OChem 1 Reactions
1/91
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
92 Terms

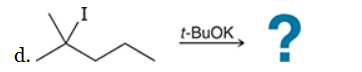
w/ strong base
w/strong base
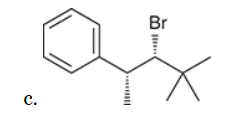
w/ strong base
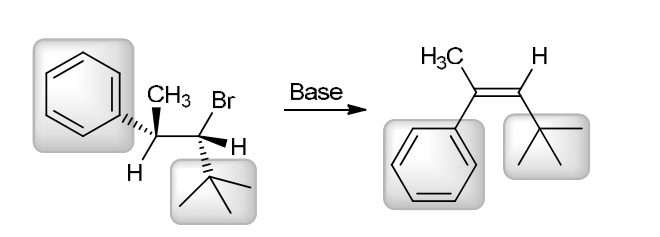
w/ strong base
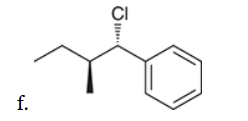
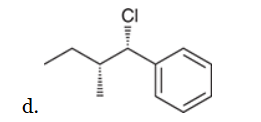
w/ strong base
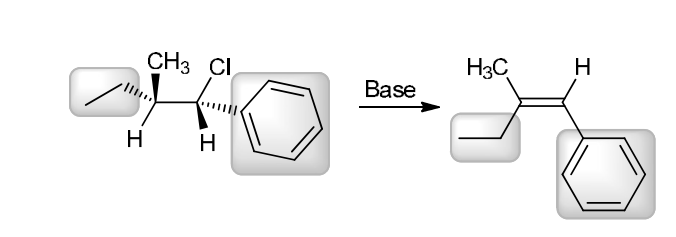
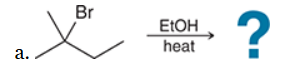
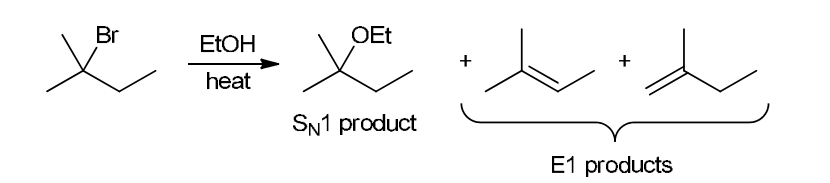
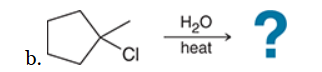
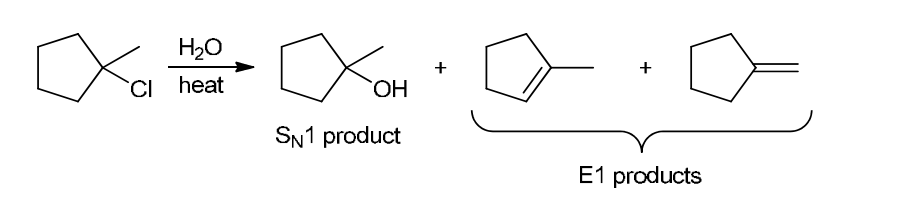
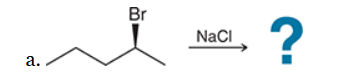
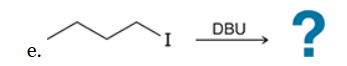
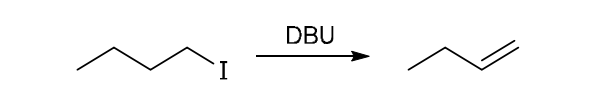
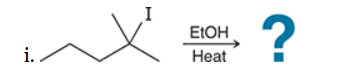
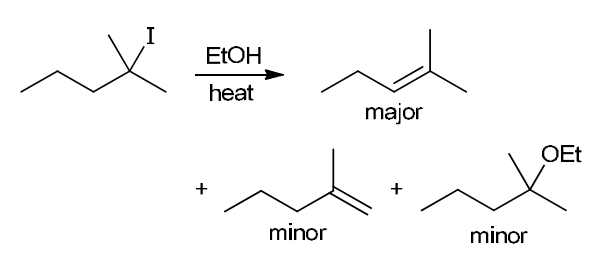
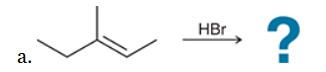
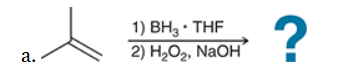
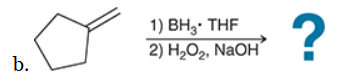
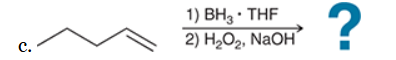
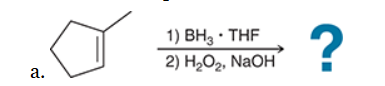
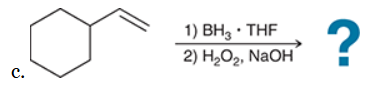
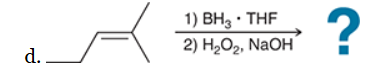
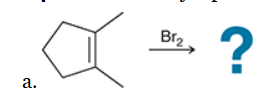
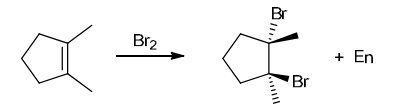
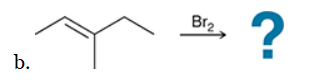
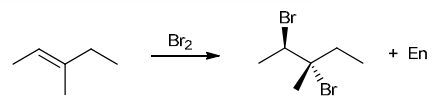
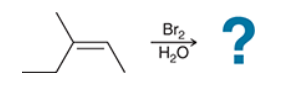
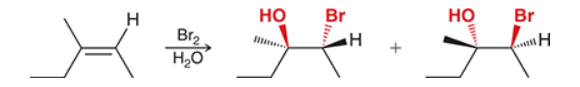
