Chapter 9:M Models of Chemical Bonding
1/52
Earn XP
Description and Tags
These flashcards cover key vocabulary and concepts related to atomic properties and chemical bonding as outlined in the provided notes.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
Ionic Bonding
The bonding that occurs when one metal transfers electrons to a nonmetal, forming cations and anions that attract each other.
Covalent Bonding
The bonding that occurs when two nonmetals share electrons to achieve stability.
Metallic bonding
The bonding that occurs between metal atoms, characterized by a sea of delocalized electrons that allow for conductivity and malleability.
Draw a Lewis Dot structure for Nitrogen and specify the sublevel of each electron depicted:

what types of atoms readily form cations?
Elements (metal) with low ionization energy readily form cations.
What type of atom readily forms anions?
Elements (nonmetal) with negative electron affinities tend to form anions
what does a negative electron affinity imply about an atom?
that energy is required to add an electron
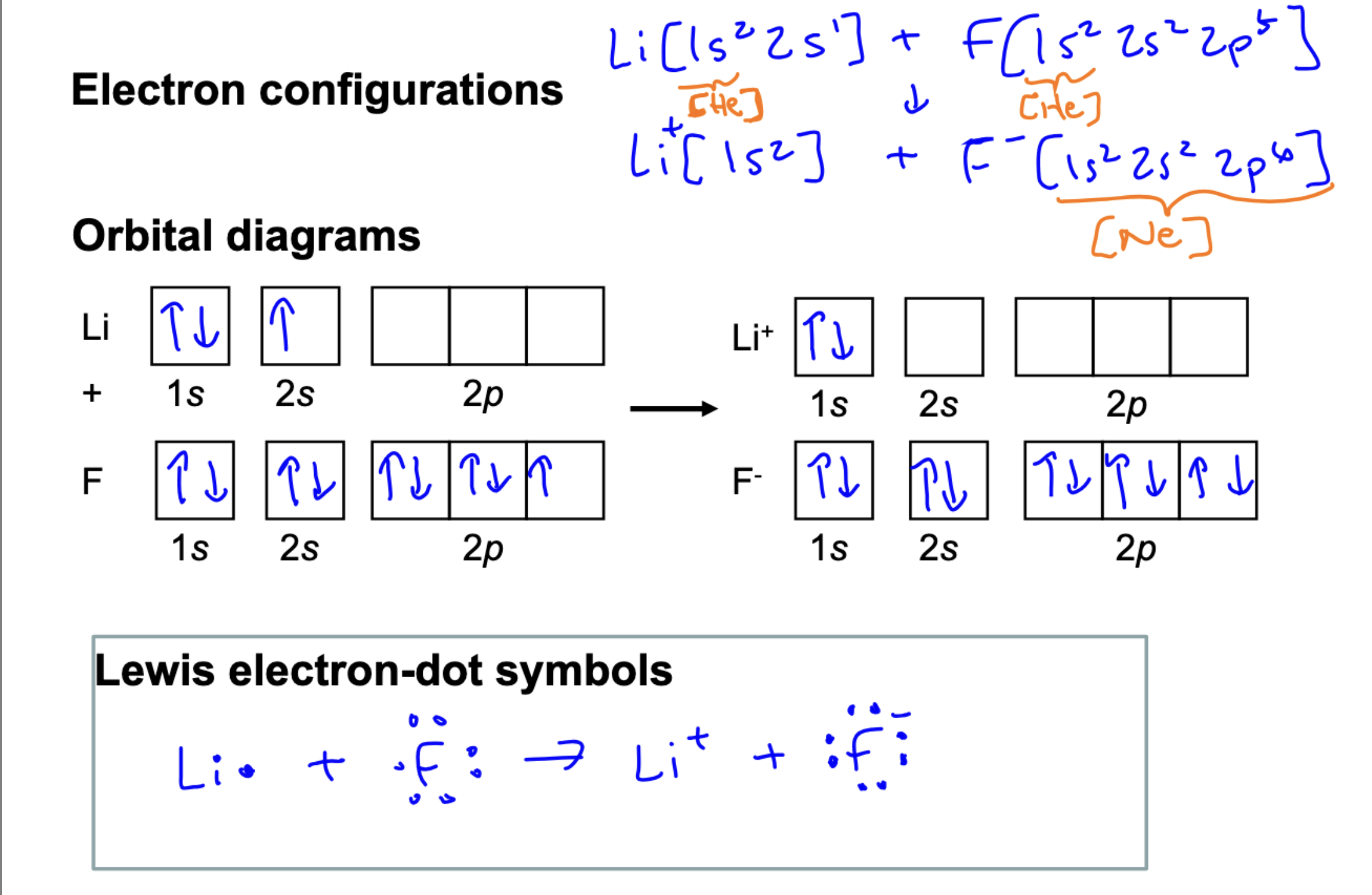
Draw an orbital diagram and a lewis dot diagram depicting: Li + F → Li+ + F-
Use Lewis symbols to depict the formation of sodium oxide (Na2O).
