Env Eng CE 3520 Final Exam
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
effects of oxygen demand on river water
(1) removal of oxygen by microorganisms during biodegradation (2) replenishment of oxygen through reaeration at the interface between the river and the atmosphere
What order is the rate of deoxygenation by organic waste?
1st order wrt Lt
Stream aeration and BOD have what type of relationship?
inverse
reaeration
rate at which oxygen is replenished and is assumed to be proportional to the difference between the actual DO in the river at any given location and the saturated values of DO
oxygen deficit
departure of the ambient DO from saturation
Kr = kd at which point of the oxygen sag curve?
critical point
mass flux density
rate of mass transferred across the plane per unit time per unit area; can result from advection, diffusion/dispersion, or a combo of both
advection
movement of a compound along with flowing air or water; depends on concentration and flow velocity
diffusion
net effect of random molecular movement caused by the molecules’ thermal-kinetic energy; causes solutes to move from high to low concentrations
dispersion
(1) turbulent: mass transferred through mixing of turbulent eddies within the fluid (2) mechanical: variations in flow pathways taken by different fluid parcels that originate in nearby locations, dominant process in GW
Why does the contaminant move slower than GW itself?
sorption to soil particles; chemical will travel at a slow rate than Va by a factor of Rf
purpose of water treatment
(1) potable: safe for human consumption, free of harmful elements (2) palatable: aesthetically acceptable (clear, no odor, no bad taste)
turbidity
measures the optical clarity or scattering and absorbance of light caused by suspended particles; WHO <5 NTU, US < 0.1 NTU; GW has <1 NTU
particles
larger than molecules, often not visible, can absorb toxic metals and organics
natural organic matter (NOM)
includes colloids and dissolved organic carbon, passes 0.45 filter
color is caused by
dissolved organic matter, natural metallic ions
apparent color: unfiltered, caused by turbidity
true color: filtered, caused by constituents
pathogens
microorganism that cause illness; includes viruses,bacteria, fungi, amoeba
indicator organism
used to monitor the microbial water quality; example: total coliform
maximum contaminant level goals
based on health and risk assessment info
maximum contaminant level
enforceable, also based on costs and technology availability
sorption
refers to either or both processes of adsorption of a chemical at the solid surface and/or absorption of a chemical into the volume (interior) of the solid
sorbate
substance transferred from the gas or liquid phase to solid
sorbent
solid material onto or into which the sorbate accumulates
sorption isotherm
relationship that describes the affinity of a compound for a solid in water or gas at a constant temperature
adsorbent types
granual activated carbon (GAC), powdered activated carbon (PAC), granular ferric hydroxide (GFH)
granular activated carbon (GAC)
removal of organic and inorganic constituents, SA=500-2500 m2/s
powdered activated carbon (PAC)
removal of seasonal taste and odor, SA = 800-2000 m2/s
granular ferric hydroxide (GFH)
used primarily for removing arsenic and selenium, SA = 250-300 m2/s
empty bed contact time (EBCT)
time required for one bed volume of influent to pass through the empty reactor
mass transfer zone (MTZ)
portion of bed in which the dissolved adsorbate concentration and the adsorption density change dramatically
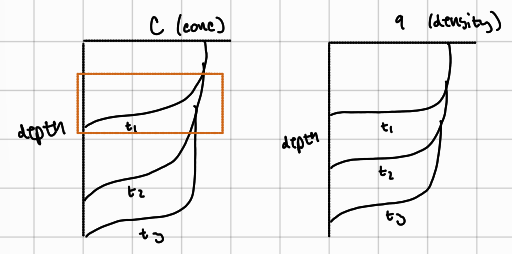
breakthrough curve
eventually the leading edge of the MTZ reaches the outlet and the concentration of adsorbate in the effluent begins to increase significantly, which causes breakthrough
hardness
used to characterize a water that doesn’t lather well and leaves scale, sum of calcium and magnesium; units mg/L as CaCO3
lime
CaO
soda ash
Na2CO3
coagulation
charge neutralization step that involves condition of colloidal matter with coagulants to destabilize particles
flocculation
aggregation of destabilized particles and formation of larger particles (flocs) for easy removal via settling or filtration
particle stability
tendency of suspended particles in water to remain separated and resist aggregation or settling
mechanism for coagulation and flocculation
compression of electrical double layer (EDL), charge neutralization, adsorption and interparticle bridging, precipitation and enmeshment
types of coagulants
inorganic metallic, prehydrolyzed metal salts, organic polymers, natural plant-based materials
Why do we add lime and soda ash?
