Immuno Week 4
1/157
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
158 Terms
what are the 2 forms of BCR
membrane bound
soluble
what is the structure of membrane bound BCR
transmembrane glycoproteins on B-cells
how many binding domains per molecule do BCRs have
2
how many binding domains per molecule do TCRs have
1
To signal the nucleus, each BCR heavy chain needs to be paired with what
Ig-α (=CD79a) and Ig-β (=CD79b)
what does Mature B-cell activation lead to
expansion
BCR will have the same Ag specificity as the parental B-cell
what are soluble BCRs
secreted antibodies
where are these antibodies secreted from
plasma cells and plasmablasts
what do B-cell clones secrete
antibodies of 1 isotype/class and 1 specificity
what decides isotype/class
AA sequence of heavy chain (Fc region)
what are the different isotypes/classes of BCRs
IgM, IgA, IgD, IgE and IgG
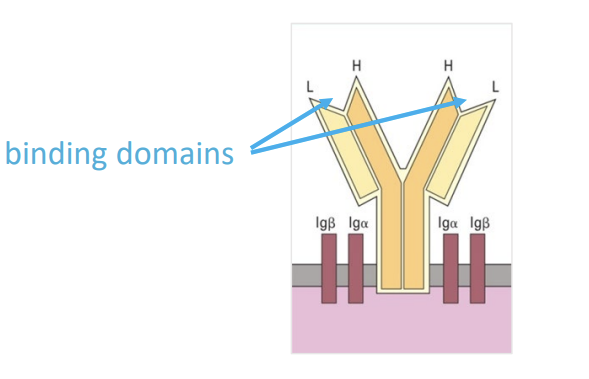
what is this
membrane bound BCR
what is this
soluble/secreted BCR
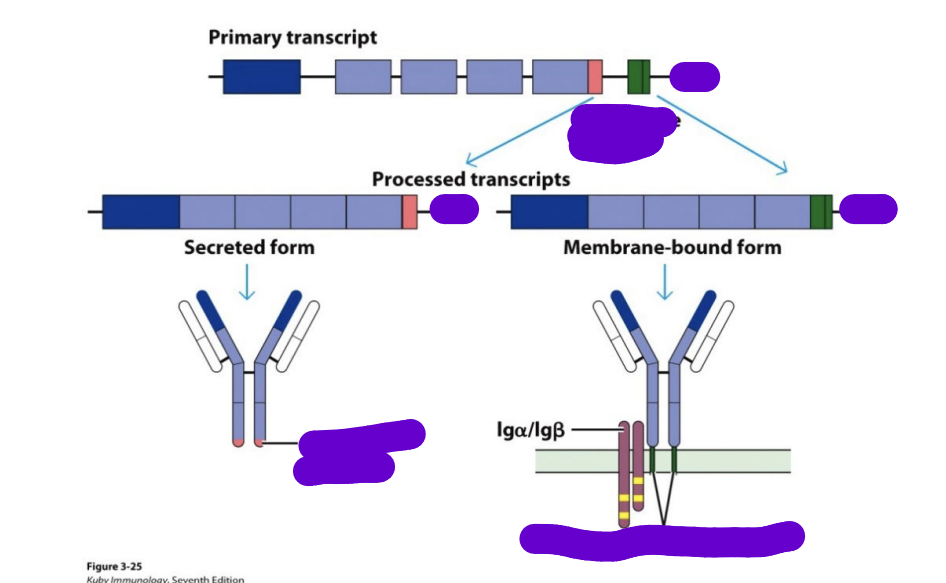
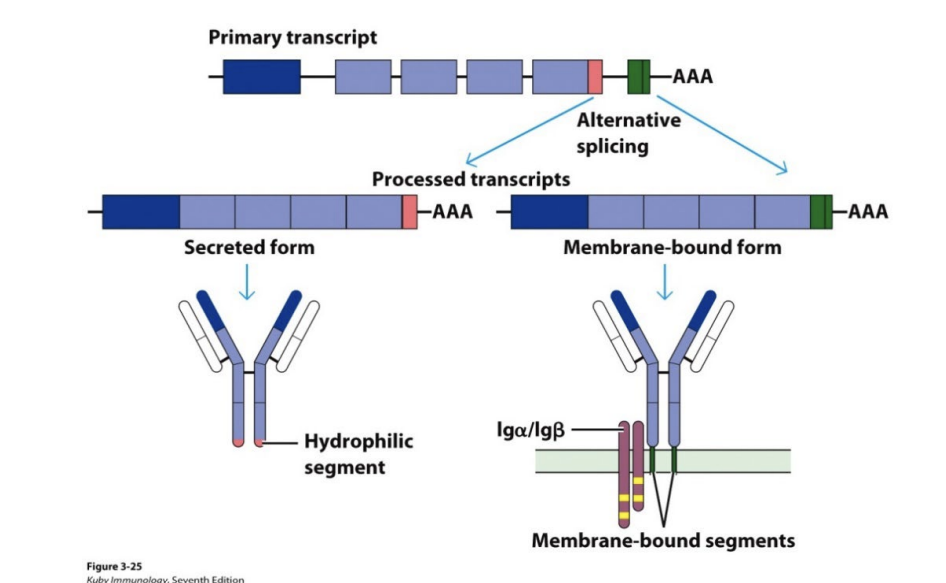
what determines if Ab is membrane bound or secreted
Alternative mRNA splicing
what do soluble Abs have
hydrophilic segment
