EXP 1: Common Laboratory Operations
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
test tube with no measurement markings
What does a “non-calibrated” test tube mean?
19.70 mL
what is the proper measurement in the diagram
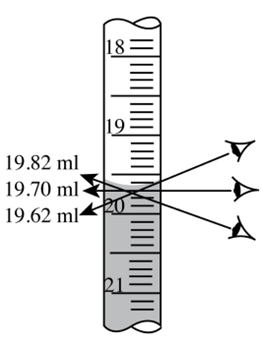
1/10
the portion of the test tube that is 2 mL is approximately __ of the test tube
1/4
the portion of the test tube that is 5 mL is approximately __ of the test tube
1/2
the portion of the test tube that is 10 mL is approximately __ of the test tube
glass rod
what piece of equipment can be used to transfer liquids from one vessel to another
to avoid spills of the liquid, especially if it is corrosive and dangerous
why should a glass rod be used to transfer liquids from one vessel to another
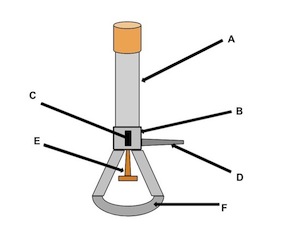
barrel
what is the name of (A)
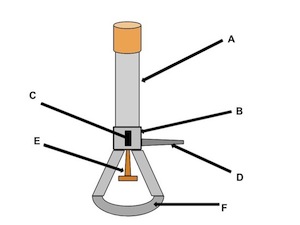
collar
what is the name of (B)
air hole
what is the name of (C)
gas adjustment knob
what is the name of (D)
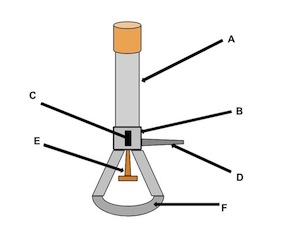
gas valve
what is the name of (E)
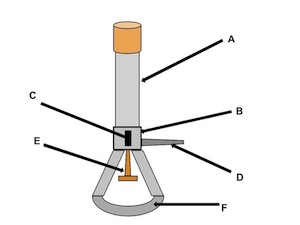
base
what is the name of (F)
(1) slightly open gas adjustment knob (2) close air holes (3) turn on lighter above the barrel
what 3 things should be done to the bunsen burner before turning on the gas
orange/yellow flame
what is the luminous flame of the bunsen burner
blue flame
what is the non-luminous flame of the bunsen burner
open air holes
how to turn luminous flame into non-luminous flame
45 degrees
what angle should a test tube be held when it is being heated above a bunsen burner
upper portion
what portion of the liquid should be heated first
to avoid the liquid from shooting out as bubbles from the test tube
why should the upper portion of a liquid be heated first before the bottom?
FeCl3
ferric chloride
NaOH
sodium hydroxide
Reddish Brownish Ferric Hydroxide (Fe(OH)3)
what is the color, name, formula of the precipitate formed between FeCl3 and NaOH
FeCl3 (aq) + 3 NaOH (aq) —> Fe(OH)3 (s) + 3 NaCl (aq)
what is the reaction between ferric chloride and sodium hydroxide
filtrate
what is the liquid that passes through the filter paper called
filtration, decantation, evaporation
three methods of separation in this experiment
sodium chloride (NaCl)
what was the filtrate
filtration
method of separating a liquid from a solid using a filter medium
supernatant liquid
liquid that is free from the precipitate during decantation
decantation
method of separating a liquid from a solid
evaporation
what method was used to prove the filtrate was NaCl
liquid evaporated, leaving white-powdery residue
what occurred after the NaCl was heated on the bunsen burner
residue
the solid resulting from evaporation
evaporating dish, wire gauze, clay flame shield
other than the bunsen burner, what equipments were used during evaporation
solid sodium chloride (NaCl)
what is the state, name, and chemical formula of the substance that remained after the NaCl was heated?