Path 1b: Cellular Responses pt 2
1/113
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
114 Terms
What is hypoxia?
oxygen deprivation
What is reduced during hypoxia?
aerobic oxidative respiration
What are possible causes of hypoxia?
- reduced blood flow (ischemia)
- inadequate oxygenation of the blood (cardiorespiratory failure)
- decreased oxygen-carrying capacity of blood
What are possible causes of decreased oxygen-carrying capacity of the blood?
anemia, CO poisoning, and severe blood loss
Depending on the severity, how may a cell respond to hypoxia?
may adapt, undergo injury, or die
What are possible physical agents that can lead to cell injury?
- Mechanical trauma
- Extremes in Temp
- Sudden changes in Atmospheric pressure
- Radiation
- Electric Shock
When might glucose or salt lead to cell injury?
when in hypertonic concentrations
When might oxygen lead to cell injury?
when in high concentrations
When might immunologic reactions lead to cellular injury?
Autoimmune - reactions to endogenous self-antigens. Can also occur in response to external agents such as microbes and environmental substances
Give an example of a subtle defect caused by genetic derangements
decreased life span of red blood cells due to single amino acid substitution inhemoglobin
sickle cell anemia
Give an example of a severe defect associated with genetic derangements
congenital malformations associated with down syndrome (due to trisomy 21, chromosomal anomaly)
What effect may variations in genetic makeup have on cells?
influence the susceptibility of cells by chemicals and other environmental insults
List nutritional imbalances that may cause cellular injury
- protein-calorie deficiencies
- vitamin deficiencies
- anorexia
- nutritional excess
What happens to cells immediately after the onset of injury?
Cells may become rapidly nonfunctional but are still viable, with potentially reversible damage.
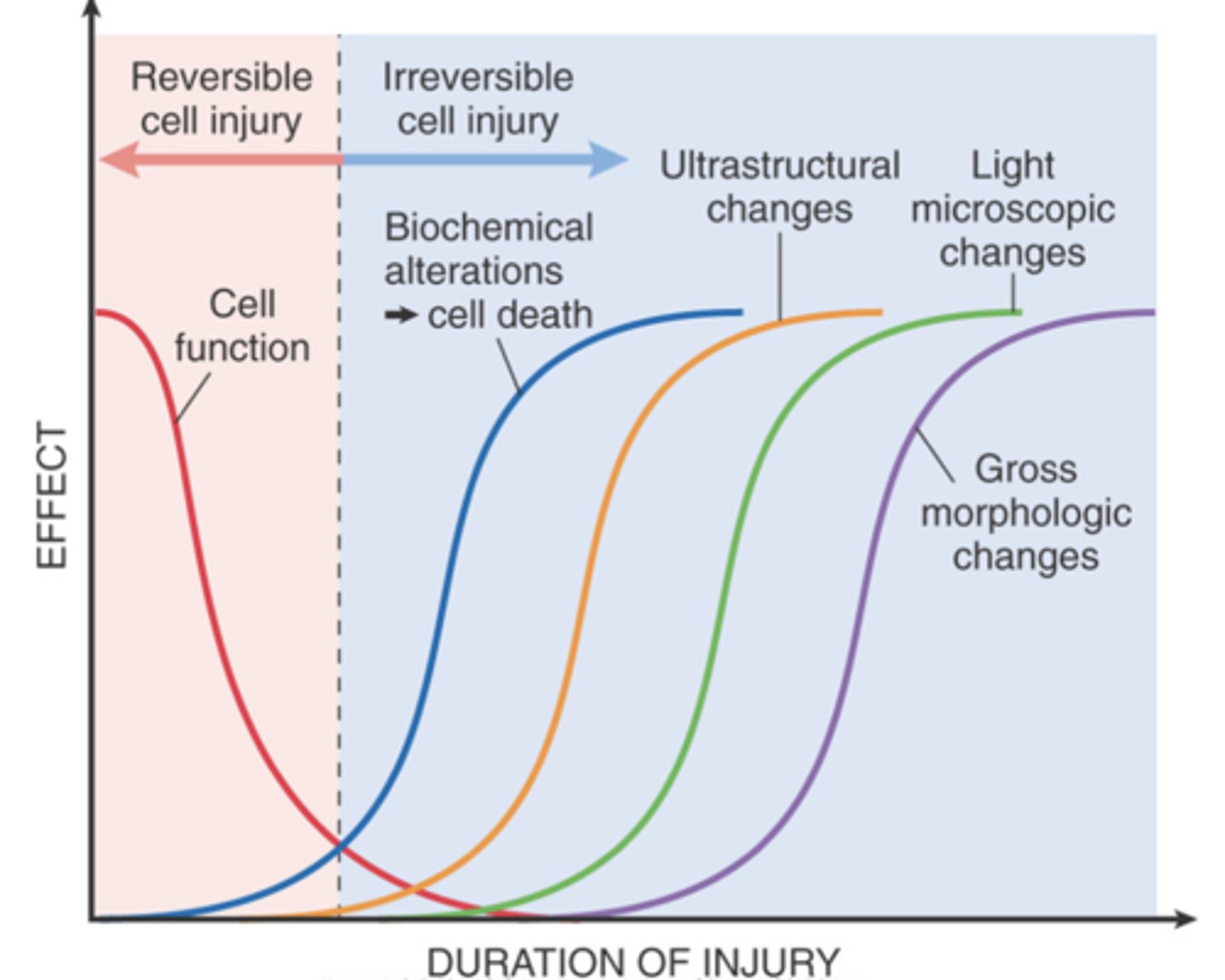
What can a longer duration of injury lead to?
A longer duration of injury may eventually lead to irreversible injury and cell death.
What typically precedes ultrastructural, light microscopic, and grossly visible morphologic changes in cell injury?
Irreversible biochemical alterations typically precede these morphologic changes.
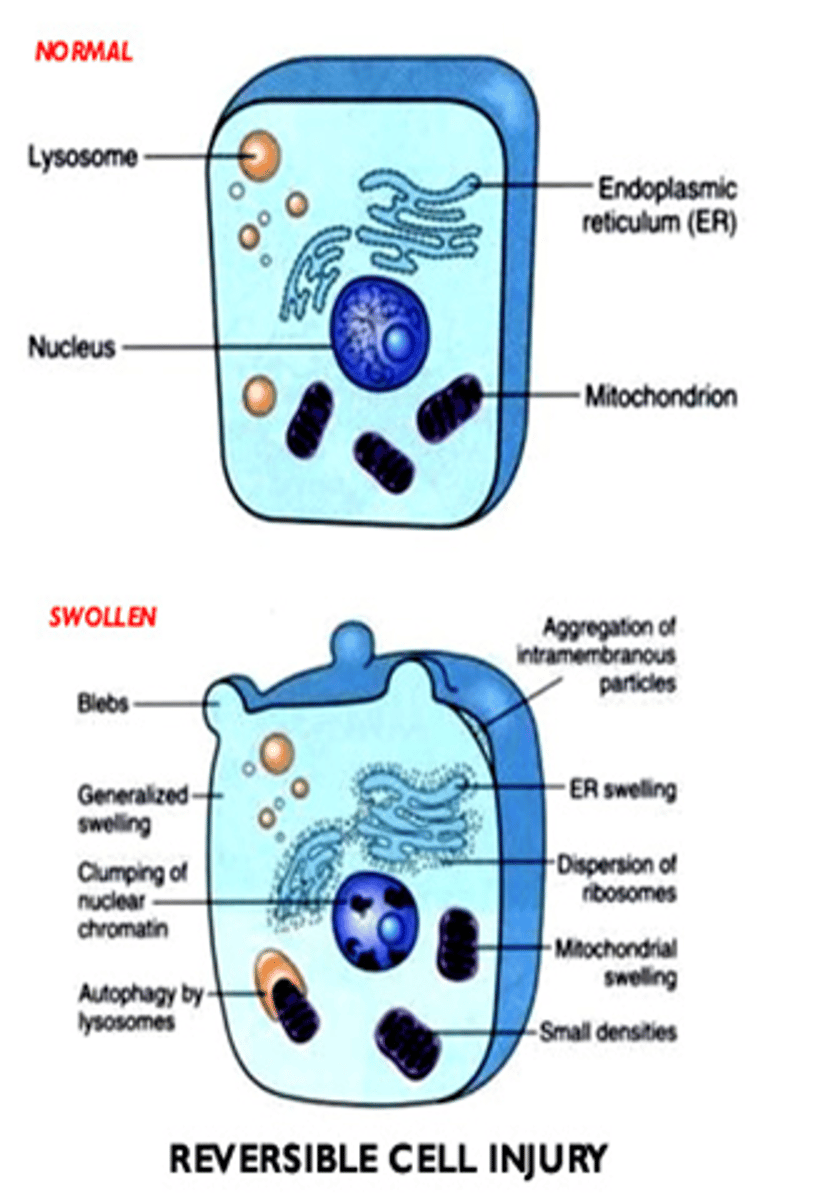
What are the generalized features of reversible cell injury?
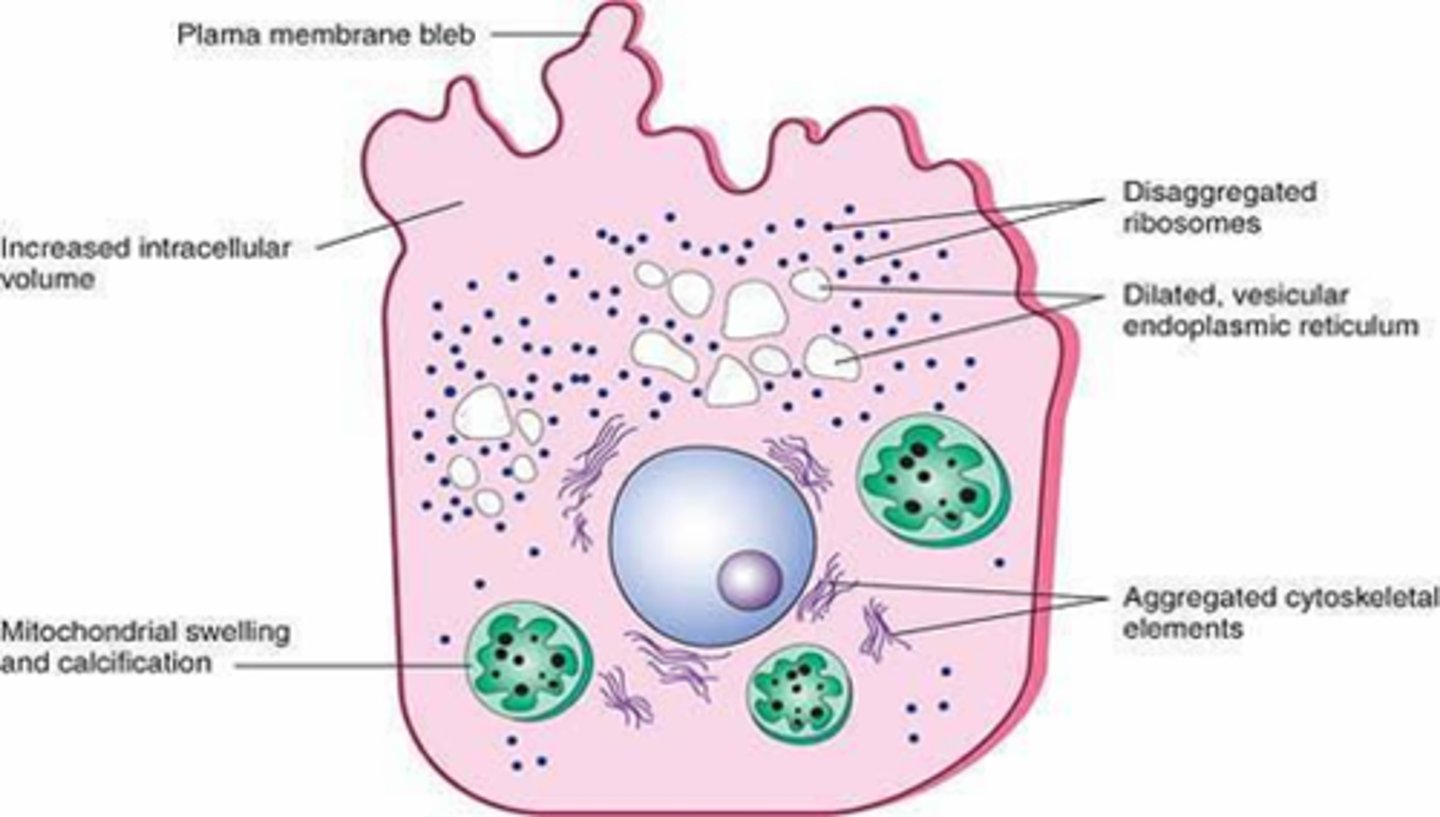
- Generalized swelling of the cell and its organelles
- Blebbing of the plasma membrane
- Detachment of ribosomes from the ER
- Clumping of nuclear chromatin
What are the sequential morphologic alterations associated with reversible cell injury?
- Decreased generation of ATP
- Loss of cell membrane integrity
- Defects in protein synthesis
- Cytoskeletal damage
- DNA damage
What are the two characteristic features of reversible cell injury?
Cellular swelling and fatty change
What causes cellular swelling in reversible cell injury?
failure of energy-dependent ion pumps in the plasma membrane
What type of injury will cause fatty change?
hypoxic injury and many forms of toxic or metabolic injury
How will fatty change due to reversible injury be manifested?
by the appearance of lipid vacuoles in the cytoplasm
In what types of cells will fatty change be seen?
hepatocytes and myocardial cells
What three characteristics will be seen in regards to an organ undergoing cellular swelling?
pallor, turgor, and increase in weight of the organ
What will be seen on the microscopic level in cells undergoing cellular swelling?