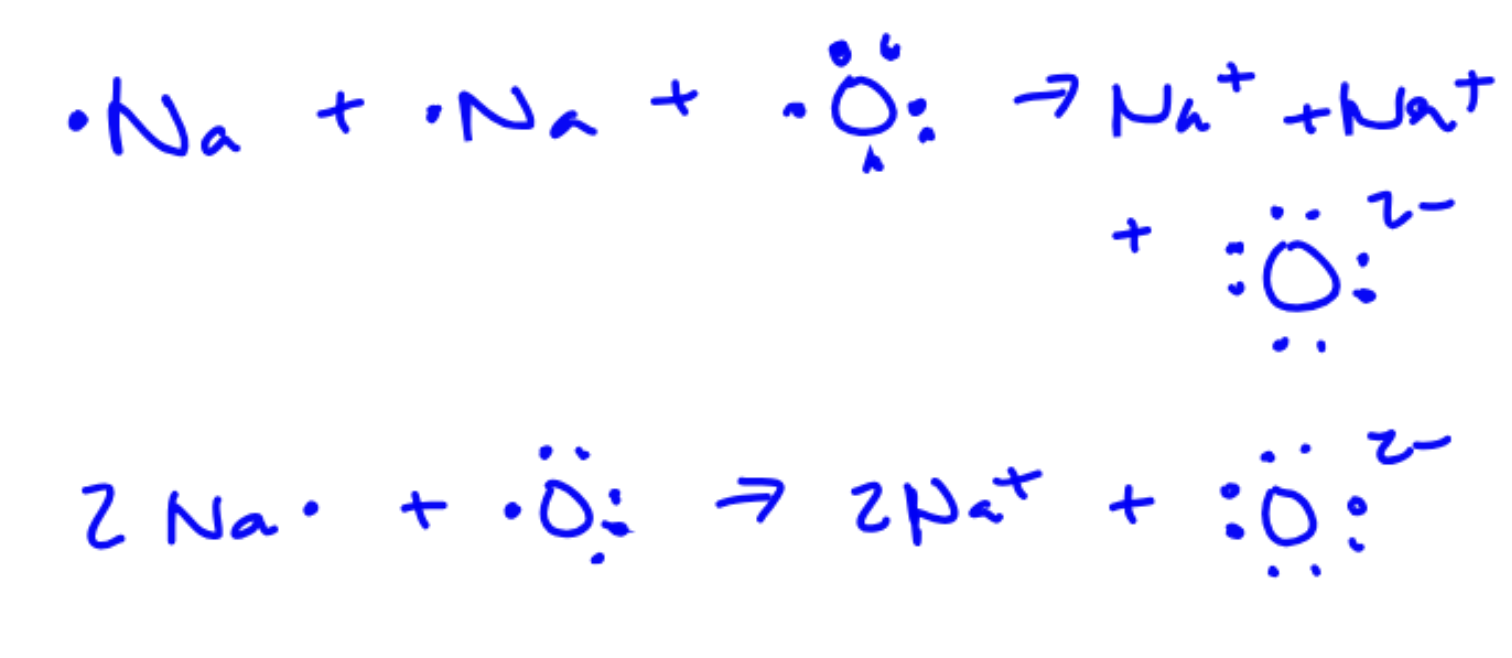
Thermodynamically what will ionic bonding alway be and why?
Ionic bonding is always exothermic (releasing heat) because ionic bonds are stabilizing thus moving from a higher energy state to a more favorable lower one releasing heat during the formation process. Although energy is required to form ions (such as ionization energy for removing electrons), the formation of the ionic crystal lattice releases a much larger amount of energy due to strong electrostatic attraction between oppositely charged ions. This lattice energy outweighs the energy input, making the overall process exothermic.
What is Lattice Energy (DELTA-H lattice)?
Lattice energy (DHolattice ) is the energy required to separate 1 mol of solid ionic
compound into gaseous ions.
Standard heats of formation?
The Standard Heat of Formation (ΔH⁰f) is the enthalpy change when one mole of a compound is formed from its elements in their standard states under standard conditions (298 K and 1 atm).
What is a Formation Equation?
A Formation Equation is a balanced chemical equation that shows the formation of one mole of a compound from its elements in their standard states, with a coefficient of 1 for the compound and no coefficient for the elements.
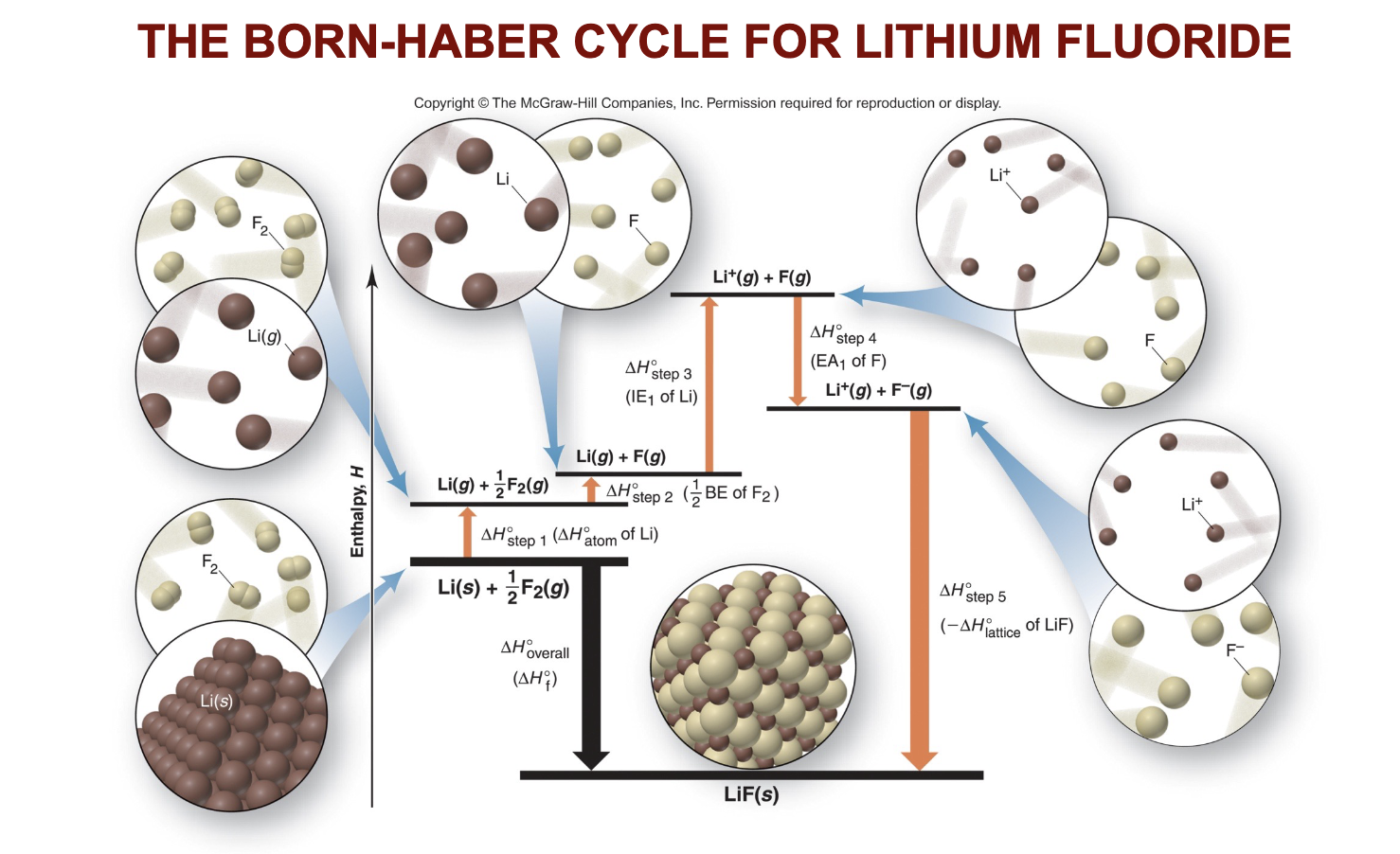
What is the Born-Haber Cycle, and why is it important for calculating lattice energy?
