Youngjae You Chemistry Exam 4
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
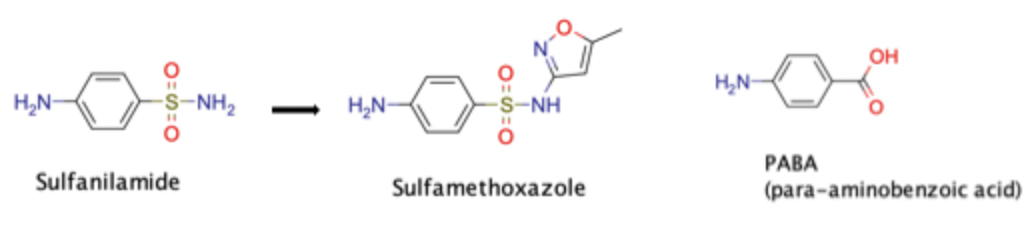
Modification of Sulfanilamide → Sulfamethoxazole
Increases the ability to mimic PABA and inhibit bacterial folic acid synthesis
Increases water solubility of drug and decreases urine precipitation
Pharmacophore
Minimum structural requirements for drug activity
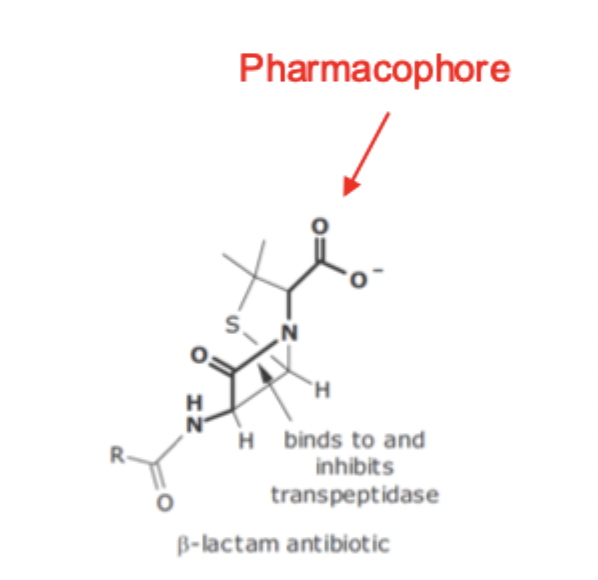
B - Lactam Antibiotics SAR
must contain ionized C. Acid
must contain intact B-Lactam ring
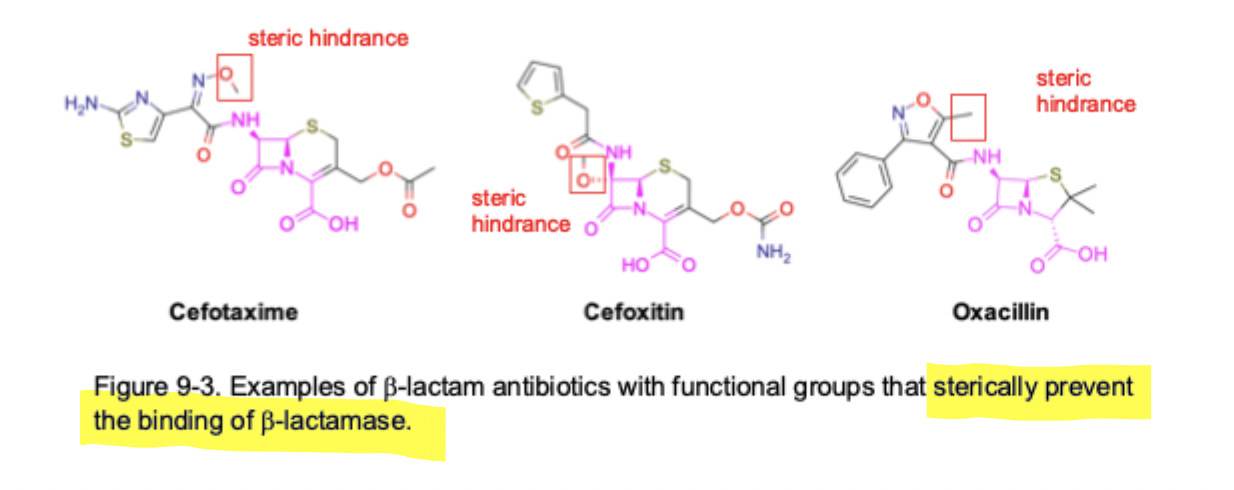
B - Lactam Antibiotics SAR to prevent B-lactamase inactivation
Steric hindrance & doesn’t interfere with binding to transpeptidase
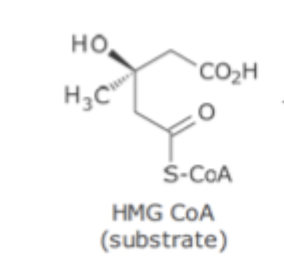
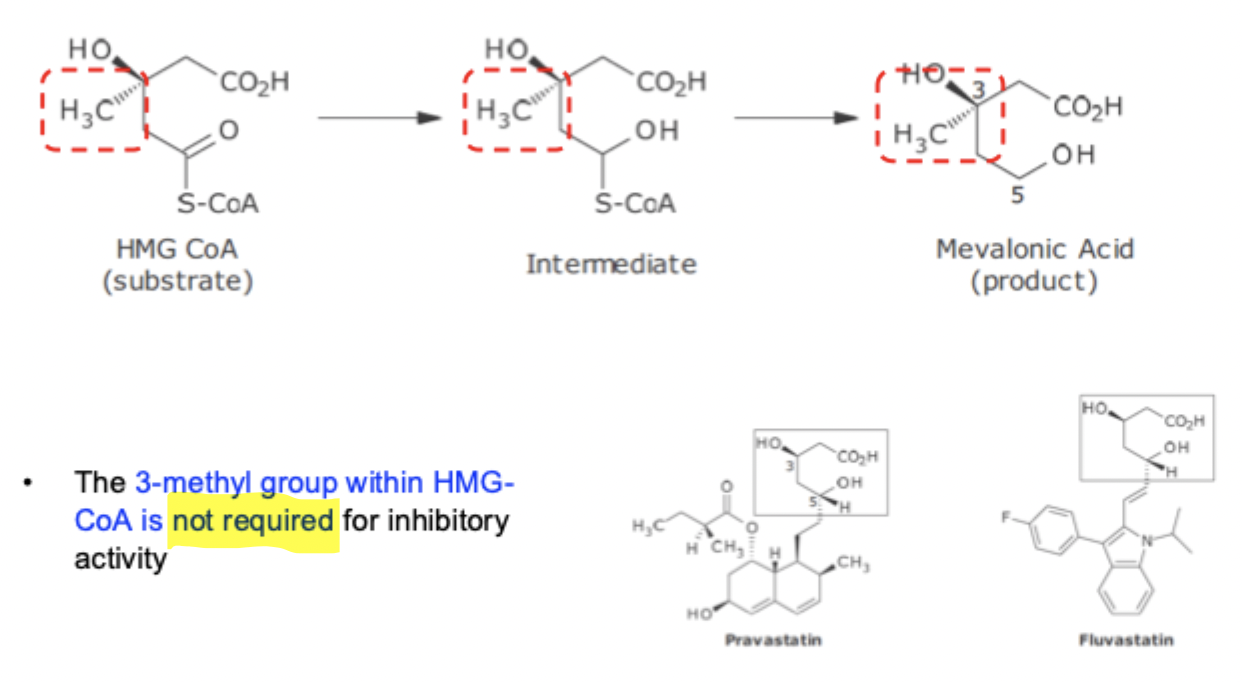
HMG-CoA Reductase SAR
Mimics substrate / product / transition state
3 - methyl group NOT required for inhibitory effect
BONUS: “Corpora non agunt nisi fixate” → binding interactions are critical to MOA of drugs and selectivity
Paul Ehrlich
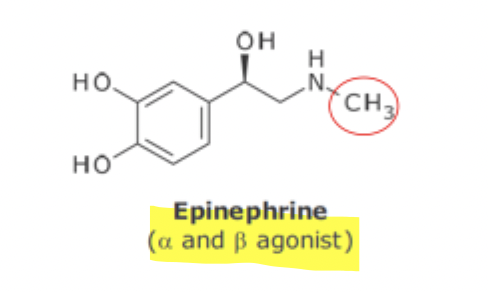