- Small clear cytoplasmic vacuoles (distended and pinched-off ER)
- Hydropic change or vacuolar degeneration
- Cells may show increased eosinophilic staining
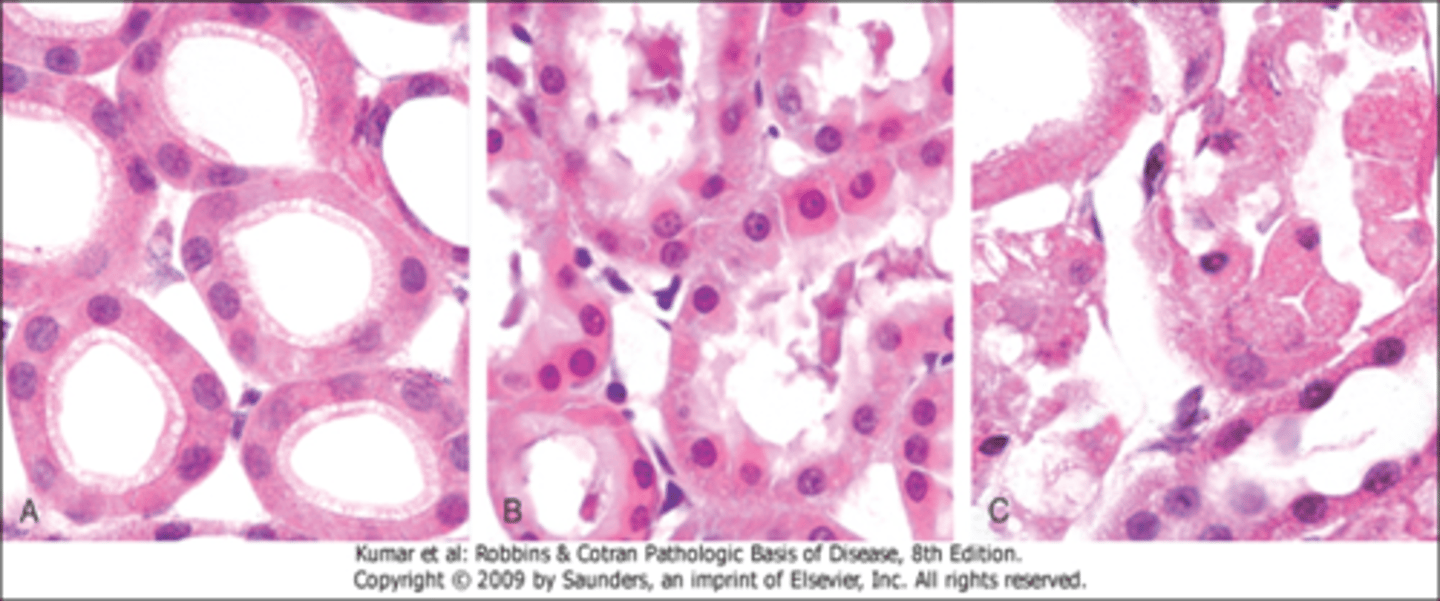
Be able to identify morphologic changes in reversible cell injury and necrosis histologically
A. Normal kidney tubules with viable epithelial cells.
B. Early (reversible) ischemic injury showing surface blebs, increased eosinophilia of cytoplasm, and swelling of occasional cells.
C. Necrosis (irreversible injury) of epithelial cells, with loss of nuclei, fragmentation of cells, and leakage of contents.
What plasma membrane alterations will be seen in a reversible injury?
blebbing, blunting, and loss of microvilli
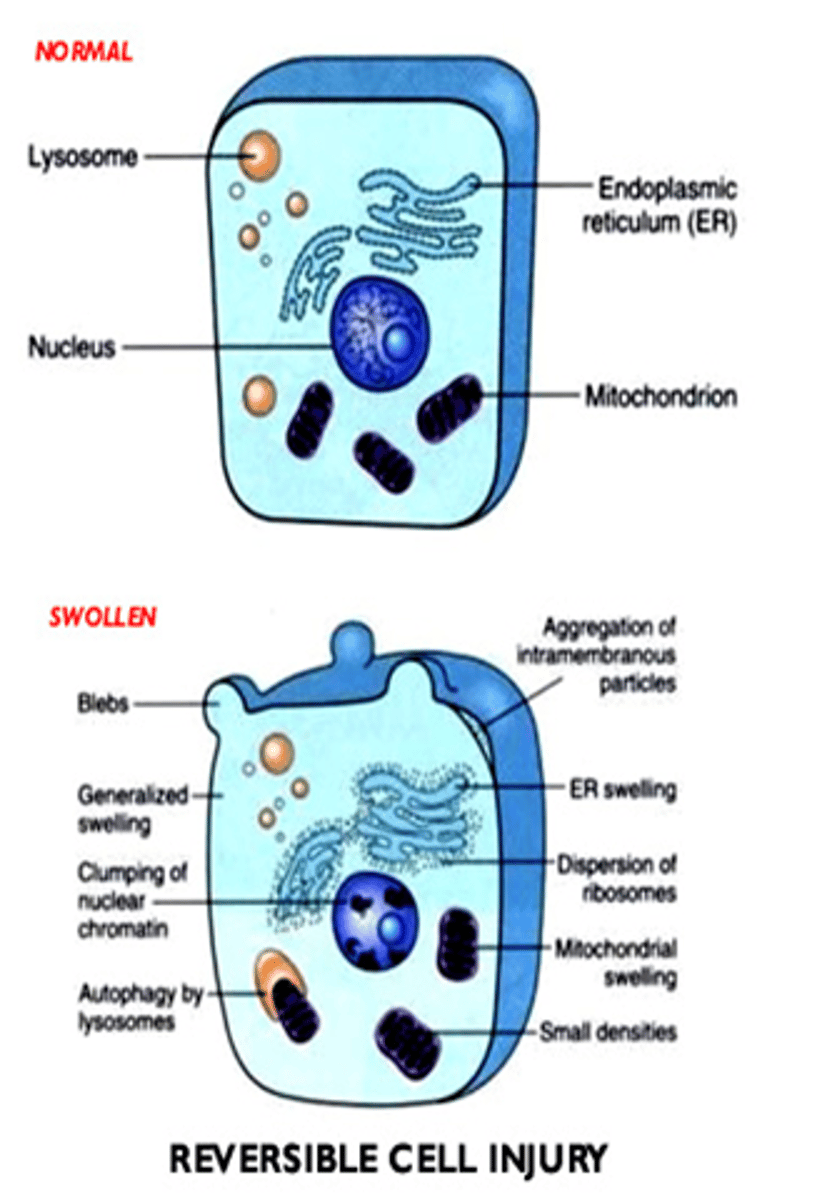
What mitochondrial changes will be seen in reversible injury?
swelling and small amorphous densities
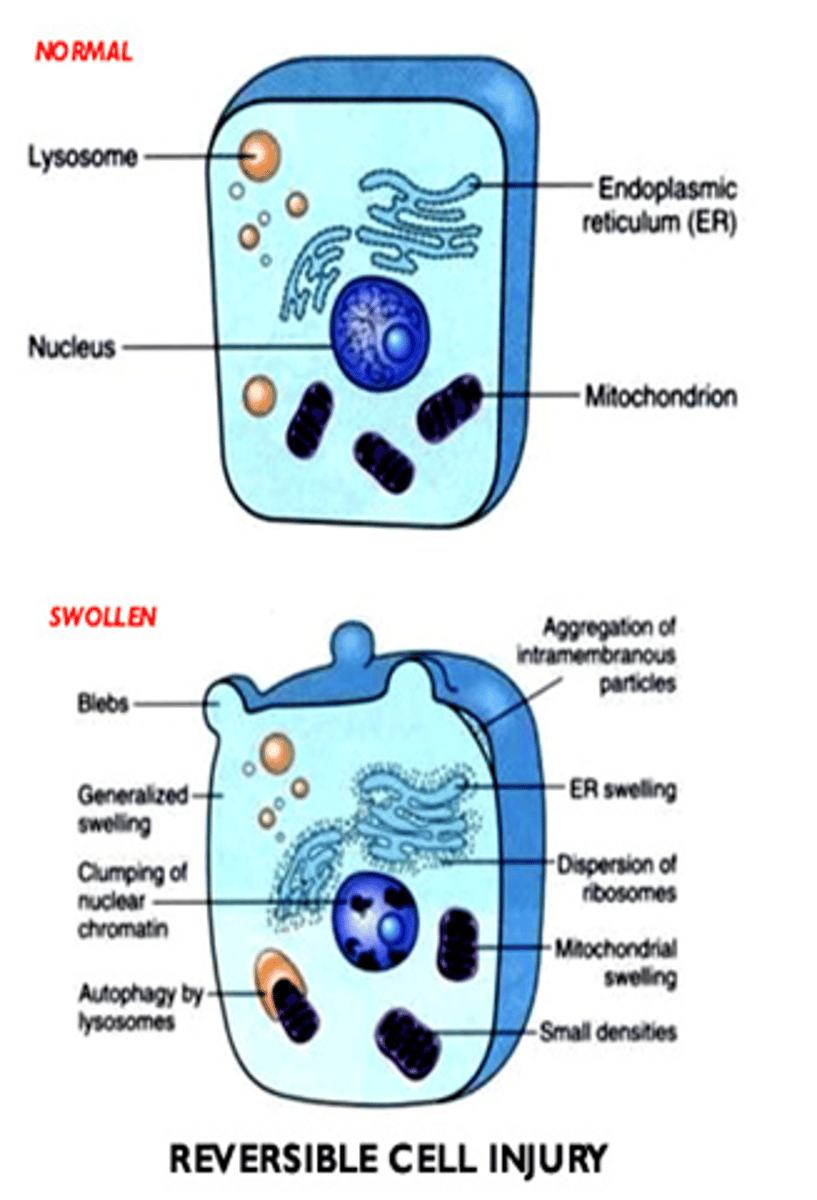
What changes will be seen in the ER in a reversible injury?
dilation of the ER, detachment of polysomes, and intracytoplasmic myelin figures
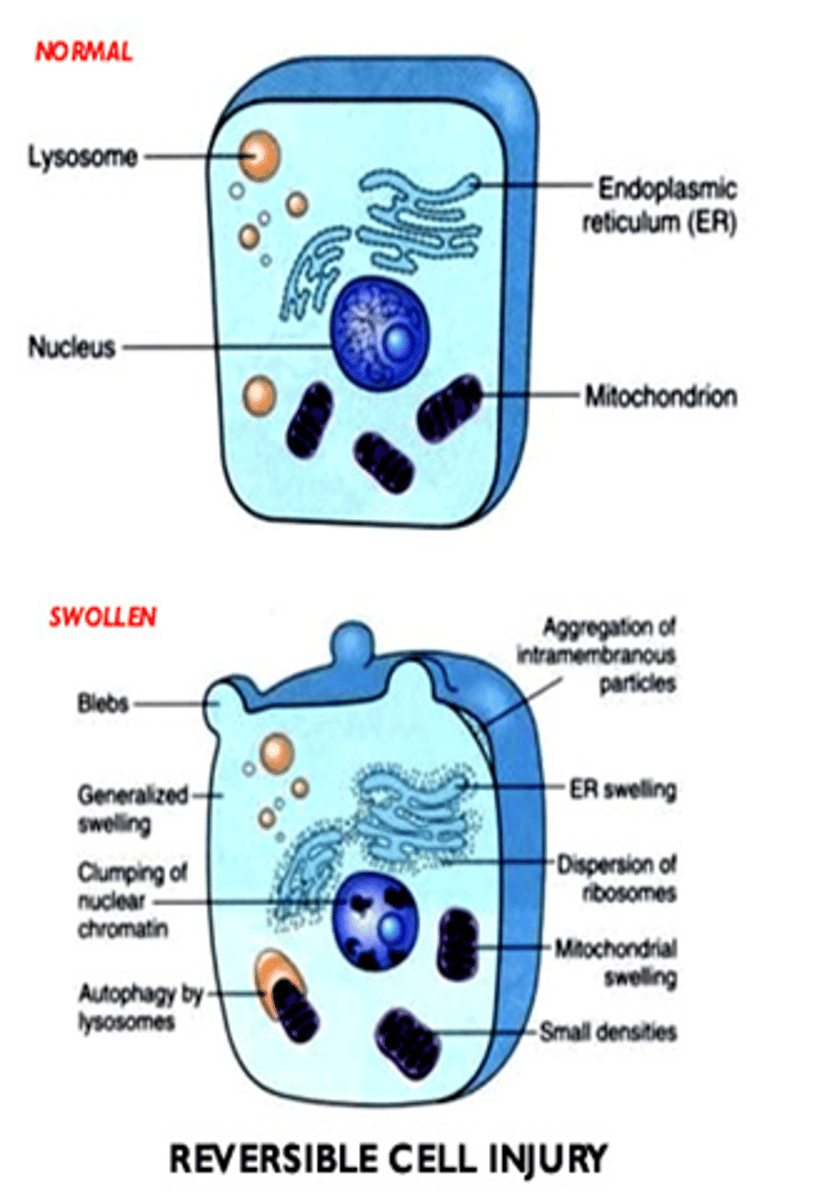
What nuclear alterations will be seen in a reversible injury?
disaggregation of granular and fibrillar elements
What will be the result of continuous damage to a cell?
cell injury will become irreversible, cell cannot recover and it dies
What are the two principle types of cell death?
necrosis and apoptosis
Understand the differences between necrosis and apoptosis
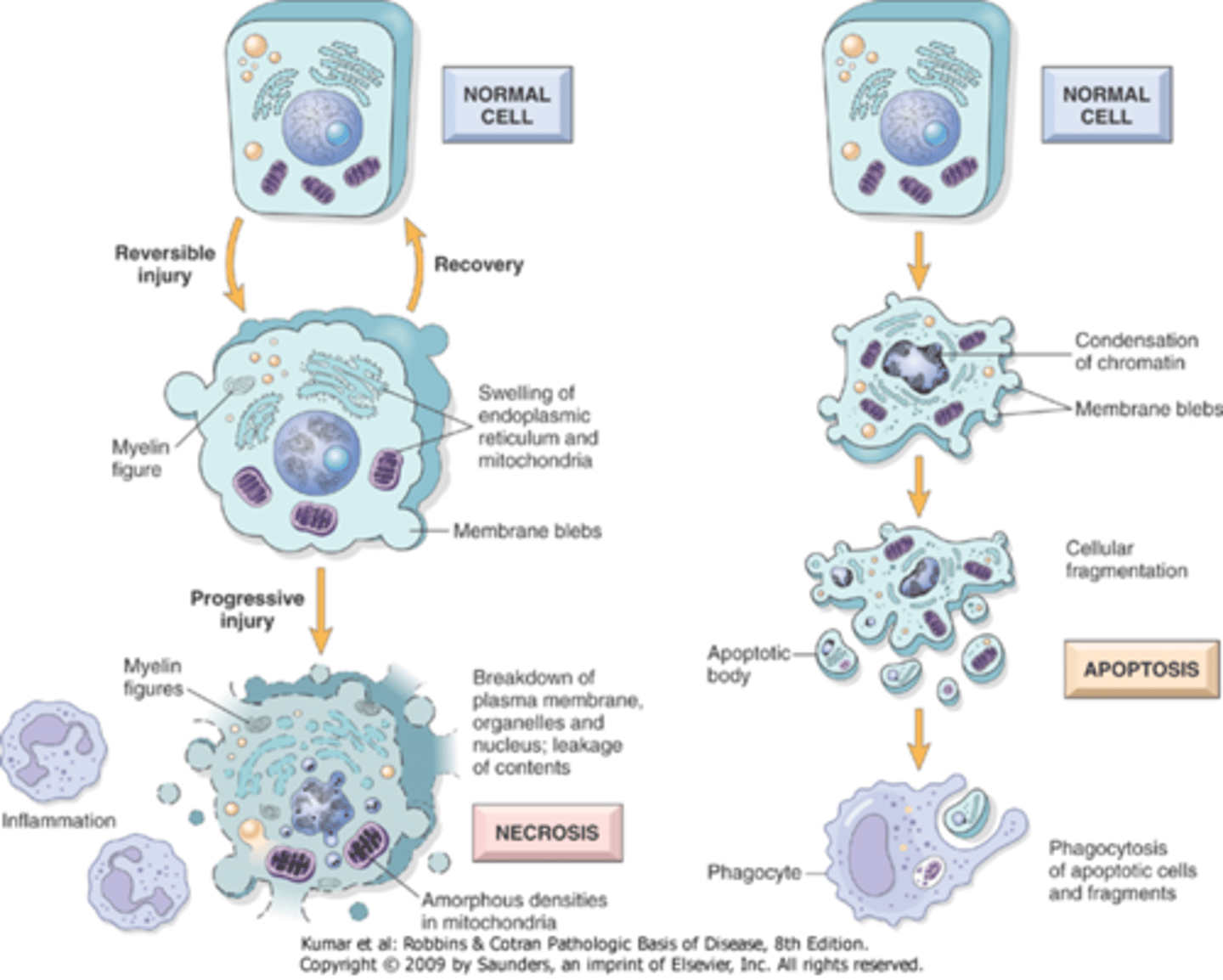
What is the key difference between apoptosis and necrosis?
