CHEM 213 POST MIDTERM 1, CHEM 213
1/938
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
939 Terms
atomic spectroscopy
The process of identifying the elemental makeup of a given sample by analyzing the wavelengths of light it emits or absorbs.
- Can be used to determine the elemental composition of a sample (usually metals)
- Heat is used to vaporize and decompose samples into atoms and ions
- The atoms of each element absorb and emit light at characteristic wavelengths that enable the analyst to determine which elements are present in the sample (quantitative analysis of complex samples).
- The amount of light that is absorbed or emitted at the characteristic wavelength enables the analyst to determine the element concentration at ppm or ppt levels (quantitative analysis). ex. trace metal analysis
- "Mature" technique with instrumentation available from many vendors
- Utilized across many industries and is instrumental in the detection of metal within a sample
ex. pharmacology, archaeology, manufacturing, mining, and forensics
advantages of atomic spectroscopy
- low cost per analysis
- easy to operate
- high sensitivity (up to ppb detection)
- high accuracy
- mostly free from inter-element interface
- wide applications across industries
limitations of atomic spectroscopy
- cannot detect non-metals
- new equipment is quite expensive
- more geared towards analysis of liquids
- sample is destroyed
Aurora Borealis (Northern Lights)
Light emission from the upper atmosphere that appear in many shapes and colors-- "nature's atomic emission" due to molecules from the sun interacting with the Earth's atmosphere
ex. the green colour is emitted by oxygen in a higher energy excited state relaxing to a lower energy excited state

Types of atomic spectroscopy
emission, absorption, fluorescence
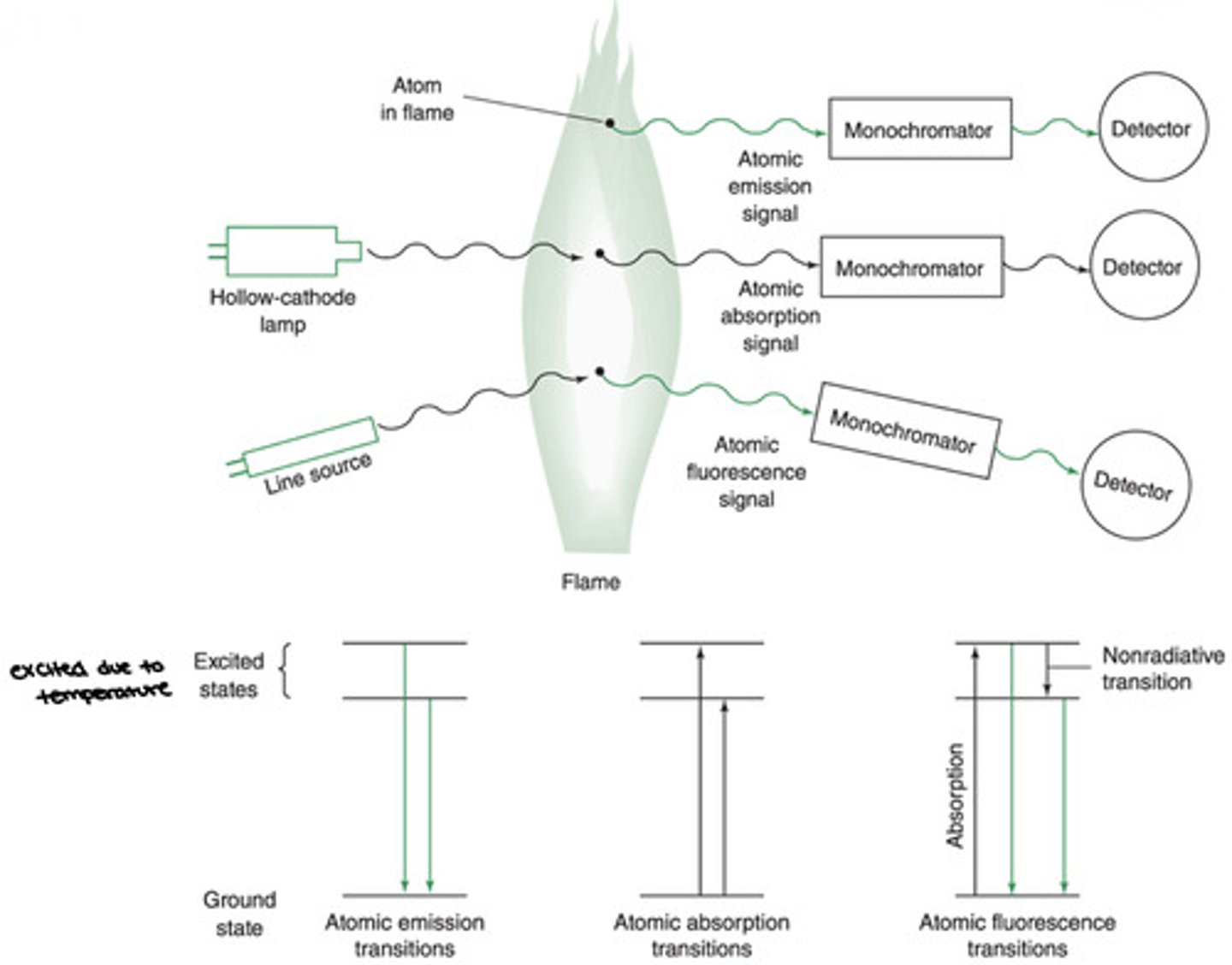
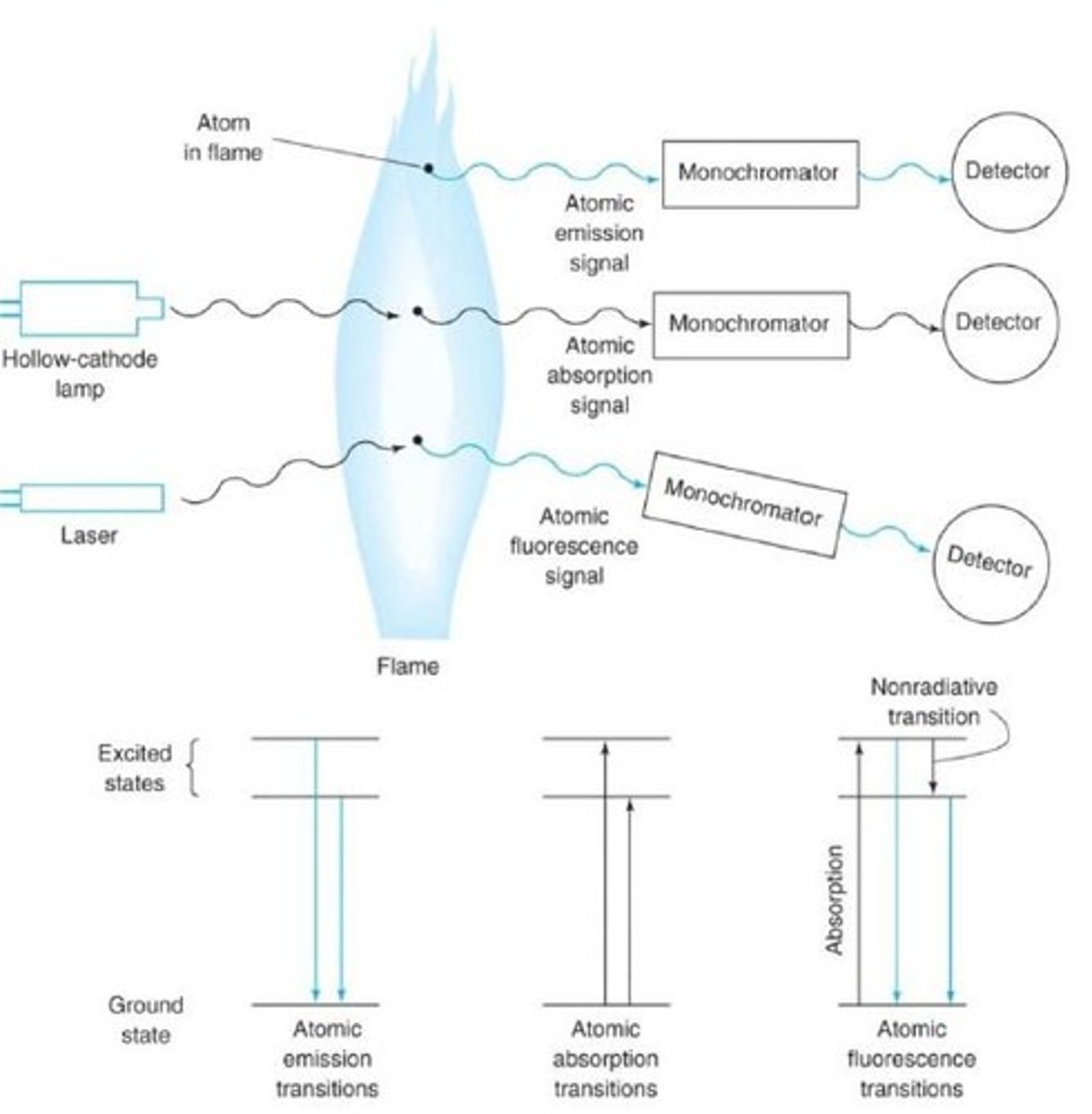
Atomic Emission Spectroscopy
A technique in which the emission of light by thermal excited atoms in a plasma, flame, or furnace is used to measure the concentration of atoms.
- collisions in a flame or plasma promote some atoms to excited electronic states from which they can emit photons to return to lower energy states
- no lamp/ external light source required
- emission intensity is directly proportional to the concentration of the element in the sample
- this is the dominant form of electronic spectroscopy
relaxation of atoms that have been excited by the flame, plasma, or furnace

Atomic Absorption Spectroscopy
A technique in which the absorption of light be free gaseous atoms in a flame or furnace is used to measure the concentration of atoms.
