3.1.12.6 buffer action
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
what are buffer solutions?
solutions that maintain a constant pH despite the addition of acid, base or water
give the two types of buffers
acidic buffers which have a pH of below 7
basic buffers which have a pH of above 7
what does an acidic buffer typically consist of?
of a weak acid (HA) + one of its salts (A-)
describe one of the two ways of making an acidic buffer
a weak acid is added to one of its salts → eg ethanoic acid + sodium ethanoate
a solution of the salt is added to a weak acid or
a solid salt is added to the weak acid
that salt dissociates completely + the acid dissociates only slightly
there is a large [HA] + a large [A-] so [H⁺] ≠ [A-]
![<ul><li><p>a weak acid is added to one of its salts → eg ethanoic acid + sodium ethanoate</p><ul><li><p>a solution of the salt is added to a weak acid or</p></li><li><p>a solid salt is added to the weak acid</p></li></ul></li><li><p>that salt dissociates completely + the acid dissociates only slightly</p></li><li><p>there is a large [HA] + a large [A-] so [H⁺] ≠ [A-]</p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/3f329816-65b1-4c10-b7da-6bbcb47d2024.jpg)
describe a second way of making an acidic buffer
partial neutralisation of a weak acid
in this case, the salt is made as a product of the reaction + some of the excess acid will still be present
a strong base is added to the weak acid → eg ethanoic acid + sodium hydroxide
there is a large [HA] + a large [A-] (all of the OH- is reacted)
explain how an acidic buffer works if H⁺ is added
if more H⁺ ions are added by adding some acid, they react with the A- ions
the acid equilibrium moves to the left to decrease the moles of H⁺
so [H⁺] remains unchanged + so does the pH
![<ul><li><p>if more H⁺ ions are added by adding some acid, they react with the A- ions</p></li><li><p>the acid equilibrium moves to the left to decrease the moles of H⁺</p></li><li><p>so [H⁺] remains unchanged + so does the pH</p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/f9961460-8649-4769-b596-f37c5e762d22.jpg)
explain how an acidic buffer works if OH- is added
if OH- ions are added by adding some alkali, they react immediately with H⁺, forming H₂O
therefore acid equilibrium moves to the right + more acid is dissociated to replace H⁺
so [H⁺] remains unchanged + so does the pH
what does a basic buffer typically consist of?
of a weak base + one of its salts
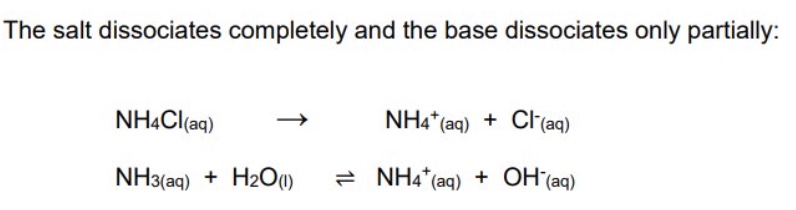
describe the two ways of making a basic buffer using the example of ammonia as the weak acid
a solution containing ammonium chloride + ammonia would make a basic buffer
the salt dissociates completely + the base dissociates only partially
or partial neutralisation of a weak base by a strong acid eg ammonia (NH₃) + HCl
explain how an basic buffer works if H⁺ is added
if H⁺ ions are added by adding some acid, they react immediately with the OH- ions
therefore the equilibrium moves to the right to replace the OH-
so [H⁺] remains unchanged + so does the pH
![<ul><li><p>if H⁺ ions are added by adding some acid, they react immediately with the OH- ions</p></li><li><p>therefore the equilibrium moves to the right to replace the OH- </p></li><li><p>so [H⁺] remains unchanged + so does the pH </p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/4064929c-b01f-4130-8920-5e1d305c6f02.jpg)
explain how an basic buffer works if OH- is added
if more OH- ions are added by adding some alkali, they react immediately with the base ions (eg in this case NH₄⁺ ions)
so equilibrium moves to the left to decrease the moles of OH- ions
so [OH-] remains unchanged, so does [H⁺] + so does pH
![<ul><li><p>if more OH- ions are added by adding some alkali, they react immediately with the base ions (eg in this case NH₄⁺ ions) </p></li><li><p>so equilibrium moves to the left to decrease the moles of OH- ions </p></li><li><p>so [OH-] remains unchanged, so does [H⁺] + so does pH </p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/d4338ee8-65ef-4b87-a8be-b9fac198e6ac.jpg)