apoptosis is regulated and non-inflammatory; necrosis causes inflammation (pathological)
What occurs to the cell during necrosis?
severe membrane damage, lysosomal enzymes enter the cytoplasm and digest the cell, cellular contents leak out
What causes the morphologic appearance of necrosis?
result of denaturation of intracellular proteins and enzymatic digestion of lethally injured cell, may take hours to develop
What happens to the surrounding tissue when necrosis occurs?
inflammation due to contents leaking from necrotic cell, cell cannot maintain membrane integrity
How will necrotic cells appear different in H&E and what causes this?
necrotic cells have loss of cytoplasmic RNA (binds to blue dye), and denatured cytoplasmic proteins (bind to red dye)
What causes the glassy homogeneous appearance of necrotic cells?
loss of glycogen particles
What are myelin figures, what type of cell are they seen in, and how are they disposed of?
whorled phospholipid masses seen in necrotic cells
Derived from damages cell membrane: phospholipid precipitates
Phagocytosed by other cells and further degraded into FA
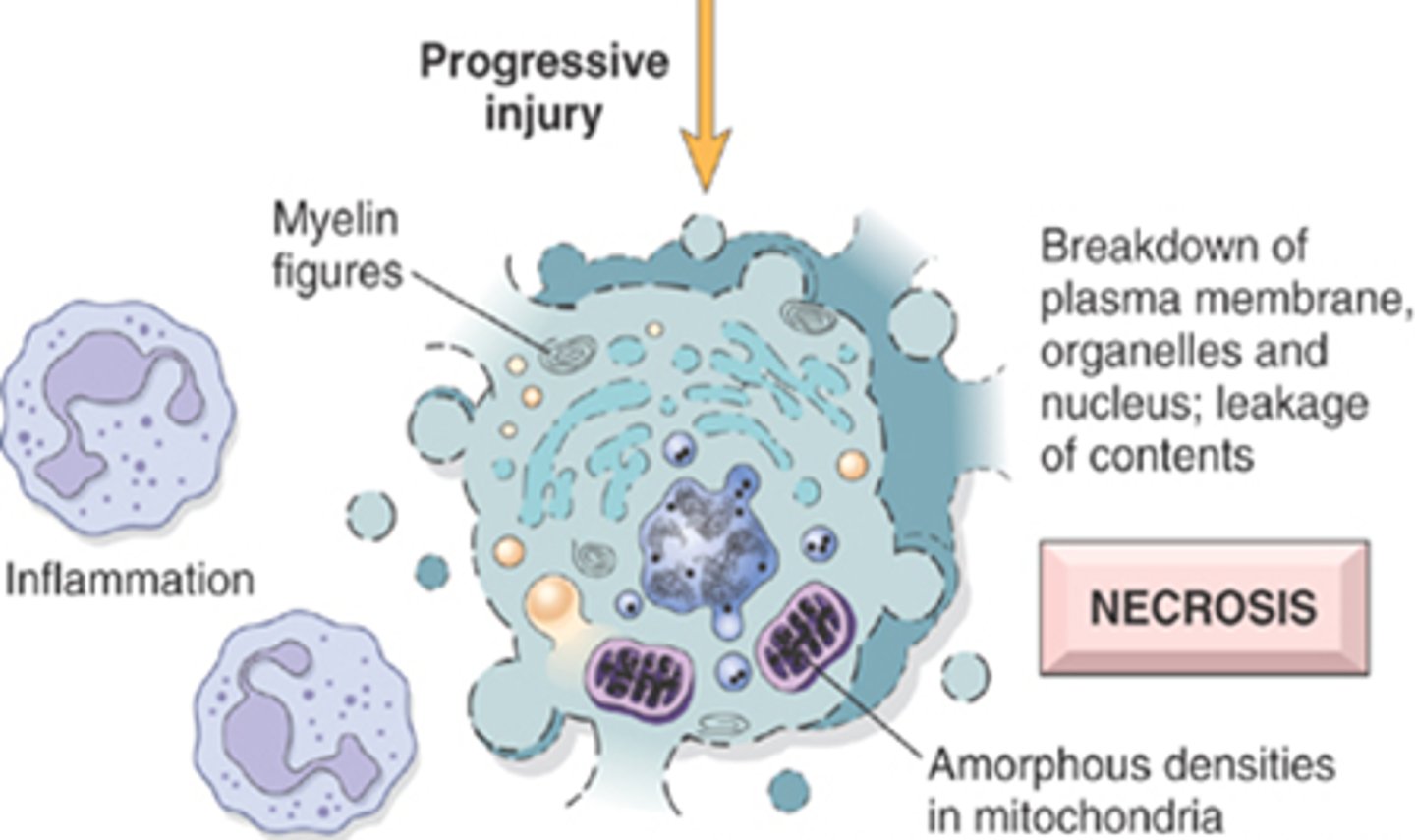
What causes the moth-eaten appearance of necrotic cells?
digestion of cytoplasmic organelles leaving a vacuolated cytoplasm
What will replace cells that have died from necrosis?
large whorled phospholipid masses (myelin figures) derived from damaged cell membranes
What causes the nuclear changes seen in necrotic cells?
nonspecific breakdown of DNA
List nuclear changes of necrotic cells
karyolysis, pyknosis, and karyorrhexis
What is Karyolysis?
fading of the basophilia of the chromatin
change that reflects loss of DNA because of enzymatic degradation by endonucleases
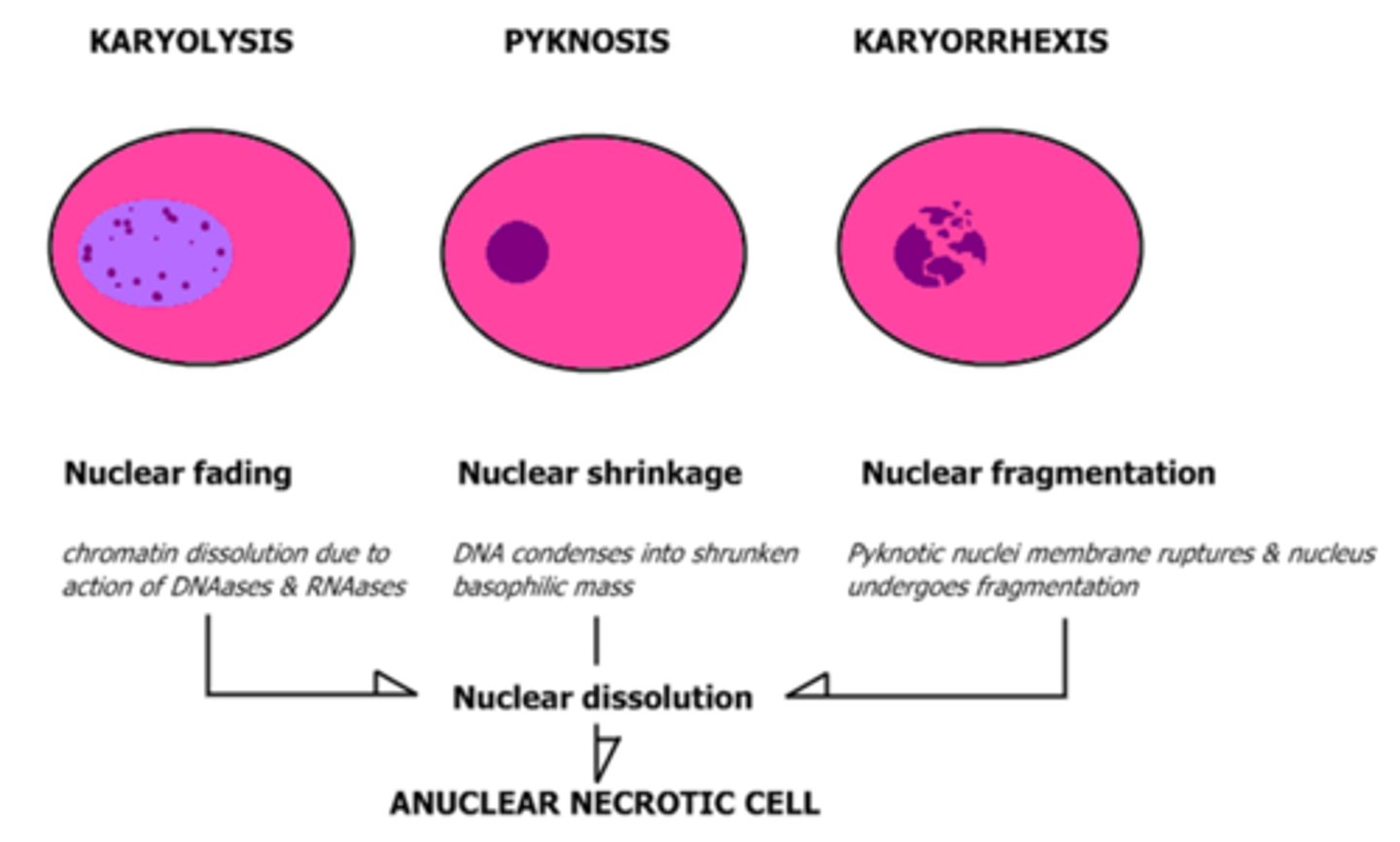
What occurs during pyknosis?
nuclear shrinkage, increased basophilia
chromatin condenses into solid shrunken basophilic mass
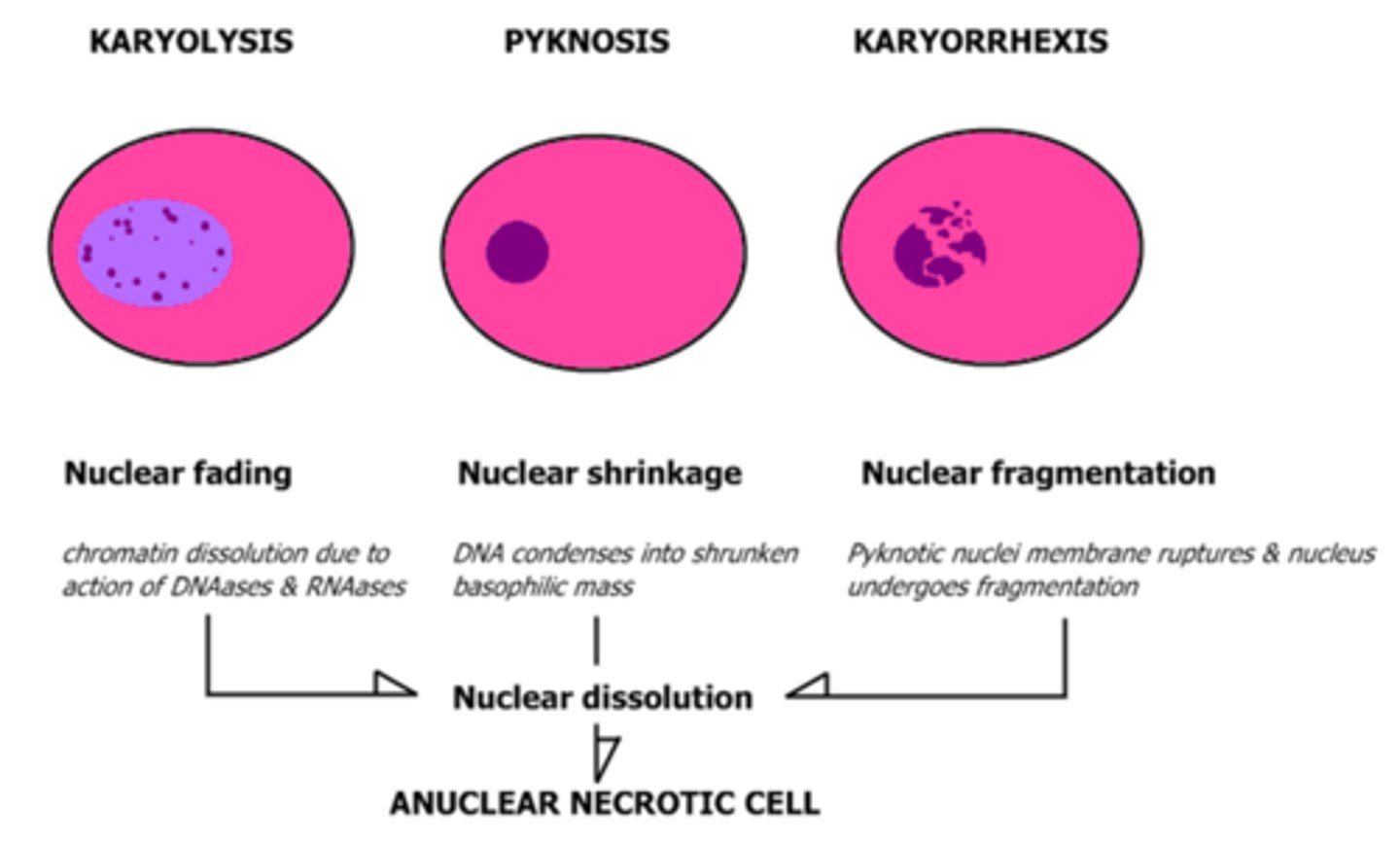
What occurs in karyorrhexis?
pyknotic nucleus undergoes fragmentation
nucleus of the necrotic cell will totally disappear in 1-2 days
What is coagulative necrosis?