what do Membrane-bound Abs have
hydrophobic transmembrane segment and AAA cytoplasmic tail
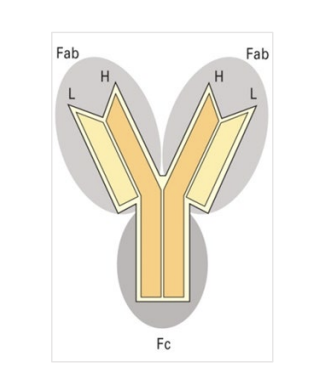
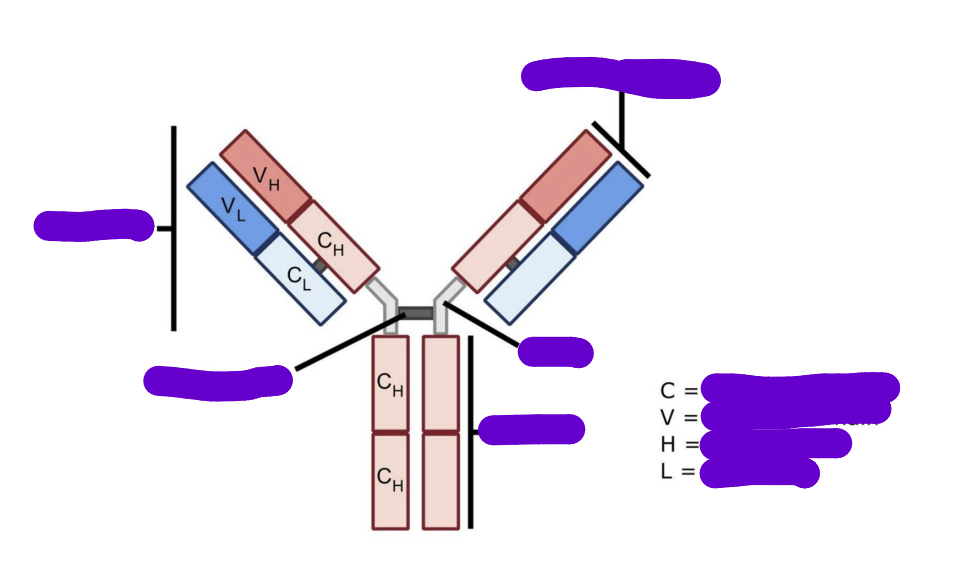
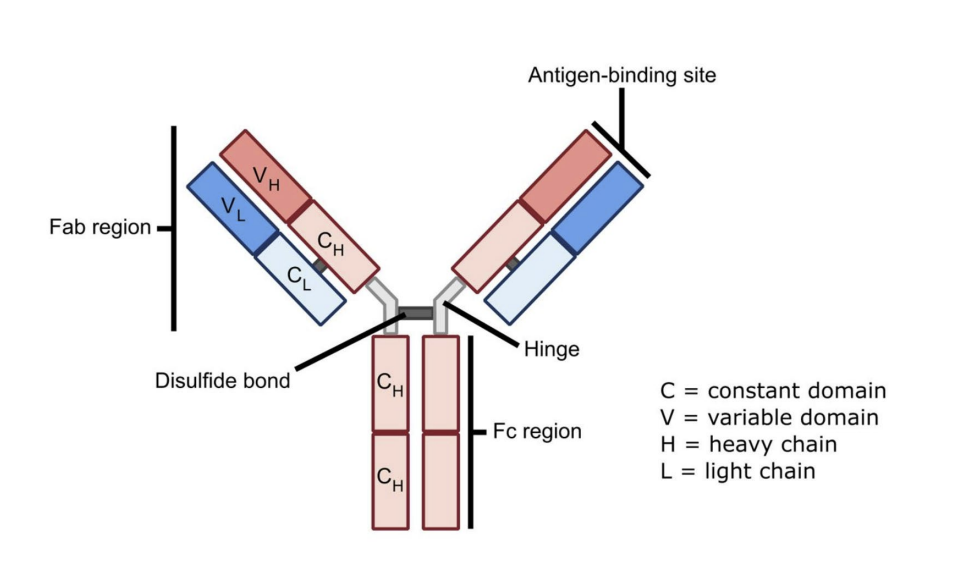
what is the structure of an antibody
Fab region
Fc region
Hinge region
Heavy-chain dimerisation through disulfide bonds
what does the Fab region contain
containing the variable region with antigen binding site
what does the Fc region contain
constant domains
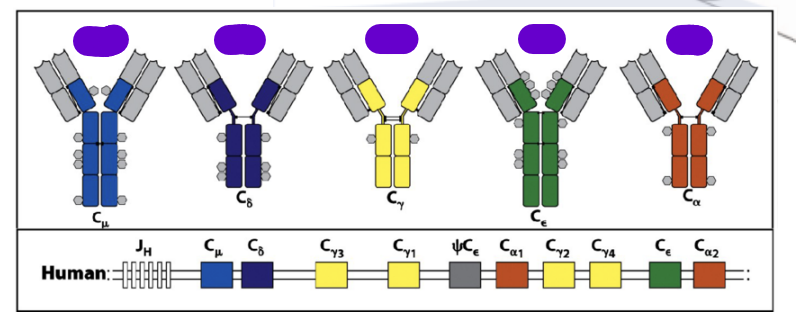
what determines the antibody isotype (Ig: M, D, G, A, E)
the genes encoding the constant heavy chain region
what isotype do all B cells produce initially
IgM
then B-cells can switch from IgM to another class
what does different gene splicing lead to
different constant heavy chains
what is the function of IgG (subclasses: IgG1, IgG2, IgG3, IgG4)
Secreted during secondary response
Major form of circulating antibodies
what is the function of IgA (subclasses: IgA1, IgA2)
Major form of circulating antibodies in external secretions
what is the function of IgE
Triggers immediate allergic reactions
what is the function of IgM
Secreted during primary response
what is the function of IgD