applications of buffers - fill in the blanks

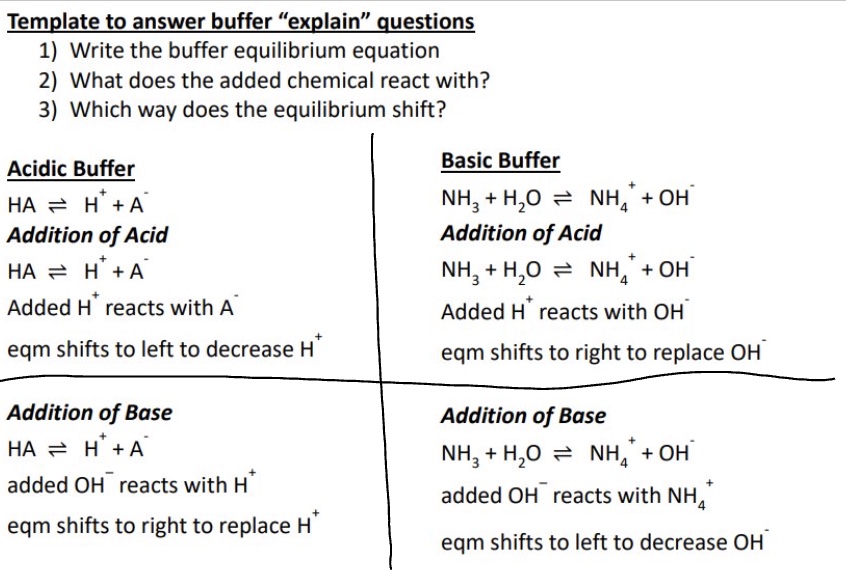
give the template for answering buffer ‘explain’ questions
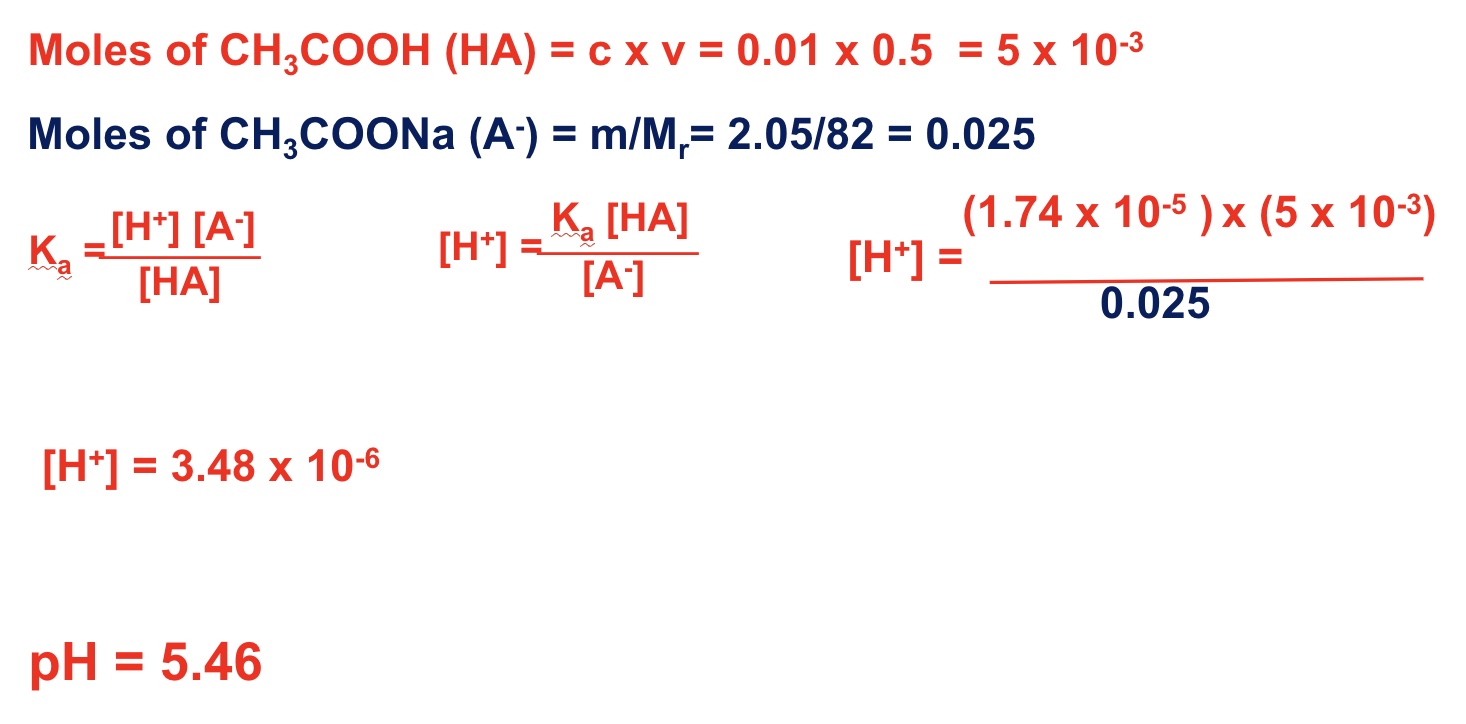
describe the method of calculating the pH of an acidic buffer thats been made using route 1: using a weak acid + its salt
rearrange the Ka expression to work out [H⁺]
remember moles can just be used rather than working out concentration
use -log to calculate pH
![<ul><li><p>rearrange the Ka expression to work out [H⁺] </p><ul><li><p>remember moles can just be used rather than working out concentration</p></li></ul></li><li><p>use -log to calculate pH </p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/f1cb6065-42ce-423d-ad3b-d8dcbfb1c86d.jpg)