architecture of dead tissues that displays a firm texture
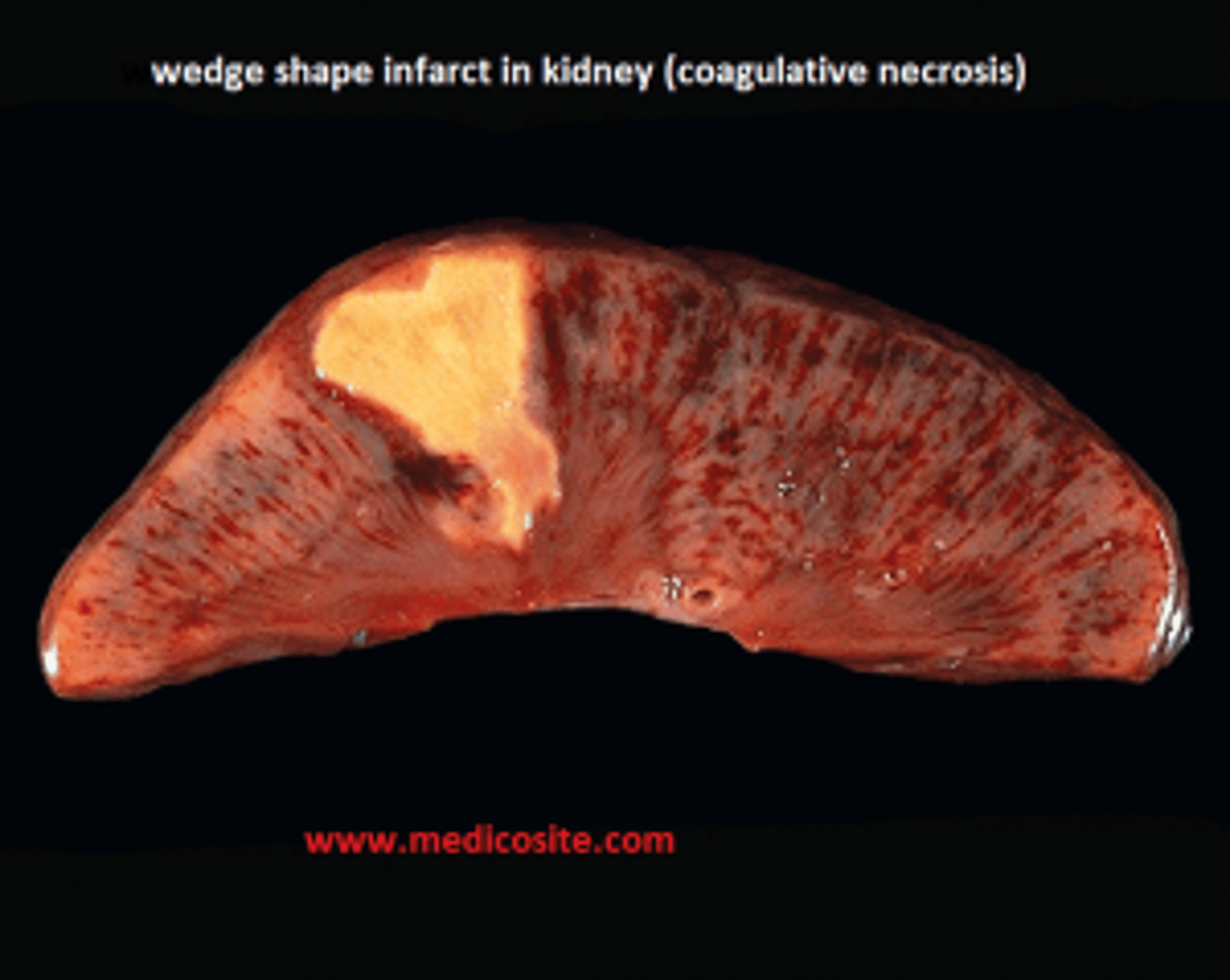
How long will coagulative necrosis persist and how is it removed?
persist for days or weeks, cellular debris removed by phagocytosis by infiltrating leukocytes
digestion of the dead cells by the action of lysosomal enzymes and leukocytes
Give an example of coagulative necrosis
ischemia caused by obstruction in a vessel may lead to coagulative necrosis of the supplied tissue
Infarct
What is an infarct, and what type of necrosis is associated?
necrosis of a localized area (caused by ischemia), can lead to coagulative necrosis
in this pick note the pink anucleate cells characteristic of coagulative necrosis
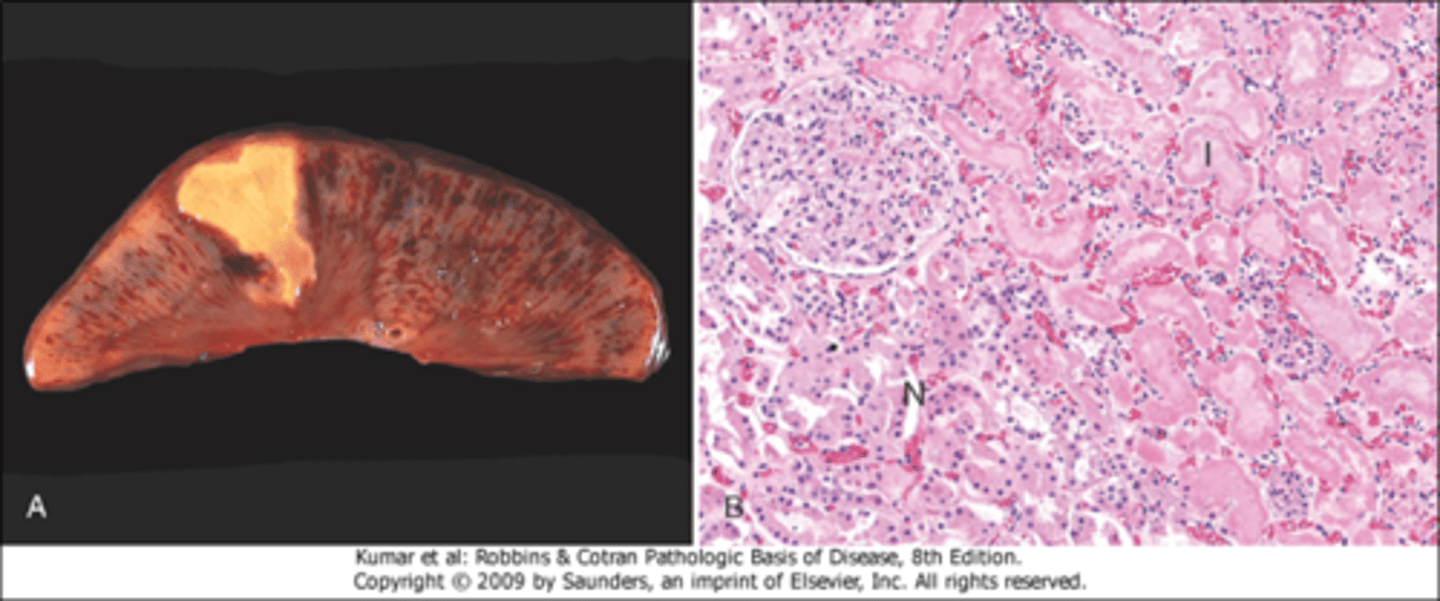
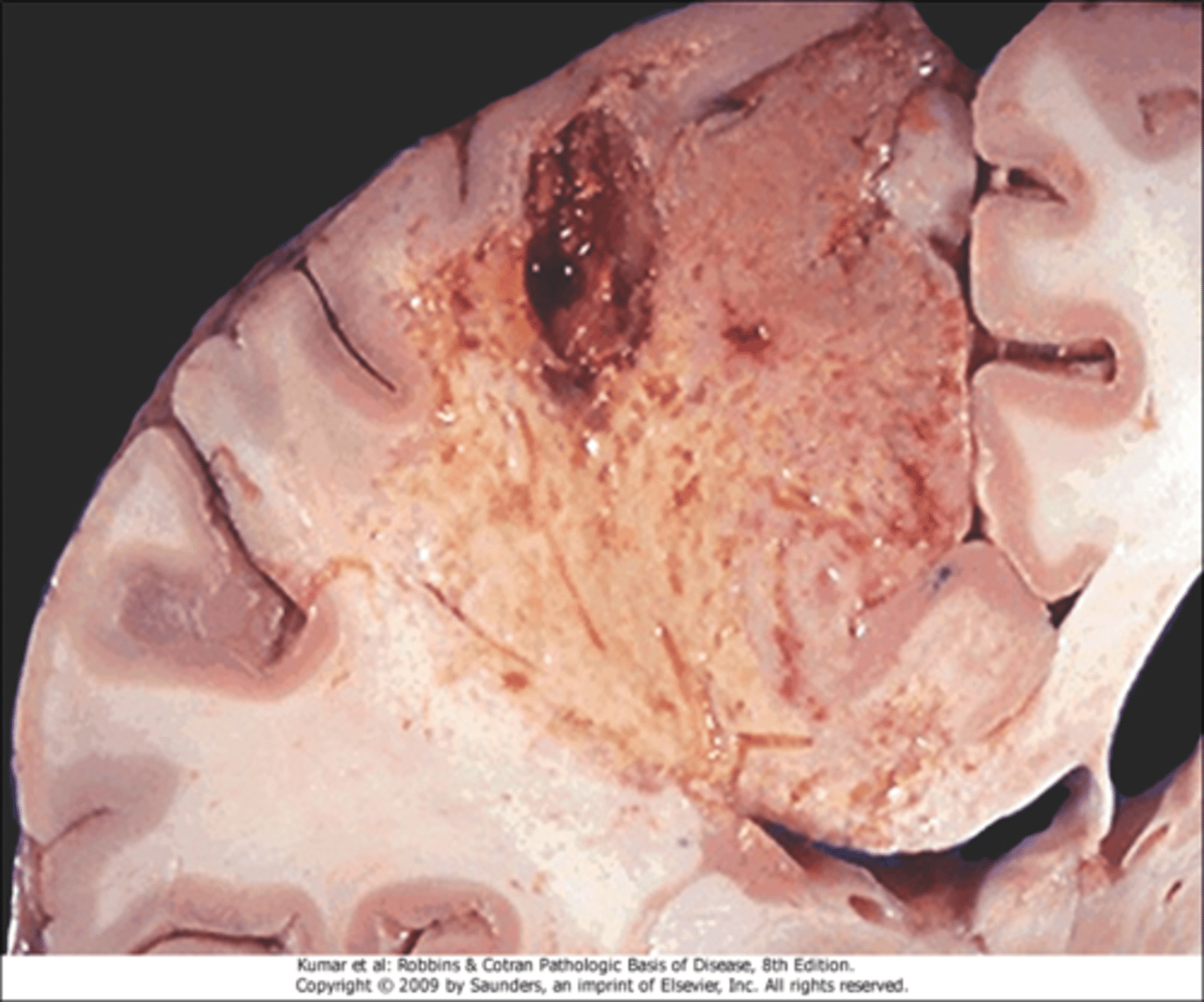
What is liquefactive necrosis?
transformation of tissue into a liquid viscous mass (characterized by digestion of the dead cells)
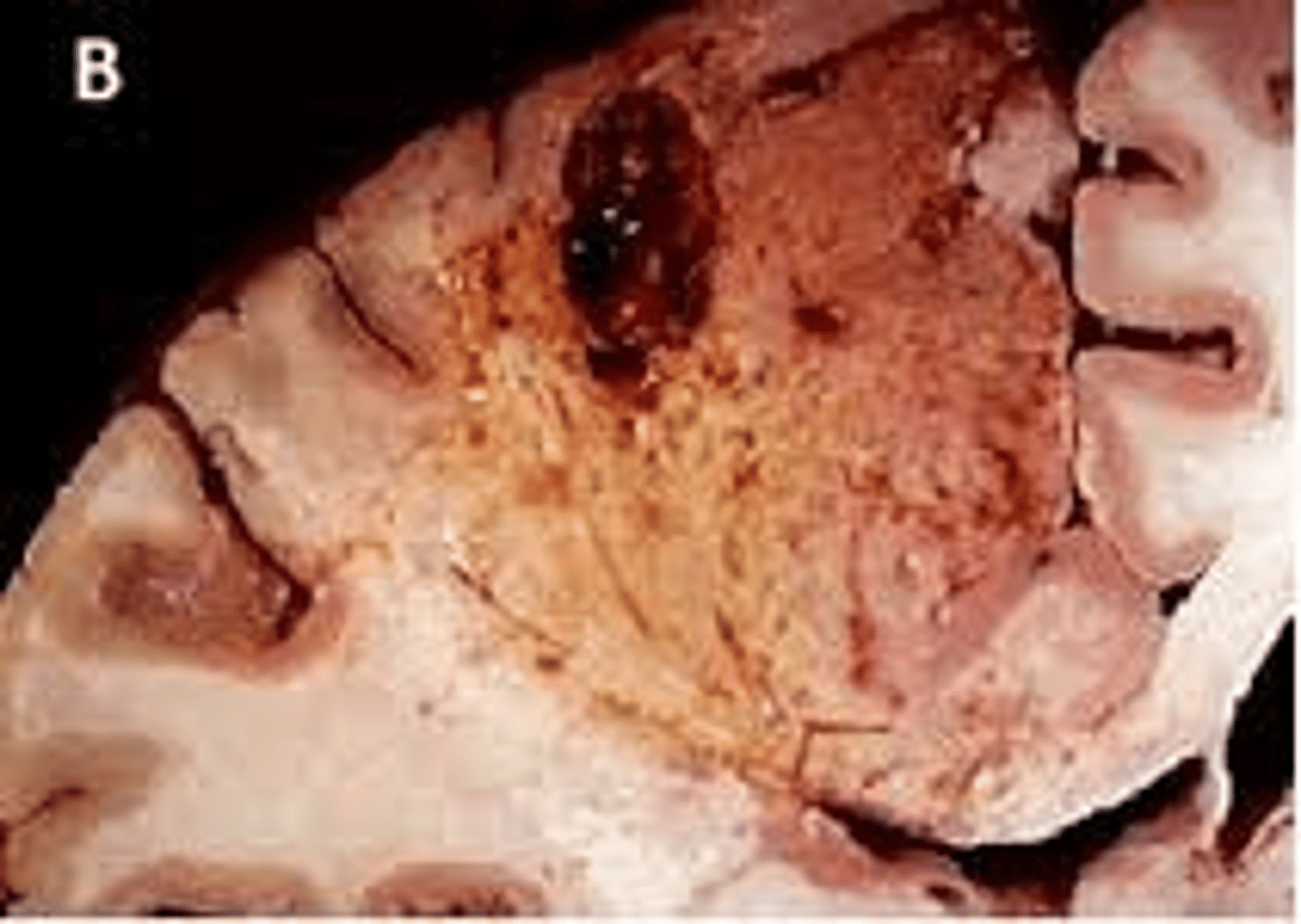
Where will liquefactive necrosis be seen?