with Br2/H2O
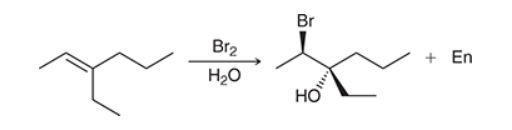
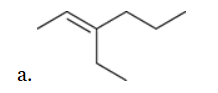
with Br2/H2O
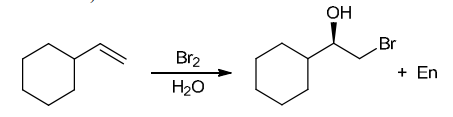
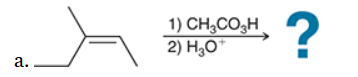
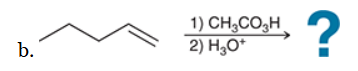
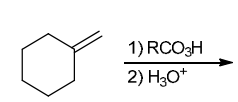
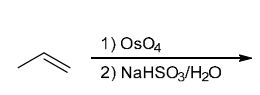
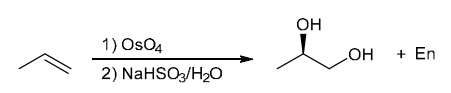
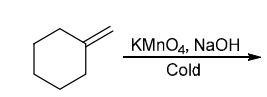
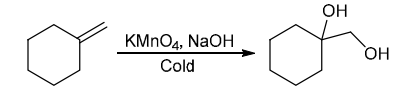
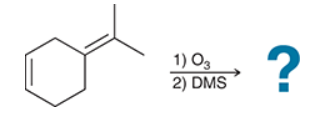
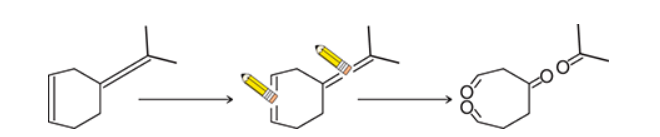
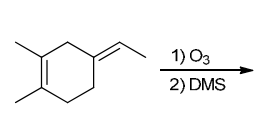
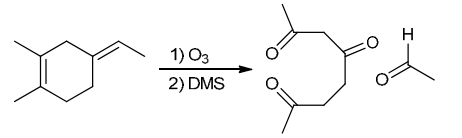
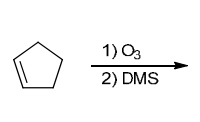
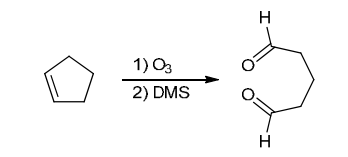
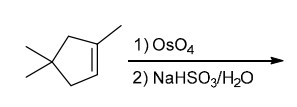
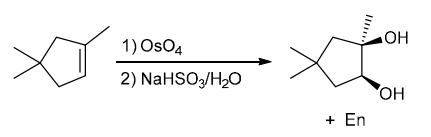
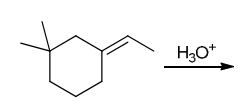
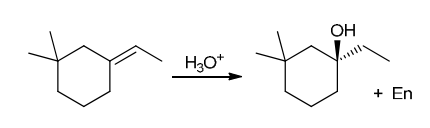
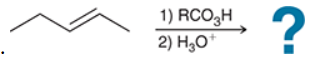
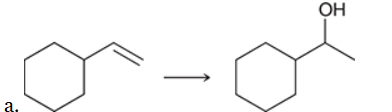
Identify Reagents
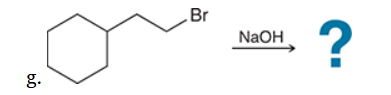
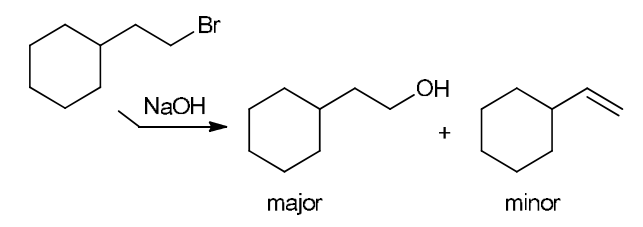
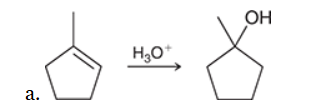
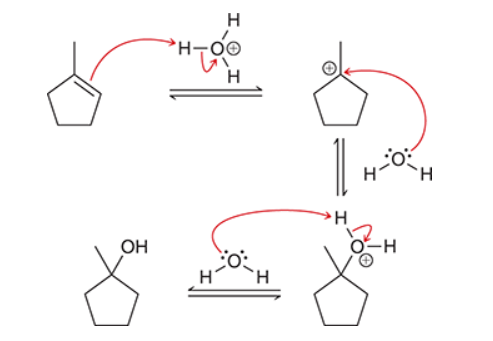
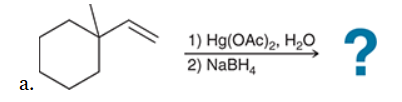
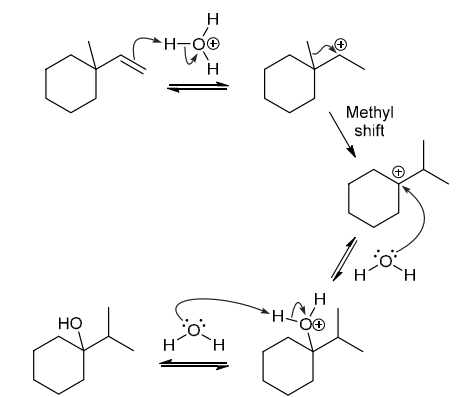
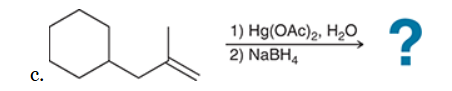
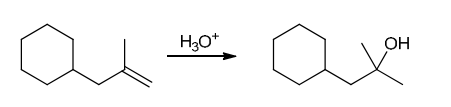
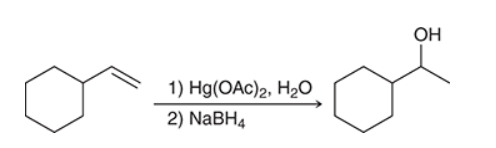