Atomic Fluorescence Spectroscopy
A technique in which electronic transitions of atoms in a flame, furnace or plasma are excited by light and the fluorescence is observed at a right angle to the incident beam
- 1000x more sensitive than AA but is not as commonly used because the equipment is more expensive-- we can observe weak fluorescence signal above a dark background, in absorption we are looking to identify small differences between large amounts of light reaching the detector
atomization
The process in which a compound is decomposed into its atoms at high temperatures
narrower
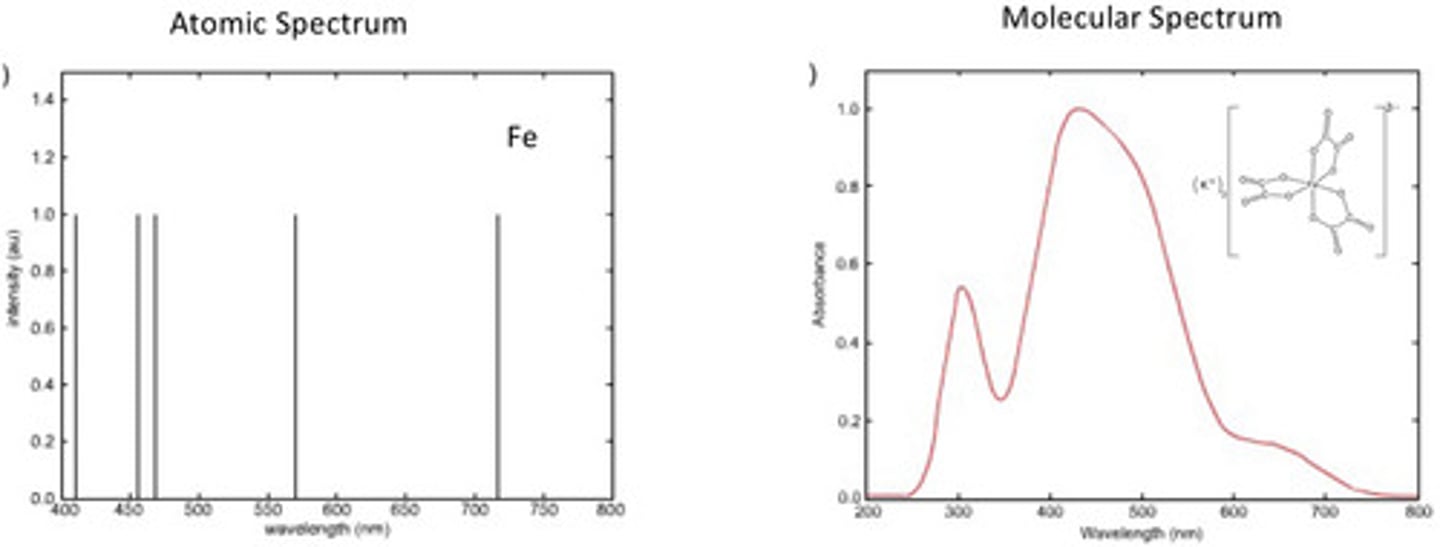
Atomic spectra have much _____________ absorption and emission bands than molecular spectra-- solids and liquids (molecular) have significantly wider bandwidths making them more prone to overlap than gaseous bandwidths
Due to collisions between molecules
Why are peaks broader for molecular spectra?
Atomic Emission
- Requires no lamp
- Collisions in the flame or plasma promote some atoms to excited electronic states from which they can emit photons to return to lower energy states
- Emission wavelength is specific to a given element
- Emission intensity is proportional to the element concentration
- Most common form of atomic spectroscopy
Atomic Absorption
- The flame or furnace atomizes samples to produce atoms that are primarily in the ground electronic state
- The atoms are illuminated at characteristic wavelengths from a hollow cathode lamp
- The atoms absorb a portion of the light atom the hollow cathode lamp
- Element concentration is calculated from the absorbance
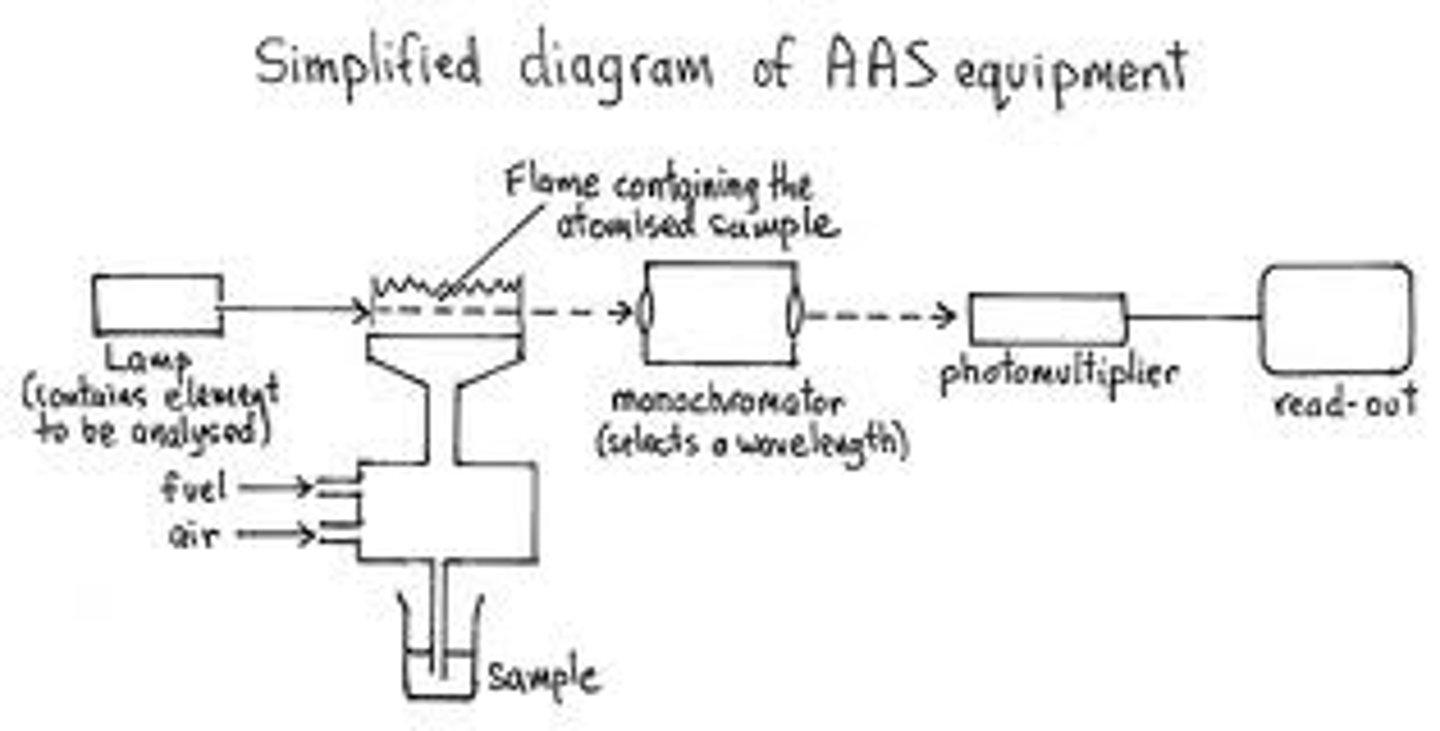
Process of Atomic Absorption Spectroscopy
1. The hollow-cathode lamp generates characteristic wavelengths that are only absorbed by the element being probed
2. Liquid samples is aspirated (sucked) into the flame
3. The atoms of the element being probed absorb part of the light coming from the hollow cathode lamp so that P < P0
4. Absorbance is measured by the detector and element concentration is calculated
Hollow Cathode Lamp
Has a tungsten anode and a cylindrical- shaped cathode containing the element of interest. The element is sputtered from the cathode into the gas phase. This process excites some of the gaseous atoms, which then emit characteristic radiation as they return to the ground state.
Hollow-Cathode Lamp Emission Spectrum
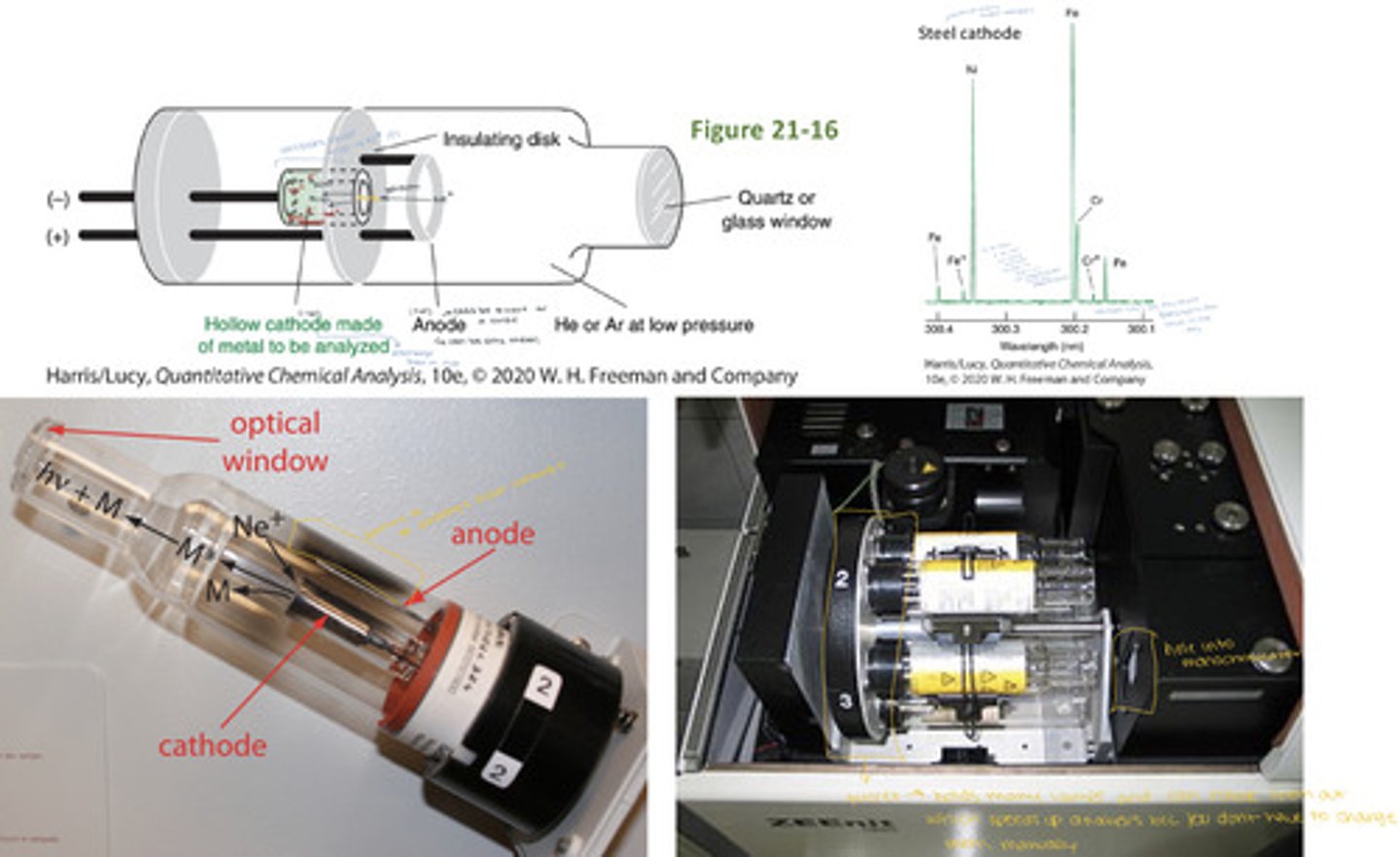
Atomic absorption experiments require lamps that produce light only at characteristic wavelengths for the element(s) being analyzed. A steel hollow cathode lamp produces light only at the characteristic wavelengths for Fe, Ni, and Cr
- The spectral resolution is 0.0001nm which is comparable to the true bandwidths
- A lab will have a collections of HCLs for various elements (because they are specialized for certain elements)
Atomic Fluorescence
The flame or author atomizer atomizes samples to produce atoms that are primarily in the ground electronic state. A line source (ex. a laser) irradiates atoms in the flame to promote them to an excited electronic state.
