Lecture 6 + 7
1/150
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
151 Terms
Primordial soup
Fundamental Ingredients: Water, heat, organic molecules, time
>200 million years of stuff sloshing around before first cell
Organic Molecules
make up 98% the dry weight of cells
Macronutrients
carbon based life forms
C is the most abundant element along with H, O, N, S, P
Micronutrients
in lower amounts: Na, K, Cl, Mg, Fe, Ca, etc
Carbin is an important building block
Stable bonds between carbons and stable bonds to many other atoms
Shape of organic molecules
3-D shape
Diverse functional groups
give organic molecules their distinct chemical properties
polarity of organic molecules
determines whether they are hydrophilic or hydrophobic
Hydrophobic
decrease solubility
regions with many C-C and C-H bonds dont form H-bonds with water
Hydrophilic
they increase solubility
most bonds involving non-C atoms can participated in H-Bonds
Four fundamental classes of macromolecules
carbohydrates
lipids
proteins
nucleic acids
what is often polymers?
bonds between carbon atoms form the backbone of some large biomolecules
carbohydrates
mostly C, H, O most or all carbon atoms are bound to oxygen (Typically: C2H2nOn)
N,P,S can also occur in functional groups
Large number of O and OH groups make sugars highly polar and soluble
Monosaccharide
a single sugar
can interconvert between linear and ring structures
isomers
same atoms in a different arrangement. The ring form is more stable and more commons
Ribose
component of RNA
core metabolic pathways
structured to utilize glucose as input
Grouping of monosaccharides
multiple monosaccharides can form disaccharides and polysaccharides through glycosidic bonds
Dehydration synthesis reaction
Two OH groups react, and one H2O molecule is lost
What are both formed from glucose?
Maltose and lactose but with different linkage
polysaccharides
longer carbohydrate polymers
can be linear, branched, or crosslinked
What is the most abundant biomolecule on earth
carbohydrates mainly due to cellulose
Lipids are composed of primarily?
C and H
possesses long hydrocarbon chains
Lipids are hydrophobic
avoid water and tend to cluster together
lipid single bond
saturated
lipid double bonds
unsaturated
lipid head group
polar
fatty acid head group
simple carboxylic acid headgroup and single hydrocarbon chain
Triacylglycerides
have a core glycerol molecule attached to three fatty acid chains
lipids are
important energy storage molecules
phospholipids
-are important components of biological membranes
-amphipathic
amphipathic
both polar and hydrophobic groups
this allows lipids to self-assemble into the lipid bilayer
phospholipid structure
A core glycerol is attached to two fatty acid chains, and to a modified phosphate group.
When bacteria is cultured?
fatty acids are extracted and analyzed in a gas chromatograph. Different fatty acids give distinct peaks in the resulting data which allows this to serve as a fingerprint for specific bacterial species
proteins
polymers of amino acids
amino acids contain
amine group
carboxylic acid group
central carbon with a side chain
(20 standard)
amino acids structure wise
has a different side chain with different chemical properties
How are amino acids linked?
linked by peptide bonds between amine and carboxylic acid groups
polypeptide
polymer of amino acids
backbone is flexible
primary structure
amino acid structure
polypeptides folding
unique 3d structure. The structure gives protein its structural and chemical properties.
Folding drive
to bury hydrophobic side chains from bulk water
what stabilizes the 3D protein structure?