The two groups being added across the alkene are H and OH. The OH group is installed at the more substituted carbon atom, so we must use conditions that give a Markovnikov addition of H and OH. This can be accomplished via oxymercuration-reduction
Identify Reagents
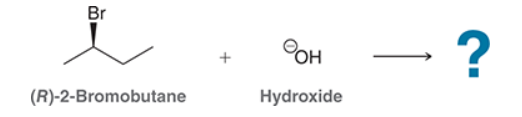
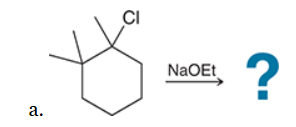
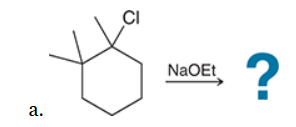
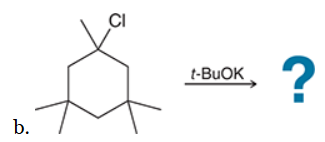
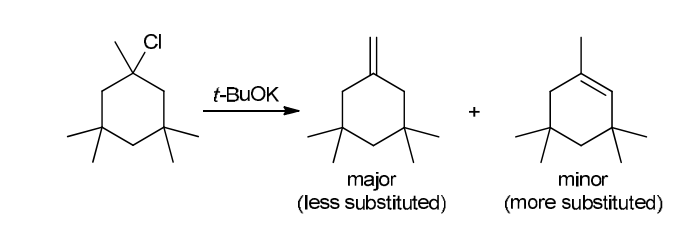
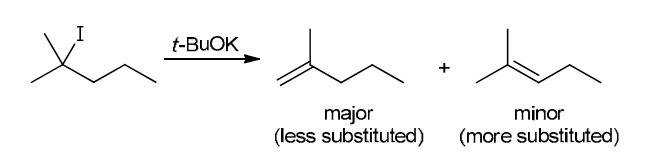
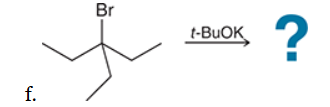
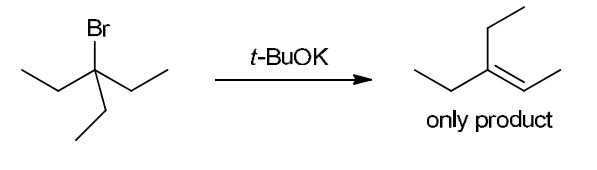
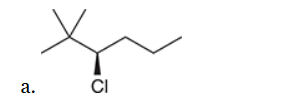
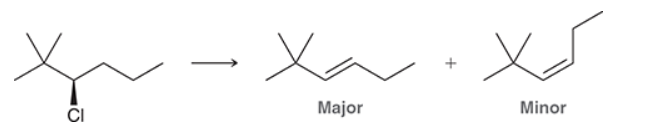
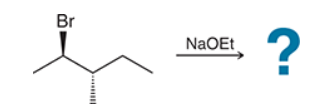
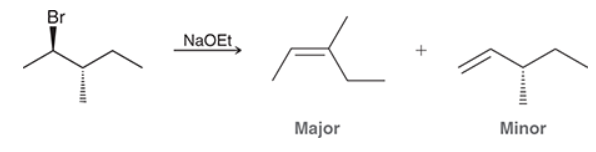
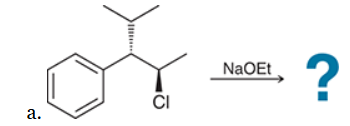
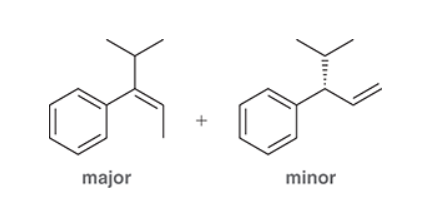
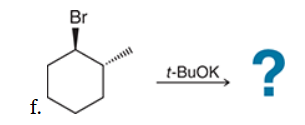
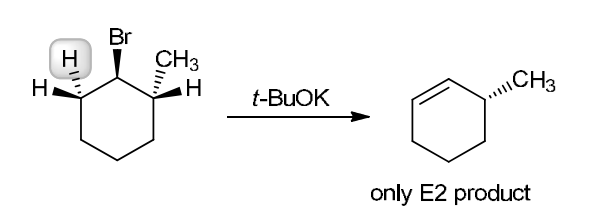
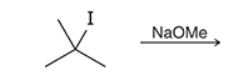
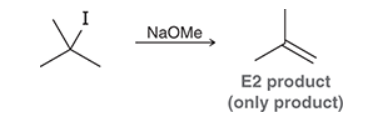
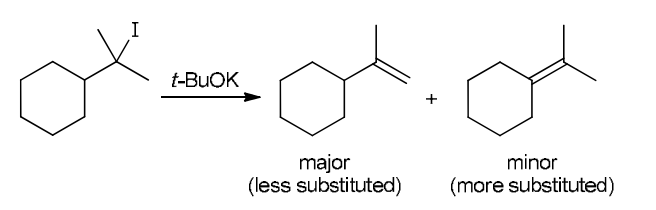
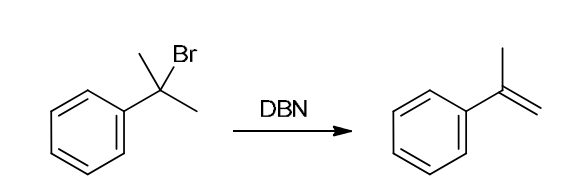
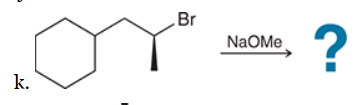
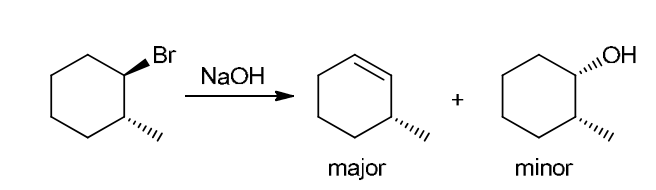
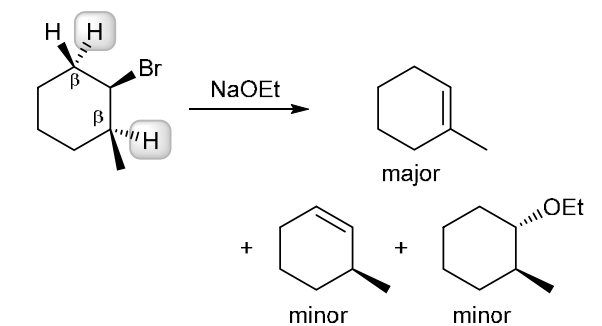
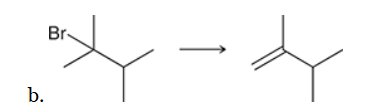
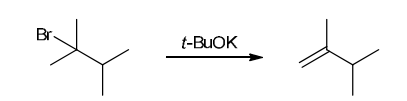