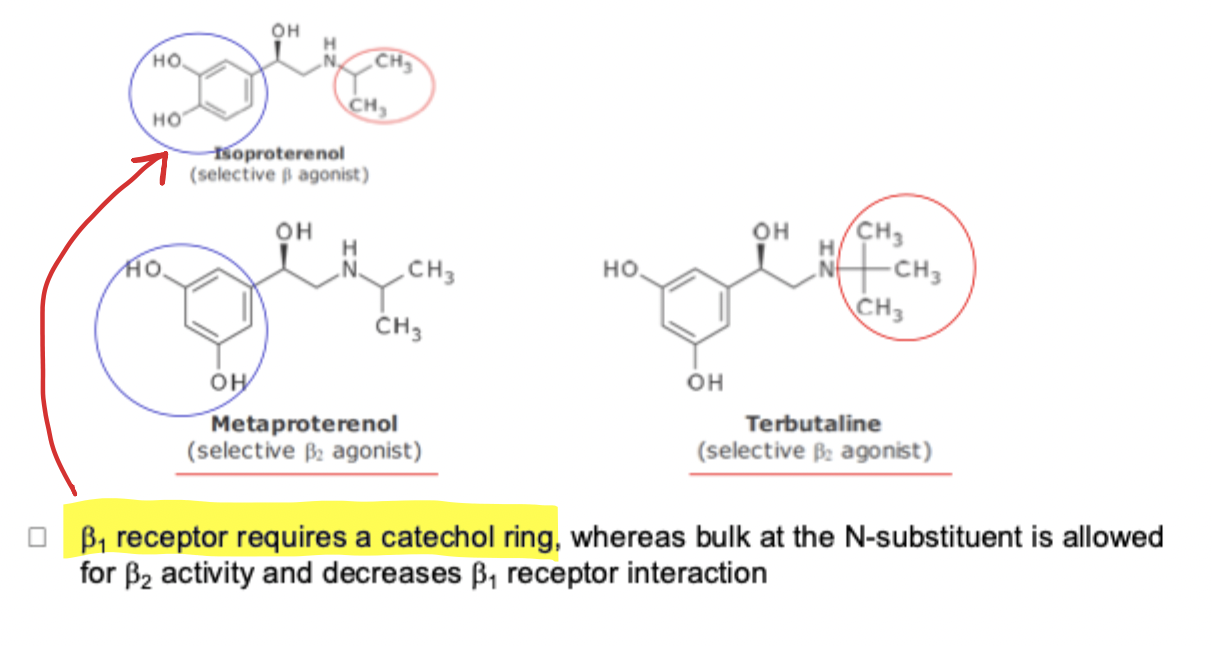
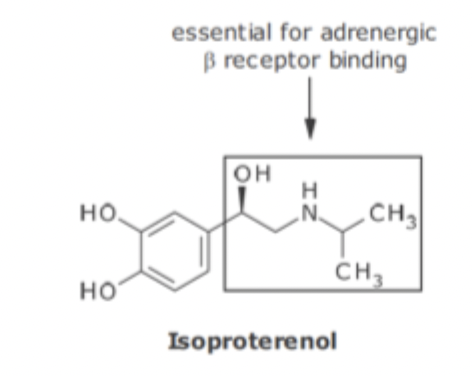
Epinephrine and Selective B Agonist SAR
replacement of N-methyl group with isopropyl group results in no A adrenergic activity but enhanced B1 and B2 activity
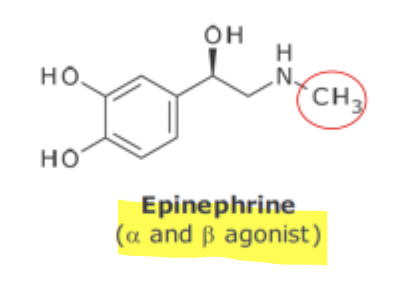
Separation of B1 and B2 activity SAR
B1 receptor → requires catachol ring
B2 receptor → requires bulk N-substitution and decreases B1 receptor interaction
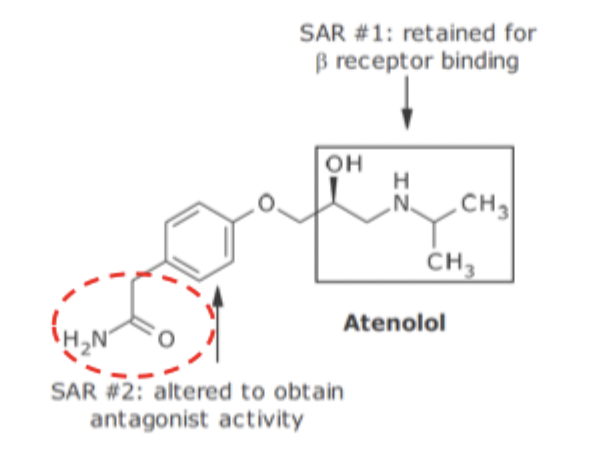
B1 Receptor Antagonism SAR
Replacement of catachol ring with para substituted phenyl ring changes it to antagonistic activity
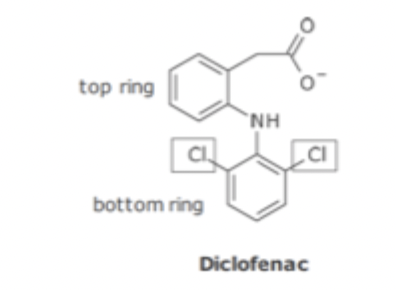
NSAID Conformational Restriction
Ortho substituents on lower ring cause conformation restriction and prevent rotation
Enhances binding cyclo-oxygenase binding
CCB Conformational Restrictions
Phenyl ring must be perpendicular to the 1,4 - DHP
steric hindrance restricts rotation and assures required conformation
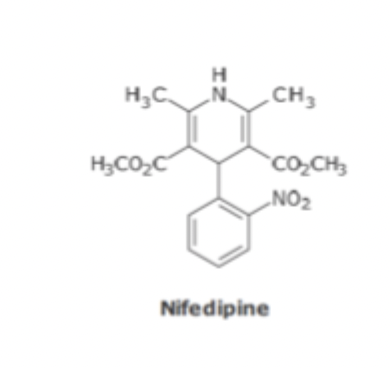
ACE Inhibitor SAR
