TM - substitution of ligands
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
what are the three monodentate ligands
Cl-, NH3 and H2O
which two are similar in size
NH3 and H2O
what happens when these ligands are exchanged
there is no change in co-ordination number
substitution may be incomplete
Co2+ and Cu2+ example
[Cu(H2O)6]2+ + 4NH3 → [Cu(NH3)4(H2O)2]2+ + 4H2O
Co2+ initially undergoes the normal deprotonation reaction with 2NH3
with four NH3 = all of ligands substituted as NH3 is better ligand
pale yellow solution formed
[Co(H2O)6]2+ + 6NH3 → [Co(NH3)6]2+ + 2OH- + 4H2O
is Cl- larger or smaller ligand
larger and charged
exchange of ligand for Cl- does?
change co-ordination number and charge of overall complex potentially
reactions
[CuCl4]2- formed, can also be done with Fe3+ or Co2+
which ligands are bidentate
H2NCH2CH2NH2 and (C2O4)2-
what ligand is multidentate
EDTA4-
why might there be no enthalpy change in substitution reaction
ligand-metal bonds have similar enthalpy to previous ligand-metal bond
same number of bonds broken and made
what is the haem complex
iron (II) complex w multidentate ligand
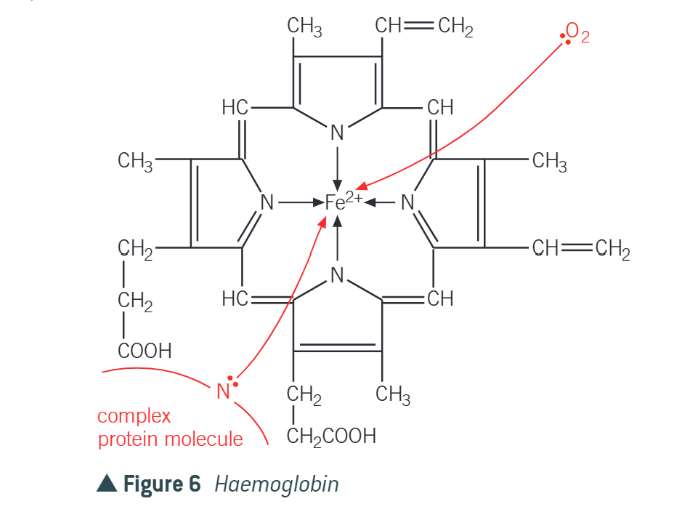
what does the haemoglobin do
oxygen forms co-ordinate bond with Fe(II) in the haemoglobin, enables oxygen to be transported around
what is the problem with CO2
carbon dioxide is a better ligand - binds more strongly to haemoglobin than oxygen does so is toxic
what is the chelate effect
the tendency of bidentate/multidentate ligands to replace monodentate ligands in complexes
why does this occur
enthalpy usually negligible as same number of same type of similar bonds being broken and formed
entropy increases/entropy change is positive as disorder increases due to more particles being formed than reactants
free-energy change is negative so reaction is more feasible