The Born-Haber Cycle is a series of steps used to calculate the lattice energy of an ionic compound. It applies Hess's Law to sum the enthalpy changes of various reactions (such as atomization, ionization, and electron affinity) to determine the overall energy change when an ionic compound forms from its elements. This cycle helps in calculating lattice energy, which is the energy released when ions in the gas phase come together to form a solid ionic lattice.
What is it and what are the steps to the Born-Haber Cycle?
The Born-Haber Cycle is a series of steps used to calculate the lattice energy of an ionic compound. It applies Hess's Law to sum the enthalpy changes of various reactions (such as atomization, ionization, and electron affinity) to determine the overall energy change when an ionic compound forms from its elements.
Steps of the Cycle:
Step 1: Atomize the Metal
Convert the solid metal into a gaseous metal
Atomize the nonmetal
by converting it from its standard state to gaseous atoms. EG: CL2 → @2CL
Step 3: Ionize the Metal
Remove the electrons from the gaseous metal atom to create a gaseous cation. (use ionization energies to calculate the DELTA-H here)
Step 4: Add Electrons to the Nonmetal
Add an electron to the gaseous nonmetal atom, forming a gaseous anion. Use the electron affinity value to calculate the DELTA-H value
Step 5: Combine Ions
combine the ions to form the final lattice. Then find the value for DELTA-Hrxn then subtract all of the values achieved up to DELTA-Hrxn to DELTA-Hrxn and then that value is your DELTA-Hlatteice of lattice energy since it is the final energy required to form the DELTA-Hrxn.
How do you apply the Born-Haber Cycle to calculate the lattice energy of calcium chloride (CaCl₂)?
Answer:
To apply the Born-Haber Cycle to CaCl₂, follow these steps:
Step 1: Atomize the metal
Convert solid calcium (Ca) into gaseous calcium atoms: Ca(s) → Ca(g) (ΔH₁ = 192.6 kJ).Step 2: Atomize the nonmetal
Convert chlorine gas (Cl₂) into individual chlorine atoms: Cl₂(g) → 2Cl(g) (ΔH₂ = 242.0 kJ).Step 3: Ionize the metal
Remove two electrons from calcium to form Ca²⁺(g): Ca(g) → Ca²⁺(g) + 2e⁻ (ΔH₃ = 1735 kJ).Step 4: Add electrons to the nonmetals
Add electrons to the chlorine atoms to form Cl⁻(g): Cl(g) + e⁻ → Cl⁻(g) (ΔH₄ = -698 kJ).Step 5: Form the lattice
Combine Ca²⁺(g) and 2Cl⁻(g) to form CaCl₂(s): Ca²⁺(g) + 2Cl⁻(g) → CaCl₂(s) (ΔH₅ = -795.0 kJ, lattice energy).
ASK PROF LEE TO BETTER EXPLAIN THIS AND WHAT I SHOULD DEDUCE FROM IT?
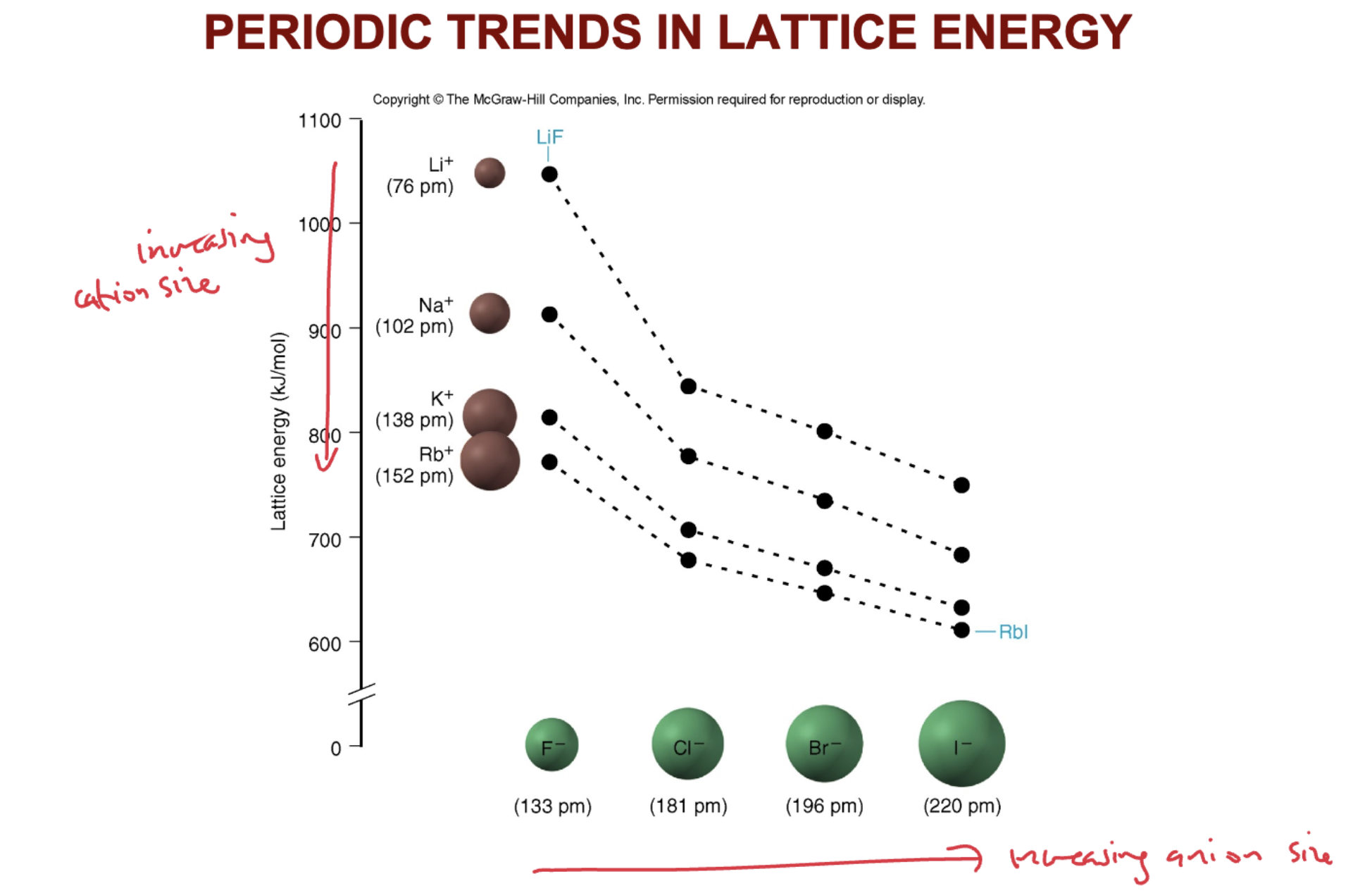
What are the PERIODIC TRENDS IN LATTICE ENERGY and thus what two factors affect lattice energy?
