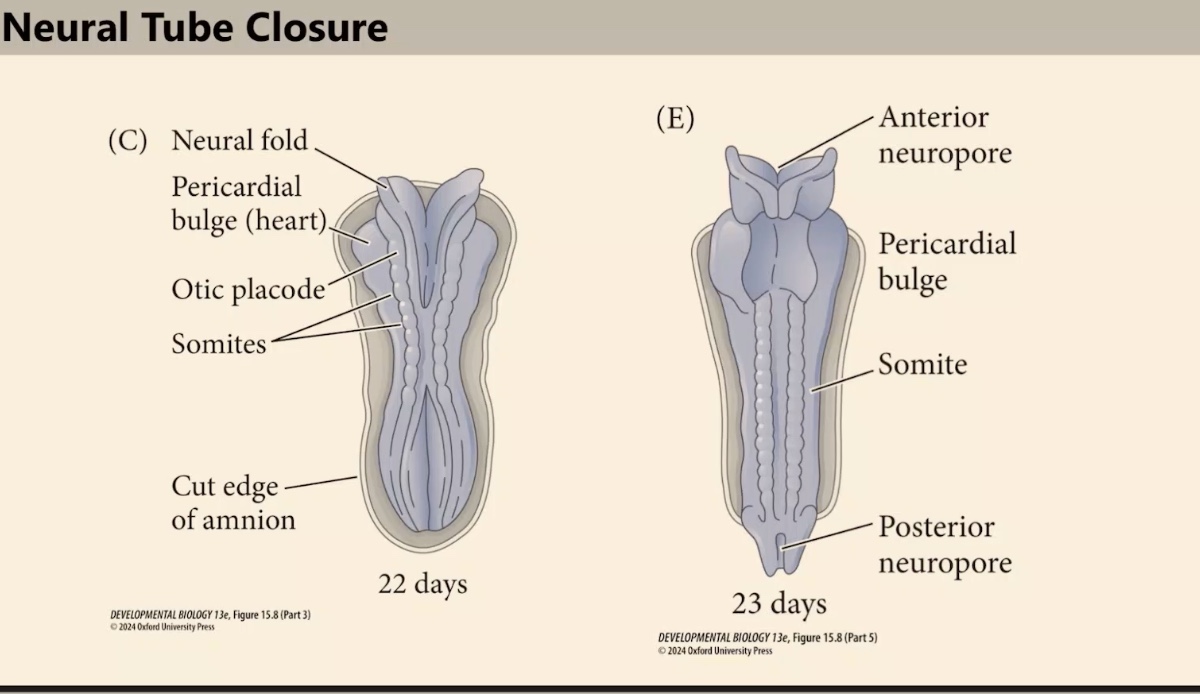
What are the names of the open canals at the ends of the neural tube during the zipping process?
The anterior neuropore and the posterior neuropore.
In the mouse example of a cephalic neural tube defect, what is the condition called and what occurs?
It is called Exencephaly. The brain develops outside the skull cavity (outside the embryo proper) and is exposed to amniotic fluid, which causes the brain to eventually degenerate.
What characterizes Spina Bifida in this context, and what is the functional result?
It is the bulging of the neural tube (the spinal cord). This results in the most posterior part of the animal becoming completely paralyzed.
Is anencephaly typically compatible with life, and how can an individual survive for a period of time with this condition?
Most cases are incompatible with life, but survival is possible (e.g., up to three years) because basic functions are maintained by the spinal cord and other very primitive structures in the neural tube, even if most of the brain is missing.
What is the result of having anencephaly regarding brain structure and long-term prognosis?
he individual lacks most of their brain; while primitive structures can maintain life for a time, the condition eventually leads to death (in the example provided, the child passed away close to age 3).
What is the physical fusion and separation of the neural tube based on?
It is based on the expression of E-cadherins and N-cadherins.
How do E-cadherin and N-cadherin facilitate the separation of the neural tube?
Cells have a higher attraction to others expressing the same kind of cadherin. This specific expression allows the tissues to feel attracted to their own type, leading to the fusion of like-cells and the separation of the tube from the surface.
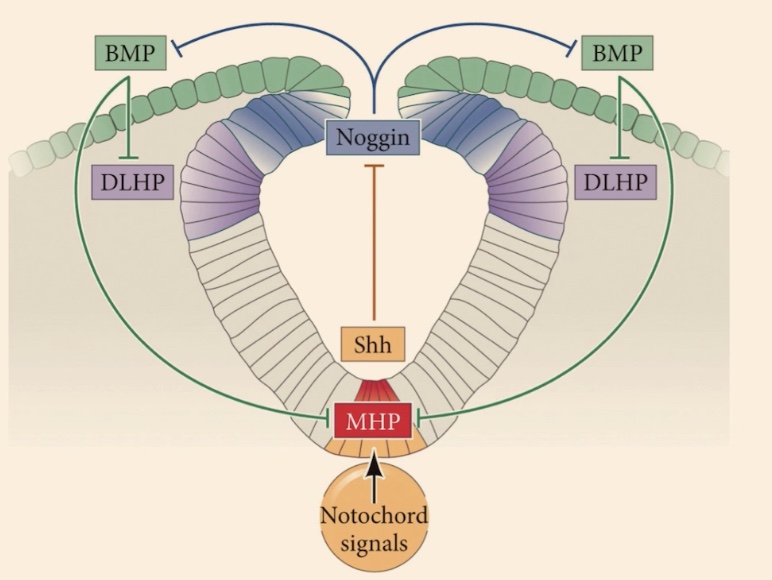
Is the neural tube an exception to polarization, and what initiates its dorsal-ventral patterning?
No, the neural tube is highly polarized in the context of dorsal-ventral patterning, which has tremendous consequences for the function of each neuron. It is initiated by 2 primary signaling centers.
What are the two primary signaling centers?
1) Epidermis (external ectoderm)
2) Notochord
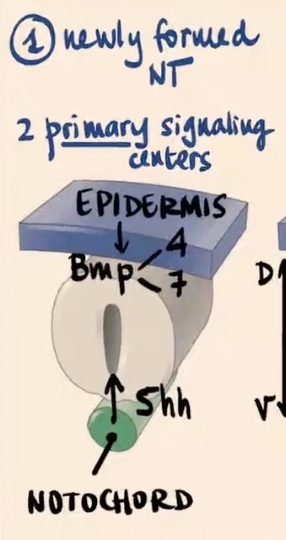
Epidermis produces and releases?
Epidermis (external ectoderm): Produces and releases BMP4 and BMP7
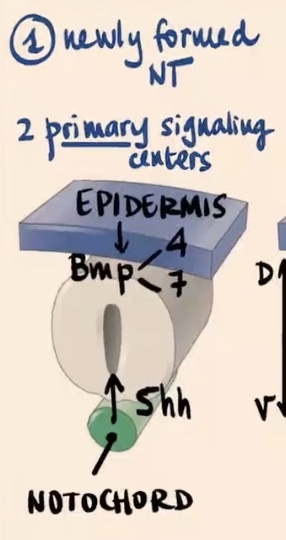
Notochord produces and releases?
Produces and releases Sonic hedgehog (Shh).
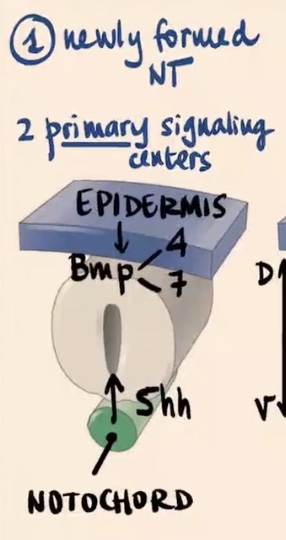
What is the main characteristic of the two families of morphogens (BMP and Sonic) in this process?
They release and secrete molecules to create gradients and create responses that are dependent on the gradient (the specific concentration the cells are receiving).
Second step of Dorso-Ventral regionalization of the Neural tube?
Formation of Secondary Signaling Centers
Describe the second step involving the floor plate and roof plate.