- Fluorescence emission from the excited state is measured-- proportional to element concentration.
there are no vibrational relaxations in atoms (i.e. no internal conversions) but there can still be intersystem crossing (between triplet and singlet states)
Atomization
The high temperature process in which a compound Is decomposed into its atoms.
- In atomic spectroscopy, the analyte is atomized in a flame, en electrically heated furnace or a plasma.
- Flames were used for decades, but they have largely been replaced by the inductively coupled plasma and the graphite furnace (flames are still common enough in teaching and service labs).
Flame Atomic Absorption Burner
- Fuel, oxidant, and sample are mixed in the premix burner before introduction to the flame
- Sample solution is drawn into the pneumatic nebulizer by the rapid flow of oxidant past the tip of the capillary
- The spray os directed against a glass bead which creates an aerosol (fine suspension of liquid particles in gas)
- The mist, oxidant, and fuel flow past baffles that promote further mixing and block large droplets of liquid.
- Excess liquid collects at the bottom of the spray chamber and flows out to a drain
- Aerosol reaching the flame contains only 5% of the initial sample
- Most common fuel-oxidizer combination is acetylene and air (temp. = 2400-2700K), hotter flames are commonly achieved using acetylene and nitrous oxide (2900-3200K)
- Flame conditions are traditionally optimized to maximize the formation of gaseous atoms
rich flame
excess fuel, excess carbons from the fuel reduce metal oxides and hydroxides--- increased sensitivity
lean flame
excess oxidant, hotter flame decompose refractory compounds more efficiently resulting in greater efficiency
free atom formation in flames
- Physical and chemical processes in the burner head and flame must occur to convert the aqueous ionic analyte into gaseous atoms:
> liquid sample is nebulizer into an aerosol
> aerosol is evaporated into a dry aerosol
> dry aerosol is vaporized into gaseous molecules
> gaseous molecules are decomposed into gaseous atoms that can be excited
- Ideally, all analyte is converted into gaseous atoms (M(g))
- Refractory compounds (oxides of Al, Is, and Mg) do not decompose and remain in there molecular form, many elements form oxides and hydroxides in the outer cone, and some metals are ionized in the flame --- these things all lower sensitivity
- Note: molecules and ions do not have the same spectra as neutral atoms
advantages of graphite furnaces over flames
1. lower minimum sample volume-- flame = 1-2mL, graphite furnace = 1-100uL
2. longer sampling time in optical path-- flame = 1 second (worse signal-to-noise ratio), graphite furnace = several seconds (you can measure the same stuff over a few seconds to get better signal-to-noise
3. easier preconcentration-- multiple aliquots onto the furnace platform and evaporated, preconcentrating the sample in the graphite furnace prior to the analysis
4. no nebulization step required (nothing can get clogged or work improperly)
note: graphite furnaces require more operator skill than flames to find the proper conditions for each sample
lower minimum sample values, longer sampling time in optical path, easier preconcentration, no nebulization of the sample required
what are the four advantages of graphite furnaces over flames?
preconcentration
Concentrating a dilute analyte to a level high enough to be analyzed--- more easily done with a graphite furnace and a flame
graphite furnace operation
- A small aliquot (1-100uL) of sample is injected into the graphite furnace through the hole at the center
- The sample is atomized by running a high current through the graphite that electrically heats the furnace to high temperatures (a stream of Ar gas prevents the graphite from igniting)
- The absorbance of the atomized sample is measured using light atom a hollow-cathode lamp
- The furnace is cooled to prepare for the next measurement ( which means it takes longer between measurements of difference things)
Graphite Furnace Heating Steps
1. Drying-- dries the sample (ex. 125 degrees celsius for 20s)
2. Charring-- decomposes the sample; breaking covalent bonds, dissociating ions, melting a bit-- can cause smoke (source of error) (ex. 1400 degrees celsius for 60s)
3. Atomizing-- atomizes the sample (turns shit into individual gas phase particles that you can shine a light though (ex. 2100 degrees celsius for 10s)
4. Cleaning-- cleans out remaining residue (ex. 2500 degrees celsius for 3s)
analytical signal = the-time integrated absorbance (peak area) measured during the atomizing step
the time-integrated absorbance (peak area) measured during the atomizing step
what is the analytical signal of a graphite furnace AA?
longitudinal graphite furnace
- less uniform vaporization
- greater memory effect (bad-- leftover sample interference from previous run)
- heat applied in the direction of the optical path
transverse graphite furnace
- heating applied perpendicular to the optical path
- maintains a more constant temperature over the sample area
- reduces the memory effect (leftover sample) interference from previous runs
superior
Reducing Matrix Smoke Interference
Problem: In the charring step, smoke due to the matrix sometimes partially blocks the light, giving rise to an unwanted background absorbance
Solution: Addition of a matrix modifier-- a substance that is added to the matrix to increase the volatility of the matrix or decrease the volatility of the analyte to suppress unwanted background signals from the matrix (i.e. makes sure that the matrix evaporations before the analyte)
ex. In an analysis of Mn in seawater, a calibration blank with no Mn gives an apparent absorbance due to optical scatter from smoke created by heating NaCl. Adding a matrix modifier (ex. NH4NO3) to the blank reduces the matrix background signal by concerting NaCl to NH4Cl and NaNO3, which evaporate cleanly instead of giving smoke.
Matrix Modifier
A substance that is added to the matrix of a sample to increase the volatility of the matrix or decrease the volatility of the analyte to suppress unwanted background signals from the matrix--- i.e. the substance ensures that the matrix evaporates before the analyte
Inductively Coupled Plasma (ICP)
A high-temperature plasma that derives its energy from an oscillating radio-frequency field. It is used to atomize a sample for atomic emission spectroscopy.
- Plasma is a state of matter in which an ionized gaseous substance becomes highly electrically conductive to the point that long-range electric and magnetic fields dominate the behaviour of the matter. High temperatures are usually needed to sustain ionization, which is a defining feature of plasma
- An inductively coupled plasma is (1) twice as hot as a combustion flame, (2) experiences fewer interferences (plasma, plasma+ and e-) and (3) is used for simultaneous, multi-element analysis via atomic emission
The advantages of the ICP spectrophotometer have lead to its widespread adoption as a replacement for flame AA instruments
argon (it is very expensive)
what gas is used to sustain the plasma?
Inductively Coupled Plasma Burner
The plasma torch is generated by an inductively coupled plasma burner.
1. High-purity Ar gas is sent in through the coolant and plasma gas inlets.
2. The oscillating magnetic field is induced by sending a radio frequency (27 or 41MHz) alternating Curren through the load coil.
3. The plasma is lit with a spark from a Tesla coil creating Ar+ and e-
4. The free e- are accelerated by the induction field where they collide with other Ar atoms and transfer energy to the entire gas (maintains temp of 6000-10000K)
5. An aerosolized sample is fed into the plasma
6. Emission from the analyte is detected using either an axial or radial detector. Sensitivity is enhanced by a factor of 3 to 20 by observing emission along the length of the plasma (axial) instead of radially across the diameter of the plasma
Inductively Coupled Plasma Sample Aerosol
How is the sample aerosol generated?
1. The sample is digested into a liquid form
2.The liquid sample is pumped slowly into the nebulizer
3. A coaxial flow of air nebulizer the sample into an aerosol that sprays into a cyclone chamber
4. The cyclone chamber:
a) removes the largest droplets
b) sends the remaining fine sample aerosol into the plasma torch
it accepts nebulizer sample and removes the largest droplets while retaining the finest aerosolized particles into the plasma torch
what is the purpose of the cyclone chamber in sample aerosolization for ICP?
graphite furnace: drying
graphite furnace: charring
graphite furnace: atomizing
graphite furnace: cleaning
nebulization
converts liquid sample to an aerosol
evaporation
converts aerosolized sample to a dry aerosol (removes all water/mositure)
vaporization
converts dry aerosolized sample into gaseous molecules
decomposition
converts gaseous sample molecules into gaseous atoms of individual species which can be detected using spectroscopic techniques
temperature affects on atomic spectroscopy
Temperature determines the degree to which a sample breaks down into atoms and the extent to which a given atom is found in its ground, excited or ionized states which influences the strength of the signal observed
- absorption is proportional to the population of neutral gaseous analyte atoms
- emission is proportional to the population of excited gaseous analyte atoms
(note that even in plasma we still have significantly more M(g) but we are forming so much M*(g) because we are getting so many particles that it doesn't matter)
Boltzmann distribution
A theoretical model that describes the distribution of kinetic energies of molecules in a gas.