This reaction involves elimination of H and Br to give the less substituted alkene, so a sterically hindered base (such as tert-butoxide) is required
Identify Reagents
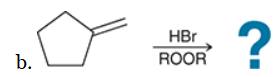
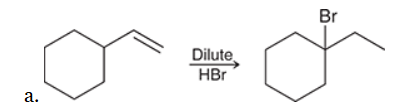
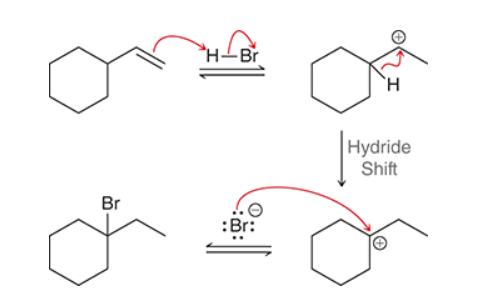
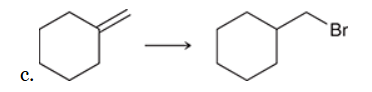
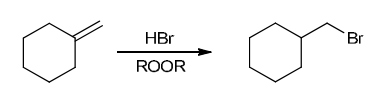
The two groups being added across the alkene are H and Br. The Br group is installed at the less substituted carbon atom, so we must use conditions that give an anti-Markovnikov addition of H and Br. This can be accomplished by treating the alkene with HBr in the presence of peroxides:

Identify Reagents
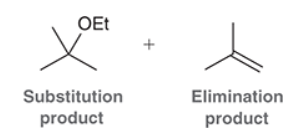
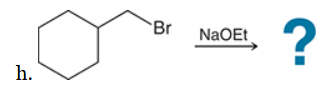
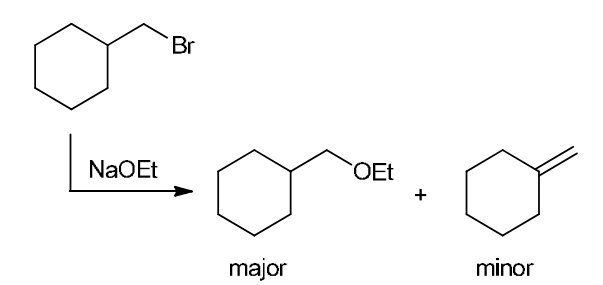
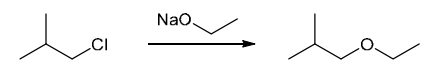
This is a substitution reaction. Our retrosynthesis begins with a disconnection at a C–O bond of the ether group. The carbon atom (at the disconnected bond below) started out as an electrophile in the given starting material, with a Cl leaving group at that position. Therefore, the oxygen atom served as the nucleophile, so we draw a negative charge on the oxygen atom to give a suitable nucleophile (ethoxide ion) The last step of the planning process is to confirm that the reaction