This is the formation of secondary signaling centers where signals are sort of copied inside the neural tube
Floor plate produces
Sonic
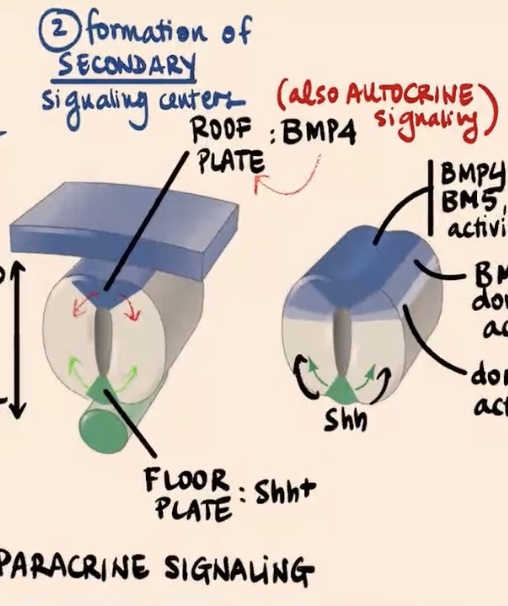
Roof plate (most dorsal area) produces
initially BMP4
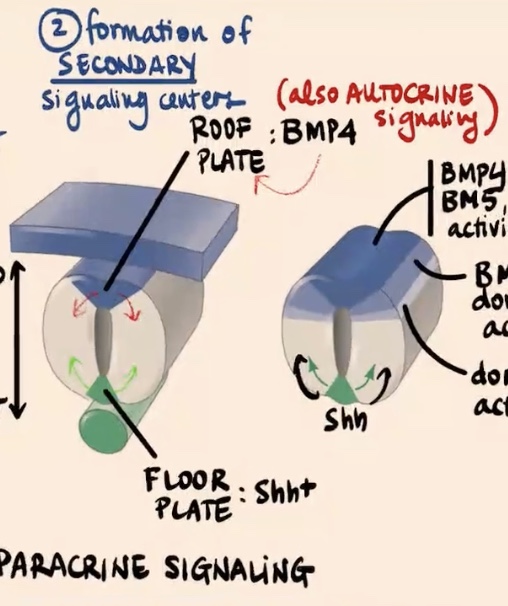
How do BMP4 and Sonic hedgehog act as signals, and what is unique about the BMP signaling case?
BMP4 and Sonic are going to act as paracrine factors (paracrine signaling). Specifically in the case of BMP signaling, there is also sort of a feedback loop where those cells are also signaling to themselves, so they are also undergoing a bit of autocrine signaling.
How do the "different blue tones" in the dorsal area represent the gradient of factors?
Cells express different factors based on their position
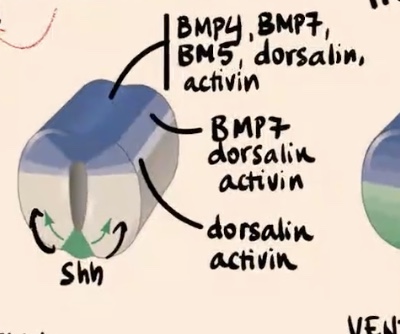
Describe the gradient of factors expressed in the most dorsal aspect of the neural tube and how it changes moving ventrally.
Cells in the most dorsal aspect of the neural tube express really high levels of BMP4, BMP7, BMP5, dorsalin, and activin. Moving away from this source
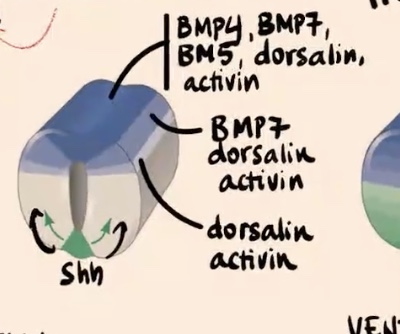
Dorsolateral side of the neural tube
Express only BMP7, dorsalin, and activin.
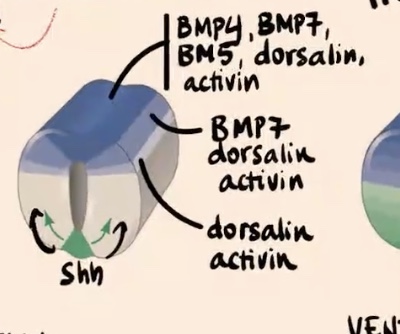
Lower shade (moving further ventrally in the tube)
Express only dorsalin and activin.
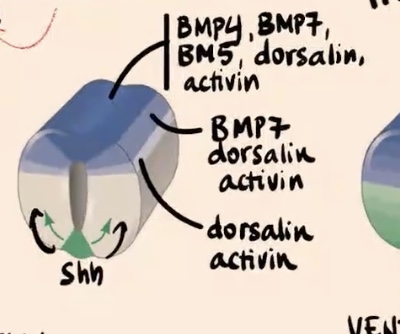
As you move away from the source,
cells of the neural tube are a bit less competent to produce all of these BMP factors.
How is the specification of different neurons integrated in the tube?
Two morphogens intersect and are integrated depending on concentration, similar to bicoid and nanos in Drosophila anterior-posterior specification. The specification of neurons depends on the concentration of Sonic and all those BMP-related factors.
What is the role of the neurons in the most ventral side of the neural tube?
They are essentially motor neurons. They send axons from the spinal cord to the muscles and control voluntary and reflex movements.
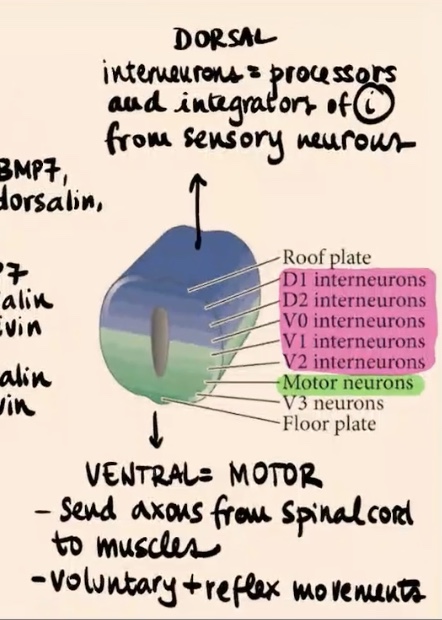
What is the role of the interneurons in the most dorsal side of the neural tube?
They are processors and integrators of information from sensory neurons (e.g., pain, heat, proprioception). They often integrate information directly in the spinal cord (sometimes not even sending it to the brain), so you move away from heat without having to think about it.
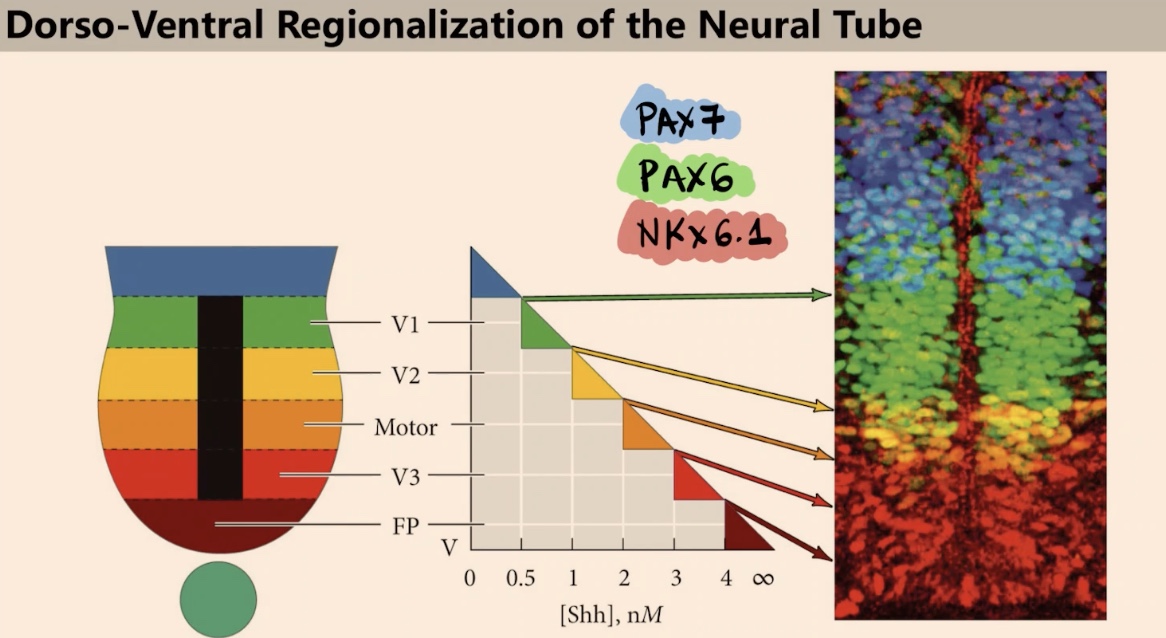
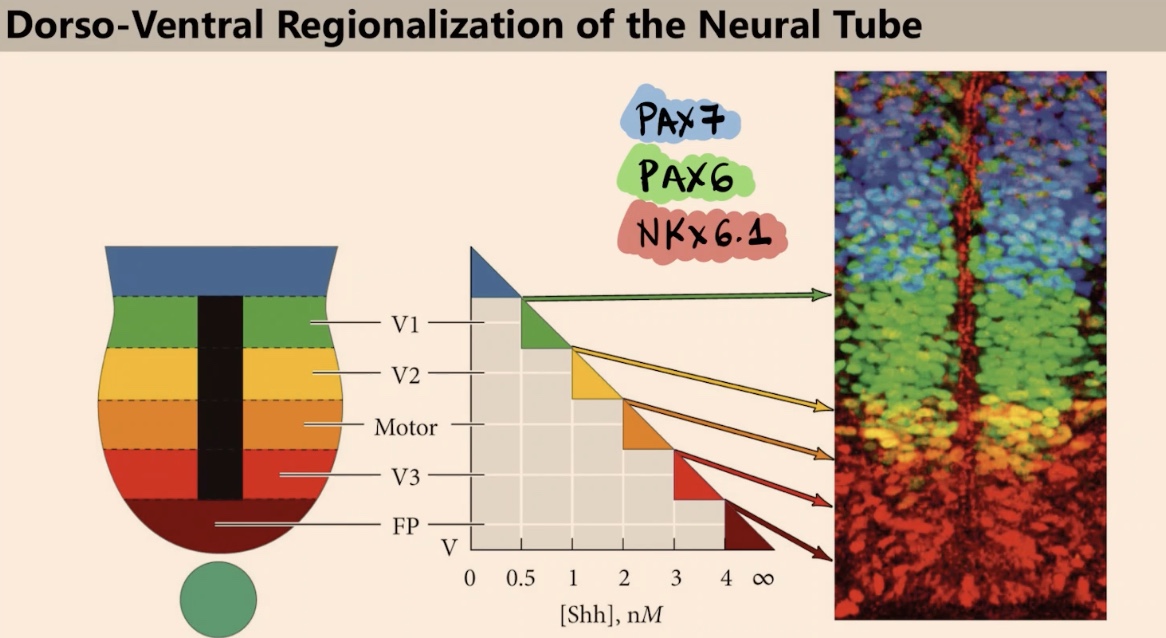
What are we seeing in the immunofluorescence signal, and how are these factors induced?