- For systems at thermal equilibrium, the boltzman distribution can be used to predict the N*/N0 = ratio of the atomized sample populations in the ground and excited electronic states

refractory compounds
species in the sample other than the analyte that do not thermally decompose in the flame and remain in their molecular form-- many form oxides or hydroxides in the outer cone
flame profile

effect of temperature on absorption and emission
- Varying temperature hardly affects the ground-state population and thus would not noticeable alter the atomic absorption signal
- Varying temperature significantly impacts the excited-state population and would have a noticeable affect on emission intensity
plasma is used for atomic emission because the temperature is more stable and it is so hot that there is also already a substantial population of excited-state atoms (recall: 6000-8000K)
effect of temperature on atomic absorption signal
temperature hardly has any impact on analyte atoms in the ground-state population (because the majority of atoms are in the ground-state), thus there would be no significant effect on the atomic absorption signal
effect of temperature on atomic emission signal (intensity)
temperature greatly changes the population of analyte atoms in the excited state (because there are few there to begin with) and thus a significant change in atomic emission signal intensity would be observed
effect of energy difference and temperature on population of excited states
the higher the energy of the transition (lower wavelengths), the lower the N*/N0, and the greater the effect of temperature--- when the energy difference between the ground-state and the excited-state is large, their are more atoms in the ground state so the excited state fraction is small, thus, temperature has a greater impact excited-state populations
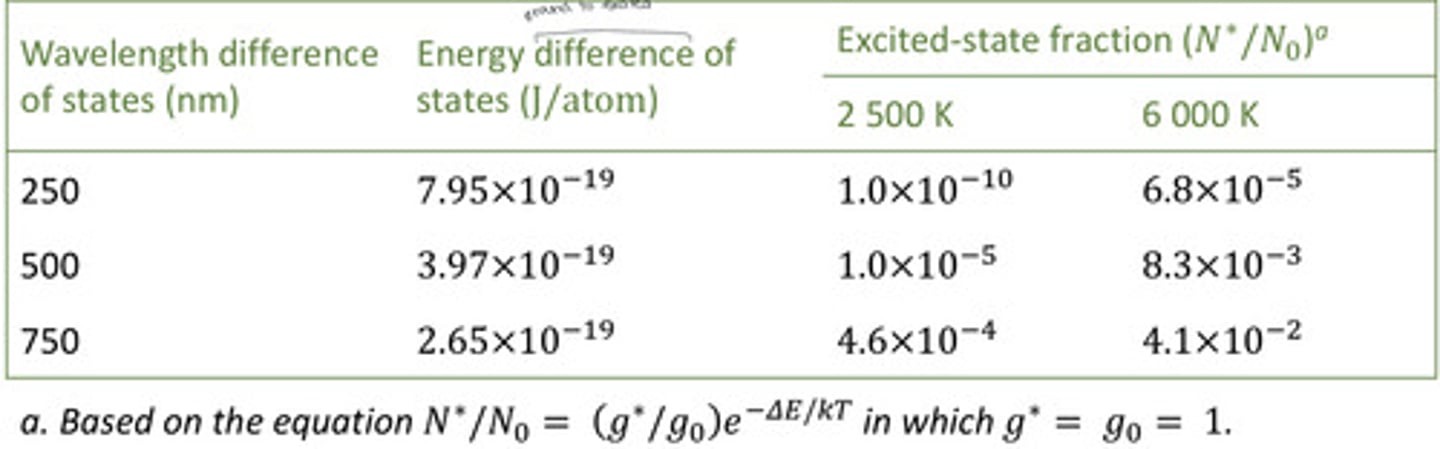
hollow-cathode lamp
- Filled with low-pressure Ne or Ar (1-5 Torr)
- The cathode is made of the element to be analyzed (ex. a lamp for Ni would have Ni as the cathode)
A voltage of ~500V applied between the anode and cathode ionizes the Ne
> Positive Ne+ ions are accelerated toward the cathode
> Accelerated Ne+ ions "sputter" some of the cathode atoms into the gas phase; M(s) + Ne+ (g) --> M(g)
> These cathode atoms are excited by collisions with high-energy electrons; M(g) collides with e- ---> M*(g)
> These excited-state electrons relax back to the ground state by emitting light; M*(g) --> M(g) + hv
The cathode atom emission:
> is the light emitted by the hollow-cathode lamp
> is emitted at characteristic wavelengths with narrow line widths
> has wavelengths that perfectly match those absorbed by the analyte because the cathode and analyte atoms are the same element
> different analyses require different lamps
> most instruments hold 3-8 lamps on a rotating support
> some lamps contain multiple elements in cathode
because the cathode of the lamp is the same element as the analyte
why does the wavelength of light being emitted by a hollow-cathode lamp in atomic absorption spectroscopy perfectly matched to the wavelength absorbed by the analyte?