Lattice energy is affected by ionic size and ionic charge. As ionic size increases, lattice energy decreases because the ions are farther apart. As ionic charge increases, lattice energy increases due to stronger electrostatic attraction between oppositely.
How does the lattice energy of NaCl compare to KBr?
The lattice energy of NaCl is greater than that of KBr. This is because Na⁺ is smaller than K⁺, leading to a stronger electrostatic attraction between Na⁺ and Cl⁻, resulting in a higher lattice energy. K⁺, being larger, results in weaker attraction with Br⁻, so the lattice energy of KBr is lower than NaCl.
Draw a grpahical depitcion of Lattice energies starting from LiF and ending at RbI
What does Coulomb's Law say about lattice energy, and how does it relate to electrostatic energy?
Coulomb's Law states that lattice energy is directly proportional to the product of the charges of the cation and anion, and inversely proportional to the sum of their radii. The equation also reflects electrostatic energy, which is the energy resulting from the force of attraction or repulsion between charged particles.
Mathematically, Coulomb's Law for lattice energy is:
Lattice energy ∝ (cation charge × anion charge) / (cation radius + anion radius).
The stronger the electrostatic attraction (higher charges, smaller ions), the higher the lattice energy.
Mechanical Properties of Ionic Compounds
Ionic compounds are typically hard, rigid, and brittle due to the strong electrostatic forces between ions. (inonic metal are typically ductile and can bend not brittle)
Draw a visual depictuion with cations and anions as to why the structure of ionic solid alows it to be brittle and seperate easily when force is applied to one side of it? THREE STEPS
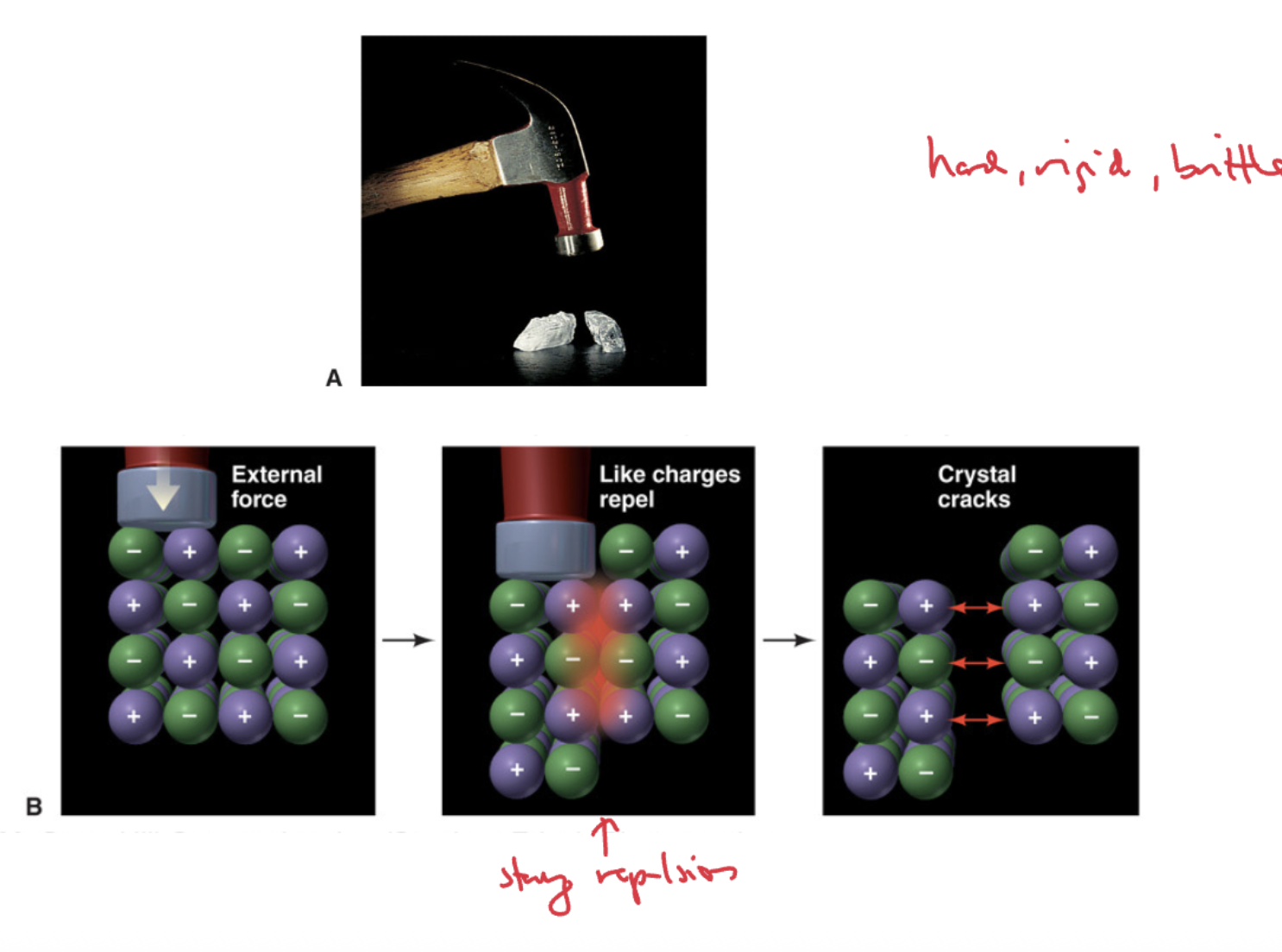
Electrical Properties of Ionic Compounds
Solid ionic compounds conduct electricity only when melted or dissolved in water, as ions are free to move.