We see a signal in the nucleus recognizing Transcription factors. These transcription factors are induced by different combinations of sonic and BMP.
What are the three transcription factors discussed in this image, and why do we see six colors instead of just three?
The three factors are PAX7, PAX6, and NKX6.1. We see six colors because of the overlap between colors. For example, the yellow area represents cells expressing both NKX6.1 and PAX6.
What is the advantage of using morphogen signals like Sonic and BMP to specify multiple types of neurons?
Morphogens are useful because you only need two signals to specify multiple different types of neurons, instead of needing a lot of different signals.
What is the key takeaway regarding transcription factors and the different types of neurons (interneurons vs. motor neurons)?
Different combinations of transcription factors will produce different phenotypes in cells. This distinguishes cells in the interneuron phenotype (able to incorporate information) from the motor neurons, which are a "completely different beast."
What governs the anterior-posterior regionalization of the neural tube, and how does it function?
It is highly, highly dependent on Hox genes, the same genes from Drosophila. Once segmentation is completed, they provide identity to each segment through co-linear expression in the closed neural tube.
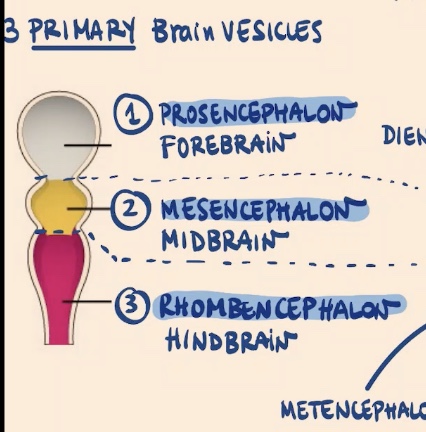
In the most early differentiation, what are the three primary brain vesicles that can be recognized in the most anterior side?
1) Prosencephalon (most anterior)
2) Mesencephalon
3) Rhombencephalon
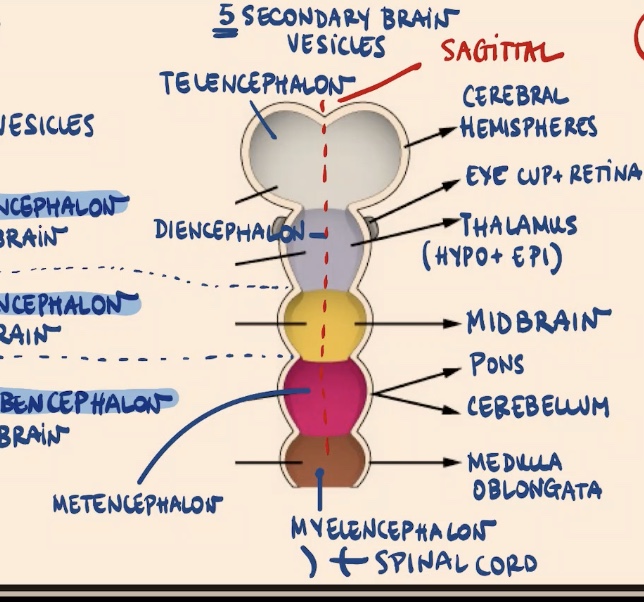
Which primary vesicle subdivides later in development to form the Telencephalon, and what does it give rise to?
The Prosencephalon subdivides. The Telencephalon is the first secondary vesicle, and it gives rise to the cerebral hemispheres.
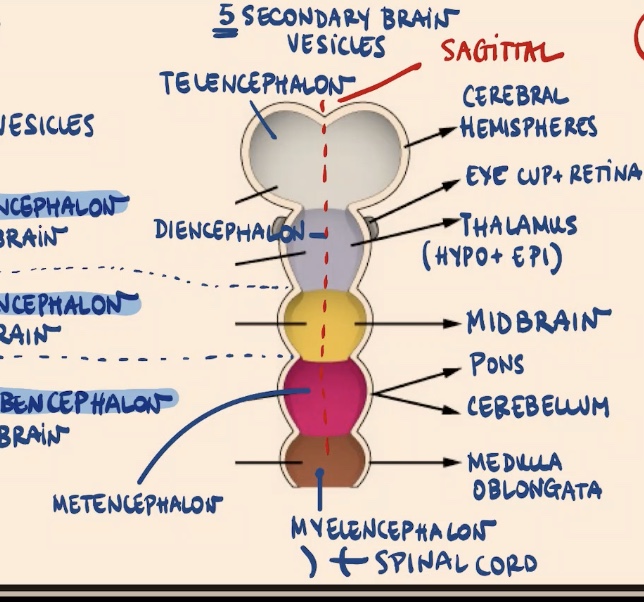
What are the derivatives of the Diencephalon?
1) Eye cup + retina.
2) Thalamus (both parts: hypo and epithalamus).
What is the specific developmental origin of the eye and retina?
They are regions of the brain that have prolonged and extended out of the main vesicle (Diencephalon) and inverted. The retina is an inverted region of the brain.
Does the Mesencephalon subdivide, and what is its adult derivative?
No, the Mesencephalon is going to stay as it is. It gives rise to the adult midbrain.
Which primary vesicle subdivides into the Metencephalon and Myelencephalon, and what are their derivatives?
The Rhombencephalon subdivides:
Metencephalon: Gives rise to the pons and cerebellum.
Myelencephalon (the most posterior structure of the brain): Gives rise to the medulla oblongata.
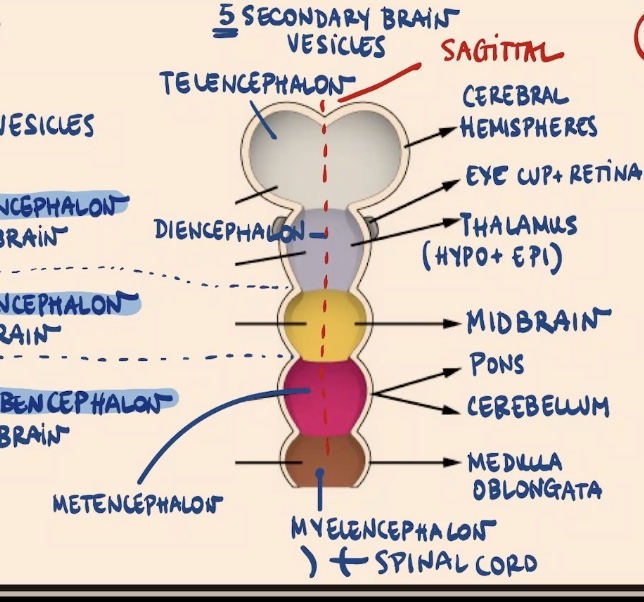
What are flexures, and what are the four important ones?
Flexures are curvatures in the anterior part of the neural tube:
Sagittal flexure.
Cephalic flexure.
Pontine flexure (between the mesencephalon and rhombencephalon derivatives).
Cervical flexure.
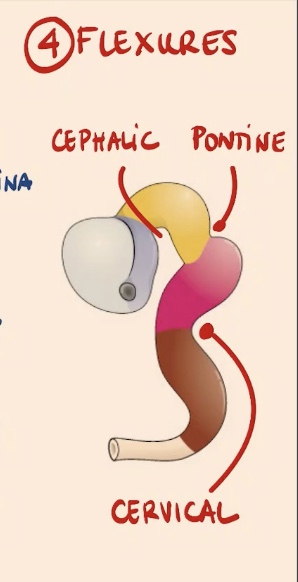
What structure is located in the most posterior region of the neural tube?