from the analyte species (ex. if the analyte is nickel, the cathode is made of nickel, steel cathodes have Fe, Ni, and Cr, so they will produce light at wavelengths characteristic to Fe, Ni, and Cr)
what material is the cathode made of for a hollow-cathode lamp being used in atomic absorption spectroscopy?
emission, absorption
It is important that the temperature be very stable in atomic ______________ spectroscopy. Temperature variation is less critical in atomic _________________ spectroscopy.
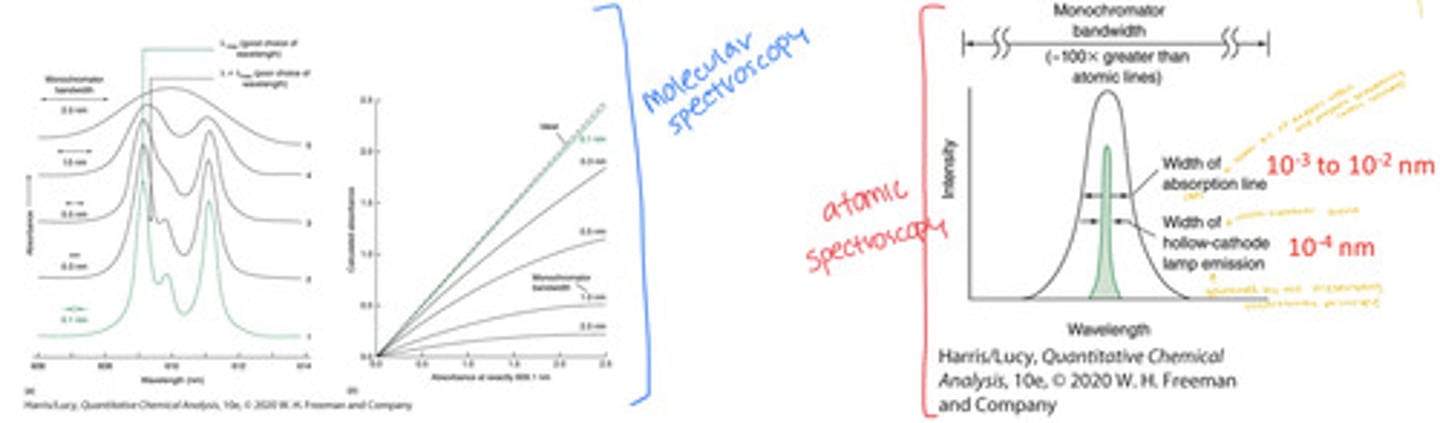
Natural Atomic Linewidths
Recall from UV-visible spectroscopy instrumentation discussion of monochromator bandwidth. Bandwidth should be one-fifth as wide as the absorption peak to achieve acceptably small distortion in peaks and Beer's law
The line width of the radiation source must be substantially narrower than the line width of the absorbing sample. But atomic line widths are narrow.
the width of HCL emission must be narrower than the width of the absorption line of the analyte
HCL and AA instrumentation
hollow cathode lamps (ex. made of steel) may emit a number of lines, all of these lines go through the flame and they get sorted out and one line gets selected by the monochromator and the rest are rejected alongside the flame (or furnace) emission
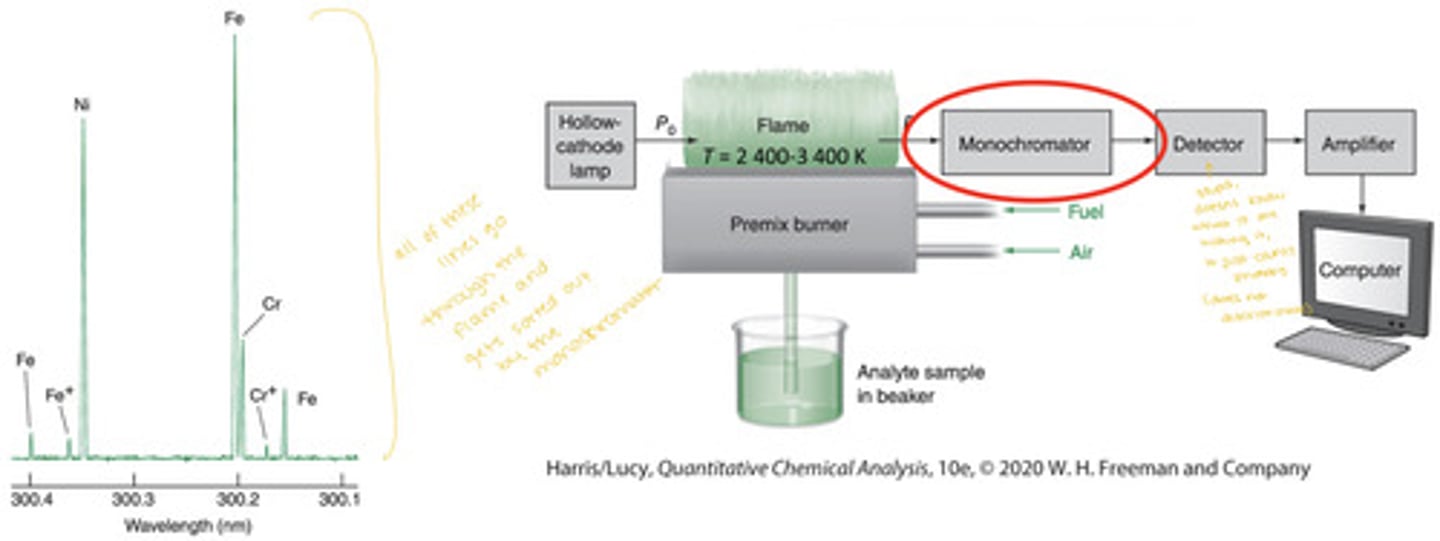
background and flame emission
- Background correction is necessary in atomic spectroscopy to distinguish the analyte signal from absorption, emission, and optical scattering of the sample matrix, the flame, plasma, or graphite furnace
- Background correction is critical for electrothermal graphite furnaces due to residual smoke from charring
- Flames can absorb and flames can emit
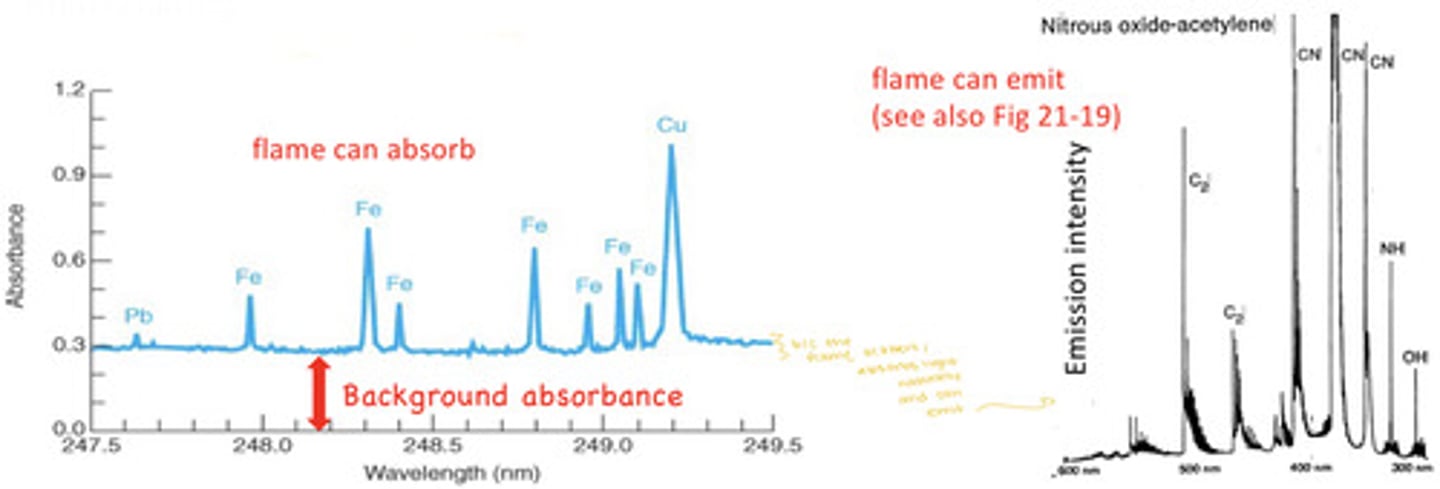
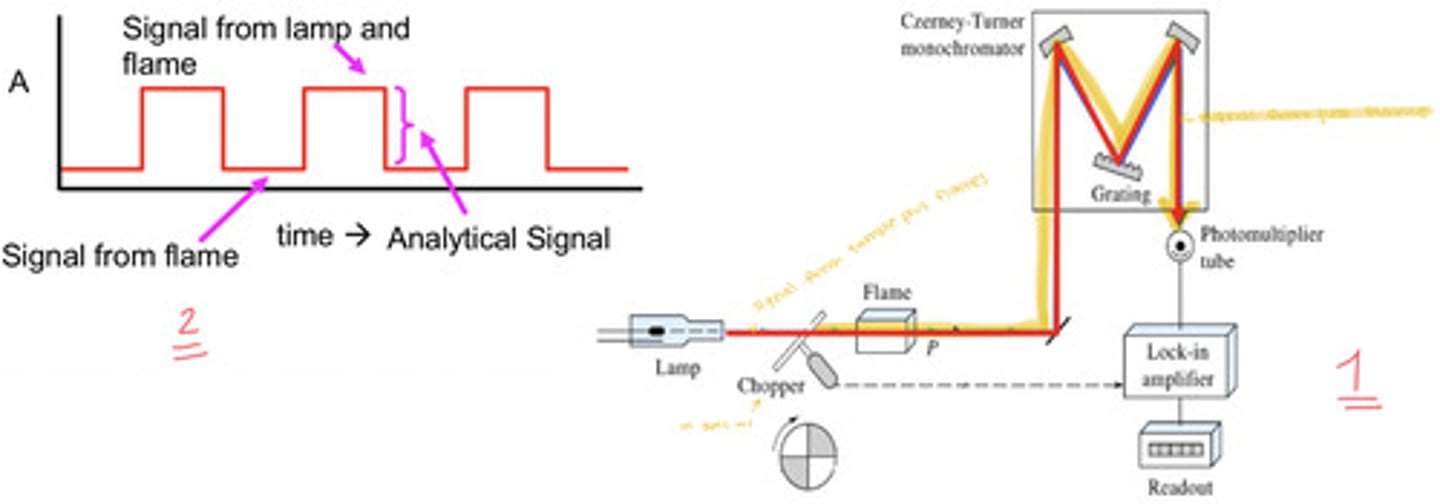
correcting for flame emission (and furnaces with lots of residual smoke)
correct via source modulation; use a mechanical beam chopper (most common) or pulse the lamp on and off (can be detrimental to lifetime of the HCL so we generally avoid that)
- corrects from flame emission only, not for scattering or background absorption from the sample
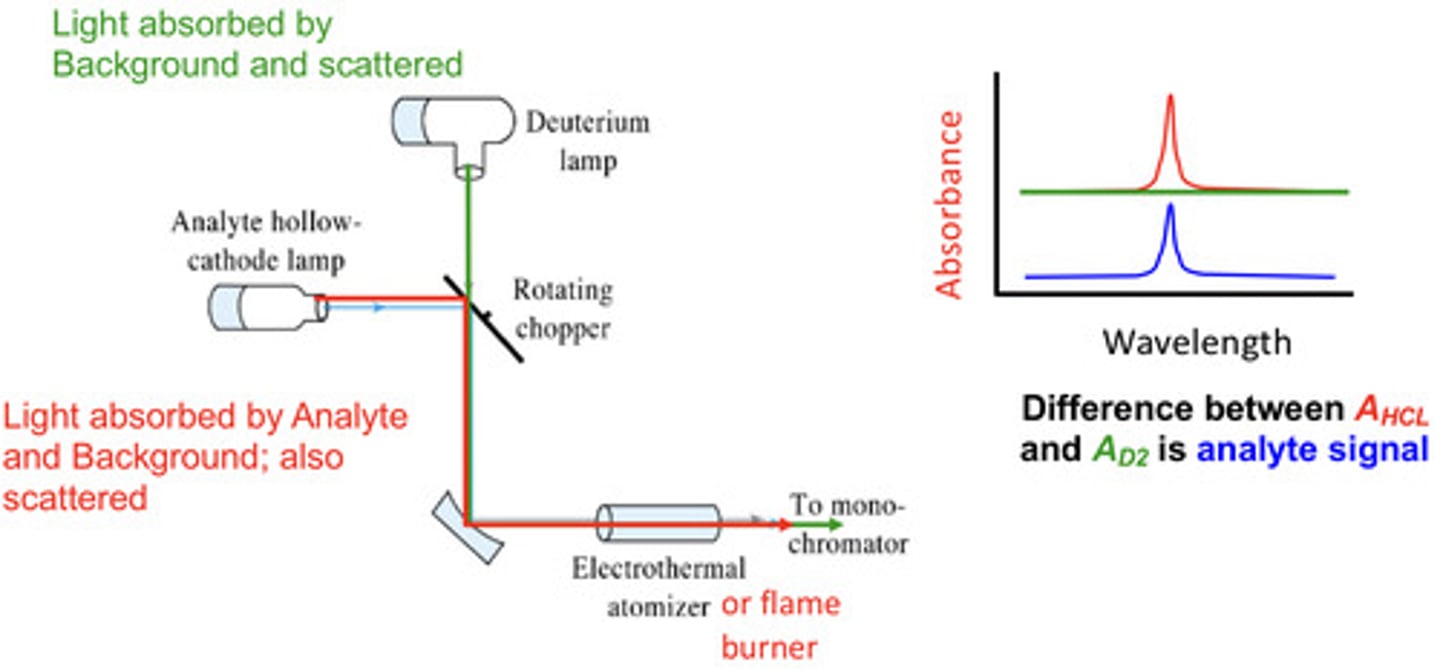
correcting for scattering and broad background (flame or furnace)
correct using a deuterium lamp/broadband lam-- subtract the absorbance measured with the deuterium lamp from the measured with the hollow-cathode lamp
1. mechanical beam chopper
2. pulse the HCL on and off (this can damage it tho so maybe don't)
how can flame emission and graphite furnace smoke interferences be corrected?
deuterium background correction (measure with broadband (deuterium lamp) then measure with narrow band (HCL) then subtract broad from narrow to yield the analyte signal
how can you correct for scattering and broad backgrounds in flame or furnace AAS?
Zeeman effect
the splitting of spectral lines into multiple components when the atoms are in a magnetic field
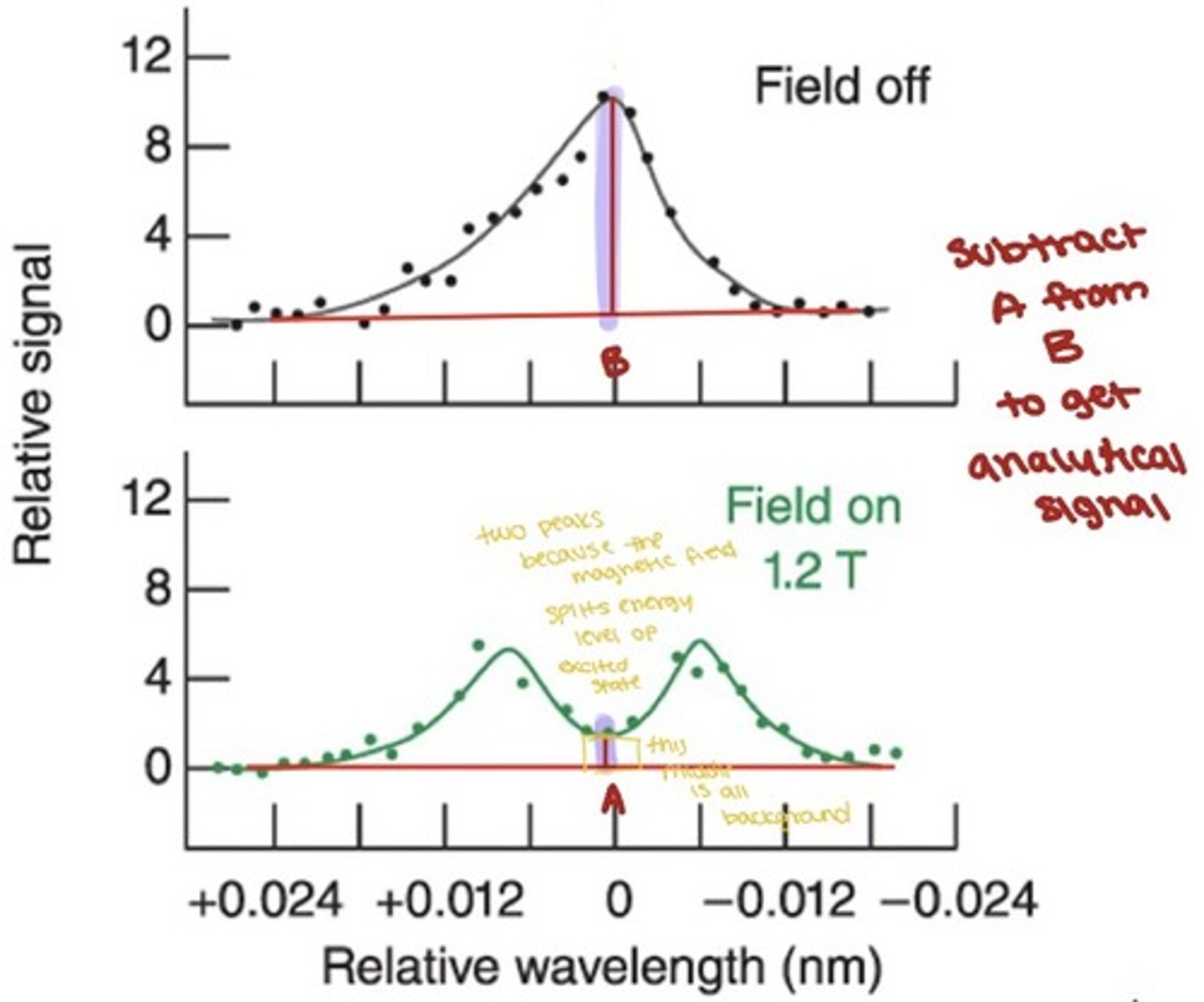
Zeeman effect background correction
For a graphite furnace, the Zeeman effect can be used to provide a background correction.