Needs FG that can interact with zinc atom on enzyme active site
Lipinski’s Rule of 5

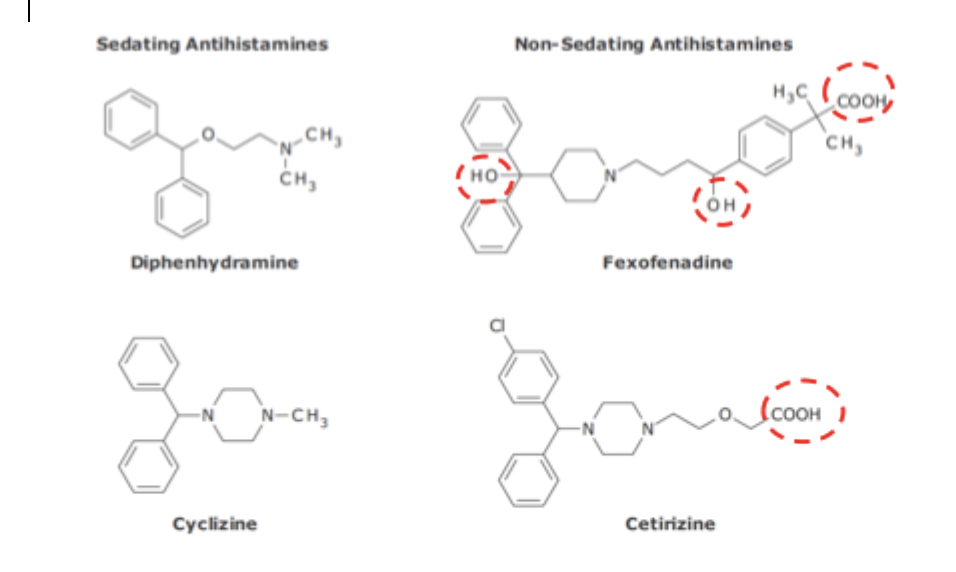
Sedating vs. Non-sedating Antihistamines
Diphenhydramine & cyclizine have lipid solubility enough to cross the BBB whereas fexofenadine & certirizine don’t
contain alcohol groups that make them more water soluble
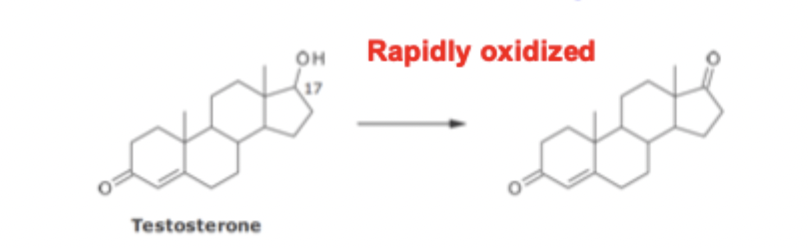
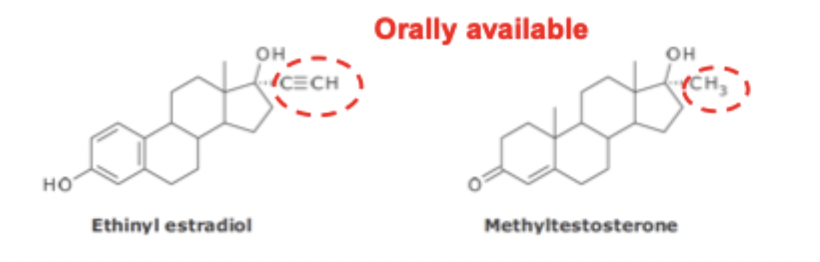
Metabolism of Estrogens & Androgens SAR
cannot be administered orally because they are rapidly oxidized
17a substituent blocks the C17 Hydroxyl from oxidation and allows these drugs to be administered orally
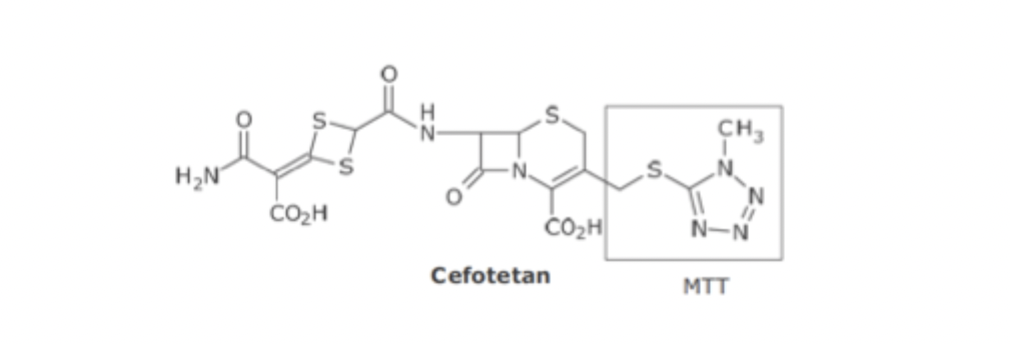
Adverse reaction of Cefotetan
The MTT group was associated with acute alcohol intolerance and serious bleeding
Once linked to this FG, drugs containing MTT were discontinued
Tetracycline & Fluoroquinolone Drug Interactions
dicarbonyl groups can chelate with Ca, Mg, Al, and Fe in the gut
this leads to very poor water solubility and decreased absorption
do not take vitamins/dairy that contain these metal ions