The spinal cord.
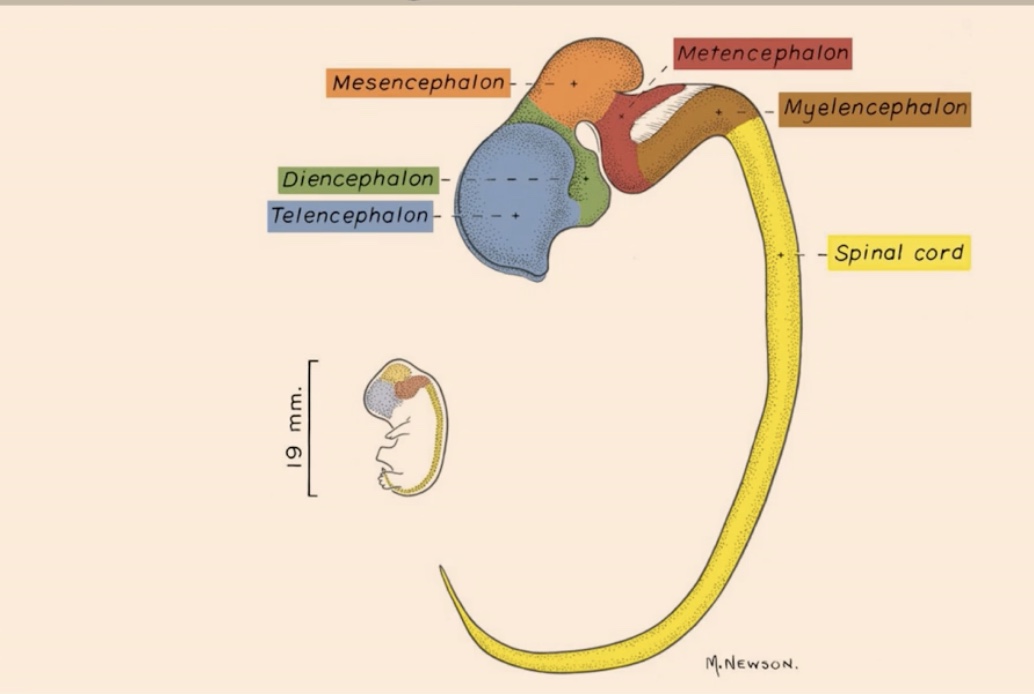
List the structures shown in the realistic representation from most anterior to most posterior
1. Telencephalon
2. Diencephalon
3. Mesencephalon
4. Metencephalon
5. Myelencephalon
6. Spinal cord (in the most posterior area)
What governs the specification of different brain regions (Telencephalon, Diencephalon, etc.) and the spinal cord?
The specification of all these regions is related to the expression of different combinations of Hox genes. In the hindbrain, you can see a transition between the most 3' elements of the Hox cluster and then transitioning to others (like 3 and 4).
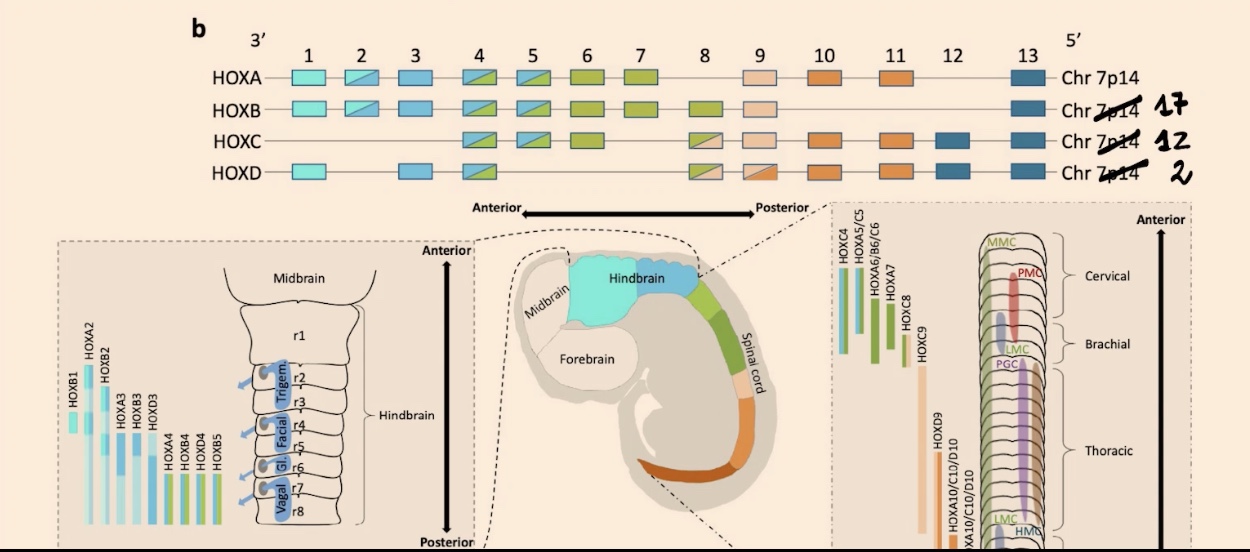
How is the spinal cord organized, and what is the functional result of its Hox gene gradients?
If you perform in-situ hybridization, you see beautiful gradients that define innervation in your limbs, including both your hindlimbs and forelimbs.
Why do some scientists refer to the neural crest as a "fourth" germ layer, and what are its major derivatives?
Because it gives rise to many different, vital structures. Key derivatives include the jaws, face, skull, and sensory ganglia.
What is the embryonic origin of the neural crest, and how is the structure itself described in terms of duration?
It is an ectodermal derivative. The neural crest as a structure is transient, even though its derivatives are not.
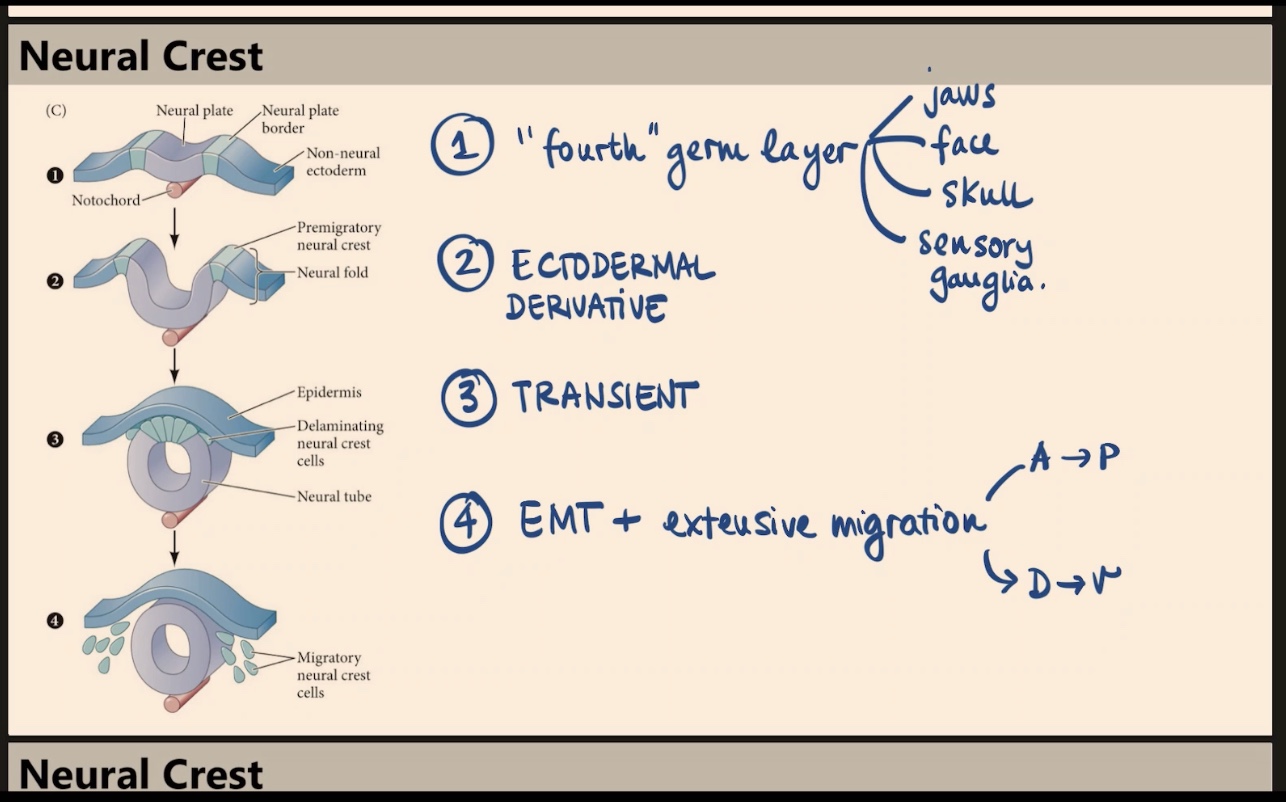
According to the diagram, which specific region of the early embryo gives rise to the delaminating neural crest cells?