Thermal Properties of Ionic Compounds
Ionic compounds generally have high melting and boiling points due to the strong attractions between ions.
When is thr bond stronger between small ions or big ions
small ions the smaller the bond length the stronger
Which of the following pairs has the element with larger (more negative) lattice energy listed first?
CsCl, BaS
CsCl, LiCl
CaS, CaO
SrO, BaO
BaS, CaS
SrO, BaO
What is the total energy change (ΔH) when oxygen (O) is converted to O²⁻?
To calculate the total energy change, you must add EA₁ (the electron affinity for the first electron) and EA₂ (the electron affinity for the second electron). Thus, ΔH = EA₁ + EA₂.
What is a Covalent bond?
A Covalent bond is a chemical bond in which two electrons are shared by two atoms. It is usually observed when a nonmetal bonds to a nonmetal.
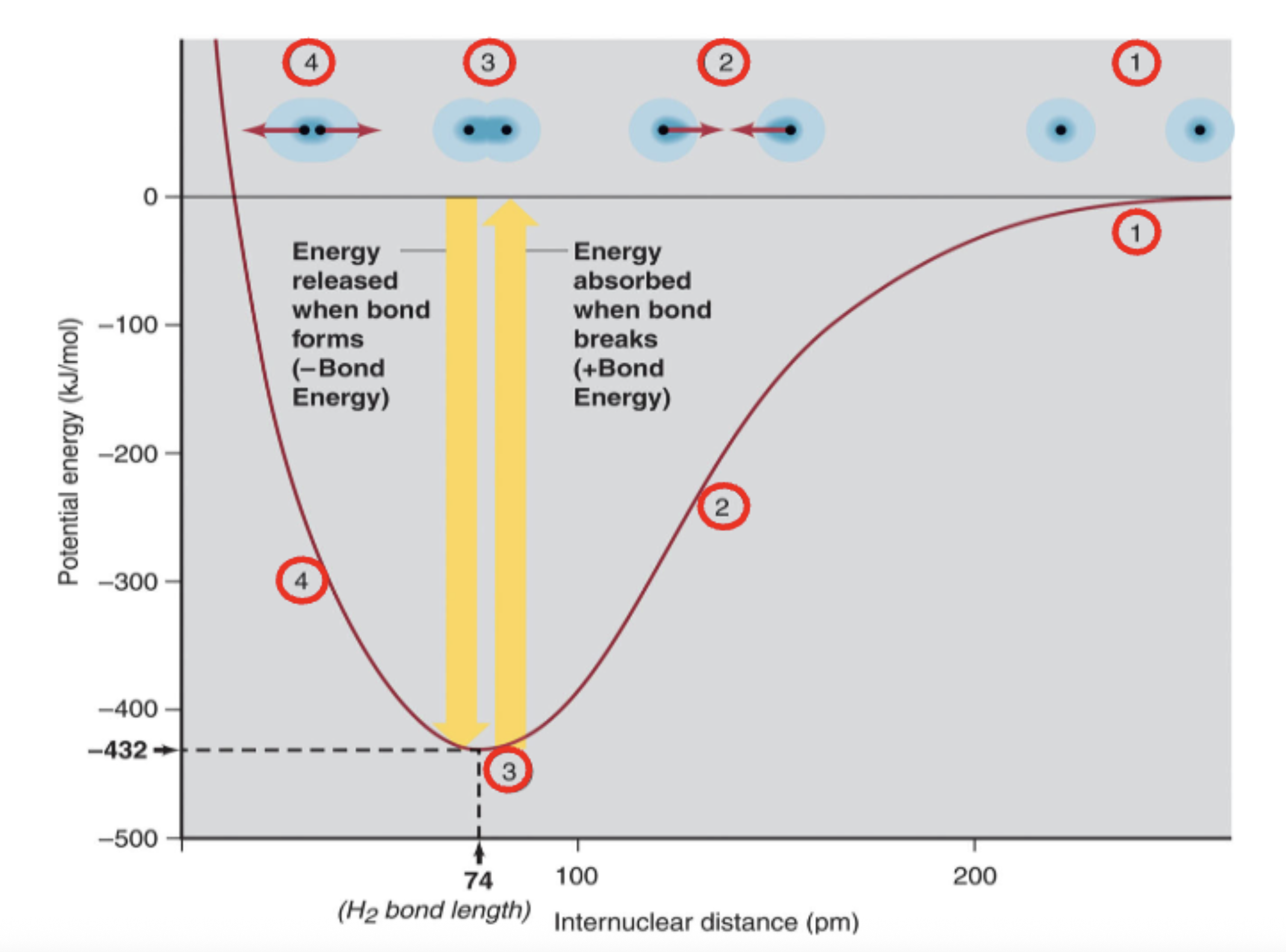
Explain this
1) far apart ions hardly feel each other
2) more potential energy because of energy required to keep ions apart since the ions feel attractive forces
3) They reach an equilibrium distance (MAX PE)
4) If they get closer, they face repulsion due to overlapping electron clouds and portons in nucleus.