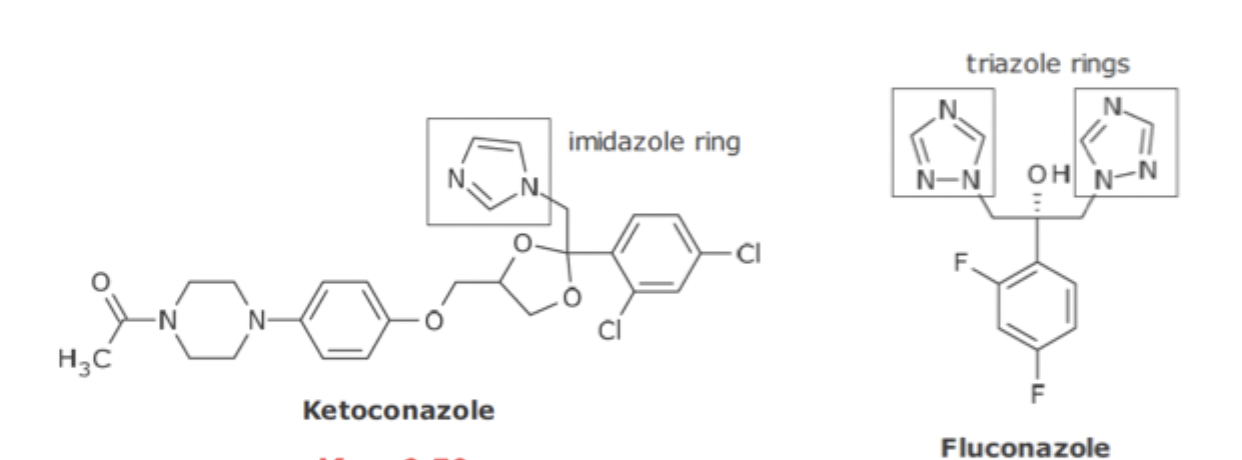
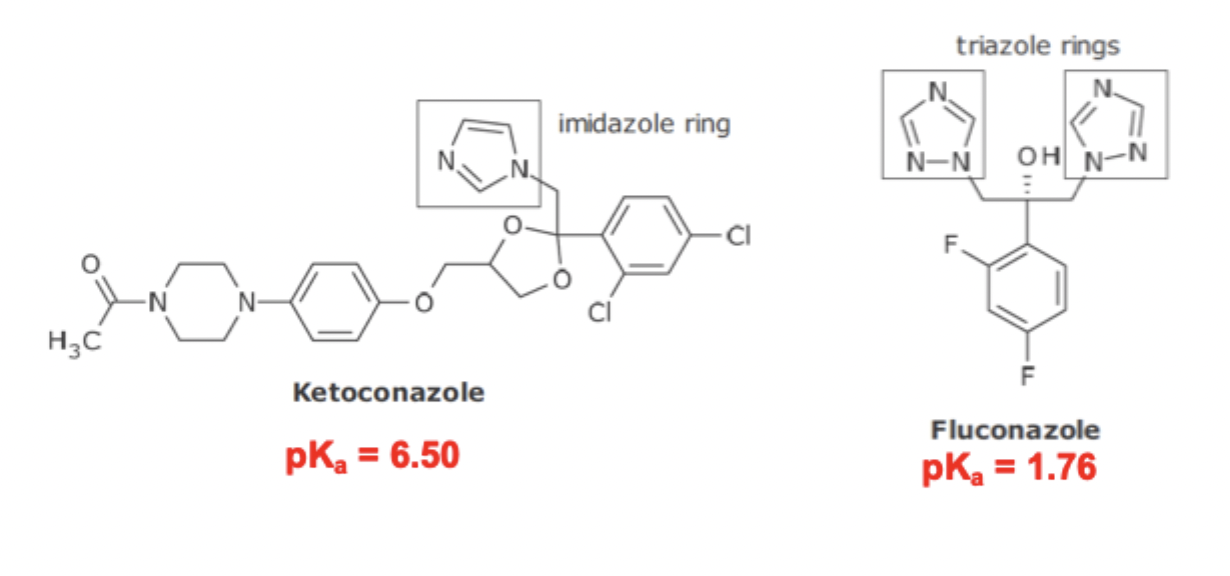
Water/Lipid solubility of Azole Antifungal Agents
Imidazole ring is pH dependent, meaning increasing pH can decrease ionization and water solubility, from DDI
The two triazole rings are not pH dependent and therefore safer to use in every situation
Acid / Base Plasma Protein Displacement Interaction
Acids
Bind substantially to plasma albumin through ionic interactions
Bases
Bind moderately to a1-acid glycoprotein
may be be clinically relevant
Lead Compound

Lead Compound Discovery
Virtual screening
Fragment-based drug design
Drug repurposing
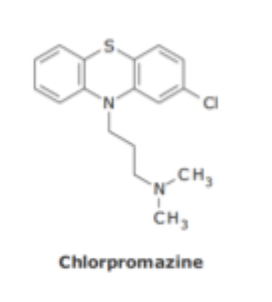
Phenothiazine Antipsychotics SAR
Conformational restriction requires aliphatic amine to lie on the same side as the chlorine on the aromatic ring
Z isomer more potent then E isomer

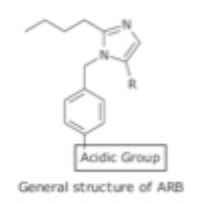
ARB SAR
substitution of C. Acid with another acidic group such as tetrazole gives several benefits
Tetrazole less likely to undergo metabolism
is more lipophilic
