S2
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
The total number of protons and neutrons in the nucleus of an atom
The number of protons in the nucleus of an atom.
Atoms of the same element with the same number of protons but different numbers of neutrons.
The weighted mean mass of the atoms of an element relative to 1/12 of the mass of a carbon-12 atom.
A spectrum consisting of discrete lines at specific wavelengths corresponding to electron transitions between energy levels.
A spectrum that contains all wavelengths of electromagnetic radiation with no gaps.
The energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous 1⁺ ions.
What EM radiation is released when an electron goes transitions to the n=1 energy level in hydrogen?
UV
What EM radiation is released when an electron goes transitions to the n=2 energy level in hydrogen?
Visible light
What EM radiation is released when an electron goes transitions to the n=3 energy level in hydrogen?
IR
Radioisotope
An isotope with an unstable nucleus that undergoes radioactive decay.
Alpha particle (α)
A helium nucleus consisting of 2 protons and 2 neutrons, emitted during radioactive decay.
Beta particle (β⁻)
A high-speed electron emitted from the nucleus during radioactive decay.
Carbon-14 dating - application
A method used to determine the age of organic materials based on the decay of carbon-14.
Electromagnetic spectrum
The range of all types of electromagnetic radiation arranged according to frequency or wavelength.
Ground state vs excited state
Ground state: the lowest energy level of an electron in an atom.
Excited state: any higher energy level an electron occupies after absorbing energy.
Ion
A charged particle formed when an atom or group of atoms gains or loses electrons.
Cation
A positively charged ion formed by loss of electrons.
Anion
A negatively charged ion formed by gain of electrons.
Equation relating frequency and wavelength
c=λν
c (speed of light (in a vacuum)): 3.00 × 108 m s−1
λ (wavelength):
ν (frequency):
First ionisation energy
The energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous 1⁺ ions.
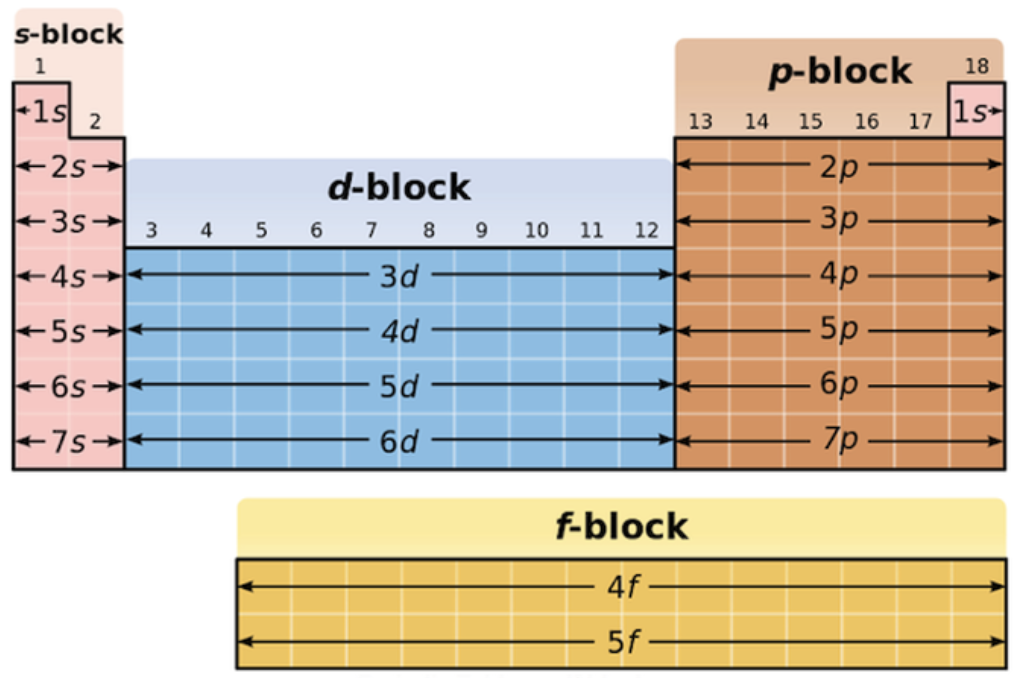
Sublevels (subshells) - and location periodic table
s, p, d, f
Atomic orbital
A region of space around the nucleus where there is a high probability of finding an electron.
Pauli exclusion principle
An orbital can hold a maximum of two electrons with opposite spins.
Aufbau principle
Electrons occupy orbitals of lowest energy first before filling higher energy orbitals.
Hund’s rule
Electrons occupy degenerate orbitals singly with parallel spins before pairing.
Electron spin
A property of electrons describing two possible orientations (↑ or ↓), giving rise to magnetic behavior.
Quantum numbers
Values that describe the position and energy of an electron in an atom (principal, angular momentum, magnetic, spin).
In what way do electrons have energy?
Energy is quantized; electrons occupy discrete energy levels rather than continuous values.
Stability and its relation to energy in particles
Lower energy states are more stable; systems tend to move toward minimum energy.
s orbital
A spherical orbital centered on the nucleus (holds a maximum of 2 electrons).
p orbital
A dumbbell-shaped orbital with two lobes oriented along axes (set of three orbitals, max 6 electrons).
Photon
A discrete packet (quantum) of electromagnetic radiation with energy proportional to frequency.
Bohr’s model
Electrons occupy fixed energy levels (shells) around the nucleus and emit/absorb energy when transitioning between levels.
Limitations of Bohr’s model
Only works for hydrogen-like atoms (one electron).
Does not explain fine spectral details or electron behavior in multi-electron atoms.
Inconsistent with quantum mechanics (no electron probability).
Exceptions to the Aufbau principle - and why
Chromium (Cr):
Electron configuration: [Ar] 3d⁵ 4s¹ (instead of 3d⁴ 4s²)Copper (Cu):
Electron configuration: [Ar] 3d¹⁰ 4s¹ (instead of 3d⁹ 4s²)
Half-filled and fully filled d sublevels have lower energy due to reduced electron–electron repulsion and increased stability.
Factors affecting ionisation energy
Nuclear charge (more protons → higher attraction → higher IE)
Distance from nucleus (greater distance → lower IE)
Shielding effect (more inner shells → lower IE)
Electron configuration (sublevel/repulsion effects)
Limit of convergence (don’t need to know definition, just understand concept)
The point at which spectral lines converge, corresponding to the ionisation energy (electron completely removed).
Successive ionisation energies
The energies required to remove electrons one at a time from gaseous atoms; large jumps indicate removal from a new (inner) shell.