10SC3A1 - Chemistry
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Structure of the atom
Charges:
Proton = positive
Neutral = no charge
Electron = negative
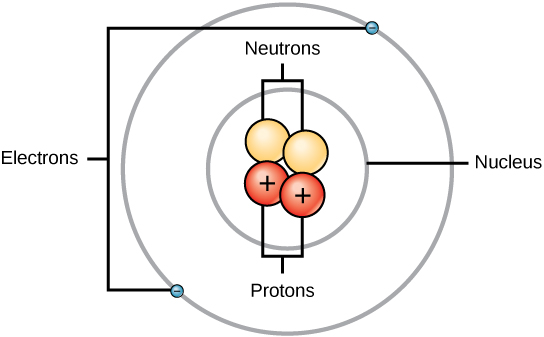
Subatomic Masses, Z, A.
Proton = 1
Neutron = 1
Electron = 1/1800
Z = number of protons
A = mass numbers
Metal form: anions or cations
Cations
Non metals form: anions or cations
Anions
Valence Electrons, Accepting and donating.
Valence electrons are transferred in ionic compounds through the donation of electrons from metals (forming positive cations) to non-metals (forming negative anions).
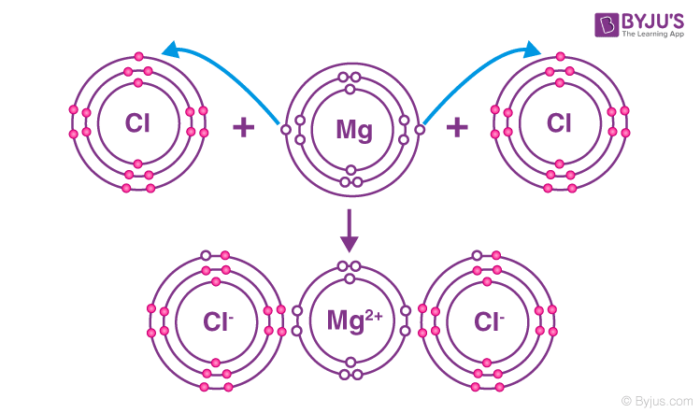
Ionic Compounds (+ Latice) (+Characteristics)
An ionic compound is a compound that is formed by ionic bonding. Ionic bonding occurs through a process called electron transfer, where one atom gives electrons to another. This causes one to be positive and the other to be negative, combining them together
Metal and Non metal bond together
Ionic compounds form giant 3D ionic lattice structures in their solid state. These structures consist of regular, repeating arrangements of alternating positive and negative ions, held together by strong electrostatic attractions that act in all directions. In the same ratio
-Charge cannot move about, → they are not good a conducting electricity
-They are hard but brittle
They have a neutral charge
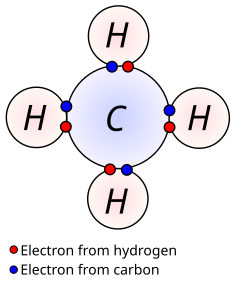
Covalent Compounds
Octect Rule:
Tendency of atoms to prefer 8 electrons in their outershell
Covalent bond:
Forms when atoms share electrons in order to satisfy the octet rule.
Valency charge of a period table
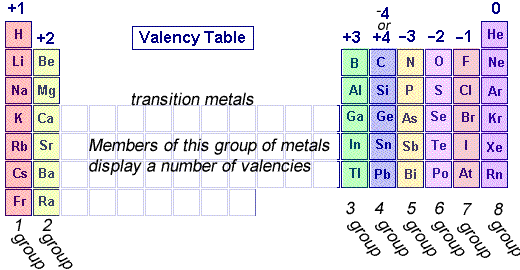
Valence electrons and electron stability
Atoms gain or lose electron to achieve a stable electron configuration, valence electrons are electrons located in the outermost shell, possessing the highest energy level
Metallic Bonding
The atoms of a metal arrange themselves so that they are abled to slide over one anothe.