focal bacterial infections and occasionally in fungal infections
What is the gross appearance of liquefactive necrosis?
creamy yellow due to dead leuocytes and purulent matter
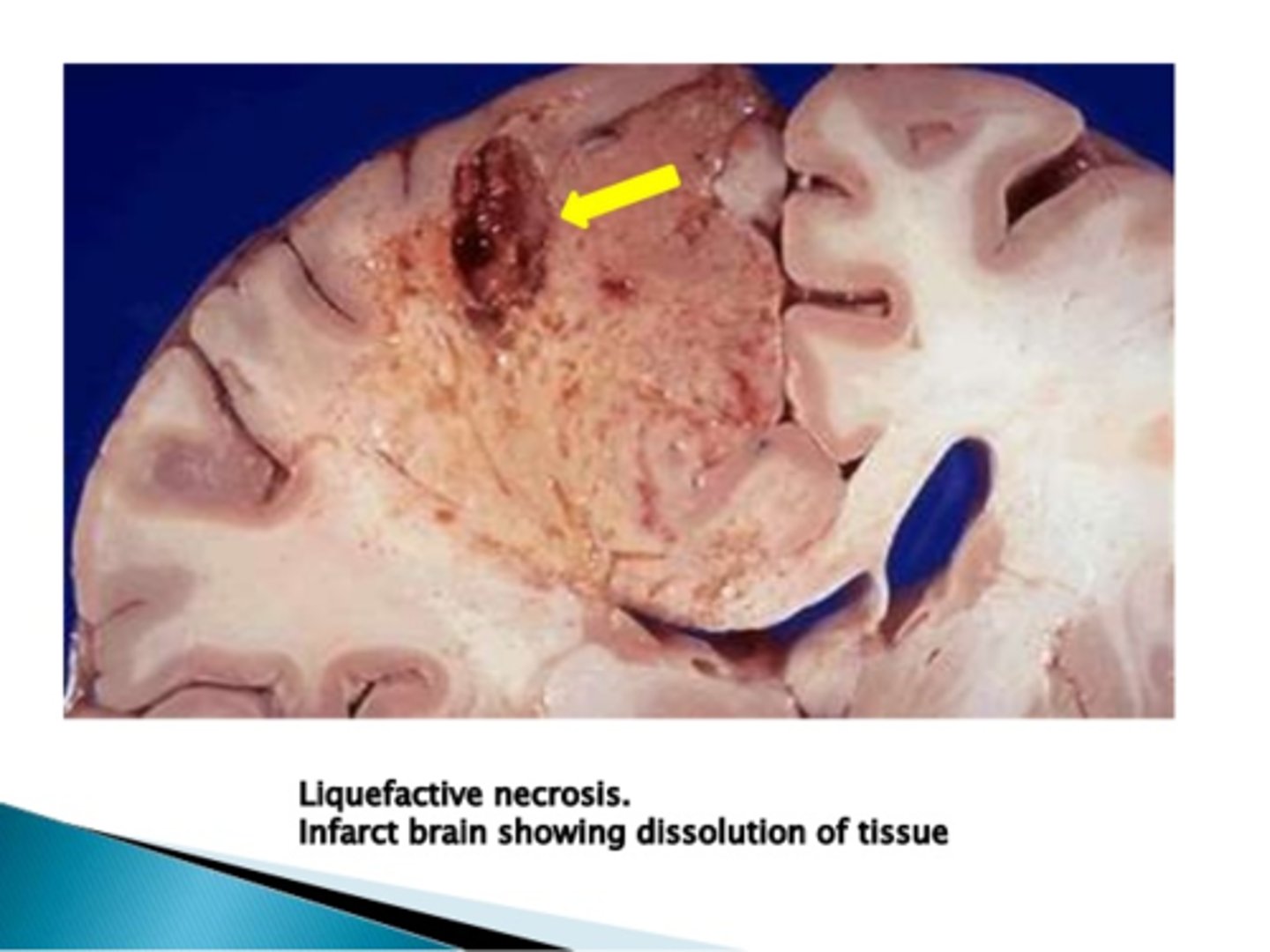
What type of necrosis is hypoxic death of cells in the CNS?
liquefactive necrosis
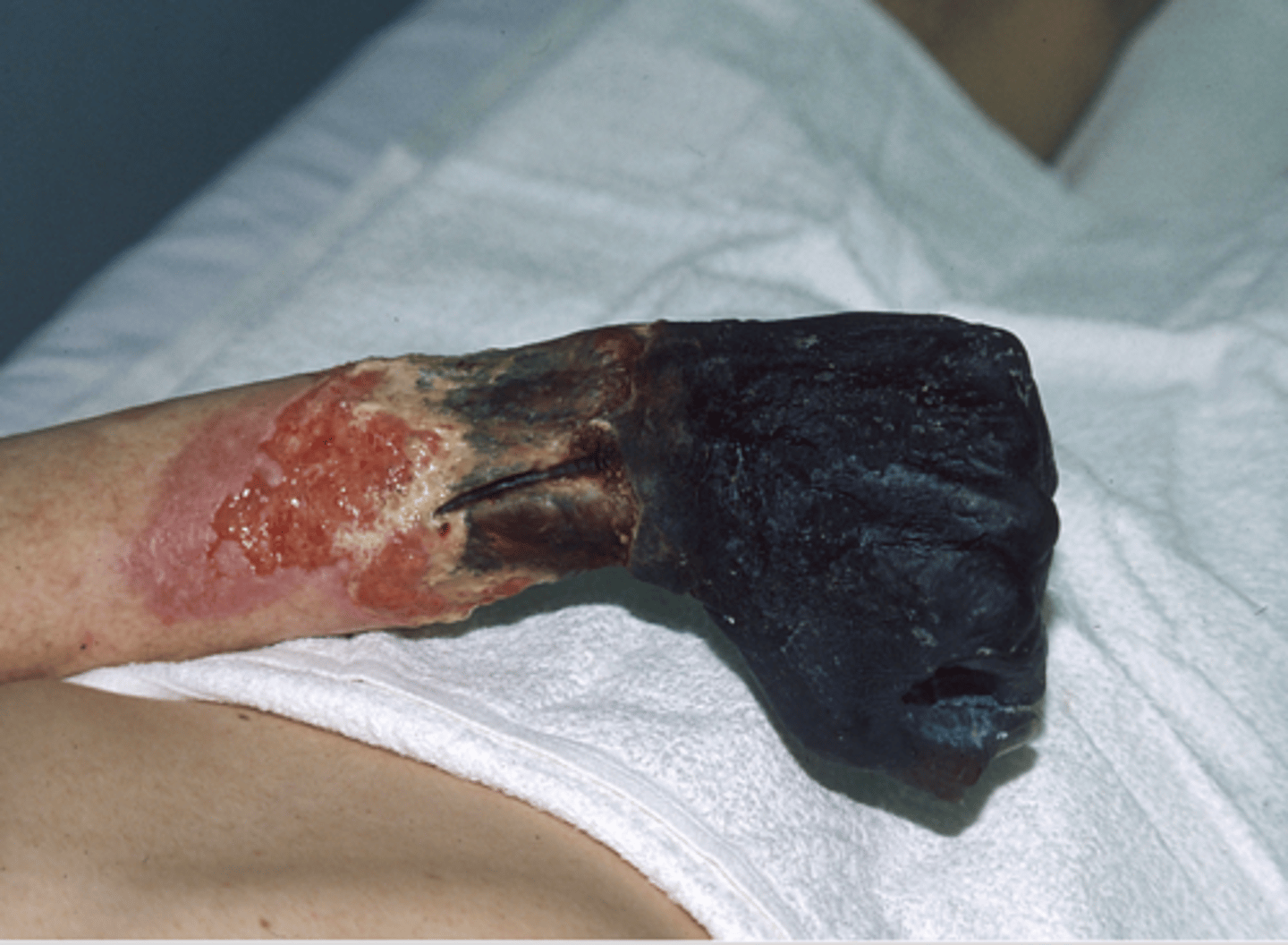
What area of the body will gangrenous necrosis most likely be seen?
limb (usually lower limb)
What is the typical cause of gangrene?
loss of blood supply to the area leading to necrosis
What type of necrosis would be seen in a gangrenous situation in which there was not bacteria?
typically coagulative necrosis involving multiple tissue planes
think wet gangrene - liquafactive
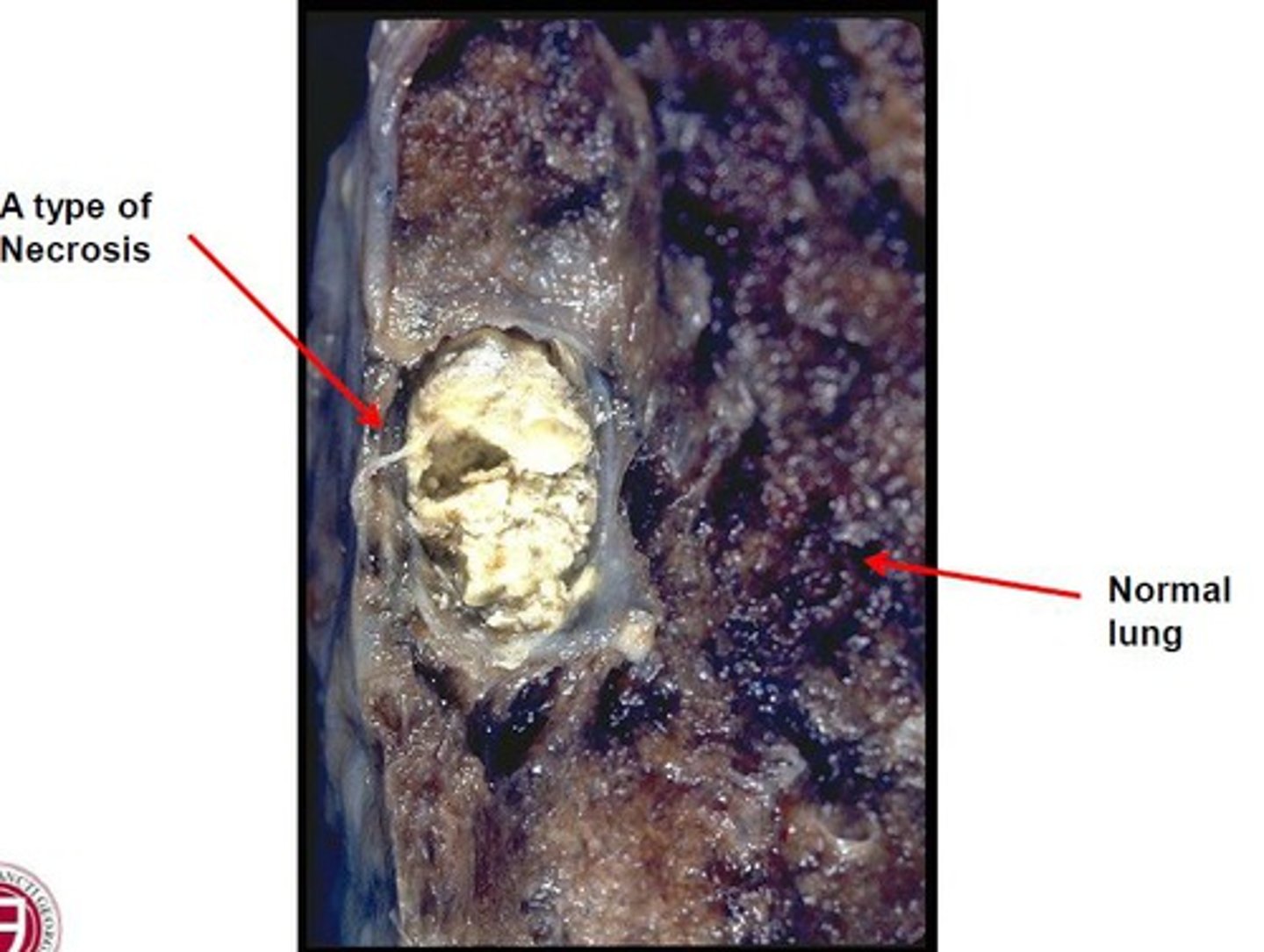
Where is caseous necrosis most often encountered?