- A magnetic field is cyclically applied to the sample in the furnace parallel to the light path.
- The magnetic field splits the analyte peak into two peaks, neither of which can absorb at the probe wavelength
- When the magnetic is off, bath analyte and background signals are measured, when the magnet is on, only background signals are measured
- The analyte signal is obtained by subtracting the on signal from the off signal
- The correction is made at the specific analysis wavelength, not over a broad band like the D2 lamp methods
Plasma Atomic Emission Instrumentation
does not require any lamps and can measure up to 70 elements simultaneously
- uses a polychromatic instead of a monochromaticr
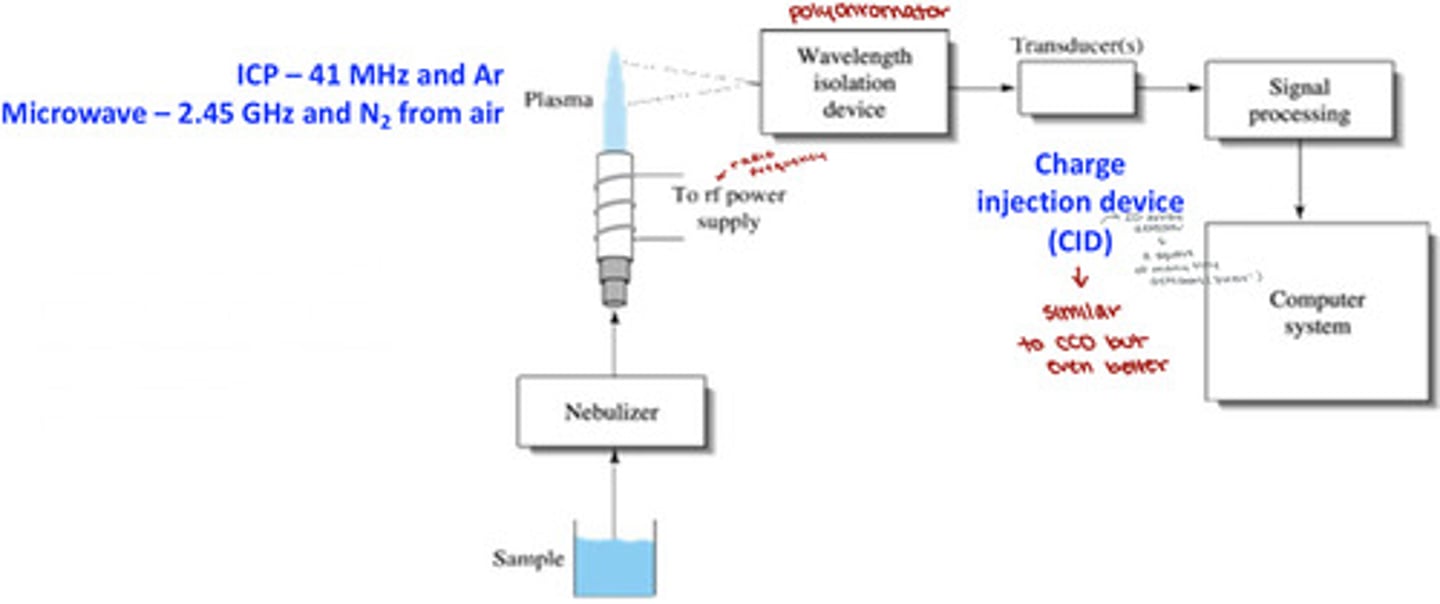
polychromator for ICP-AES (inductively coupled plasma-atomic emission spectrometer)
light emitted by a sample in the plasma:
1. enters the polychromator
2. is separated into a 2D array of wavelengths by a prism and orders by a grating
3. different wavelengths/orders are focused on difference sensing pixels of the 262,000-pixel charge injection device (CID) detector array (512x512 detectors)
4. emission from all elements in plasma is detected simultaneously
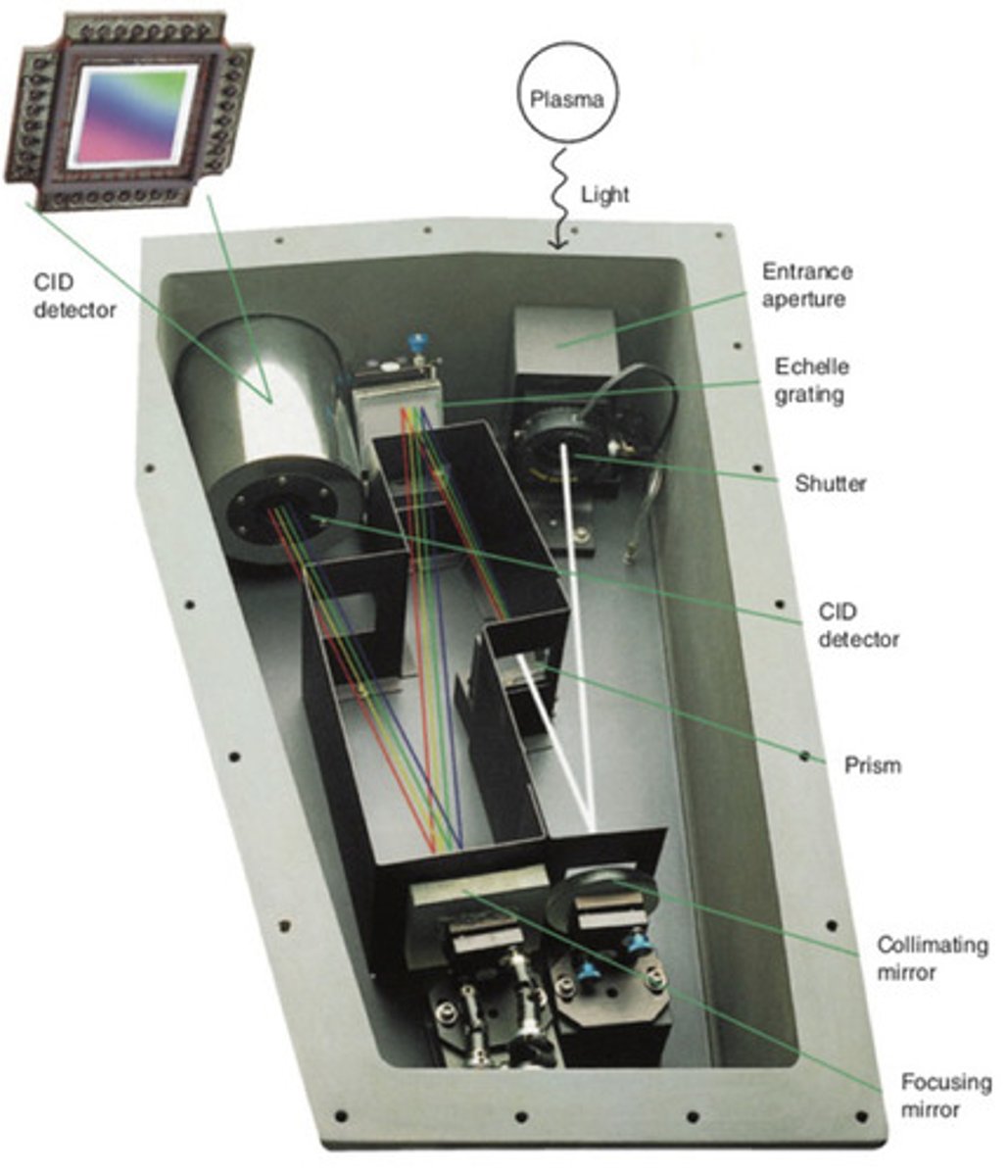
Multielement detection with a CID detector
A charge injection device (CID) array detector offers superior detection characteristics relative to charge couple device (CCD) array detectors.
1. CID pixels can be read individually at any time (not possible with CCD detectors)
2. CID detectors can measure a dim spot adjacent to a bright spot
3. Background correction is done automatically by the machine
CID detector
aka. charge injection device array detector-- offers superior detection characteristics relative to CCD array detectors; used in atomic emission (particularly with ICP-AES)
- CID pixels can be read individually at any time, CID detectors can measure a dim spot adjacent to a bright spot, and background corrections are done automatically
interference
any effect that changes the signal while analyte concentration remains unchanged
types: spectral, chemical, ionization
spectral interference
In atomic spectroscopy, any physical process that affects light intensity at the analytical wavelength. Created by substances that absorb, scatter, or emit light of the analytical wavelength.-- unwanted signals overlapping with the analyte signal
- the solution is to pick a difference line
chemical interference
In atomic spectroscopy, any chemical reaction that decreases the efficiency of atomization-- chemical reactions decreasing the concentration of analyte atoms
- the solution is releasing agents (preferentially reacts with interferent)
ionization interference
in atomic spectroscopy, loss of signal as a result of ionization of analyte atoms-- ionization of analyte atoms decreasing the concentration of neutral atom
- the solution is the use of an ionization suppressor
releasing agent
ionization suppressor
standard addition
same
The method of standard addition compensates for many types of interference by adding known quantities of standard (that is the same species as the analyte) to the unknown in its complex matrix.
standard addition compensates for the interferences by ensuring that t he calibration standards experience the ______ matrix as the analyte
virtues (benefits) of the inductively coupled plasma
An inductively coupled plasma eliminated many common interferences.