Exact function unknown
does the fetus produce Abs
no
what is the exception to this
Late pregnancy B1 B-cells of fetus produce low affinity IgM
how does the fetus receive protection
Passive IgG from mother crosses placenta, providing some protection from infection
how else is maternal IgG transferred to the offspring
through colostrum & breast milk
what does IgM do
Activates classical complement pathway
Opsonisation of pathogens and phagocytosis
what is IgM associated with
immune response to antigenically complex blood-borne infections
10 binding sites per molecule (2 on each, pentamer)
what % of normal human serum Immunoglobulins does serum IgM make up
10%
what is the major site of IgM production
Plasmablasts in spleen
where is IgD found
Transmembrane Ag receptor Found on mature B-cells
what is the half life of IgD
Very-low concentration in plasma, half-life is 2-3 days
what does IgD do
Research suggests involvement in immune tolerance and mucosal defence against pathogens
what is the main Ig isotype in blood
IgG (3-4 week half life)
what does IgG do
Opsonizes pathogens
activates classical complement
induces phagocytosis
what are the 4 subclasses of IgG
IgG1, IgG2, IgG3, IgG4
how do IgG subclasses vary
based on the hinge region that joins the constant regions of the heavy chain
when is IgA produced
during a secondary response to Ag
where is IgA present
Predominates in mucosal surfaces
where can IgA opsonise Ags
in the respiratory, GI & reproductive tracts
what form do IgAs exist
in monomer form and dimer form
what is IgA bound to
poly Ig receptor on basolateral side of epithelial cells
when is IgA Transported to apical side
for secretion on mucosal surfaces
what is IgE usually bound to
Fc receptors of mast cells or basophils
what does IgE do
Provide immunity to parasites e.g. helminths, venom
Also evoke responses to allergens – mast cell degranulation
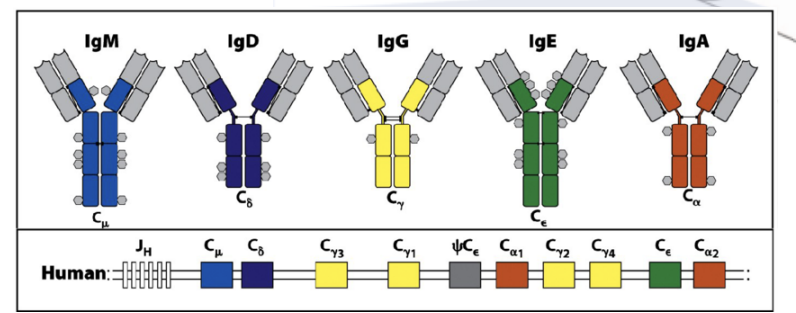
what do All naïve B-cells express
plasma membrane-bound IgM and IgD (= BCR)
when are Mature B-cells activated
upon encounter with a specific antigen
what happens after activation
they begin to proliferate and produce secretory IgM and IgD
how does isotype switching occur
Further activation by Ag or other stimuli (e.g. CD40L on T helper cells, cytokines) can rearrange genes encoding the constant region of the heavy chain
what do CD4 T-cells do