describe the method of calculating the pH of an acidic buffer thats been made using route 2: using an excess weak acid + strong base
work out moles of HA
work out moles of OH- which is the moles of A-
work out leftover/excess moles of HA
rearrange Ka expression to work out [H⁺]
use -log to work out pH
![<ol><li><p>work out moles of HA</p></li><li><p>work out moles of OH- which is the moles of A-</p></li><li><p>work out leftover/excess moles of HA </p></li><li><p>rearrange Ka expression to work out [H⁺]</p></li><li><p>use -log to work out pH</p></li></ol><p></p>](https://knowt-user-attachments.s3.amazonaws.com/ac6e318f-c3b0-4523-9778-5cc25077b4e4.jpg)
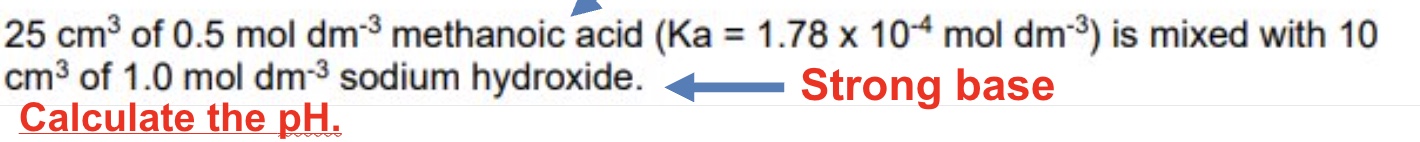



outline the effect of adding a strong base (OH-) to a buffer
it moves equilibrium moves to the right which:
decreases moles of HA
increases moles A-
outline the effect of adding a strong acid (H⁺) to a buffer
it moves equilibrium to the left which:
increases moles of HA
decreases moles of A-
describe the method of working out the new pH when a strong acid or base is added to a buffer solution
work out moles of strong acid/base being added + so the moles of H⁺/OH⁻
work out the new moles of the salt (A⁻) → ie does it decrease/increase
work out the new moles of the acid (HA)
→ ie does it decrease/increase
work out [H⁺] by rearranging Ka expression
use -log to work out pH

using the info of the buffer solution given, work out the change in pH
change = - 0.04


using the info of the buffer solution given, work out the change in pH
change = + 0.03

give the 3 possible calculations involving mixtures of weak acids + strong bases, and state when each one is used
weak acid in excess + strong base → BUFFER CALCULATION
weak acid + strong base in excess → MIXTURE CALCULATION
half equivalence point reached (ie when half of moles of acid/base have been neutralised)→ pH = pKa