enhances oral absorption
allows for better charge distribution
enhances stability and binding interaction to receptor
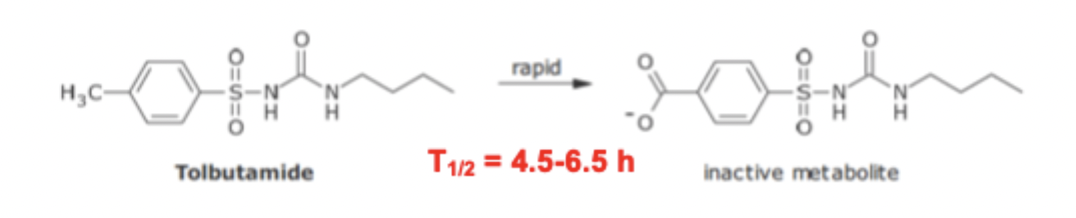
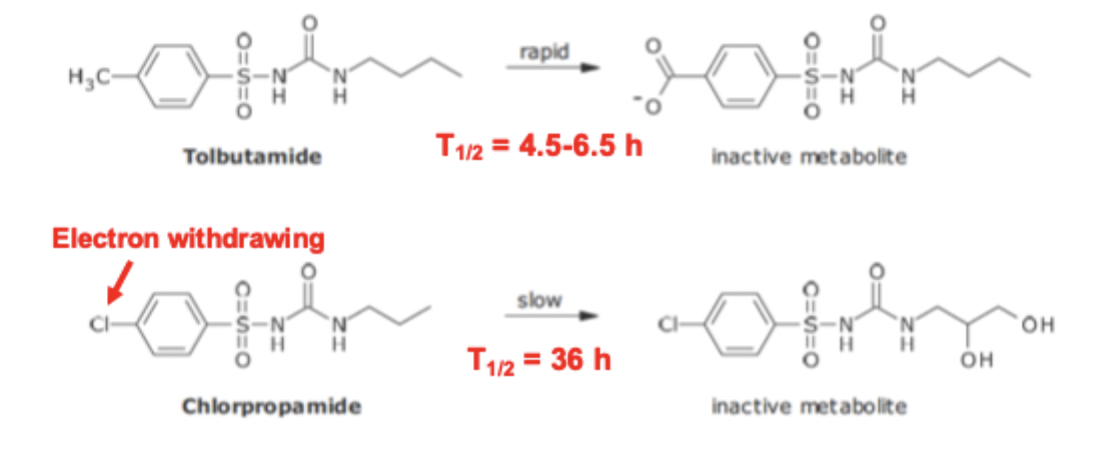
Sulfonyl Urea variation of FG SAR
para methyl is rapidly metabolized
changing the group to a chloro FG prolongs half life
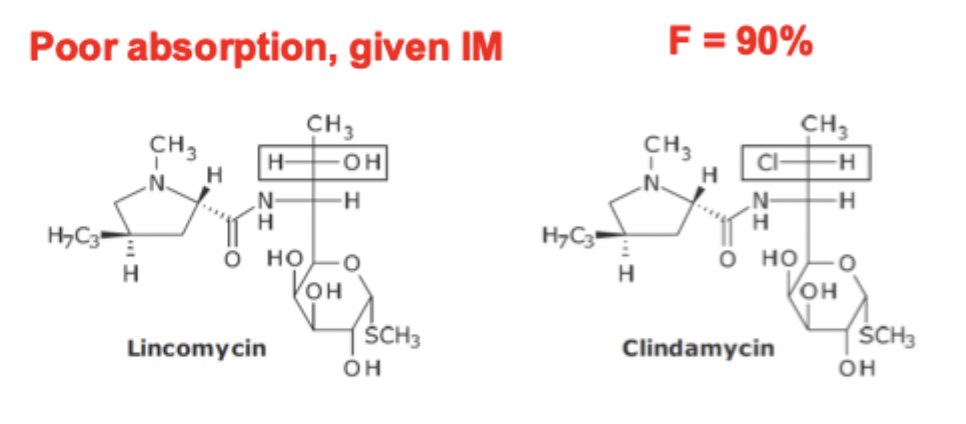
Lincoasamide variation of FG SAR
Replacement of -OH group with -Cl group
enhances lipophilicity
crosses bacterial membranes better
better oral absorption
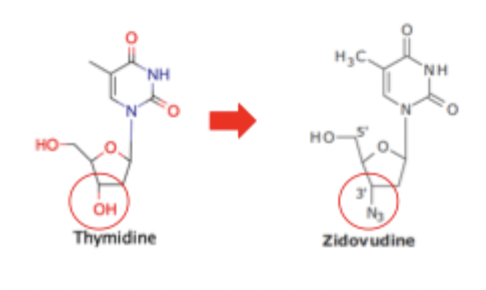
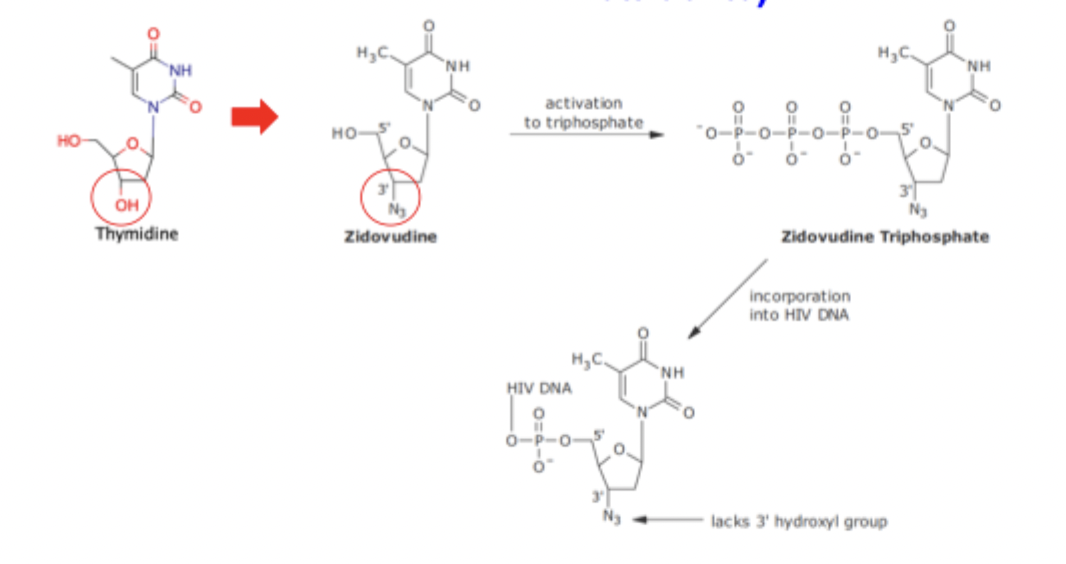
Zidovudine Inhibition of Reverse Transcriptase SAR
Zidovudine mimics nucleic acids but with a removed 3’ OH group for an azido group inhibiting function
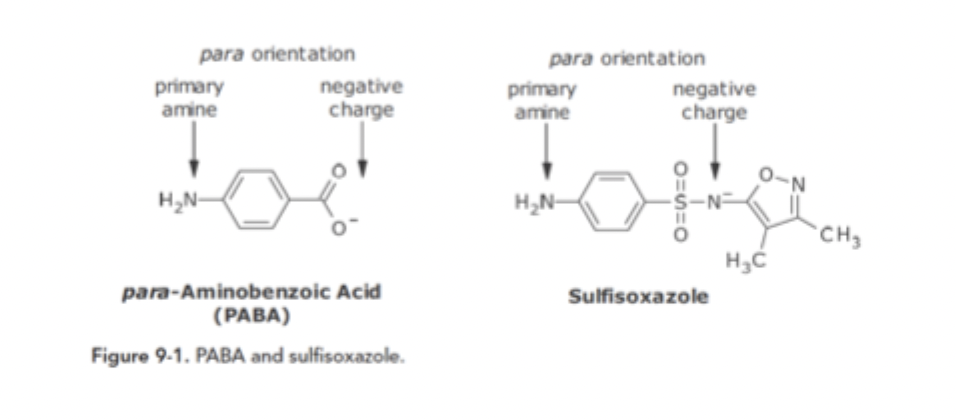
Sulfonamide Anti-metabolite Variation of FG SAR
Sulfisoxazole contains groups mimicking PABA but with an additional group that inhibits bacterial enzymes
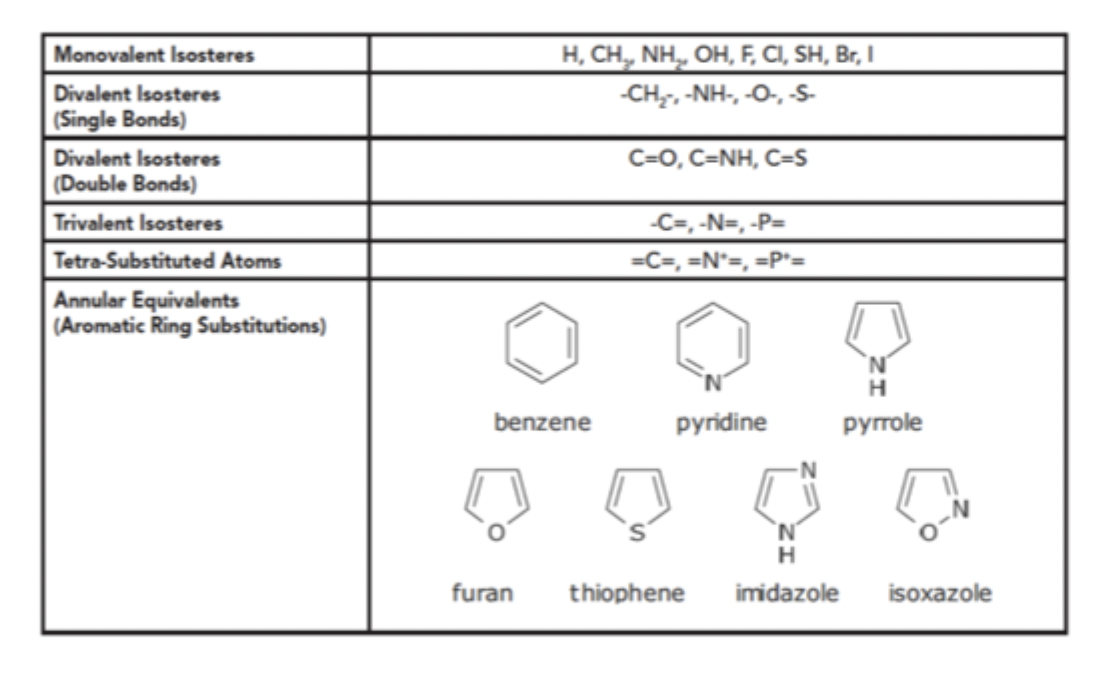
Isosteres
Compounds and Groups of atoms that contain same number and arrangement of electrons