- The plasma is twice as hot as a conventional flame
- The residence time of analyte in the plasma is about twice as long
- Atomization is more complete and the signal is enhanced
- Formation of analyte oxides and hydroxides is negligible
- Calibration curves are linear over five orders. of magnitude compared o three orders of magnitude in flames and furnaces
methods of background correction
- pulsed source-- flame AA
- D2 lamp-- flame AA, GF AA
- zeeman correction-- GF AA
- on chip CID-- ICP AES
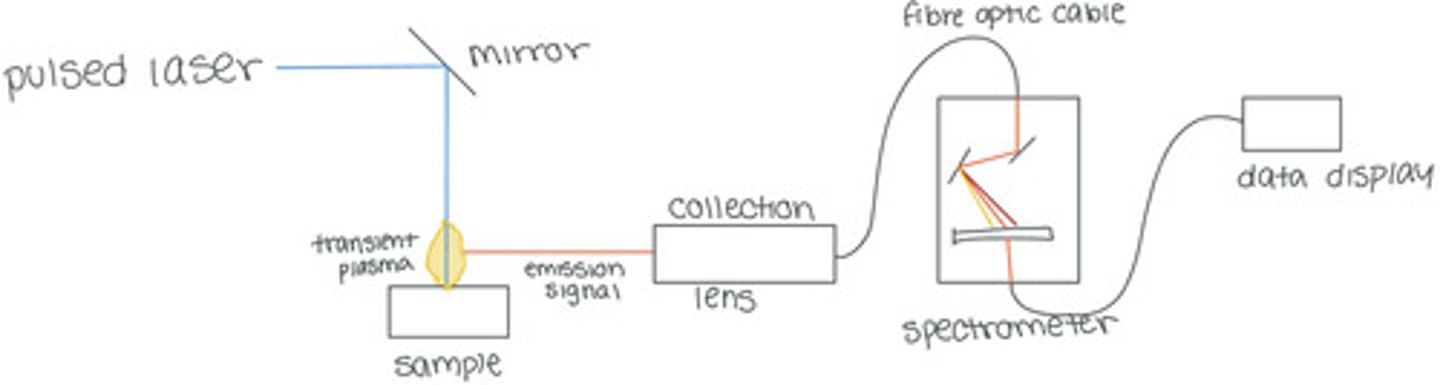
sampling by laser ablation
A pulsed laser is focused on to a microscopic spot on a solid sample, creating an explosion of particles, atoms, electrons, and ions into the gas phase
ex. when a pulsed laser (213nm, 2.7mJ of energy per pulse, repetition rate of 20Hz) is focused onto a spot with a diameter of 50mm-- with each pulse:
- a power of ~50GW/cm^2 is delivered to the surface
- an explosion occurs, sending portions of the surface (particles, atoms, electrons, and ions) into the gas phase-- i.e. a short-lived plasma
- material is removed to a depth of 0.02 to 5mm by each pulse
THE EMISSION FROM THE VAPORIZED ATOMS IS ANALYZED TO DETERMINE THE ELEMENTAL COMPOSITION OF THE SURFACE
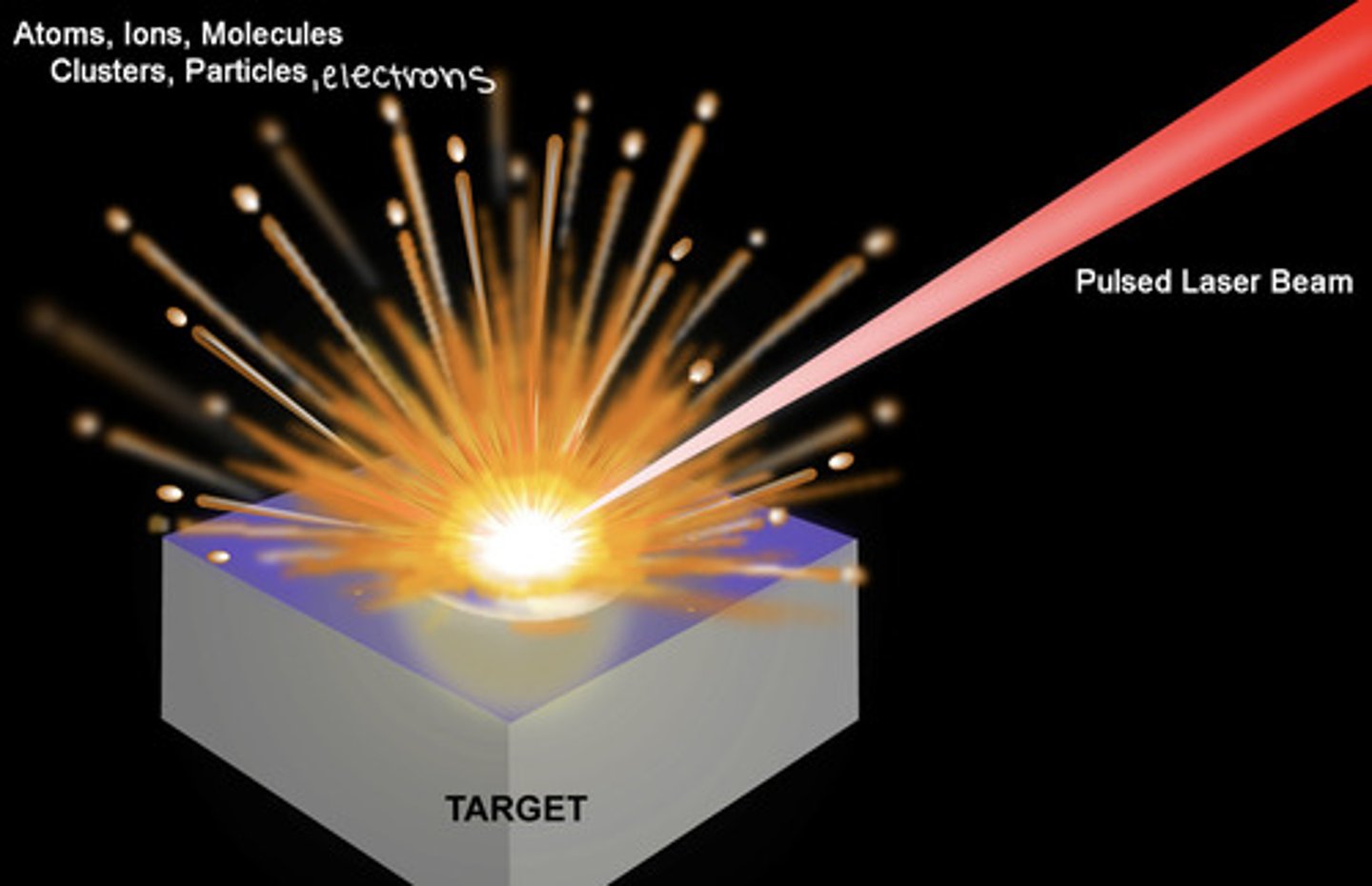
Laser-Induced Breakdown Spectroscopy (LIBS)
This type of spectroscopy records the atomic emission spectrum of the temporary plasma created by laser ablation.
In laser induced spectroscopy:
- each laser ablation pulse produces a short-lived plasma that is initially so hot that the continuum background masks the atomic line signals
- as the plasma expands and cools, the signal from discrete atomic emission lines rises above the continuum background
- to avoid the initial background interference, the detector is gated to delay measurement of atomic emission signals until several microseconds after the laser pulse
by gating the detector to delay measurement a few microseconds so the plasma can expand and cool slightly before measuring the atomic emission signals
how does LIBS (laser-induced breakdown spectroscopy) avoid the initial background interference caused by the initially excessive heat of the transient plasma?
pulsed laser
A pulsed beam of monochromatic light that delivers energy in controlled amounts-- used in the laser-induced breakdown spectroscopy to laser ablate the sample creating a transient plasma from which atomic emission signals can be detected to determine the elemental composition of the surface layer of the ablated object
continuous laser
A continuous beam of monochromatic light
light amplification by stimulated emission of radiation
what does LASER stand for?
the emission from vaporized atoms in the transient plasma created by laser ablation (to determine the elemental composition of the surface)
what is analyzed when laser-induced breakdown spectroscopy?
laser-induced breakdown spectroscopy
Semiquantitative measurement of elements in a surface by vaporizing a small patch with a short laser pulse and measuring atomic emission from the plasma above the surface.
Atomic Emission Spectroscopy on Mars (using LIBS)