electrostatic interactions between charged side chains, hydrophobic interactions, hydrogen bonds, covalent disulfide bonds between cysteine side chains
protein secondary structure
local folding of the backbone, stabilized by hydrogen bonds among backbone atoms
protein tertiary structure
three dimensional structure of a single polypeptide chain
proteins quaternary structure
interactions between multiple folded protein chains
-oligomeric state is often linked to function
proteins can be chemically modified
this influences function ((phosphorylation, glycosylation, etc)
protein function
determined by structure and primary sequence
Changes to the primary sequence can disrupt folding or function
nucleic acid
nitrogenous base, phosphate (negatively charged), deoxyribose
nucleotides are linked by
phosphodiester bonds between 3’ carbon and 5’ carbon
how do bases interact in nucleic acids
hydrogen bonding patterns forming base pairs
adenine: thymine
guanine: cytosine
this allows strands to hybridize, they run antiparallel directions
Metabolism
encompasses all the chemical reactions inside a cell
exergonic (energy)
reactions are spontaneous and release energy
endergonic (energy)
reactions consume energy
catabolism (biological molecules)
reactions that break down complex molecules into smaller components, releasing energy
anabolism (biological molecules)
reactions that build complex molecules from simple components, req energy input
autotrophs
convert inorganic CO2 into organic carbon molecules
heterotrophs
use existing organic molecules as nutrients (originally created by autotrophs)
phototrophs
energy captured by light
chemotrophs
energy from breaking chemical bonds
organotrophs (chemical)
use organic compounds
lithotrophs (chemical)
use inorganic sources
chemoautotrophs
chemical reactions provide energy, and inorganic carbon serves as the carbon source
chemoheterotrophs (most organism/bacteria)
break down organic molecules for energy and as a carbon source
photoautotrophs
energy from light, and uses CO2 as a carbon source (cyanobacteria)
photoheterotroph
get energy from light, but cannot use CO2 as a carbon source. They must consume organic compounds (purple nonsulfur bacteria)
energy is stored
in high energy electrons
redox reactions
transfer electrons allow the cell to harness this energy
redox: oxidation reactions
remove electrons from donor molecules, leaving them oxidized
redox: reduction reactions
adds electrons to acceptor molecules, leaving them reduced
When cells break chemical bonds in organic molecules
they capture energy either by extracting a high energy electron or storing energy in the bonds of ATP
Electron carriers
bind high energy electrons and shuttle them around the cell
NAD+/NADH
nicotinamide adenine dinucleotide
NADP+/NADPH
nicotine adenine dinucleotide phosphate
FAD/FADH2
flavin adenine dinucleotide
NADH reduced form
carries a high energy electron
NAD+
can capture an electron from reduced substrates, NAD+ reducing to NADH and oxidizing the substrate
Other ways cells store energy
within high energy bonds of phosphorylated compounds especially ATP
ATP
most common currency of cellular energy
Hydrolysis
of the phosphate bond yields free energy that can be harnessed for chemical reactions or mechanical work
cleavage of phosphate from ATP
highly exergonic, it releases lots of energy. This can be couples to endergonic reactions to drive them forward.
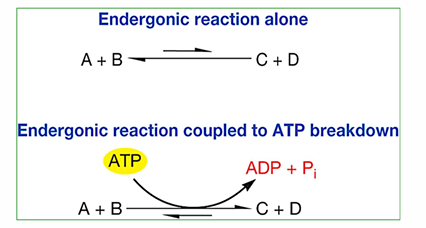
activation energy
all reactions have, energy that must be invested for them to move
Enzymes
catalyze chemical reactions by lowering th activation energy, helping the reaction move forward faster
Enzymes are reusable
not consumed in the process
Enzyme 3D fold
required for its catalytic activity
catalytic site
enzymes typically have this site that matches the shape of the substrate molecule
Enzymatic reactions
Enzymes are often dynamic.
• The enzyme may change shape upon binding of substrate(s) – the induced fit hypothesis.
• After the reaction happens, this helps kick out products to reset the enzyme.
Enzymes required specific local environmental conditions
pH, substrate concentration, temperature
Extreme temperatures or unfavorable conditions on enzymes
may cause enzymes to denature (unfold) and lose activity
Enzymes may need
cofactors or coenzymes to function
holozyme
enzyme + cofactor/coenzyme
apoenzyme
enzyme without cofactor/coenzyme
Enzymes activity is often heavily regulated by
by the cell – one mechanism is through inhibition.
Competitive inhibition
occurs when an inhibitor directly competes with substrate for binding to the active site
Sulfa drugs
an example of competitive inhibition. They look like a natural substrate and compete for binding to the enzyme. Sulfa drugs block synthesis of folate, an essential cofactor
noncompetitive inhibition
occurs when an inhibitor binds somewhere other than the active site.
allosteric regulation
Noncompetitive inhibitor binding typically changes the shape of the enzyme to prevent activity.
molecules that act allosterically
can change the shape of an enzyme to activate it
biochemical pathways
Metabolic reactions organized into multistep
metabolites
Product of one reaction is substrate for next