stimulate progeny of B-cells expressing IgM and IgD to produce Abs of different heavy chain isotypes/classes through CD40L-mediated signals and cytokines
does the new isotype bind to a different Ag
no, it will have the same binding specificity for the Ag
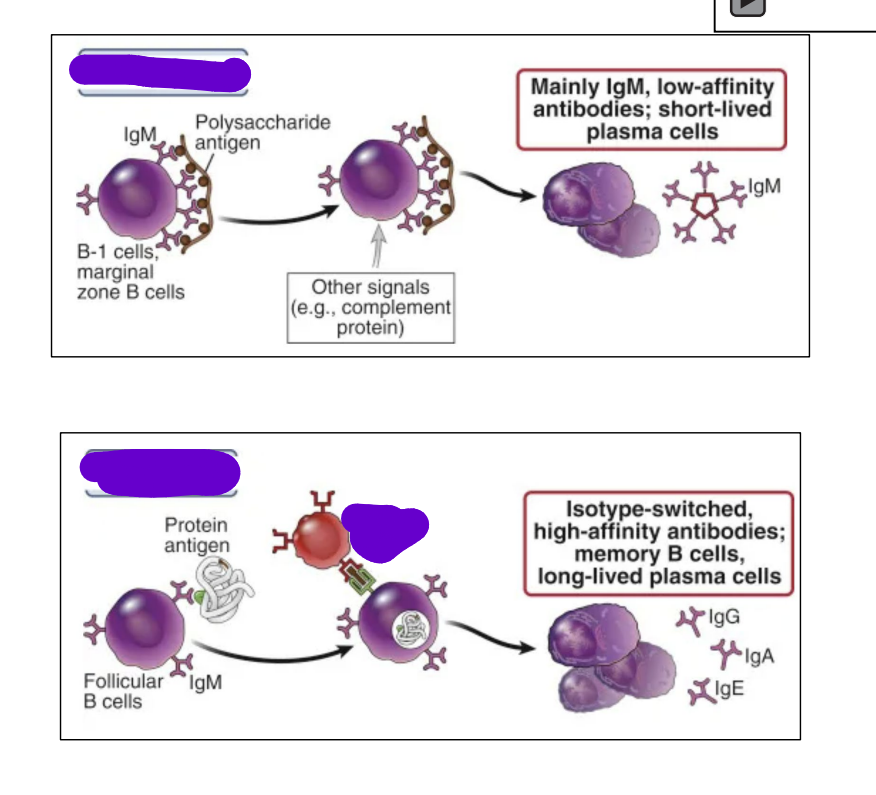
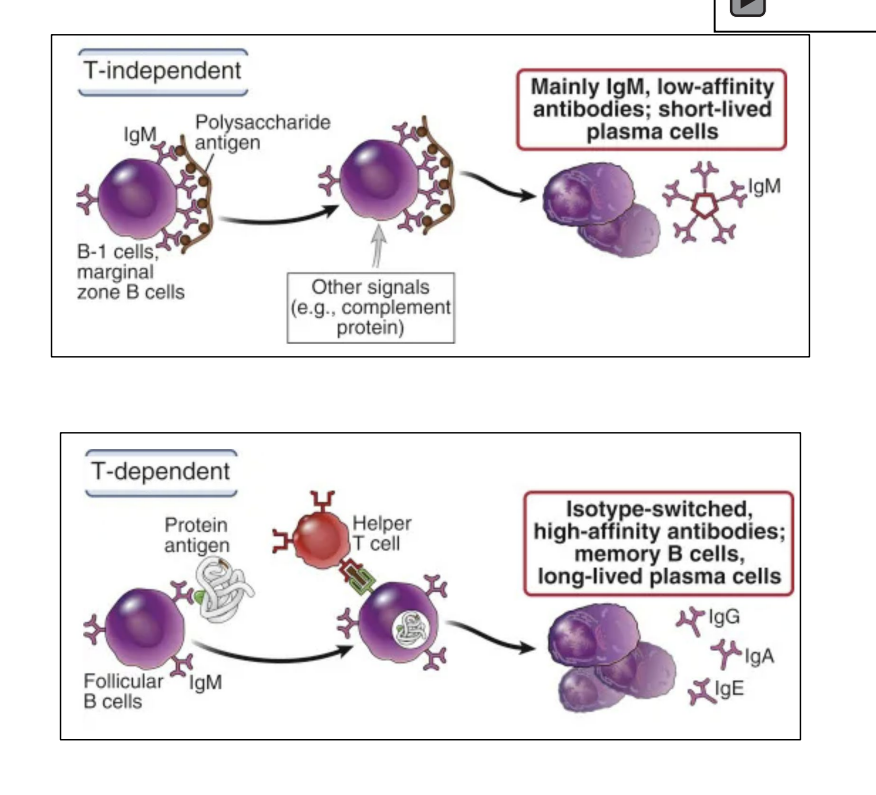
what happens in the T independent response
B cell directly activated by antigen
Production of IgM
No Helper T-cells needed
what happens in the T dependent response
Require T-cell contact to switch isotype
Antigens stimulate generation of long-lived plasma cells and memory B-cells
what is affinity maturation
During a B-cell response to an infection, Abs improve affinity to Ags
what does Activation induced cytidine deaminase (AID) do
induces point mutations which drives Somatic hypermutation
what happens to Affinity of antibodies specific for microbial proteins over time
it increases
what is the primary response
Response on first exposure to antigen
Smaller amount of antibody produced in primary immune response
what is the secondary response
Subsequent exposures to same pathogen
Increased heavy-chain isotype switching and affinity maturation
can all Abs activate complement
no