The Mars Science Laboratory rover, Curiosity, uses laser-induced breakdown spectroscopy to analyze the elemental composition of rocks and stuff on the surface of Mars.
1. The ChemCam laser-induced breakdown spectroscopy unit measures the chemical composition of rocks and soil up to 5m away
2. Scientists select a target with the rover's high resolution telescope
3. A 1.067-mm-wavelength laser then fires a series of pulses through the telescope to vaporize a ~0.4mm-diameter area
4. Each pulse creates a luminous plasma of atoms
5. Optical emissions from the plasma are collected through the telescope to generate a profile of elemental composition of the irradiated area
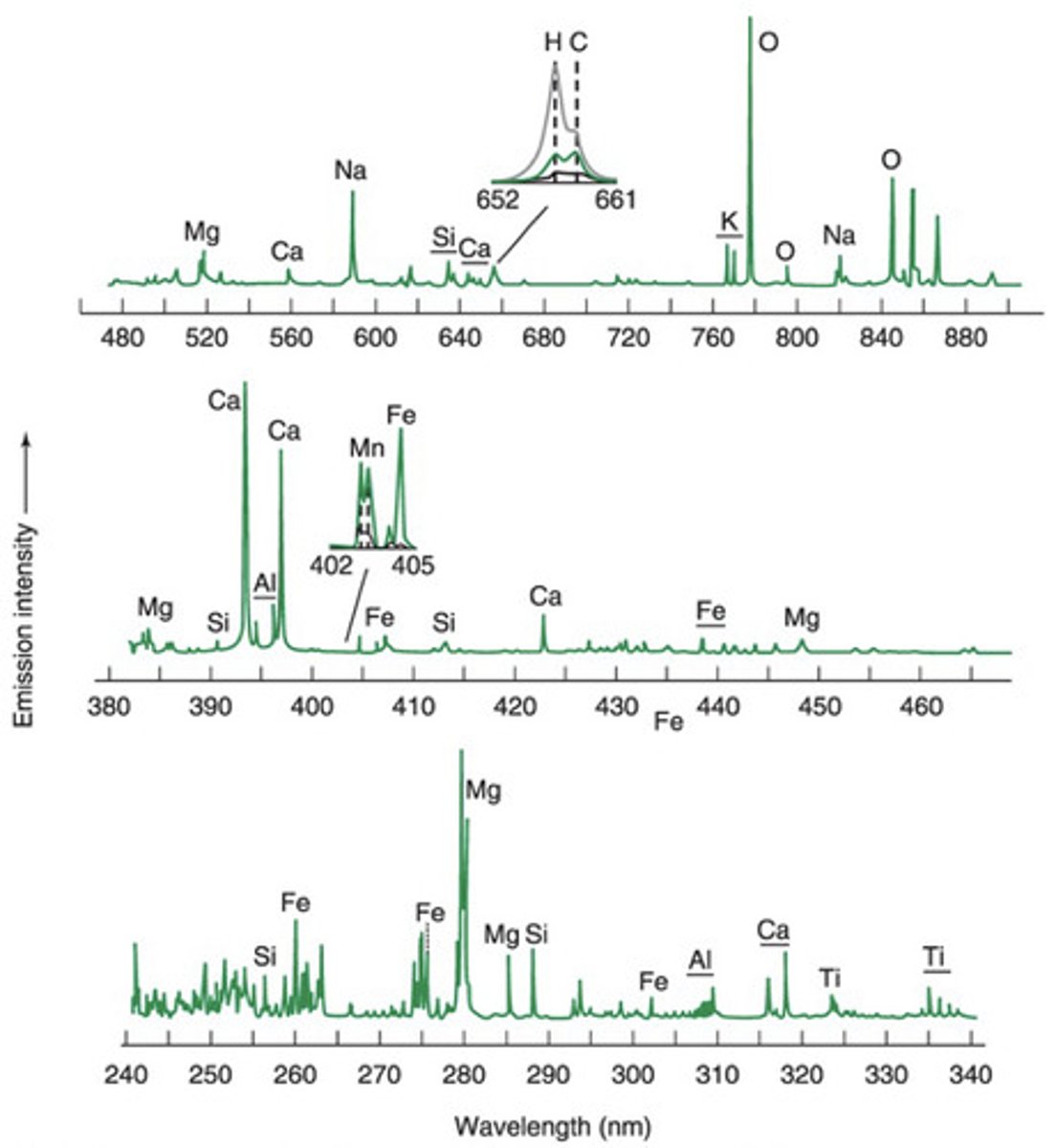
Atomic Emission Spectra from Mars
- These spectra were collected using laser-induced breakdown spectroscopy (LIBS)
> Strong atomic emissions are observed for abundant elements like magnesium, calcium, sodium, and iron
> The sharpness of atomic emissions (narrow bandwidths) allows weaker nearby signals to be observed
> The top inset shows emissions from hydrogen and carbon
> Hydrogen arises from waters of hydration with amorphous particles, allowing estimation of at the amount of water on Mars
> The lower inset shows emission from Mn-rich minerals, revealing that the Martian atmosphere was once wetter with considerably more free oxygen than today
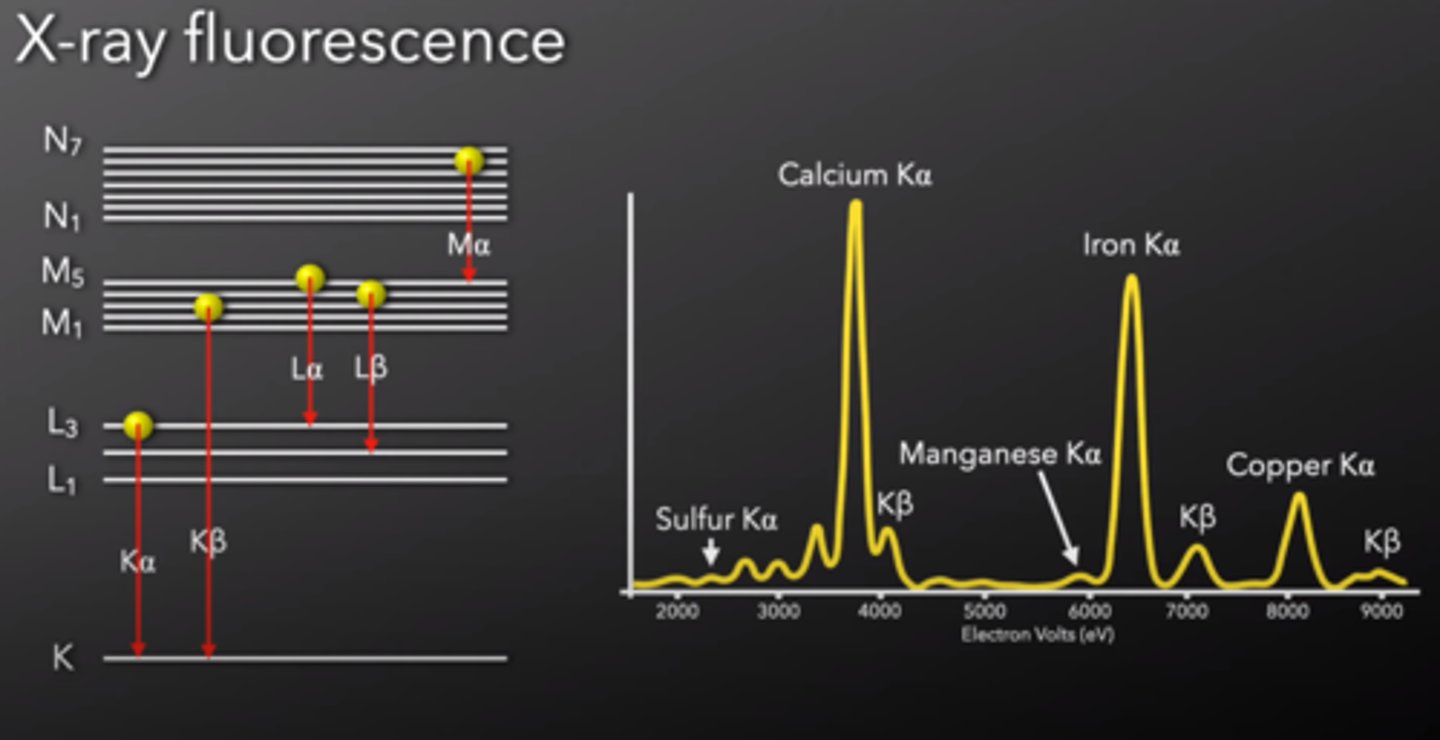
X-ray fluorescence (XRF)
The emission of x-rays following the absorption of x-rays by a material
- The abscissa (x-axis) is x-ray energy in keV rather than wavelength, the y-axis is the detector response (counts per second)
- Elements are identified by their peak energies and quantified by the number of photons in each peak
X-ray fluorescence transitions
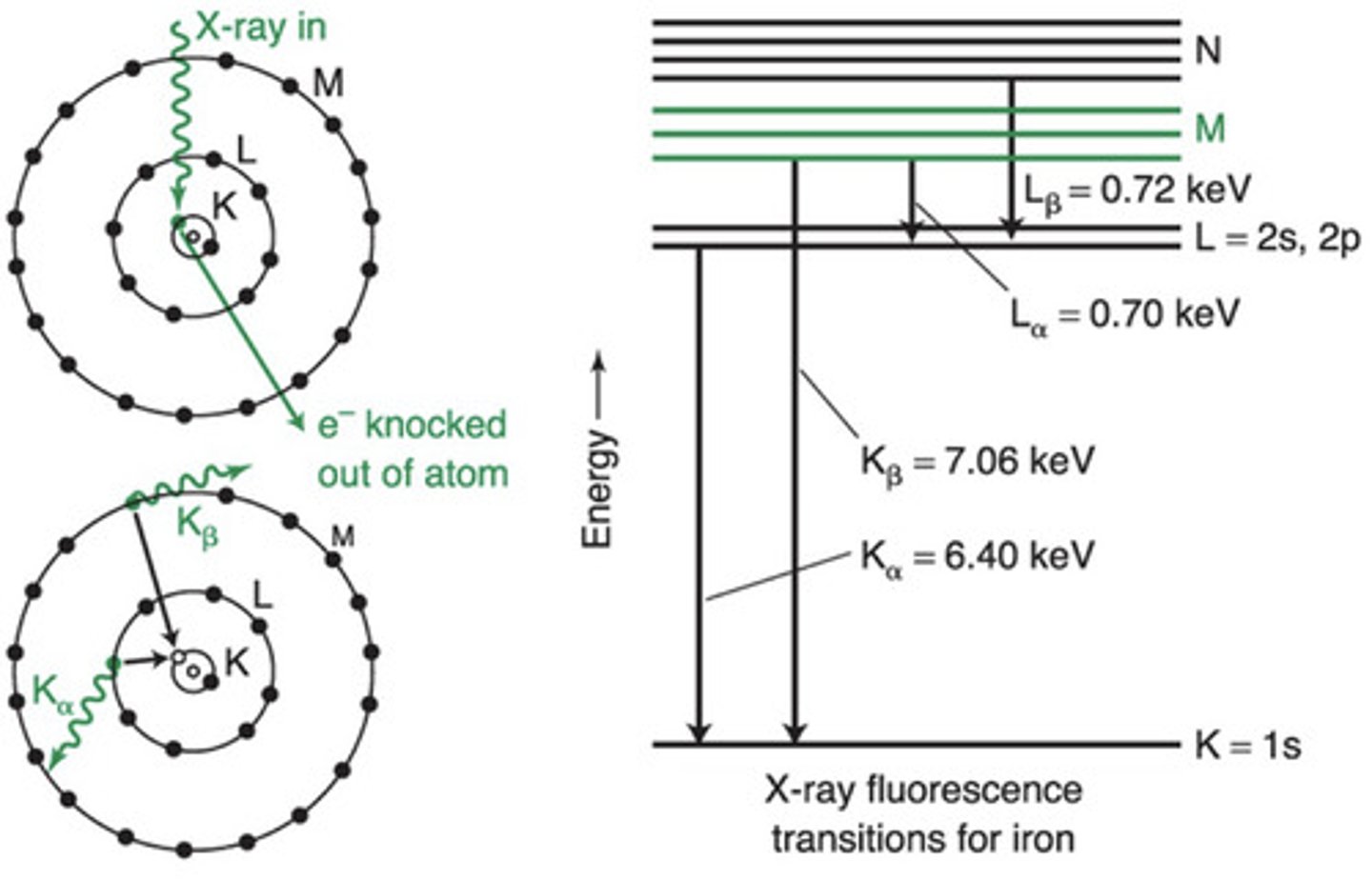
Electron shells were historically labelled K, L, M...
> K shell = the 1s orbital
> L shell = the 2s and 2p orbitals
> M shell = the 3s, 3p and 3d orbitals
> N shell = the 42, 4p, 4d and 4f orbitals
In X-ray fluorescence, an incoming x-ray knocks an electron in the K or L shell out of the atom and an electron from a higher shell falls into the vacancy (relaxes from higher energy orbital to lower energy orbital) and emits x-rays in the process
> L to K (2 to 1) = Kα X-ray
> M to K (3 to 1) = Kβ X-ray
> M to L (3 to 2) = Lα X-ray
> N to L (4 to 2) = Lβ X-ray
α = relaxation from one shell above
β = relaxation from 2 shells above
K shell
energy level closest to the nucleus-- most stable (lowest in energy)-- 1s
L shell
2s, 2p
M shell
3s, 3p, 3d
N shell
4s, 4p, 4d, 4f
Kα X-ray
emission resulting from the relaxation of an electron from the L shell to the K shell (from 2s/2p to 1s)
Kβ X-ray
emission resulting from the relaxation of an electron from the M shell to the K shell (from 3s/3p/3d to 1s)--- this is the highest energy emission (most keV) because the M shell is significantly higher in energy than the K shell
Lα
emission resulting from the relaxation of an electron from the M shell to the L shell (from 3s/3p/3d to 2s/2p)--- this is the lowest energy emission (lowest keV) because the M shell is very close in energy to the L shell
Lβ
emission resulting from the relaxation of an electron from the N shell to the L shell (from 4s/4d/4p/4f to 2s/2p)