mechanism is favorable. The ethoxide anion is a strong nucleophile and a strong base, so we must consider both SN2 and E2 pathways. In this case, we have a primary substrate and a small nucleophile, so there is very little steric hindrance and we expect an SN2 process to give the desired target molecule as the major product
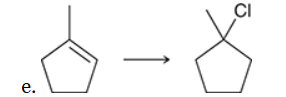
Identify Reagents
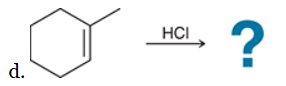
The two groups being added across the alkene are H and Cl. The latter is installed at the more substituted carbon atom, so we must use conditions that give a Markovnikov addition of H and Cl. This can be accomplished by treating the alkene with HCl:
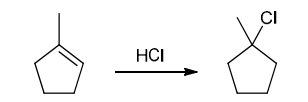
Identifying Reagents
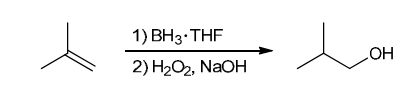
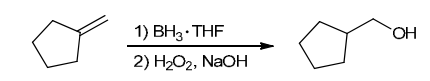
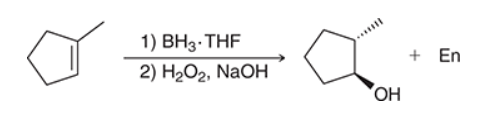
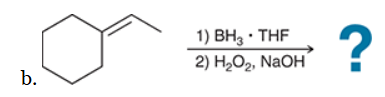
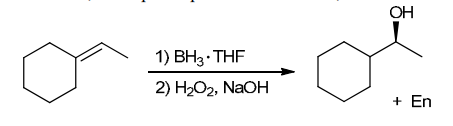
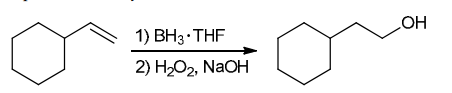
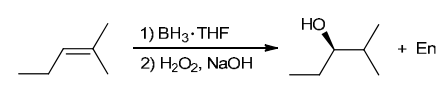
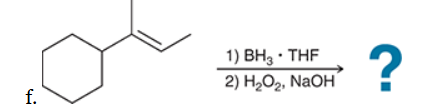
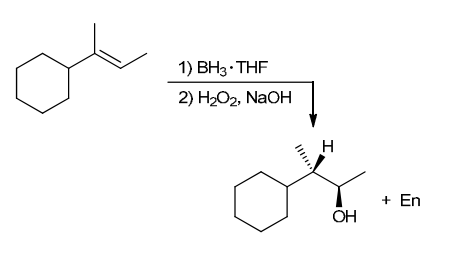
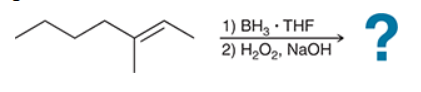
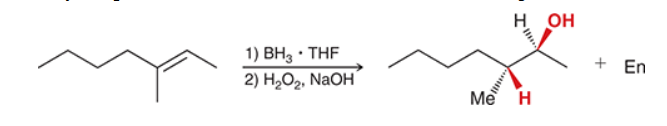
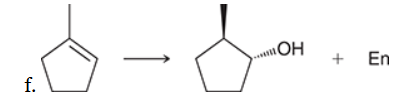
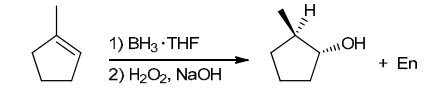