They arise from the neural plate border, located between the neural plate and the non-neural ectoderm (surface ectoderm). When the tube closes, these cells become the delaminating neural crest.
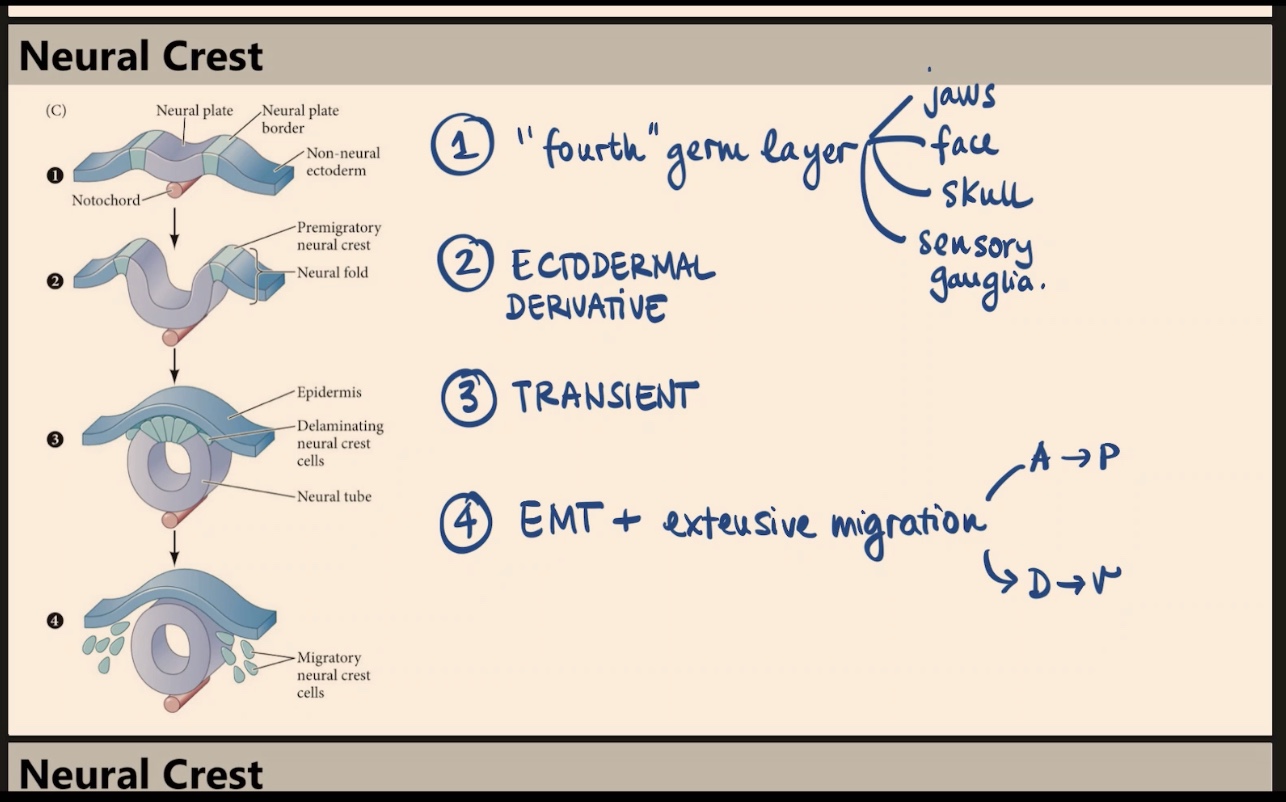
What process is involved in the formation of neural crest derivatives, and how do they migrate?
The process is EMT (epithelial to mesenchymal transition). These cells undergo extensive migration along both the Anterior-to-Posterior (A → P) side and the Dorsal-to-Ventral (D → V) axis (e.g., migrating from the dorsal neural tube all the way to the gut).
When asked about the origin of structures like pigments, why is "ectodermal derivative" not the most complete answer?
While ectoderm is technically correct, you must provide more detail for full points: pigments are an ectodermal derivative that derives from the neural crest.
Which components of the Peripheral Nervous System (PNS) are derived from the neural crest?
The neural crest gives rise to the neurons, the sensory ganglia, sympathetic and parasympathetic ganglia, neuroglial cells, Schwann cells, and other glial cells. These are identified as heavily ectodermal.
What are some of the "less obvious" structures derived from the neural crest?
Pigment cells
Facial cartilage and bones
Tooth papillae
Dermis, smooth muscle, and adipose tissue of the head and neck
Connective tissue of the salivary glands, thymus, and thyroid
Pituitary gland
What role does the neural crest play in the development of the great vessels (aorta and pulmonary)?
The neural crest is the origin of the septation (separation/division) of the great vessels.
From an evolutionary perspective, how did the division between pulmonary and systemic circulation occur?
At some point in evolution, our bodies created a separation (septation) between the pulmonary and systemic circulation by dividing the great vessels. This vital septation is derived from the neural crest.
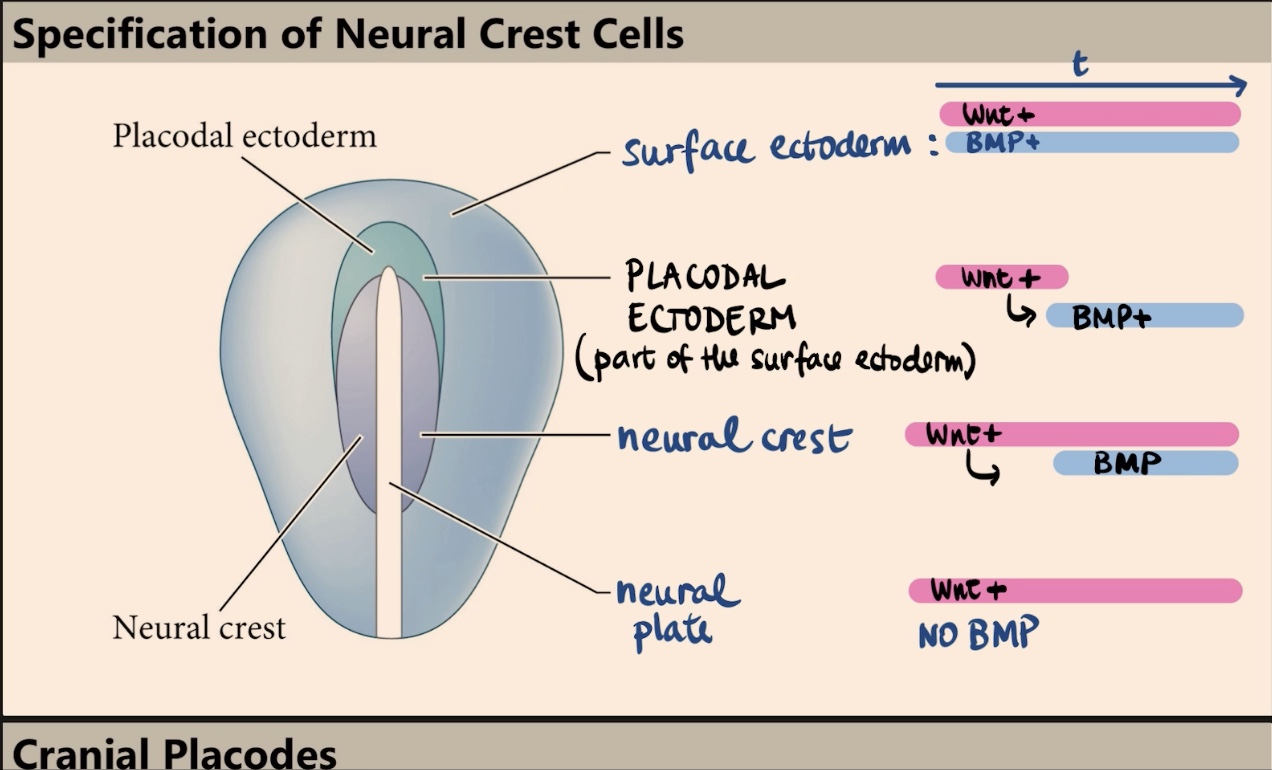
Which two molecules specify the four different fates (surface ectoderm, placodal ectoderm, neural crest, and neural plate), and what is the key factor in their outcome?
The four fates are specified by Wnt and BMP. The outcome depends on timing and exposure, specifically when and where those signals are received.
Describe the signaling profile (Wnt and BMP) that specifies the surface ectoderm.
The surface ectoderm is exposed to high levels of Wnt signaling and is exposed to BMP signaling for a really long time.