Tuberculous cavity (lungs) seen in tuberculosis infections
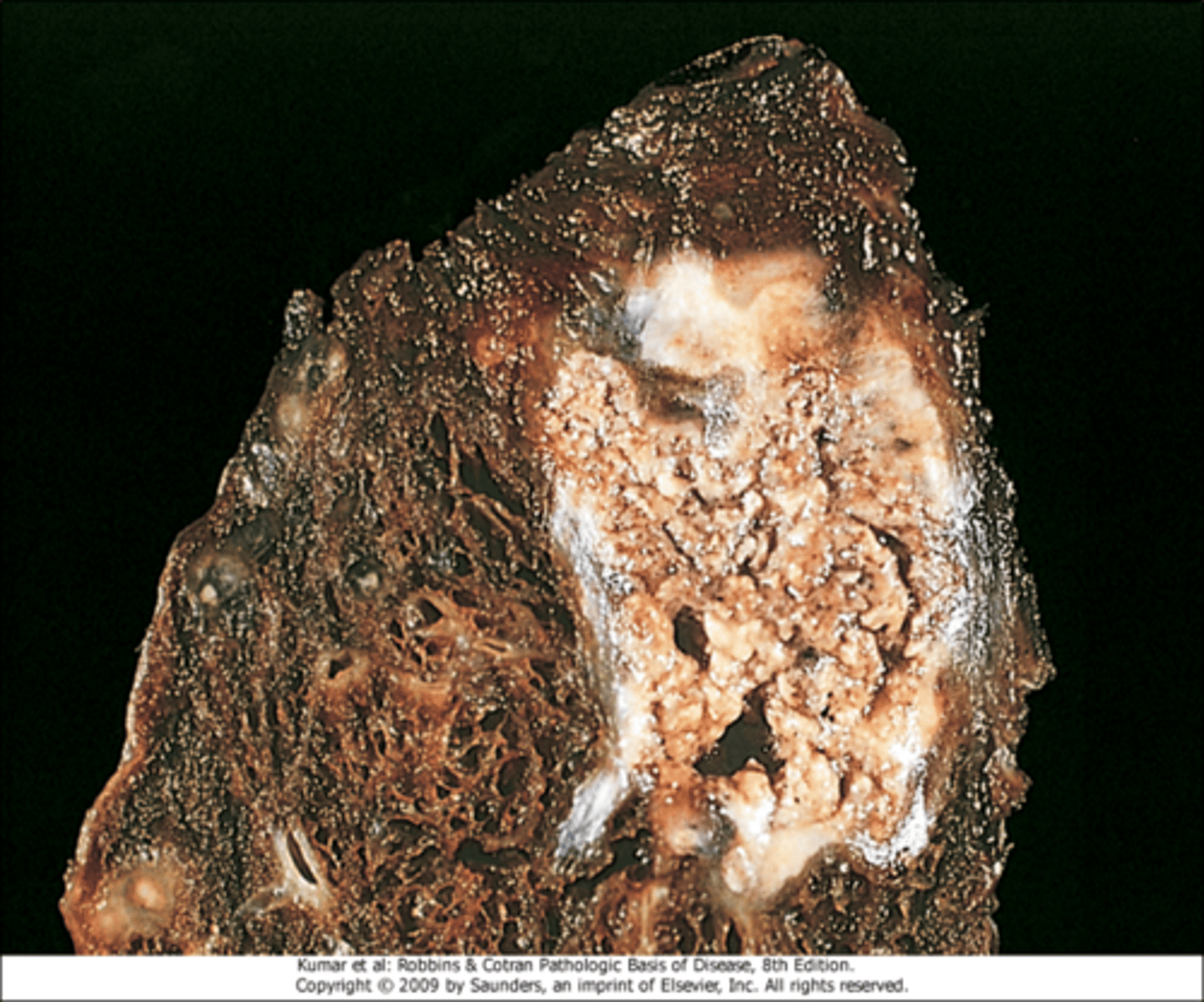
Describe the appearance of caseous necrosis
cheeselike, friable white appearance of the area of necrosis
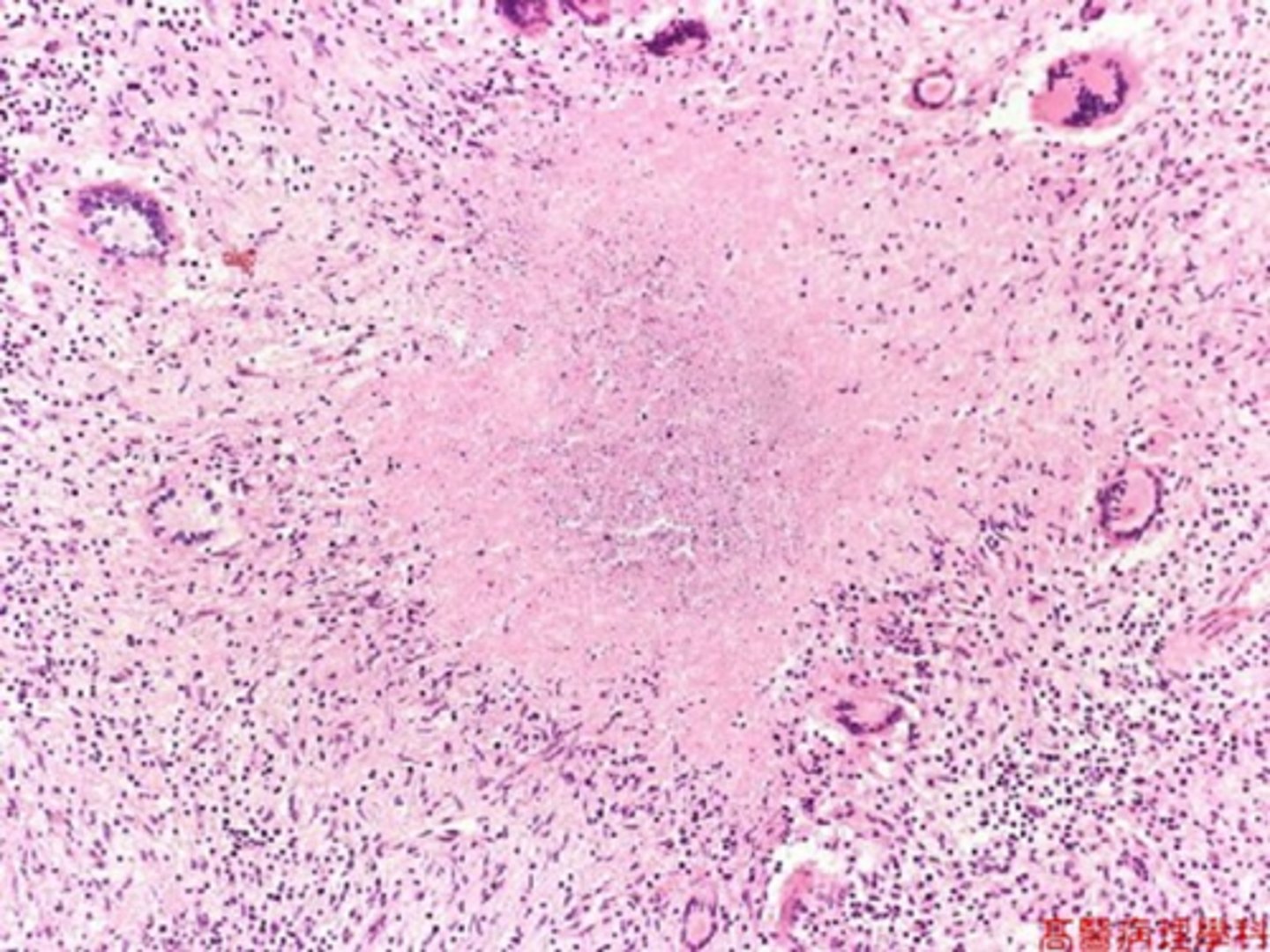
Describe what will be seen upon microscopic examination of caseous necrosis
collection of fragmented or lysed cells, amorphous granular debris enclosed within distinctive inflammatory border (granuloma)
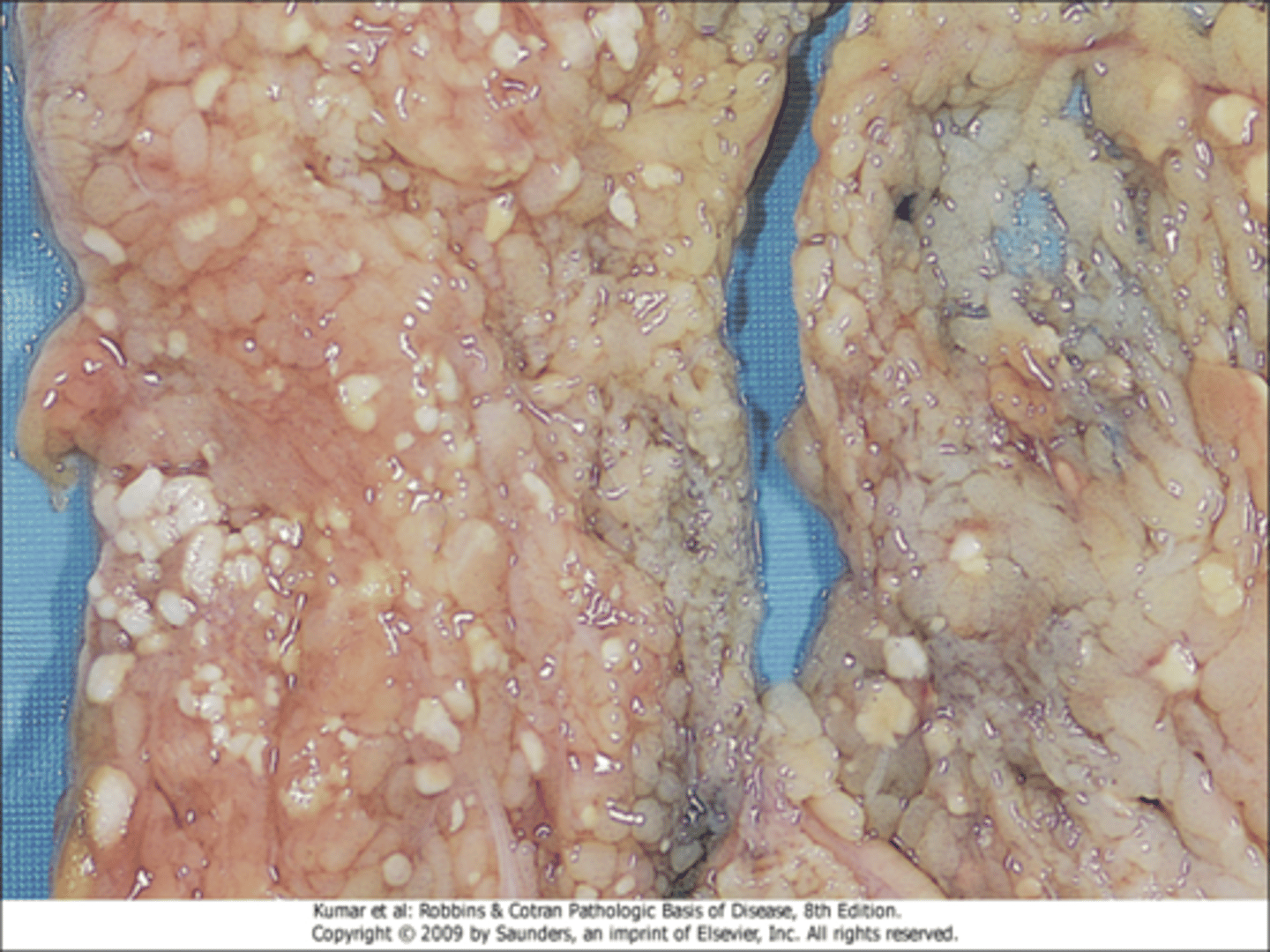
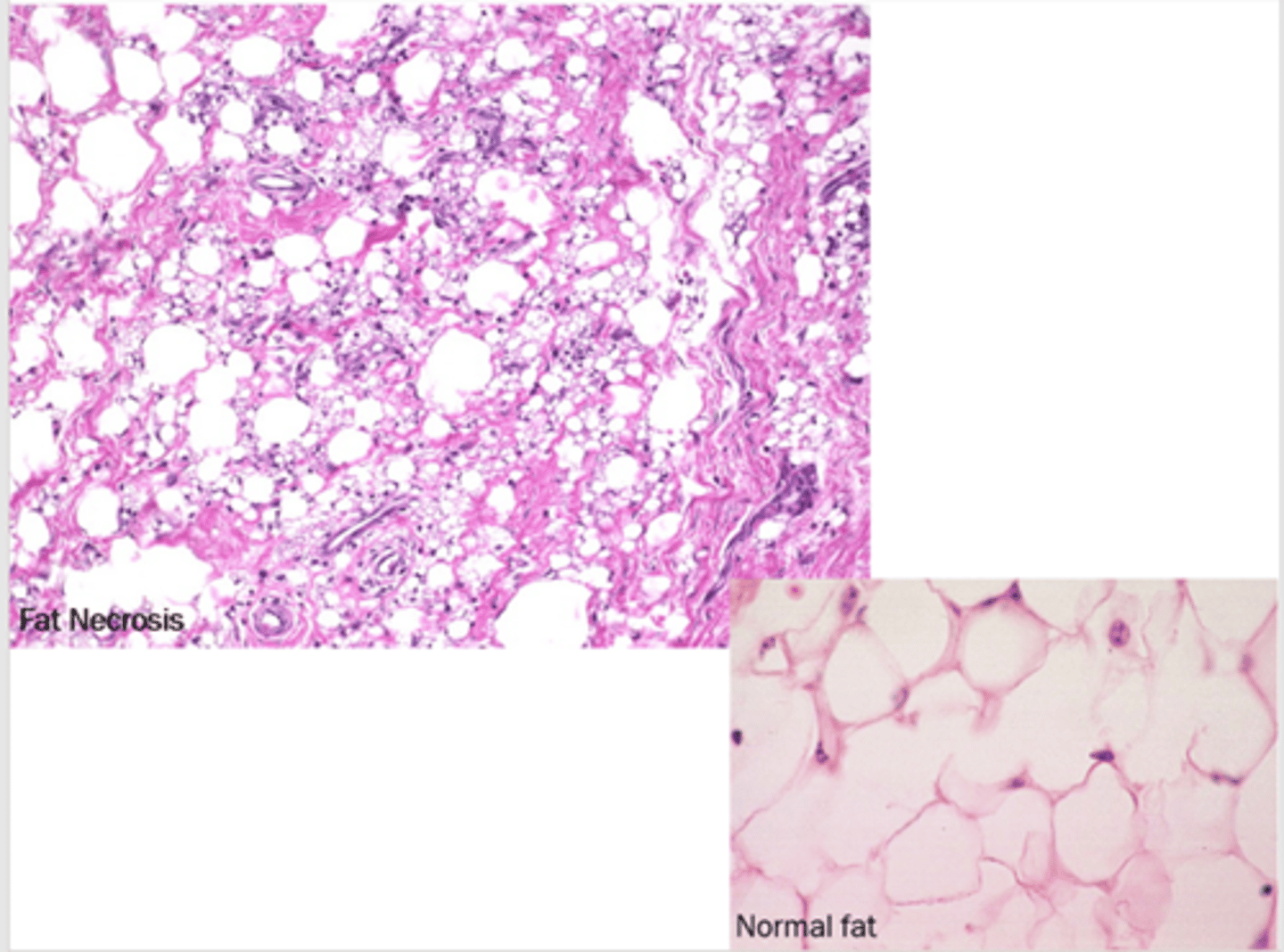
What will be seen upon examination of fat necrosis?
focal areas of fat destruction, release of activated pancreatic lipases into the substance of the pancreas and the peritoneal cavity
What will be seen upon microscopic examination of fat necrosis?