The two groups being added across the alkene are H and OH. The OH group is installed at the less substituted position, so we must use conditions that give an anti-Markovnikov addition of H and OH. Also, the H and OH are added in a syn fashion (this can be seen more clearly if you draw the H that was installed, as shown below). This can be accomplished via hydroboration-oxidation
Identify Reagents
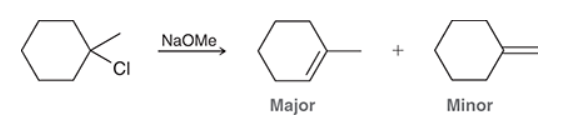
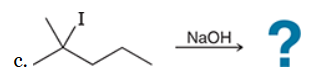
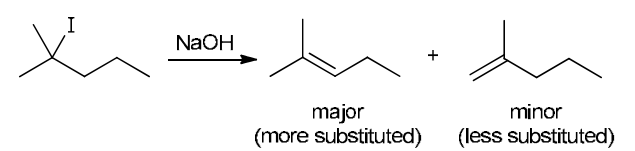
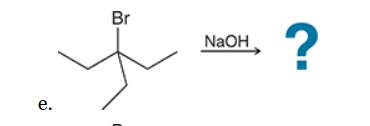
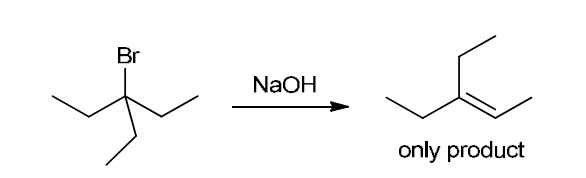
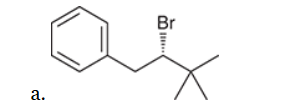
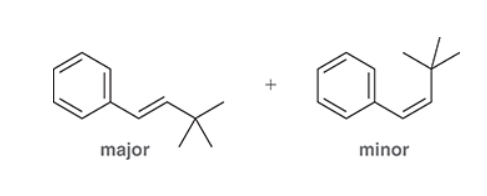
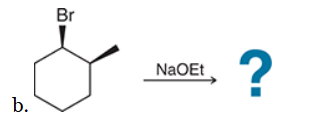
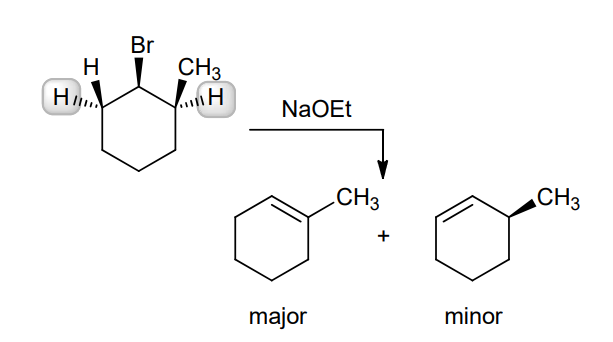
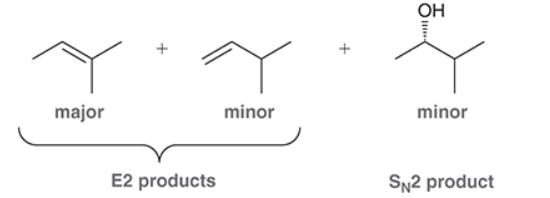
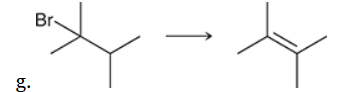
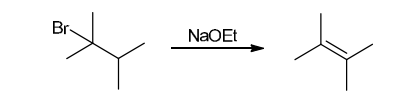
This reaction involves elimination of H and Br to give the more substituted alkene, so we must use a strong base that is not sterically hindered. We can use hydroxide, methoxide or ethoxide as the base. All of these bases are suitable, as the substrate is tertiary so SN2 reactions will not compete:
Identify Reagents
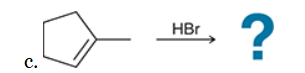
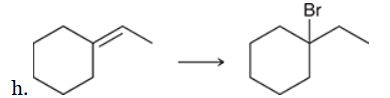
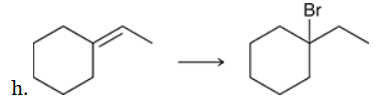