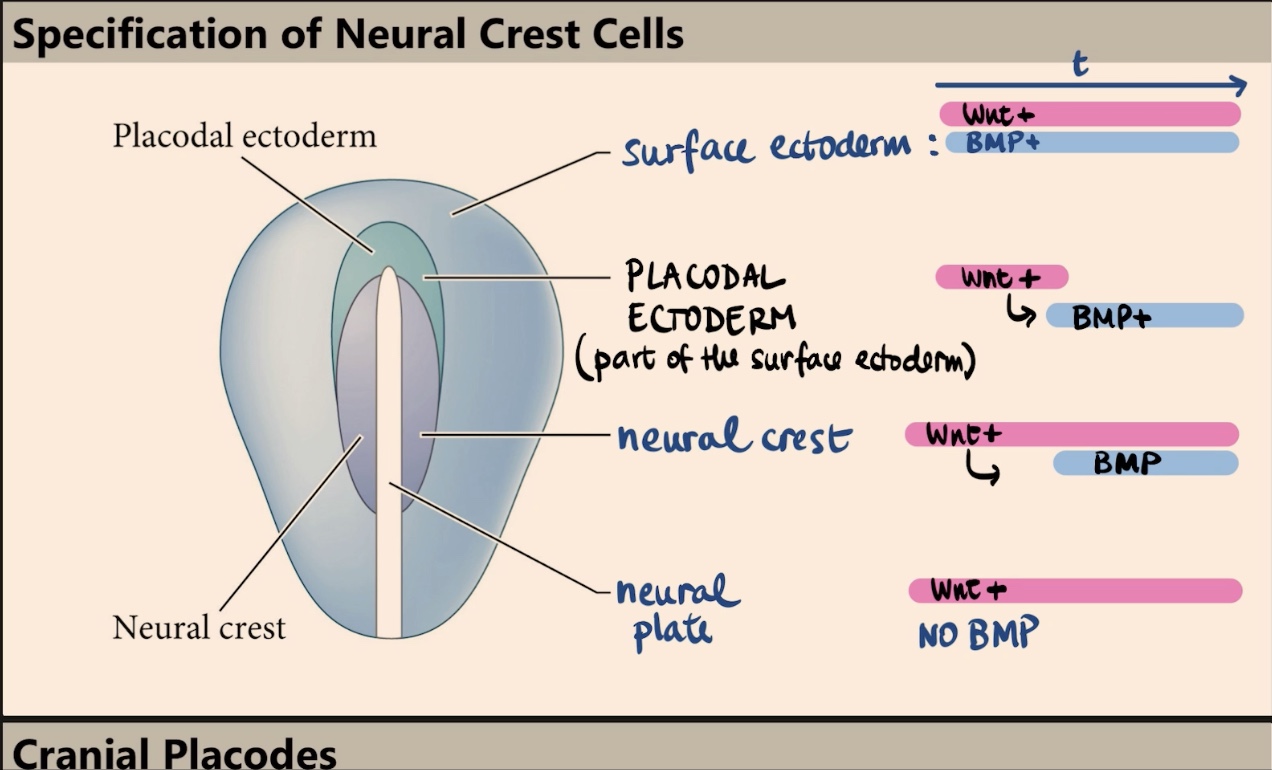
How does the neural plate differ from the surface ectoderm in terms of BMP and Wnt signaling?
In the neural plate, there is high Wnt signaling but no BMP at all.
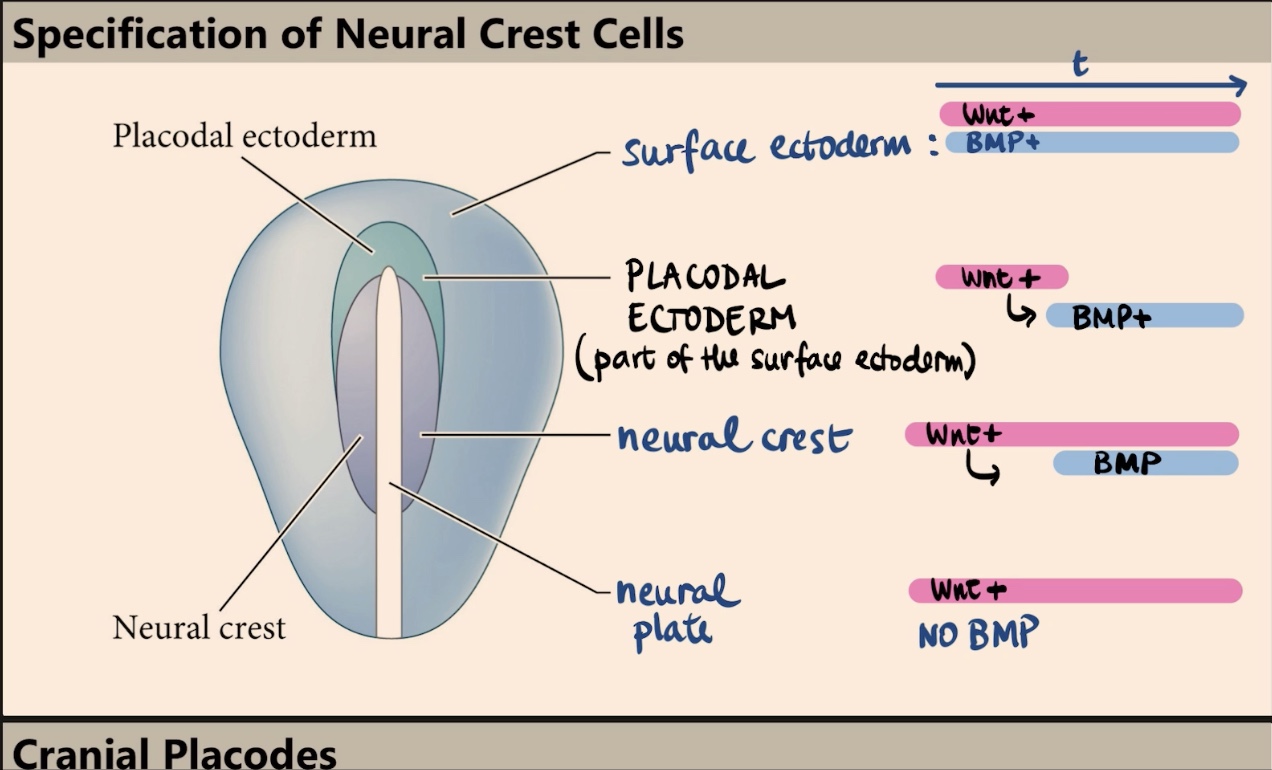
How is the neural crest specified, and why is timing critical in this process?
The neural crest is specified by a specific combination of time and exposure. They initially receive high levels of Wnt, and later on, those same cells are induced to receive/express BMP. It is the expression of Wnt followed by the later expression of BMP that specifies the neural crest.