what will Ab subclasses interact with
only with their specific Fc receptor
what is an example of this
FCRI receptor expressed only on basophils and mast cells
hence IgE can only bind specifically to these cells and activate them
what happens when micrbones produce virulence factors that can bind specifically to some Ab isotypes
they can evade complement activation and opsonization through specific isotypes/subclasses
what are the 4 main antibody functions
Neutralizing antibodies
Activation/Inhibition of Leukocytes through Fc region of receptors
Opsonizing Pathogens
Complement (C’) Activation
how do neutralising antibodies work
Soluble antibodies can bind and block pathogenic proteins or toxins
what is the result of neutralising antibodies
pathogens then cannot bind or kill cells anymore
what is an example of neutralising Abs
Ab against spike protein of COVID – virus cannot bind to epithelial cells anymore
what are most neutralising Abs
IgG in the blood, on mucosal surfaces IgA
how does Activation/Inhibition of Leukocytes through Fc region of receptors work
Specific receptors on white blood cells can bind Fc part of antibodies
how have some bacteria have developed evasion strategies (virulence factors) to avoid the immune system
by producing their own Fc binding receptors that can bind and capture Ab
how does Opsonizing Pathogens work
Bacteria coated with antibodies to signal phagocytosis
Helps eliminate infected and tumour cells
what does the Ability of cross-linking of Fc receptor do
creates additional stimulation of phagocytosis
how does complement (C’) Activation work
Antibody binds pathogen and triggers classical C’
phagocytosis, target cell lysis
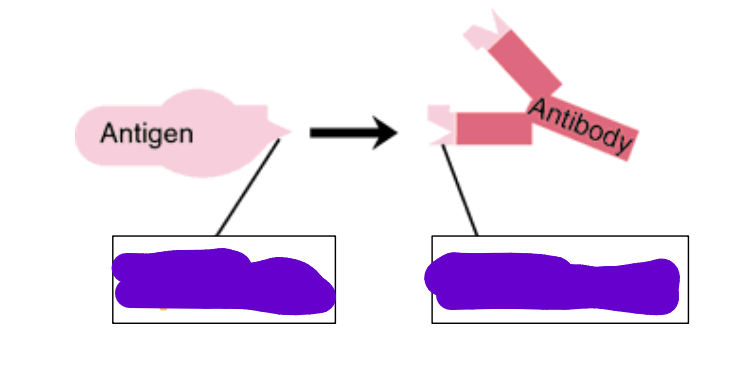
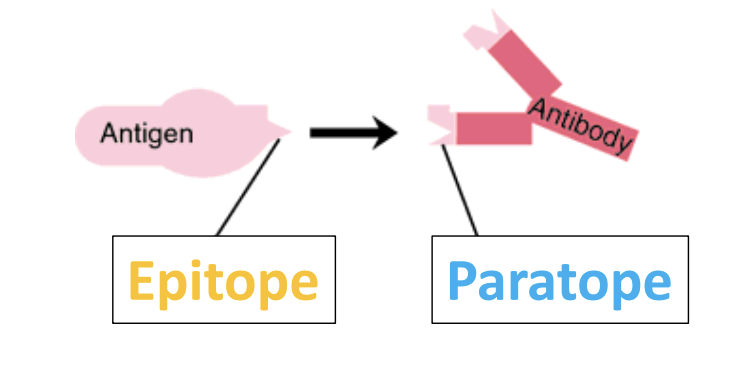
what is an epitope
Part of antigen, the peptide sequence that binds specifically to ABS (antigen binding site)
what is a paratope
Part of antibody, the sequence of the receptor that interacts with the Ag in the ABS
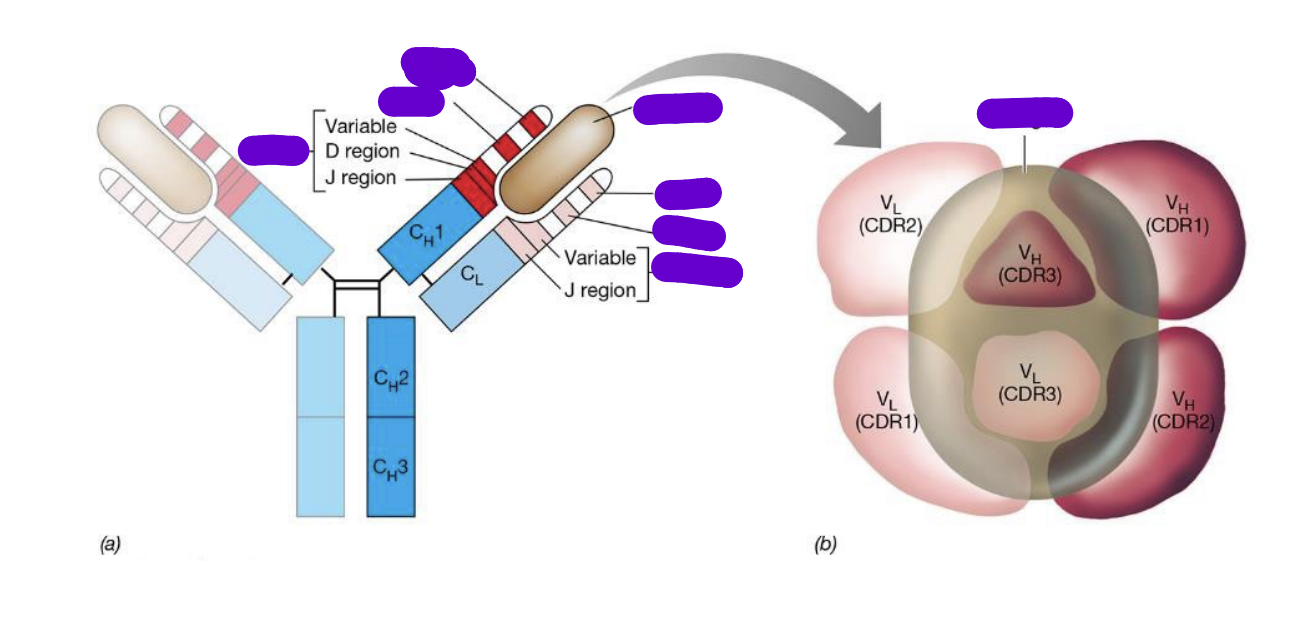
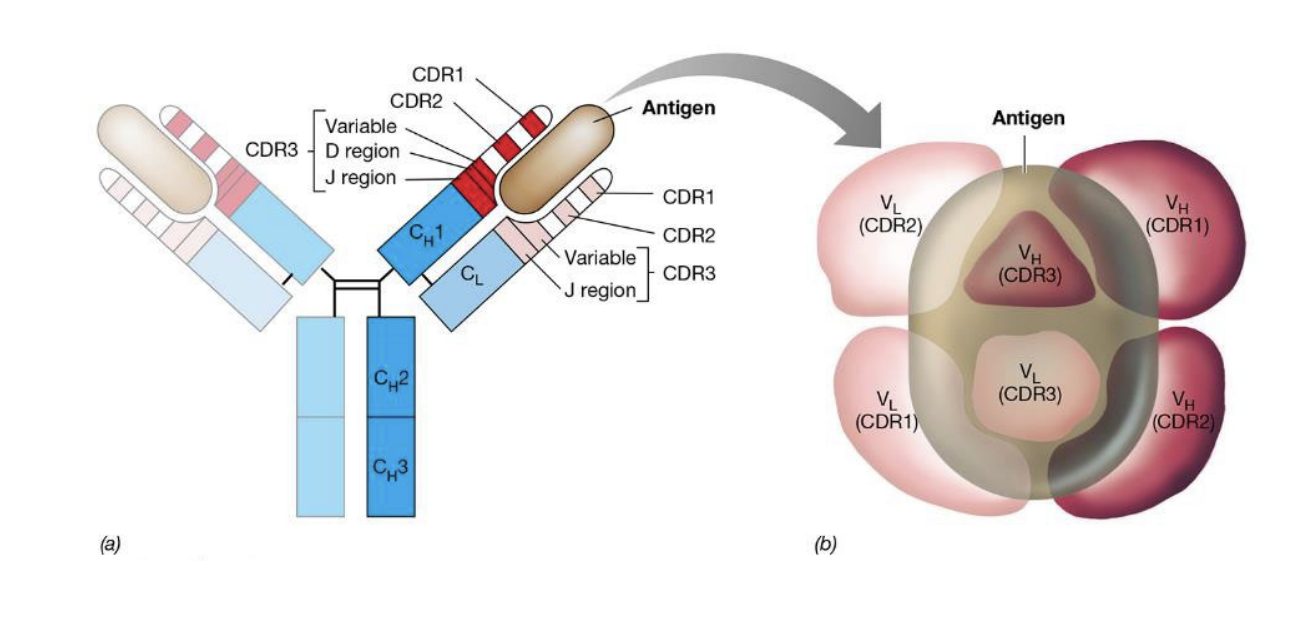
where are the 3 hypervariable regions (HV1, HV2 & HV3) located
within the variable region of both the heavy and light chain
what are HVs aka
Complementarity Determining Regions
e. HV1 = CDR1, …CDR2, …CDR3
what are HVs Critical for
providing antigen specificity for the BCR
what does CDR3 do
provides the most critical contact points between paratope and epitope