The two groups being added across the alkene are H and Br. The latter is installed at the more substituted carbon atom, so we must use conditions that give a Markovnikov addition of H and Br. This can be accomplished by treating the alkene with HBr
Identify Reagents
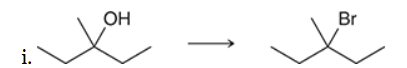
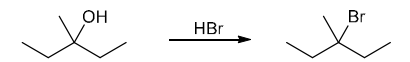
This is a substitution reaction. The substrate is tertiary, so we will need to perform an SN1 reaction. The nucleophile must be bromide, but we cannot simply treat the substrate with bromide, because hydroxide is not a good leaving group. The use of HBr will provide both the nucleophile (bromide) and the proton for converting the bad leaving group to a good leaving group (water)
Identify Reagents
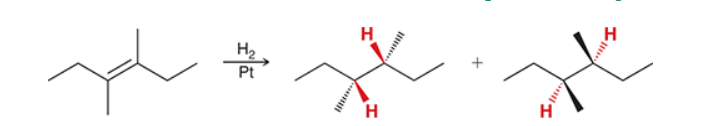
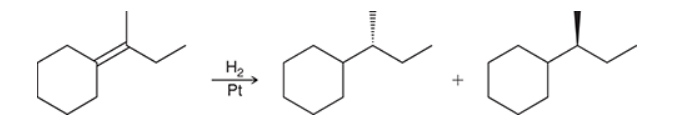
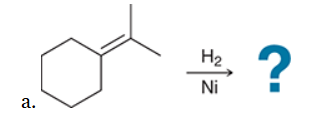
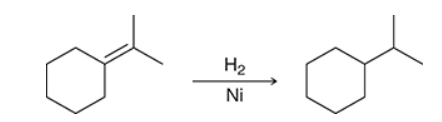
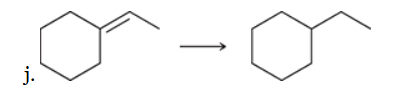
The two groups being added across the alkene are H and H, which can be accomplished by treating the alkene with molecular hydrogen (H2) in the presence of a suitable catalyst
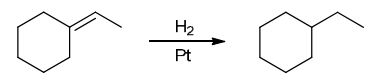
Identify Reagents
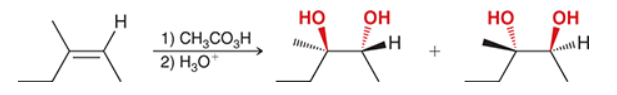
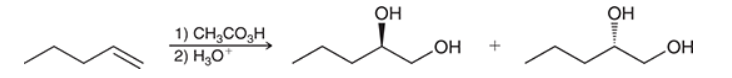
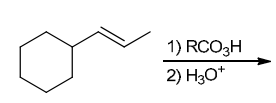