foci of shadowy outlines of necrotic fat cells, basophilic calcium deposits, and inflammatory reaction
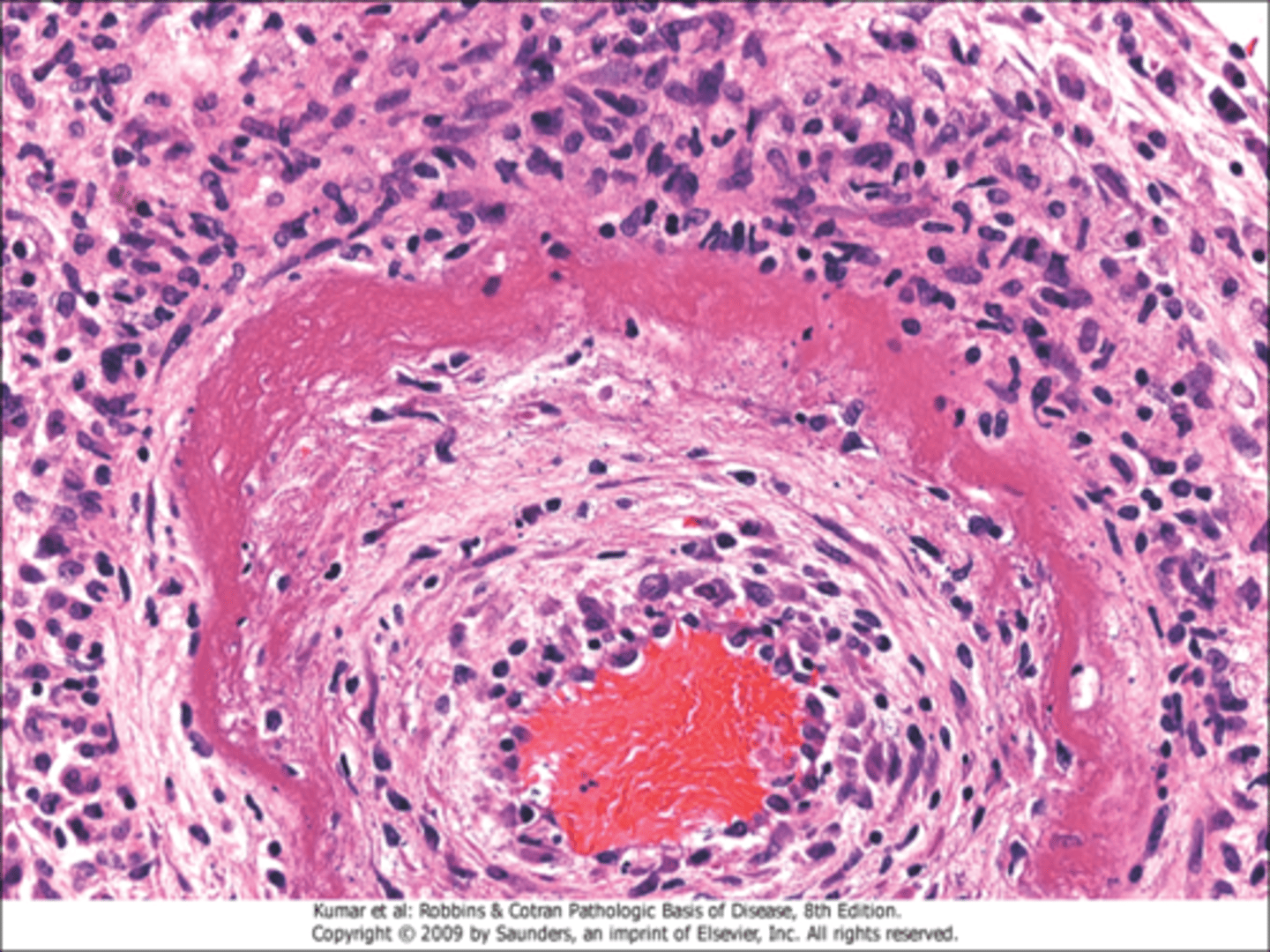
What is fibrinoid necrosis?
special form of necrosis seen in immune reaction involving blood vessels
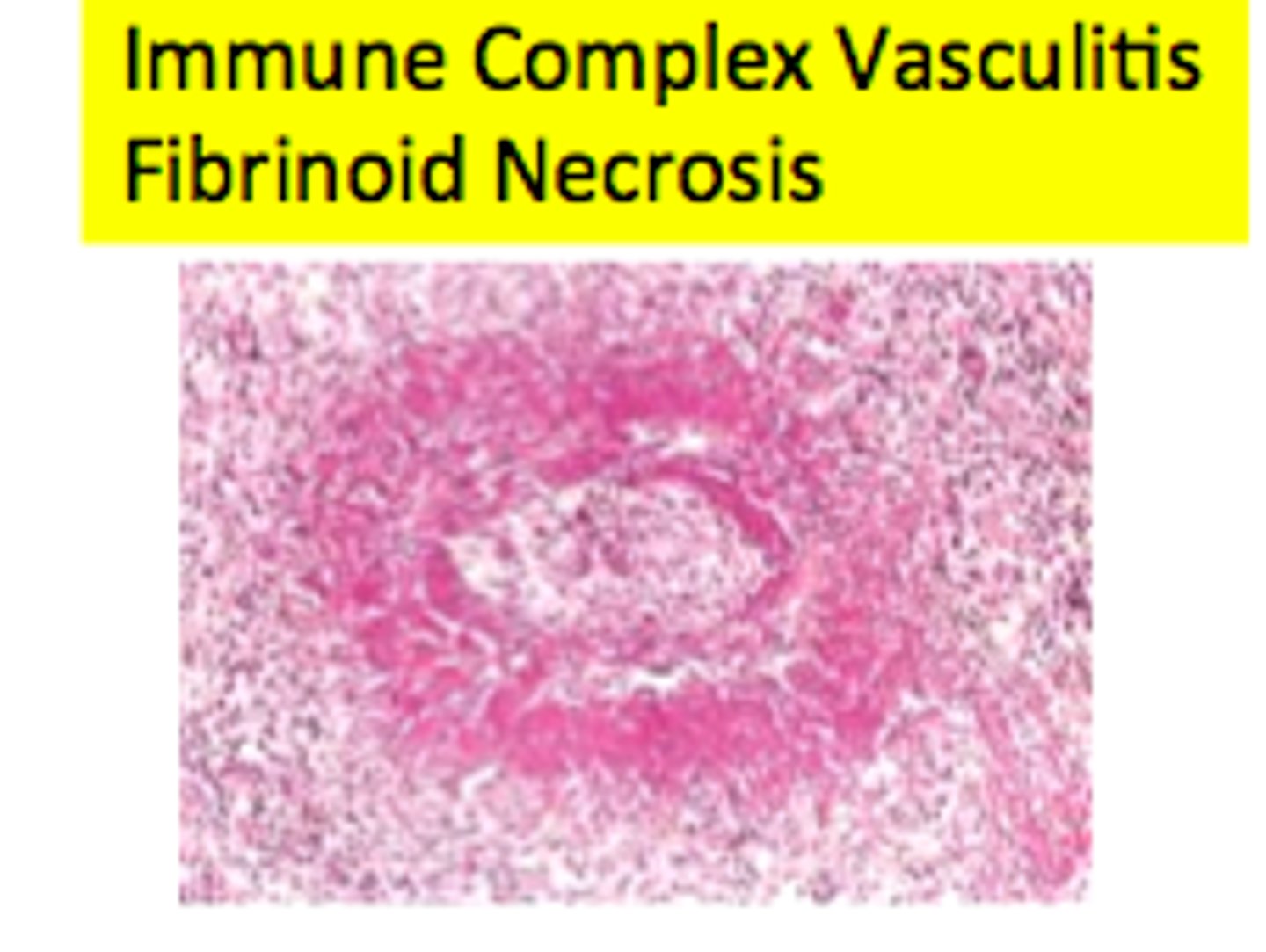
What is seen in arteries in fibrinoid necrosis?
complexes of antigens and antibodies deposited in the walls of arteries
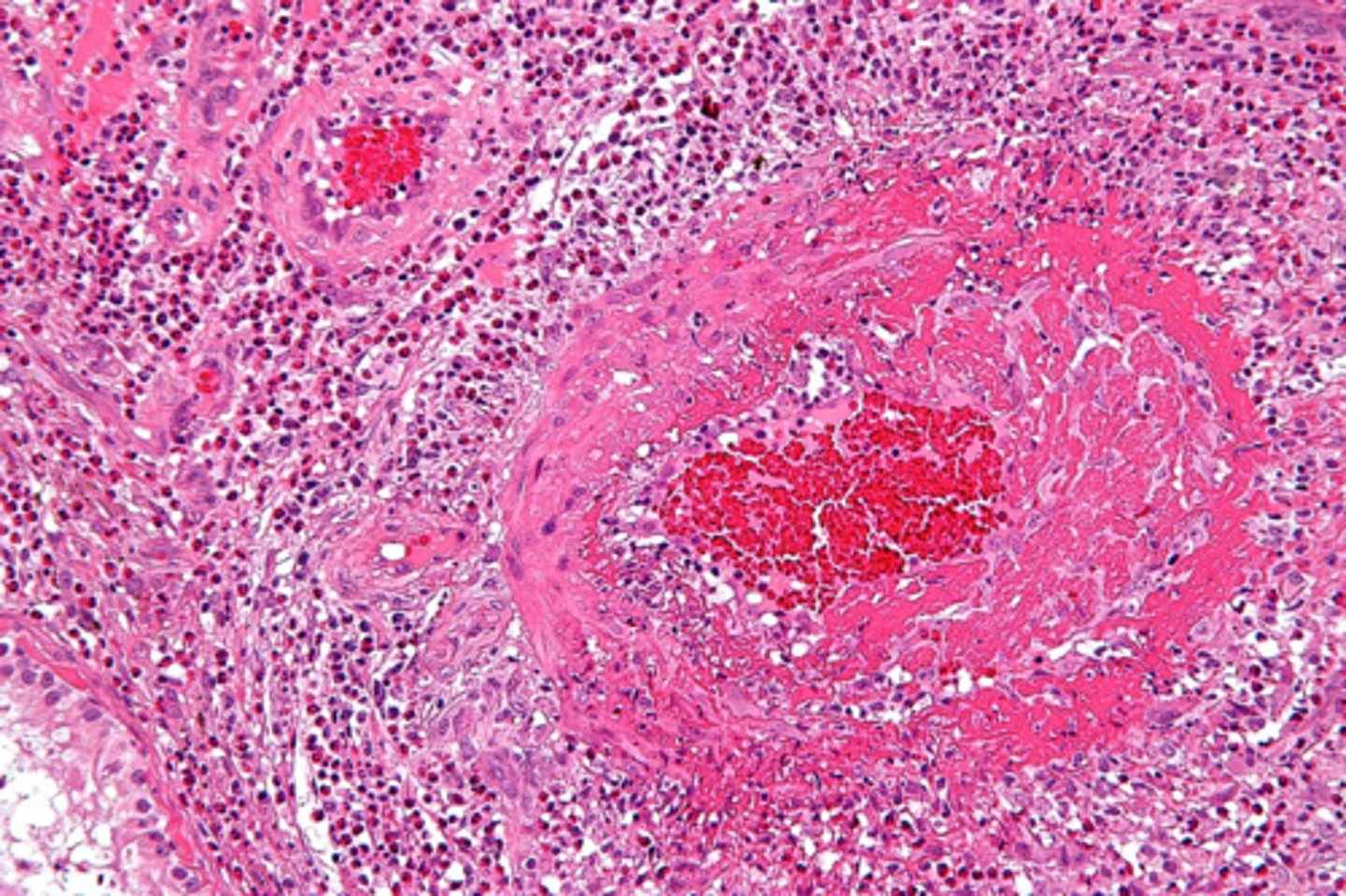
What will be seen upon microscopic examination of fibrinoid necrosis?
deposits of immune complexes and fibrin, bright pink amorphous appearance ("fibrinoid")
What does cellular response to injurious stimuli depend upon?
- nature of the injury, it's duration and it's severity
- small doses of toxin or brief ischemia: may induce reversible injury
- large doses or prolonged ischemia: instantaneous death, slow irreversible injury leading to cell death
What is an example of the difference between skeletal and cardiac muscle in terms of cell injury
skeletal muscle deprived of it's blood supply can be placed at rest and preserved while cardiac muscle cannot
What mechanisms do cell injury result from?
different biochemical mechanisms
act on several essential cellular components such as mitochondria, cell membranes, and DNA in nuclei
What is the major and secondary pathway in which ATP will be made?
major: oxiadtive phosphorylation of adenosine diphosphate
secondary: glycolytic pathway, works in absence of oxygen and uses glucose
What results from oxidative phosphorylation and where does this occur?
reduction of oxygen through electron transfer system of mitochondria
How does the glycolytic pathway work?
generates ATP in the absence of oxygen, uses glucose derived from body fluids or hydrolysis of glycogen
What type of injury is ATP depletion associated with?
both hypoxic and chemical injury
What are the major causes of depletion of ATP?
reduced supply of oxygen and nutrients, mitochondrial damage, and actions of toxins (ex: cyanide)
Why is ATP depletion detrimental?
involved in virtually all synthetic and degradative processes within the cell
depletion of just 5-10% can have widespread effects
How will depletion of ATP affect the sodium potassium pump?
reduced activity of sodium potassium pump, sodium accumulates within the cell, water will enter leading to cell swelling and dilation of the ER
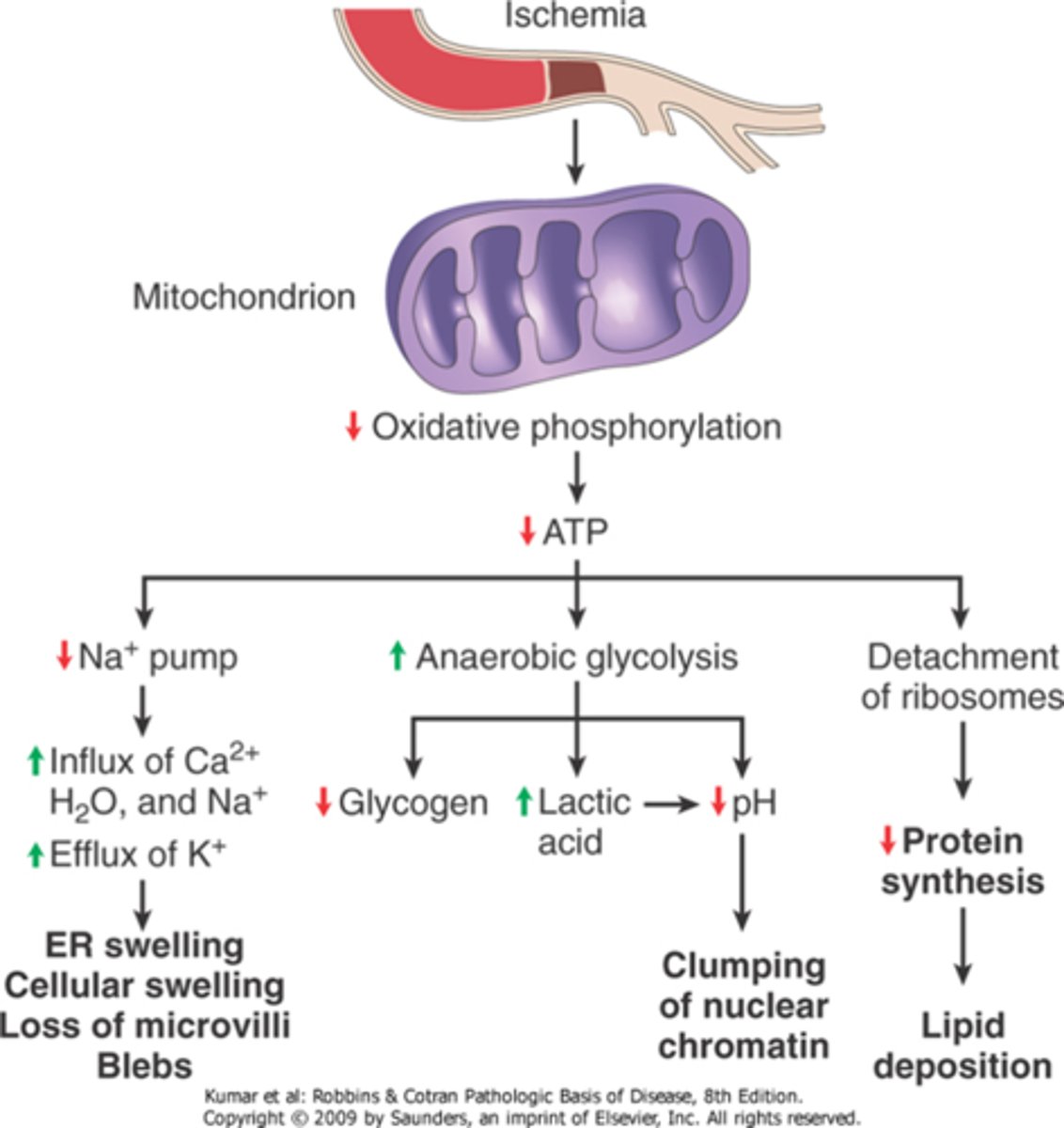
How does ischemia affect oxidative phosphorylation?