the natural alkalinity of the water source isn’t sufficient and we need to maintain appropriate pH
granular (sand) filtration
solid-liquid separation process for removal of colloidal and suspended particles; final particle removal process (tertiary)
slow sand filtration
appropriate for rural communities due to simplicity, land availability, and low energy requirement; cycle of filtration and regeneration (top 1-2 cm of sand scraped off and cleaned)
rapid filtration
cycle of filtration (water flows down and particles trapped) and backwash (water flows up to flush out particles from bed)
sedimentation
process in which a majority of particles will settle by gravity in a reasonable time and be removed
discrete particle settling
occurs when particles are discrete and don’t interfere with one another; described by Stokes’ or Newton’s laws
When settling velocity is > OR what % of particles are removed?
100%
Why is water disinfected at almost the last stage?
economical, less formation of disinfection byproducts and disinfectant residuals
which free chlorine component is more effective for disinfection
HOCl (why pH<7 is desired)
lime removes
(H2CO3 or CO2 + Ca CH + 2x Mg CH + Mg NCH + 30 mg/L CaCO3) x 56/100
ash removes
NCH x (106/100)
C x t product
inactivation of microorganisms is controlled by concentration of disinfectant and time of contact; equal to dosage of disinfectant; higher value means more effective
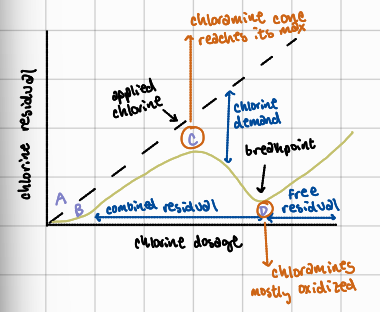
A to B
chlorine reacts first with the reducing agents present and develops no measureable residual
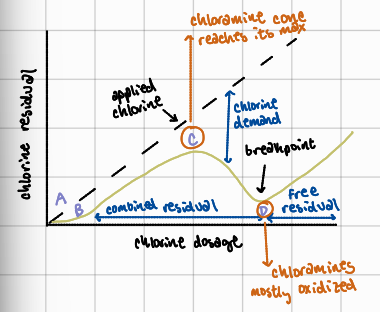
B to C
chlorine continues to be added, reacting with ammonia and forming chloramines; chlorine residual begins to appear, but as combined residual, not free chlorine
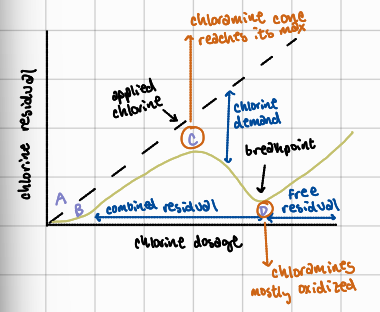
C to D
with further chlorine addition past point C, chlorine begins oxidizing chloramines and other nitrogenous compounds, causing the chloramine residual to drop
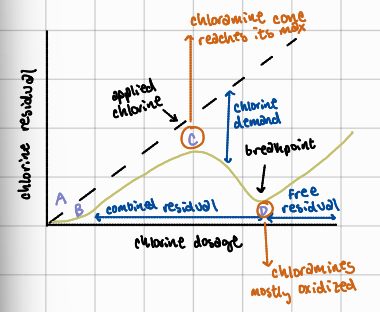
after D
further chlorine addition results in accumulation of a stable free chlorine residual which is most effect for disinfection
What is the importance of the breakpoint chlorination curve?
helps water treatment operators identify optimal chlorine dosage to ensure effective disinfection; ensures chlorine residuals are in the effective “free chlorine” range to reliably eliminate pathogens
Chick’s law assumes the rate of disinfection reaction is what order?
pseudo first order
CH
= min(alkalinity, TH)