When a bond breaks what is the delta-E vs when a bond forms
Bond energy:
breaking bonds → +
forming bonds → -
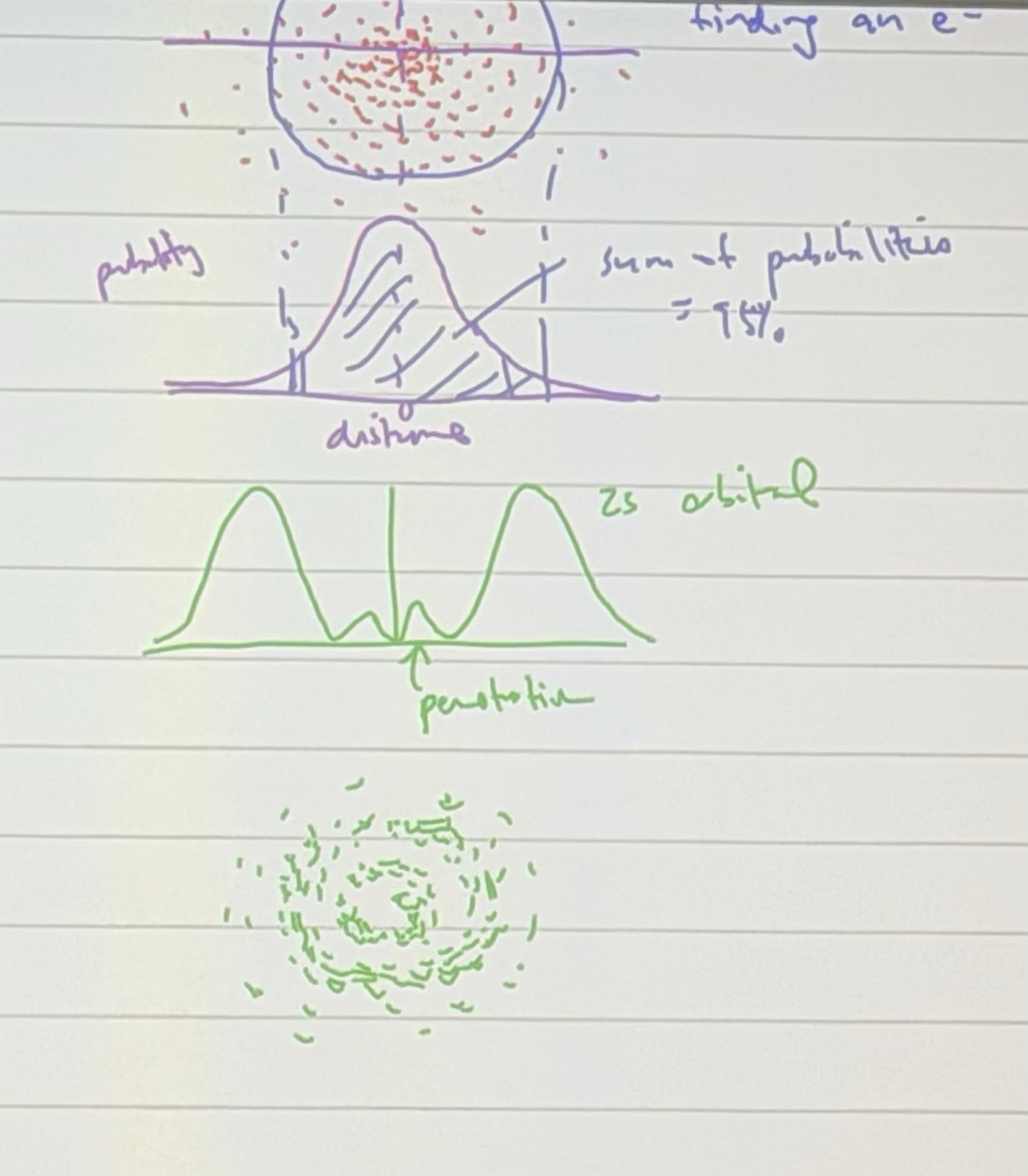
Draw a helpful diagram to determine the probability of finding an electron in an atom
During covalent bonding what hapens to the orbitals?
The orbitals of the bonding atoms overlap, creating new molecular orbitals that lower the potential energy of the system. Going from two differnet waves to one wave
What is the equilibrium distance suggest and give us?
The min distance between two covalently bonded atoms before the protons repel one another and it suggests the bond length
What electrons are involved in bonding?
Only the valence electrons of an atom can be involved in covalent bonding (not all of
them will be participating in bonds)
What is a shared pair or bonding pair?
Atoms share electrons to achieve a full outer level of electrons. The shared electrons are called a shared pair or bonding pair. Two or three shared pairs are called double or triple bond.
How are covalent bonds drawn?
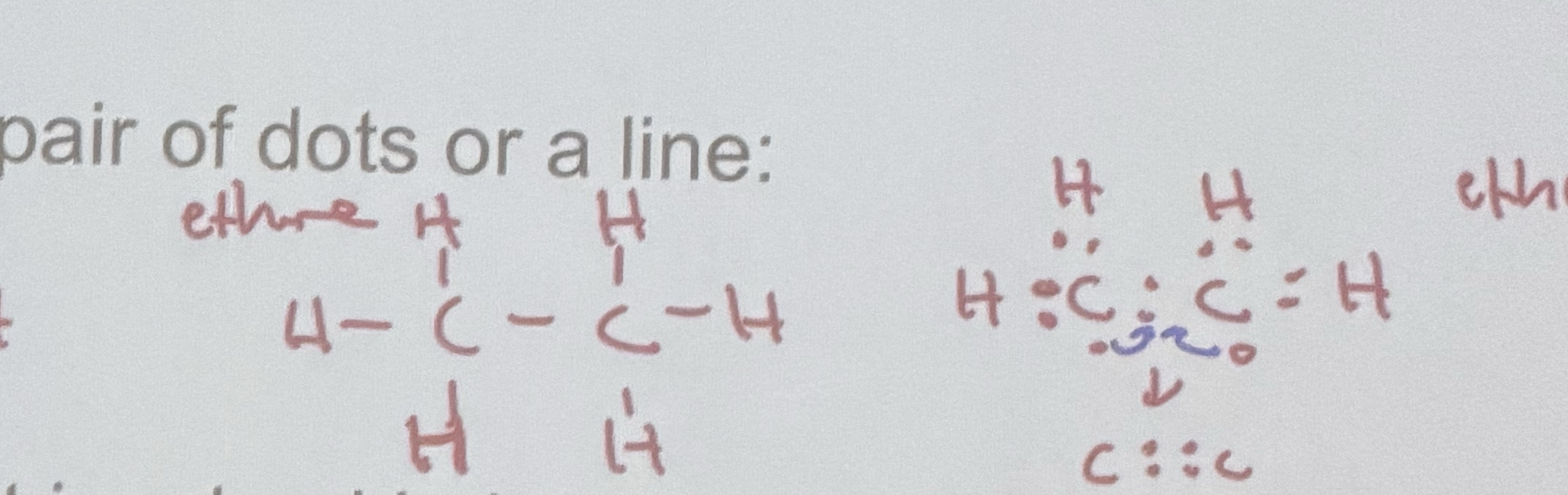
What is a lone pair?