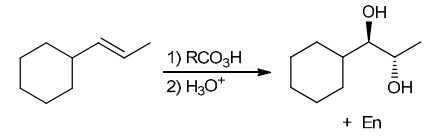
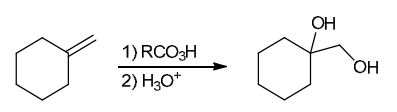
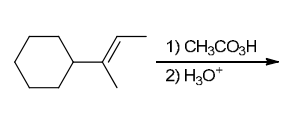
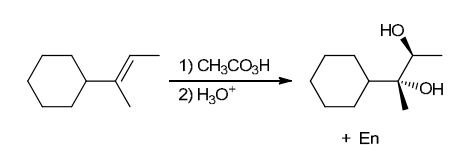
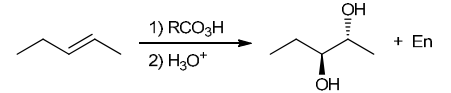
The two groups being added across the alkene are OH and OH, and the alkene has undergone anti addition (one OH group has added to the top face, and the other OH has added to the bottom face). Anti dihydroxylation can be accomplished with a two-step procedure: formation of an epoxide by treatment with a peroxy acid (RCO3H), followed by a ring-opening reaction with aqueous acid
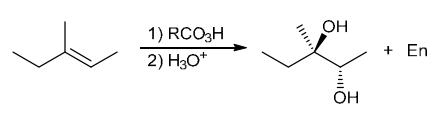

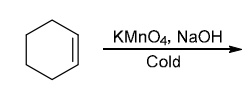
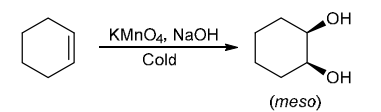
Identify Reagents
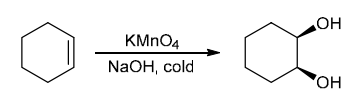
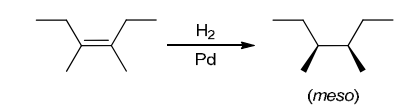
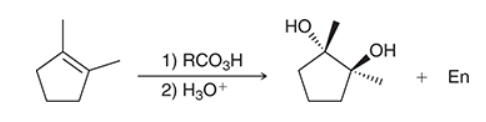
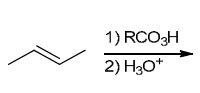
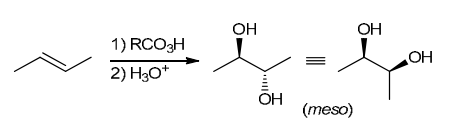
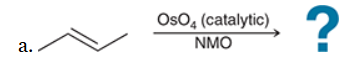
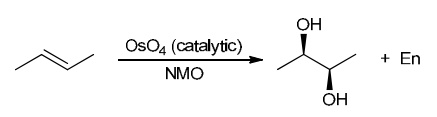
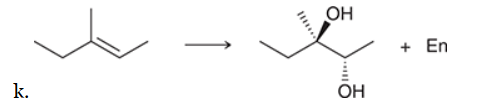
The two groups being added across the alkene are OH and OH, and the alkene has undergone syn addition (both OH groups have been added to the same face, resulting in an achiral meso product). Syn dihydroxylation can be accomplished by treating the alkene with KMnO4, or OsO4