oxidative phosphorylation ceases without O2, decrease in cellular ATP, increase in adenosine monophosphate, and glycogen stores are rapidly depleted
How can ATP depletion affect the Ca2+ pump and how will this change with prolonged depletion?
influx of Ca2+ damages intracellular organelles
prolonged worsening depletion of ATP: detachment of ribosomes from the rough ER, reduced protein synthesis (dissociation of polysomes)
What may be the result of oxygen or glucose deprivation within a cell?
proteins may become misfolded which will trigger a cellular reaction leading to cell injury or death
How would a cell respond to irreversible damage to the mitochondrial and lysosomal membranes?
cell necrosis
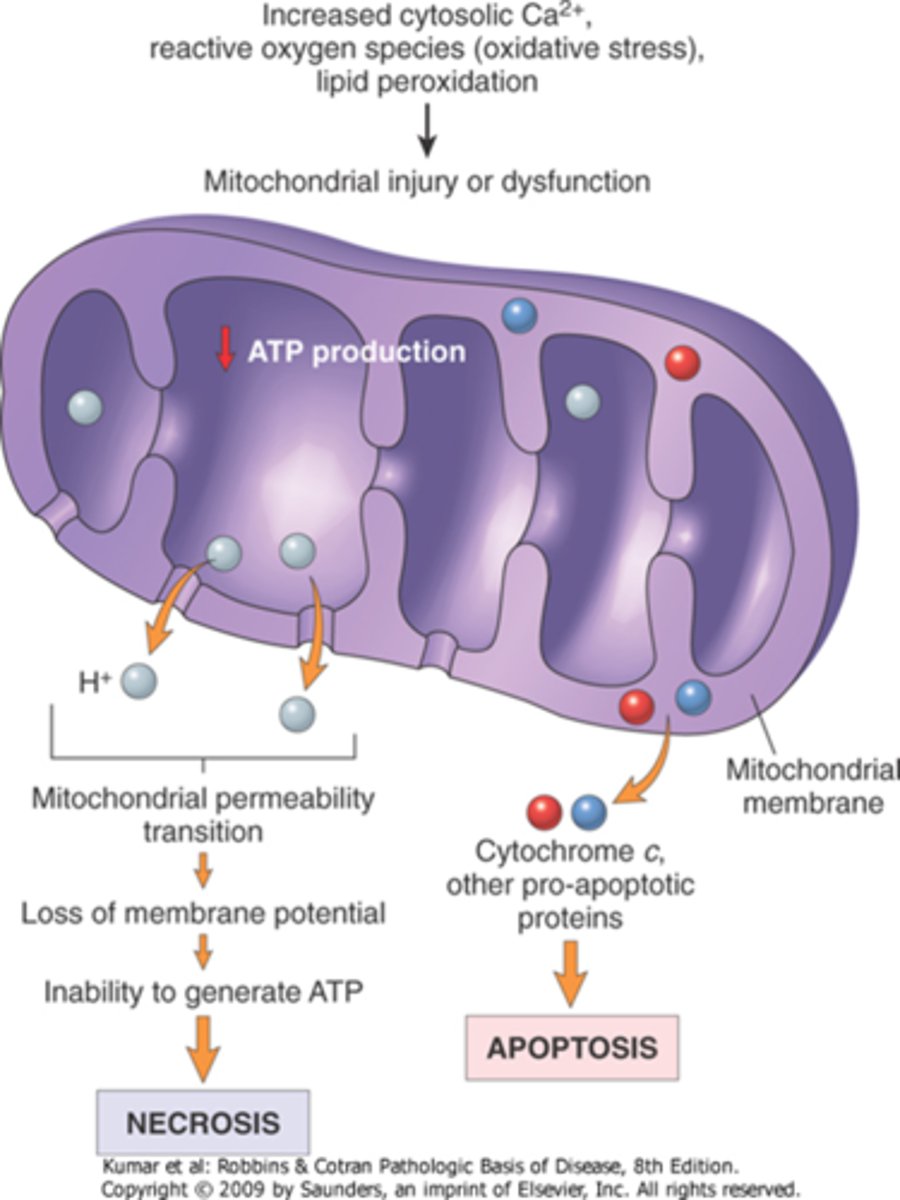
How will ischemia affect the mitochondria and it's functions
Functional and morphologic consequences of decreased intracellular ATP during cell injury.
The morphologic changes shown here are indicative of reversible cell injury. Further depletion of ATP results in cell death, typically by necrosis. ER, endoplasmic reticulum.
List examples of things that can cause damage to mitochondria
increases of cytosolic Ca2+, reactive oxygen species, and oxygen deprivation
can also see mutations in mitochondrial genes cause of some inherited disease
How will formation of a high-conductance channel affect the mitochondria?
opening of the channel leads to a loss of mitochonrial membrane potential, results in failure of oxidative phosphorylation and depletion of ATP, will lead to necrosis of cell
How do mitochondria activate apoptotic pathways?
cytochrome c and caspases indirectly activate apoptosis-inducing enzymes
increased permeability of the outer mitochondrial membrane, leakage of proteins into the cytosol, death by apoptosis
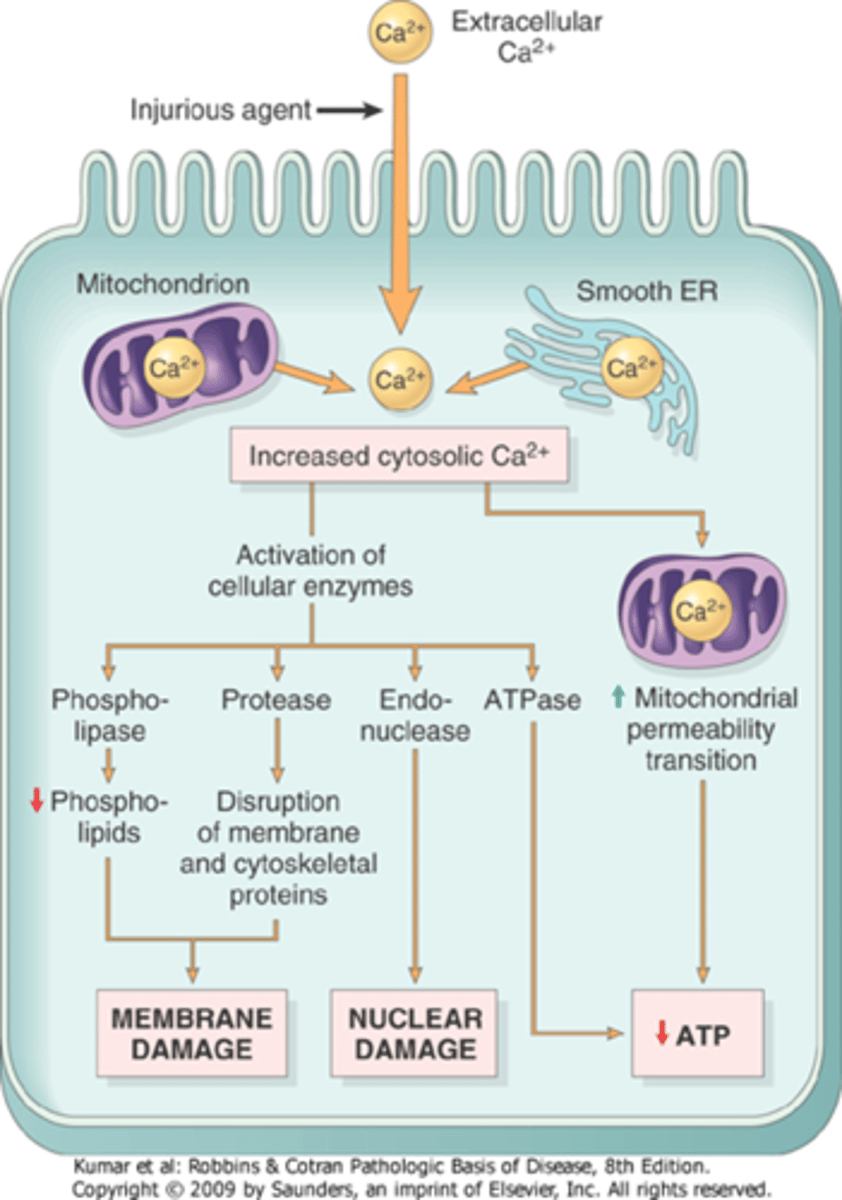
Where is intracellular calcium sequestered?
in mitochondria and the ER
What affect would increased cytosolic Ca2+ levels have?
activates a number of enzymes, has deletrious cellular effects, and phospholipases (membrane damage)
What will be released when there are increased cytosolic Ca2+ levels?
proteases: break down both membrane and cytoskeletal proteins
endonucleases: responsible for DNA and chromatin fragmentation
ATPases: speed up ATP depletion
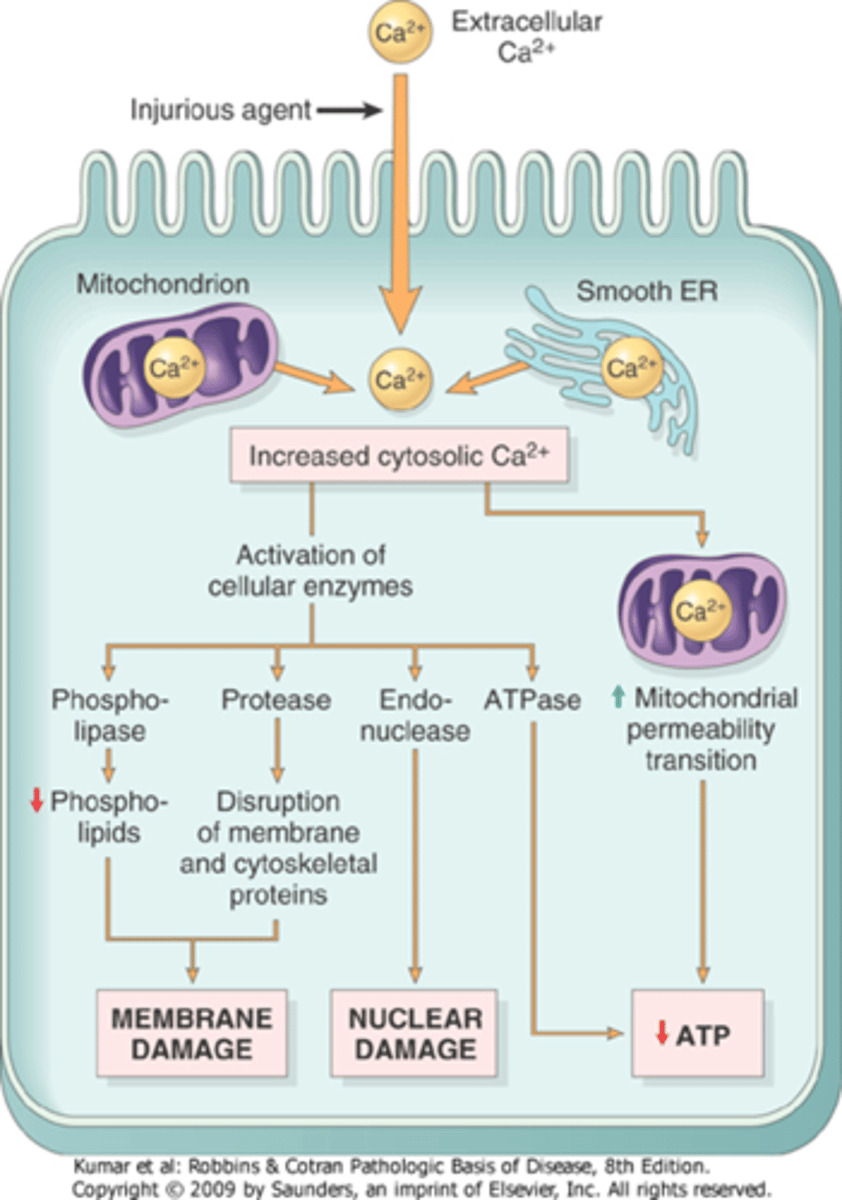
What will be the effect of increased intracellular Ca2+ levels?
induction of apoptosis: direct activation of caspases increasing mitochondrial permeability
What are free radicals?
chemical species that have a single unpaired electron in an outer orbit, energy is created by this unstable configuration
How is the energy in free radicals released? Provide an example
- Through reactions with adjacent molecules
- Ex: inorganic or organic chemicals/proteins, lipids, carbs, and nucleic acids q
Where are reactive oxygen species made within cells?
produced normally in cells during mitochondrial respiration and energy generation, also produced in large amounts by leukocytes (neutrophils and macrophages)
How are free radicals degraded?
Through enzymes, spontaneous decay, and antioxidants. (cellular defense systems)
In what ways can free radicals be made that are not normal to physologic processes?
UV light, xrays, and ionizing radiation (hydrolyzes water into OH and hydrogen free radicals), and enzymatic metabolism of exogenous chemicals or drugs
What type of reaction will lead to free radicals?
Reduction-oxidation reactions that occur normally in metabolic processes
When will rapid bursts of reactive oxygen species be seen?
during inflammation: made by activated leukocytes
What role do transition metals play in generation of free radicals?
iron and copper donate and accept free electrons during intracellular reactions
Ex: Fenton reaction (H2O2 + Fe2+ → Fe3+ + OH + OH
What is the function of nitric oxide?
important chemical mediator, acts as a free radical
Where is nitric oxide generated within the body?
endothelial cells, macrophages, neurons, and others
How are free radicals removed?
unstable and decay spontaneously
multiple nonenzymatic and enzymatic mechanisms in cells remove free radicals
iron and copper minimize levels by binding ions to storage and transport proteins (transferrin, ferritin, lactoferrin, and ceruloplasmin)
enzymes act as free radical-scavenging systems
How can free radicals affect membranes of cells?
leads lipid peroxidation within plasma and organellar membranes in presence of O2
leads to oxidative damage: double bonds of FA in membranes are attacked by O2 derived free radicals