Limited and not very applicable to drug molecules
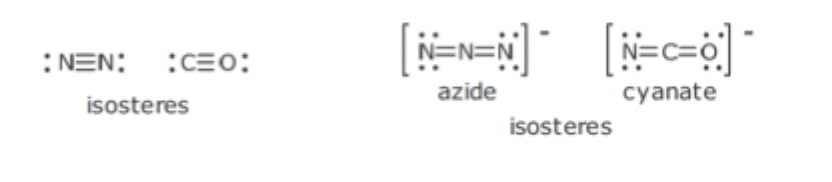
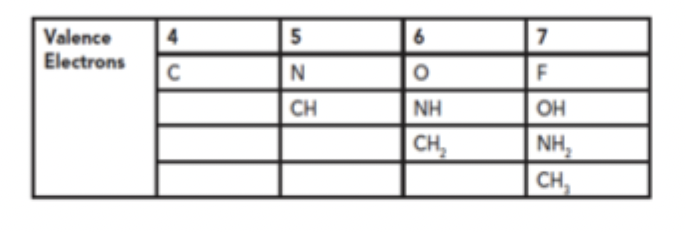
Grimm’s Hydride Displacement Law
organized groups of atoms according to valence atoms and allowed for different number of atoms
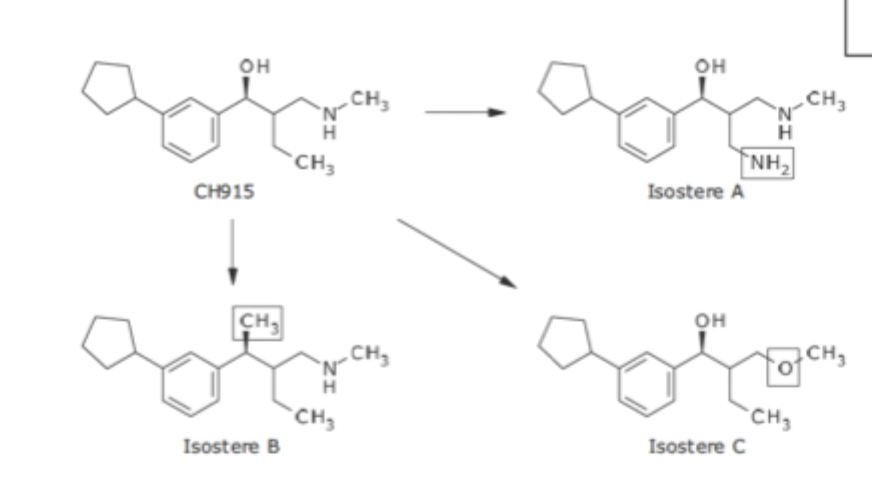
Erlenmeyer Isosteres
Expanded the table of isosteres
considered:
ionization
neutral molecules
water solubility
lipid solubility
electronegativity, steric size, shape
Friedman Bioisosteres
Functional groups or molecules that have similar chemical and physical properties and produce broadly similar biological properties
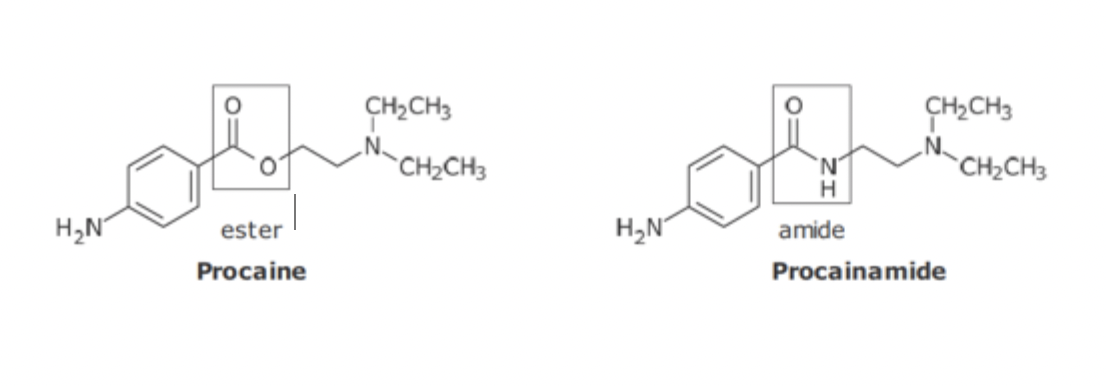
Anti-Arrhythmic isosteric replacement
Ester undergoes rapid hydrolysis, poor F, short duration
N substitution decreases metabolism, and increases F
Nonclassical Isosteres
Do not follow the guidelines set by Grimm and Erlenmeyer
Larger in size, rarely have same # of atoms, and can vary in valence electrons
Homologation