Metallic bonding consists of a regular, three-dimensional lattice of positively charged metal ions (cations) surrounded by a "sea" of delocalized valence electrons. These free electrons are shared among all atoms, acting as electrostatic glue that holds the structure together.
Link properties of metals to their structure
High electrical conductivity → delocalised (free) electrons move through the lattice
High thermal conductivity → free electrons transfer kinetic energy quickly
Malleable (can be hammered) → layers of metal ions can slide without breaking bonds
Ductile (can be drawn into wires) → same sliding layers allow stretching
High melting/boiling point → strong electrostatic attraction between positive ions and delocalised electrons (metallic bonding)
Strong/dense → closely packed lattice of positive ions
Shiny (lustrous) → free electrons reflect light
Naming Ionic Compounds
State the cation first (Metal)
State the anion second (Non - metals)
Different States of oxidation (Polyvalency)
Metals can have different oxidation states as they can have multiple valencies, we represent that with roman numbers in parenthesis, so balance out the formula.
Polyatomic Ions
Other ions consist of a group of atoms with a net charge. Since these ions are composed of multiple atoms held together by covalent bonds
, they are called polyatomic ions.
They also participate in forming ionic compounds just like monoatomic ions, but they are held together by covalent bonds.
Four Types of Solids
-Covalent Network Substances
-Molecular Covalent
-Metallic Solids
-Ionic Solids
Describe the bonding structure of a covalent compound
Covalent compounds form when two or more elements share pairs of electrons so that each has a full valency shell
Covalent bonds share two electrons, one from each atom.
Molecular Covalent
Structure: Atoms are covalently bonded within molecules, but the overall structure is held together by intermolecular forces
Forces between molecules: Weak dispersion forces (and sometimes dipole forces / hydrogen bonding)
Key idea: Strong bonds inside molecules, weak forces between molecules
Characteristics:
Low melting and boiling points (weak intermolecular forces)
Often gases or liquids (or soft solids)
Poor electrical conductivity (no free charged particles)
Covalent Network Substances
Structure: Atoms are covalently bonded in a continuous network (no separate molecules)