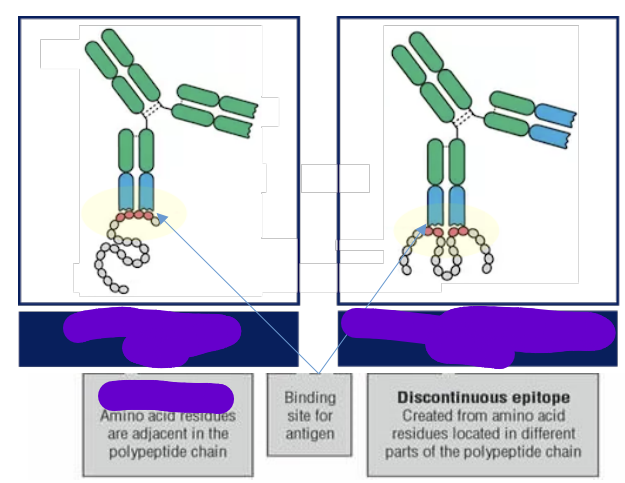
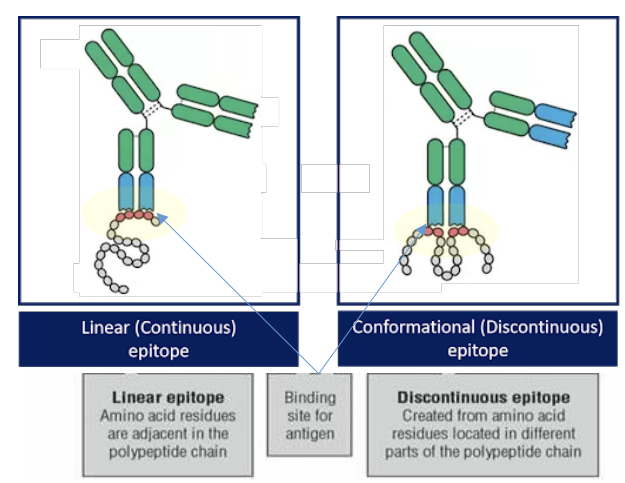
what are the 2 types of epitopes
linear (continuous) epitopes
conformational (discontinuous) epitopes
what is the structure of linear epitopes
All amino acids of the epitope binding to the paratope are next to each other
what is the structure of linear epitopes
Amino acids located in different parts of the amino acid chain come in proximity to form an antigen
why are AAs located in different parts in linear epitopes
depends on the folding of the native protein which may come from 2 or more stretches of aa sequence
what is affinity
Measures strength of interaction between epitope and antigen binding site
what is avidity
Measures overall strength of an antibody-antigen complex
what 3 major parameters is avidity dependent on
Binding affinity of Ab for the epitope
Valency of both the Ab and Ag
Structural arrangement of proteins in the complex
what is valence
The number of binding sites on an Ab or Ag
what valence is IgG
bivalent (valence = 2) because they are monomers with 2 paratopes
what valence is IgM
decavalent (valence = 10) because they are pentamers with 2 paratopes each