Extending a hydrocarbon chain by successively adding methylene groups
Increases size and lipophilicity of FG
Ex. N - Homologation of epinephrine makes it beta selective
In Vitro, in Vivo, In Silico
Outside an organism, like a petri dish
Inside living organism
On a computer
In Vivo and In Vitro, activity is dependent on …
Concentration
PK Route
IV Bolus
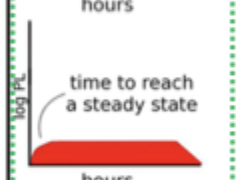
PK Route
IV Infusion
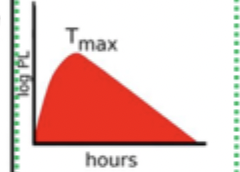
PK Route
Oral tablet or capsule
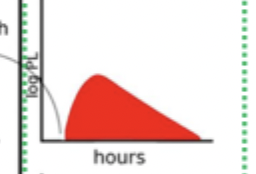
PK Route
Enteric coated tablet or capsule
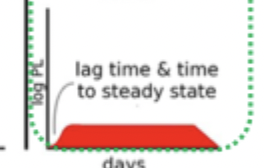
PK Route
Transdermal patch
______ is the driving force for drug activity / response
Drug Plasma Concentration
Bioavailability
Fraction of initial dose that reaches circulation after first pass effect
F = AUC oral / AUC iv
Volume of Distribution (Vd)
Proportionately relates amount of drug in body with measured plasma concentration
High Vd = high tissue distribution
weak basic groups, low plasma binding, high lipophilicity, low ionization rates
Clearance (Cl)