Bonding: Strong covalent bonds throughout the entire structure
Characteristics:
Very high melting and boiling points (strong bonds everywhere)
Very hard and strong
Low electrical conductivity (no free electrons or ions, except graphite)
Naming Covalent Molecular Compounds
State prefix only for the second element, unless required for the first element.
Identify whether the compound is a molecular compound first, this should be a non-metal with a metal.
List of prefixes
1 → mono-
2 → di-
3 → tri-
4 → tetra-
5 → penta-
6 → hexa-
7 → hepta-
8 → octa-
9 → nona-
10 → deca-
Grams to Mols
To convert grams to moles, divide the given mass of the substance (in grams) by its molar mass (in g/mol).
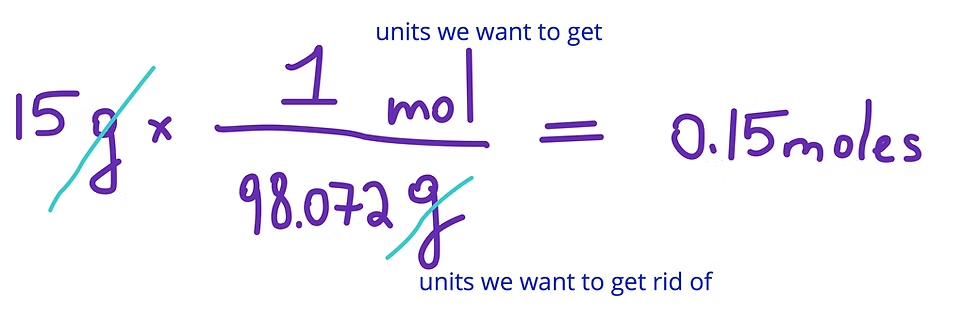
Concentration
How much substance is dissolved in a Solvent
Solute/Solvent
Solvent, Solute, Solution
Solvent: the substance that dissolves the solvent
Solute: the substance that gets dissolved
Solution:the mixture of solvent + solute
Molar Mass
Summing the average atomic masses, e.g H2o = 18.015
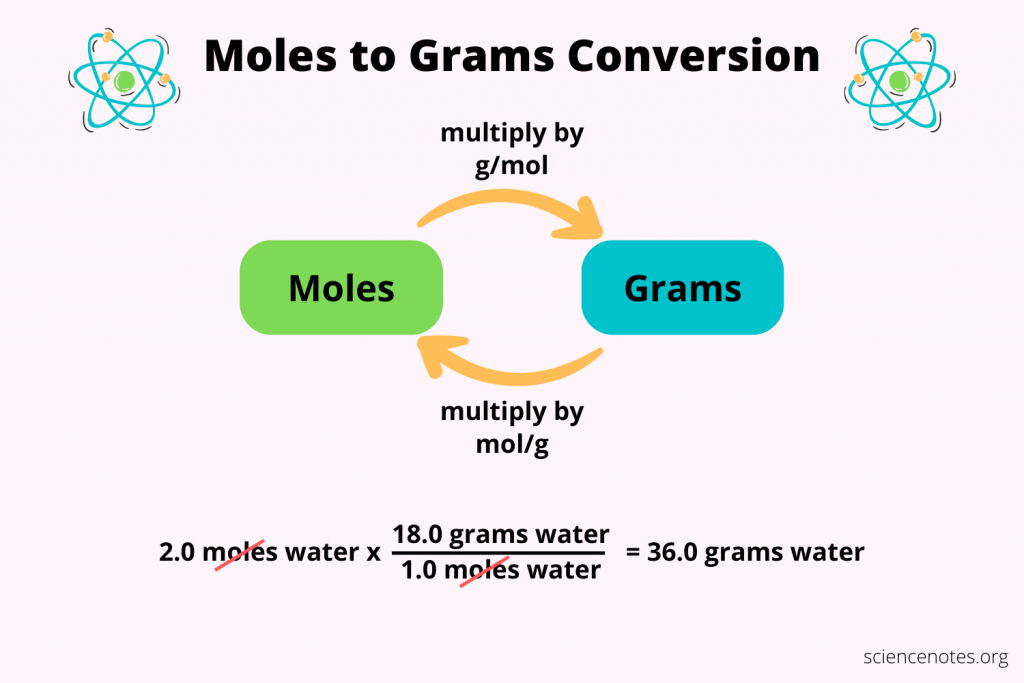
Molarity
Molarity = #Mols solute/ litres of solution.
Example 1.25M = 1.25 mol KCL/ 1 Litre KCL
Generalised formula for precipitation reaction
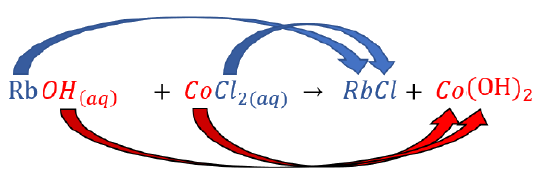
metal + non mentals, acids + bases
salt
How do you know if a product is soluble or insoluble?
What does aq? s? l? and g mean?
What do we do once the solubility has been checked?
Check the solubility table.
aq = aqueous, s = solid, l = liquid, g = gas
-Make sure to balance the equation by checking the charges
Acids vs Bases (Chemical Formula)
Acid has H+ and an Anion
Bases has a hydroxide (OH)- with a metal
Types of double displacement reactions:
Precipitation:
AB(aq) + CD(aq) → AD (aq) + CB (s)
Neutralisation:
Acid (aq) + Base (aq) → salt (aq) + water (l)
Precipitation
swap ions + check solubility
Acid–base
salt + water
Acid–metal
salt + hydrogen
Acid–carbonate
salt + water + CO₂
Electron Configuration - Elements properties
An elements chemical properties are controlled by its valence electrons
Elements in the same group have similar chemical properties because they have the same number of valence electrons.
It is to be filled clockwise, one at a time. Do not start pairing immediately.
Row Number
Column Number
How many shells it contains
Number of valence electrons.