An outer-level electron pair that is not involved in bonding is called a lone pair, or unshared pair.
What is bond energy in the context of a covalent bond and what makes bond energy stronger?
Bond energy (BE) is the energy required to break a bond. The stronger the bond, the higher the bond energy. (Bond strength ∝ Bond energy)
What is bond length and how does it vary across the periodic table?
Bond length is the distance between the nuclei of the bonded atoms. Bond length increases down a group in the periodic table and decreases across a period from left to right.
How does bond energy change across the periodic table?
Bond energy generally shows the opposite trend to bond length: it increases across a period and decreases down a group.
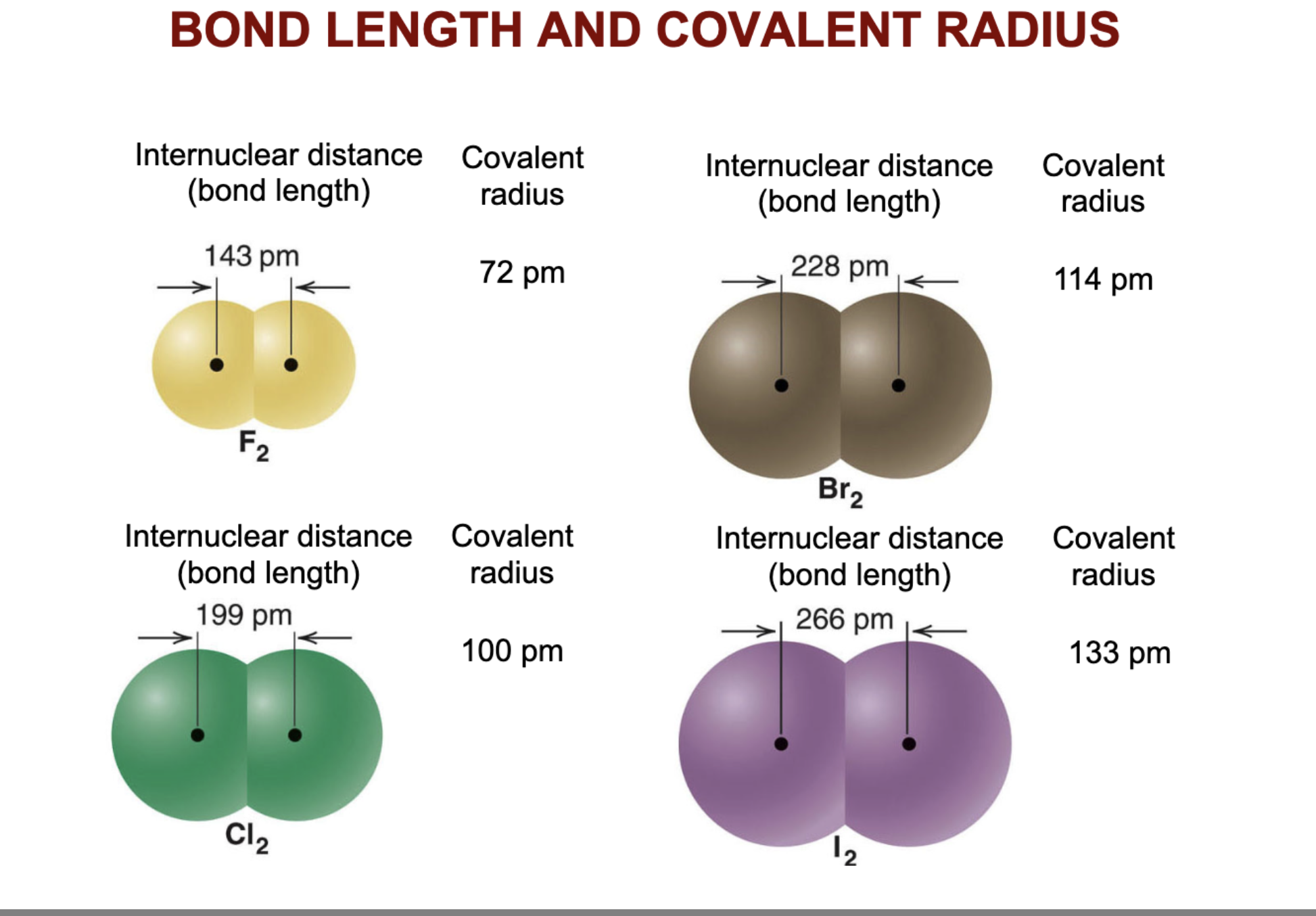
which compound has the strongest bond energy?
F2
What is the energy change when bonds are broken and formed?
When the bonds are being broken, the energy value is positive, and when they are formed, the energy value is negative.