Volume of plasma which drug is totally removed over a specified period of time
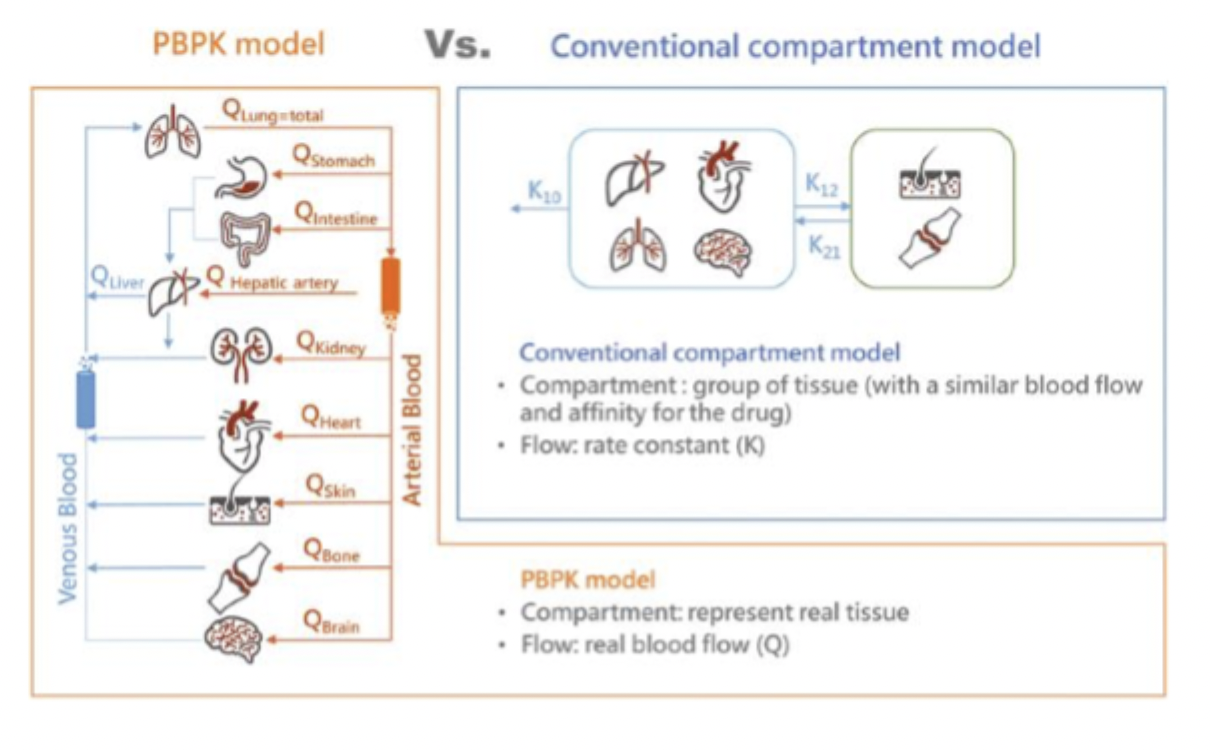
PBPK model vs Conventional Compartment Model
PBPK → real tissue and blood flow, more realistic
Compartment → more simplified, grouped tissues
BONUS: Occupancy theory, relationship between concentration and target occupancy
A.V. Hill

BONUS: Response is directly proportional to occupancy
A.J. Clark
Hill Equation
Measures the relationship between concentration and activity
Emax, EC50, C, etc..
Hammet Equation
Relates the electronic effects of substituents on the reactivity of aromatic ring compounds
Plasma Protein Binding - Weak Bases
A1-Acid Glycoprotein
becomes protonated inside tissues/lysosomes
trapped in tissue = high Vd
Plasma Protein Binding - Weak Acids
Albumin
negative charge attracts albumin binding
Stays in plasma = Low Vd
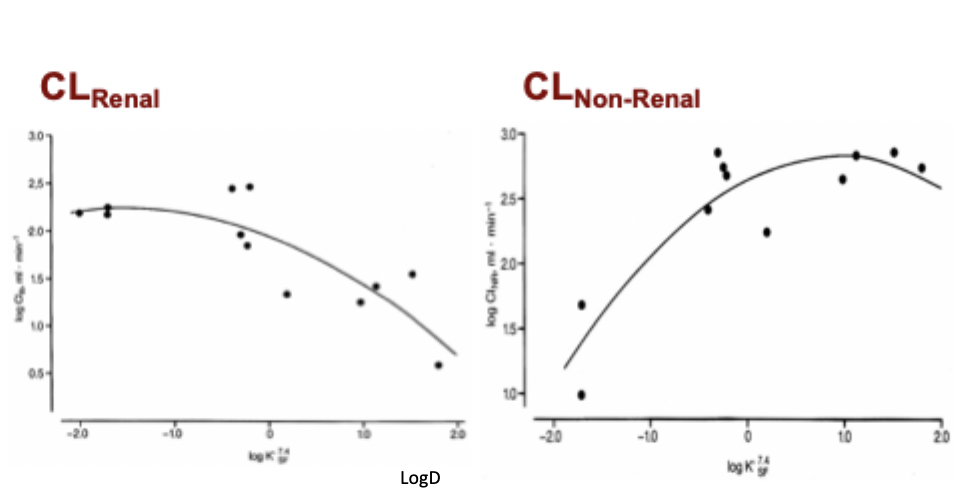
Relationship between LogD and renal clearance
High LogD = drug tissue permeable and is reabsorbed = low renal clearance = high non-renal clearance