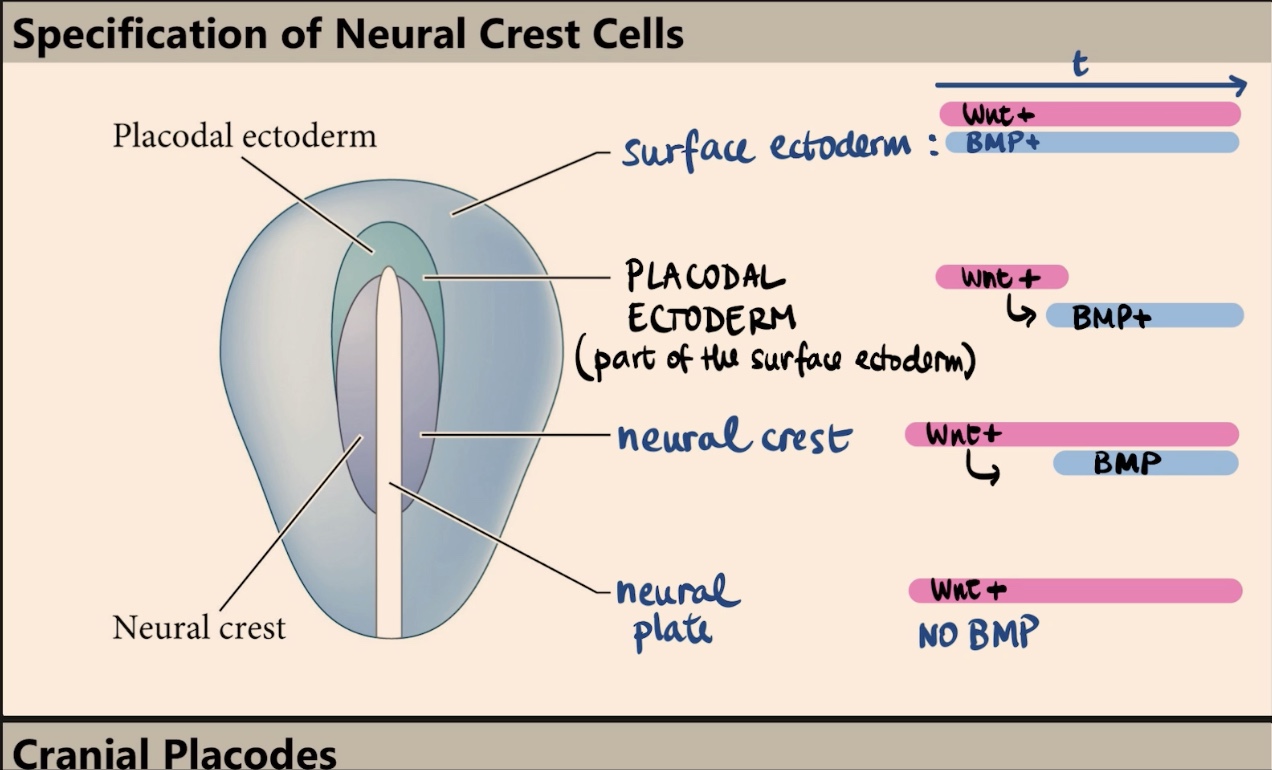
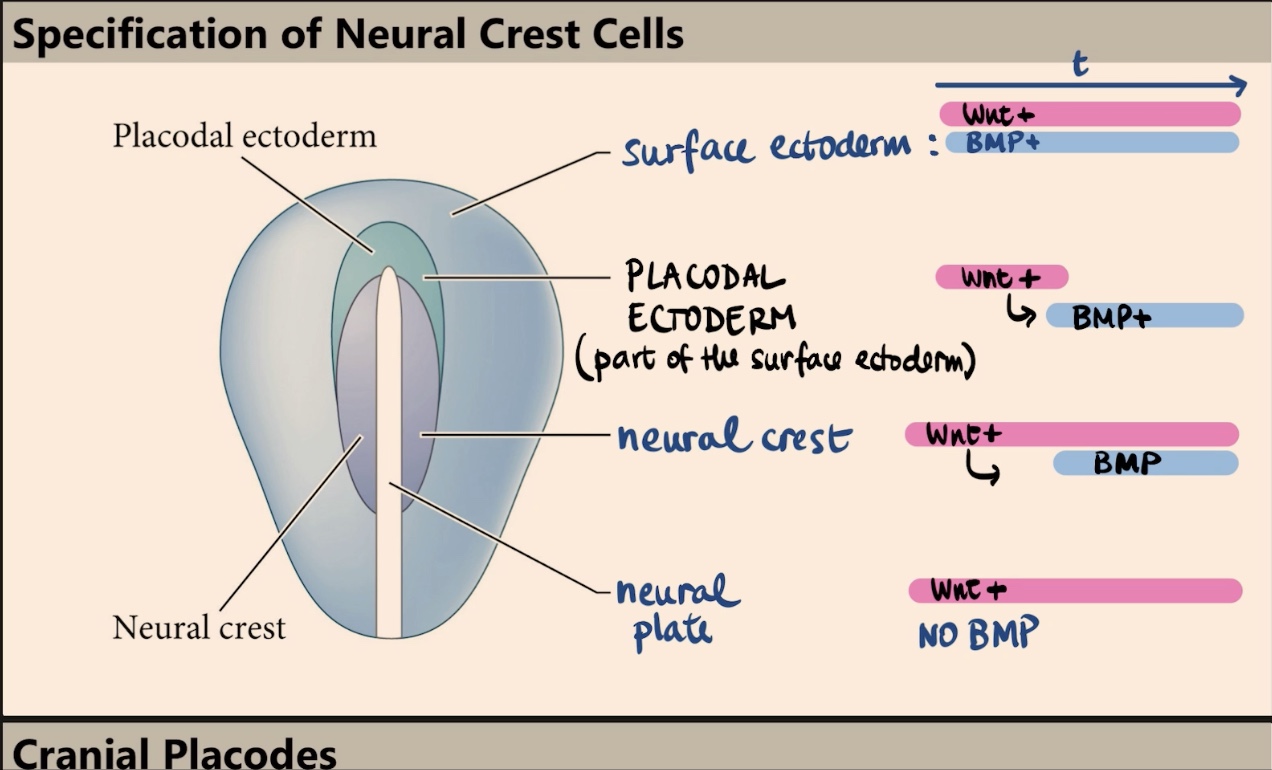
What is the placodal ectoderm, and what is its transient signaling profile?
The placodal ectoderm is part of the surface ectoderm (kind of like a border/arc). It has a transient situation where it receives Wnt only at the very beginning, and later on, it receives BMP.
What are cranial placodes, and what is their duration during development?
They are specialized ectodermal thickenings located at the border of the surface ectoderm. Like the neural crest, they are transient structures, but they give rise to the sense organs.
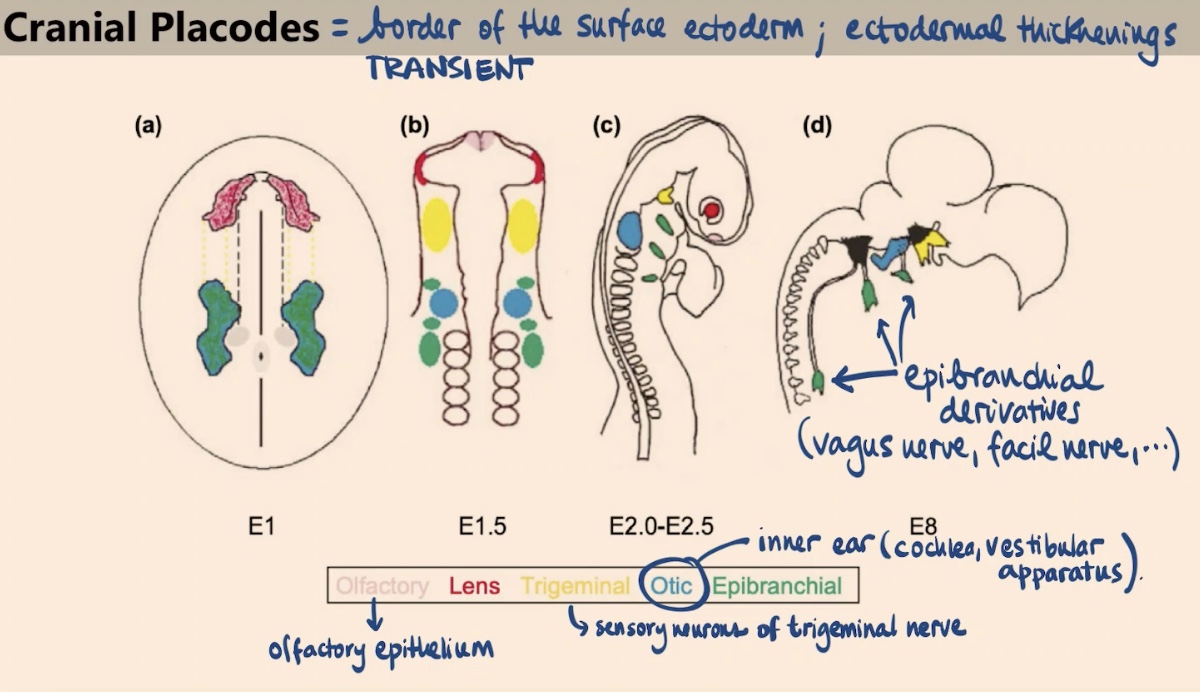
How is the specification of cranial placodes similar to yet different from the neural crest?
Like the neural crest, they appear due to a particular combination of Wnt signaling followed by BMP activation. However, the placodes involve interrupting the Wnt signaling later on.
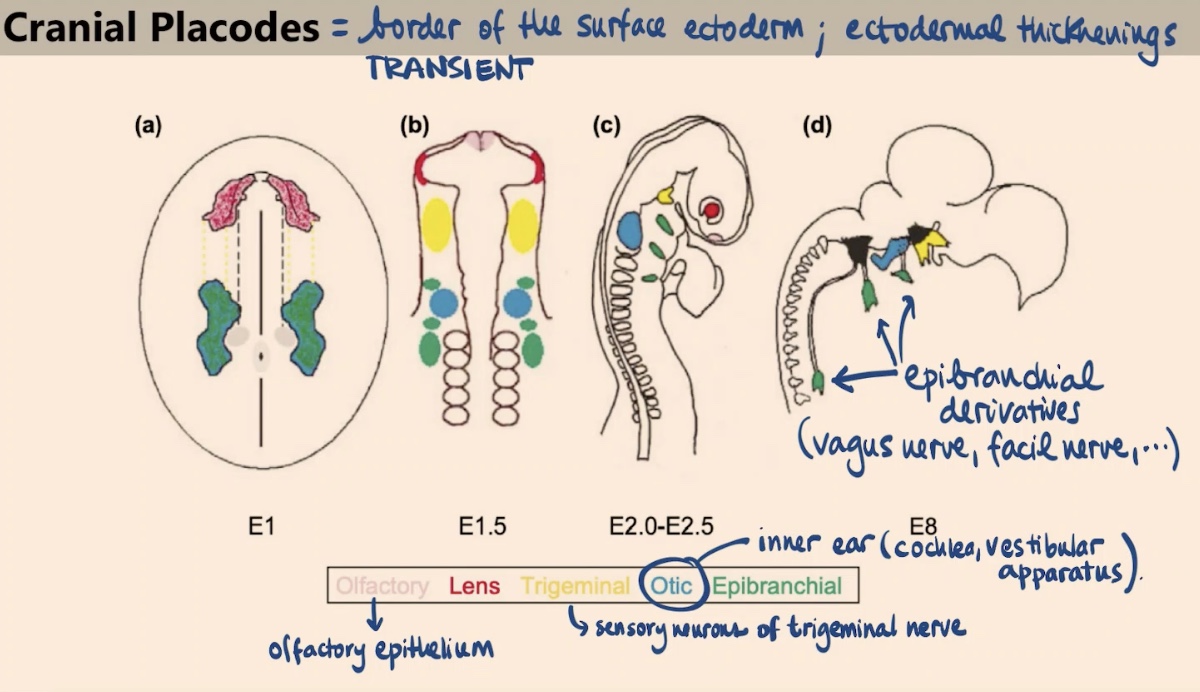
What sense organs or tissues are derived from the Olfactory and Lens placodes?