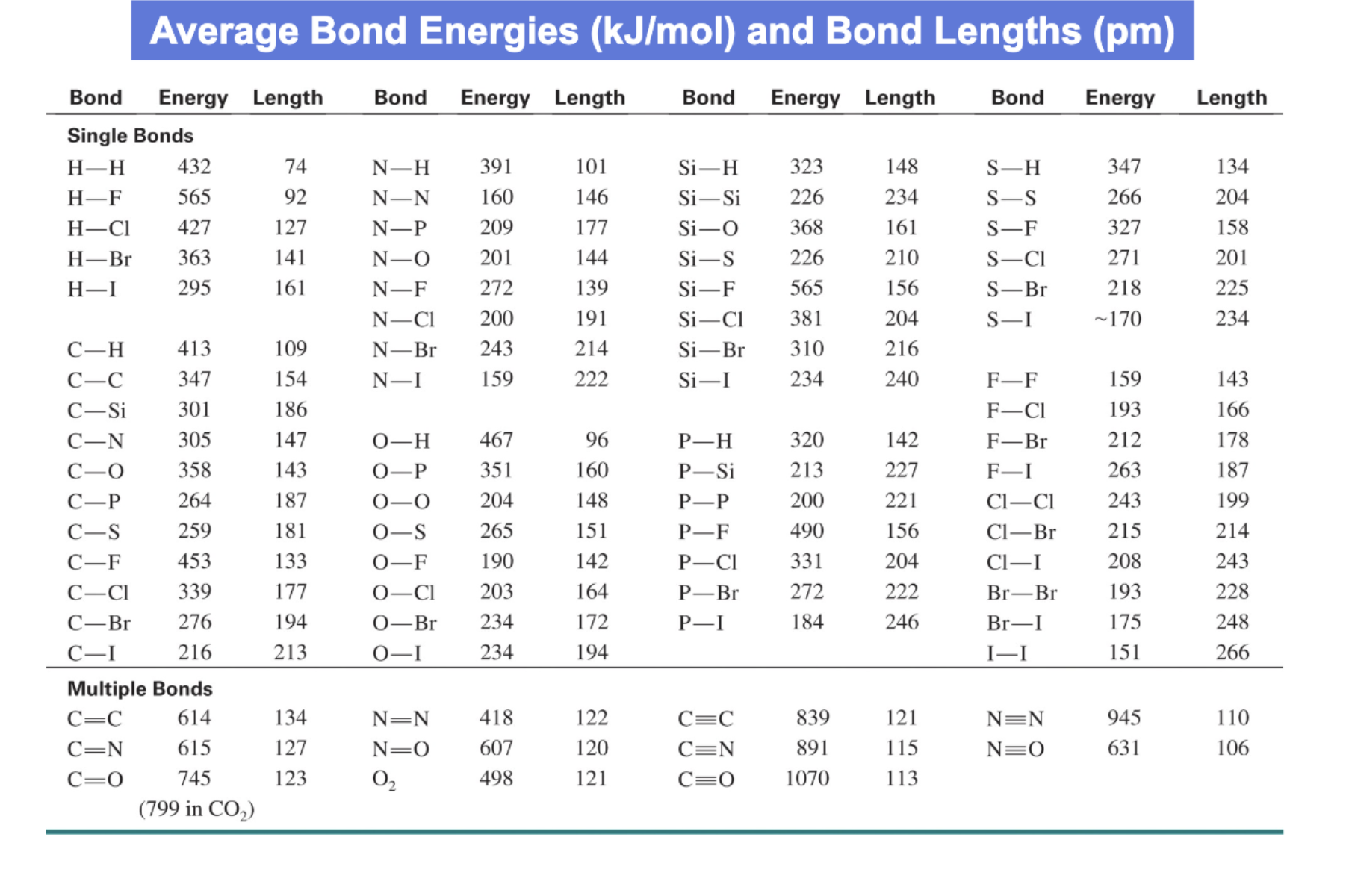
How to determine DELTA-Hrxn off of bond energies
(DELTA-Hrxn = Sum of DELTA-Hbondsbroken - Sum of DELTA-Hbondsformed)
When the bonds are being broken the energy value positive and when they are formed the energy value is negative
Average bond length table used to estimate reaction energies (DELTA-Hrxn = Sum of DELTA-Hbondsbroken - Sum of DELTA-Hbondsformed)
How To calculate the ΔH (enthalpy change) for the reaction not using bond energies:
CH4(g)+2O2(g)→2H2O(l)+CO2(g)
You can use the Hess's Law or the standard enthalpies of formation (ΔH_f°) of each reactant and product. The general formula is:
ΔHrxn =∑(ΔHf∘ products)−∑(ΔHf∘ reactants)
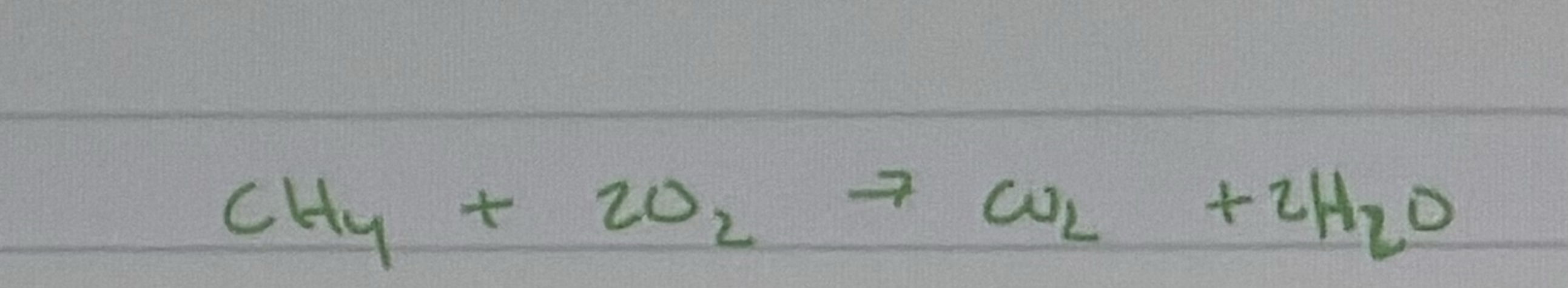

Using the periodic table, but not Tables 9.2 or 9.3, rank the bonds in each set in order of decreasing bond length and decreasing bond strength:
(a) S–F, S–Br, S–Cl
(b) C=O, C–O, CΞO
a) decreasing bond length: S-Br, S-Cl, S-F
decreasing bond strength: S-F, S-Cl, S-Br
b) decreasing bond length: C–O, C=O, CΞO
decreasing bond strength: CΞO, C=O, C–O
Molecular covalent compounds?
Molecular covalent compounds: a number of atoms covalently bonded together to form a discrete molecule. i.e. CO2(g)
What are the relative strengths of intramolecular and intermolecular forces in molecular molecules?
In molecular molecules, there are strong intramolecular forces in the form of covalent bonds and very weak intermolecular forces.
When melting a molecular compound what are the interactions that break? ie: subliming CO2
the weak intermolecular forces
Network covalent compounds:
every atom in involved in a single three-dimensional network of covalent bonds and no separate molecules will exist. i.e.
Quartz (SiO2).
Molecular vs Network COvalent Compounds Melting point and why?