Olfactory placode: Gives rise to the olfactory epithelium.
Lens placode: Gives rise to the lens in our eye.
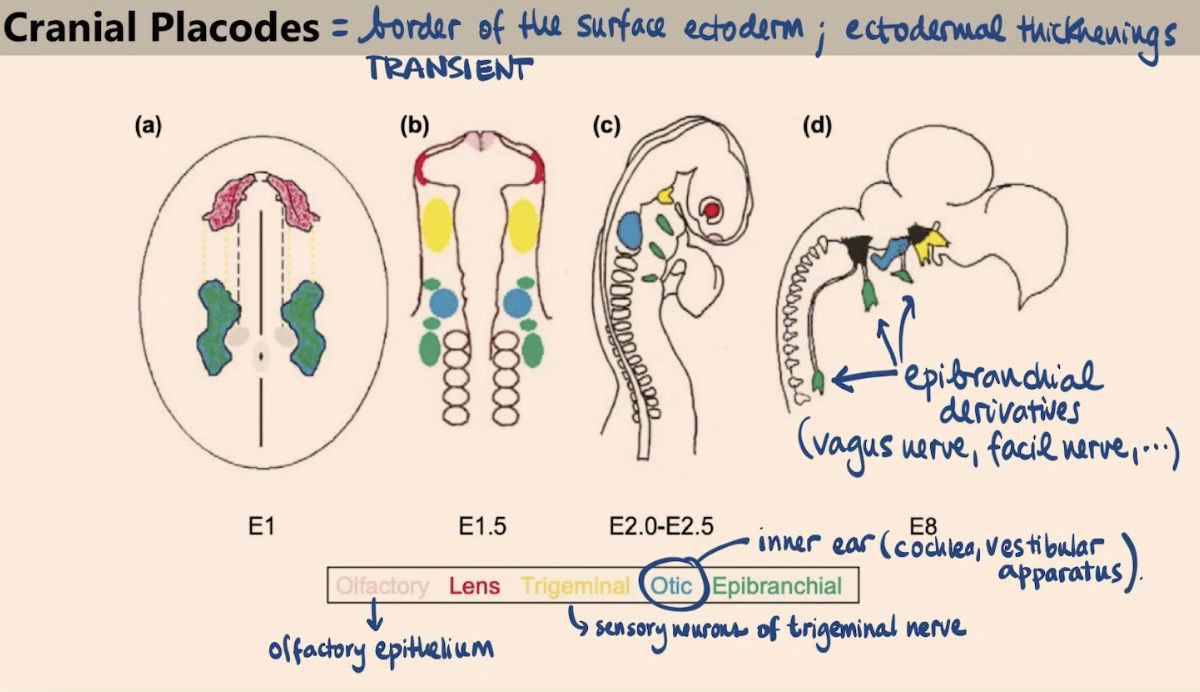
What are the derivatives of the Otic and Trigeminal placodes?
Otic placode: Forms the inner ear, specifically the cochlea and the vestibular apparatus.
Trigeminal placode: Gives rise to the sensory neurons of the trigeminal nerve.
Which nerves are derived from the epibranchial placodes?
They give rise to a variety of nerves, including the vagus nerve and the facial nerve.
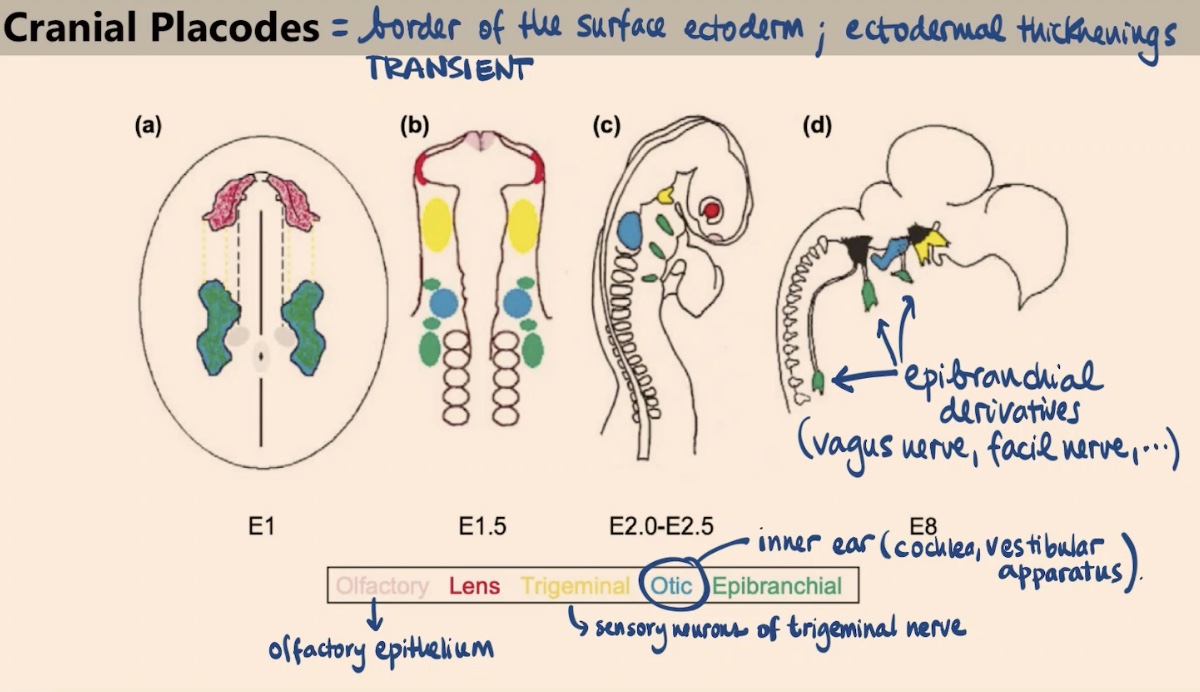
Collectively, what sensory functions do these local transient thickenings in the ectoderm provide?
They allow for the ability to smell, form part of the lens, enable you to sense things in your face (trigeminal), form the ear, and contribute to the vagus and facial nerves.
Name the four different regions into which the neural crest is divided along the anterior-posterior axis.
Cranial (Cephalic)
2. Cardiac
3. Trunk
4. Vagal and Sacral
What does the cranial neural crest contribute to, specifically regarding the pharyngeal arches and pouches?
It contributes to craniofacial mesenchyme (cartilage, bone, cranial neurons, glia, pigment cells, and connective tissue). It also builds the pharyngeal arches and pouches, which are important for forming the thymus, odontoblasts, and the bones of the middle ear and jaw.
What are the key derivatives of the cardiac neural crest?
It forms the muscular and connective walls of large arteries and the septum that separates pulmonary circulation from systemic (aortic) circulation. Depending on the arches (4 or 6), it also forms melanocytes, neurons, cartilage, and connective tissue.
In the trunk neural crest, what structures are formed via the ventrolateral migration path?
This path forms sensory neurons such as the sympathetic ganglia, the adrenal medulla, and nerve clusters around the aorta (which control functions like breathing).
Describe the dorsal-lateral migration of the trunk neural crest and its outcome.
Cells are sandwiched between the dermis and the skin to form the pigmented cells (melanocytes).
What is the primary role of the vagal and sacral neural crest?
It gives rise to the parasympathetic (enteric) ganglia of the gut, which provide the gut innervation.
What did early transplantation experiments (e.g., transplanting neural crest between birds with different beaks) conclude about neural crest cells?
They concluded that the neural crest has cell autonomous properties. If you transplant just the neural crest, the resulting skull/beak structure (e.g., a "Quack" or "Duail") follows the identity of the donor cells, proving they do not depend on the environment to determine their specific characteristics.