topic 6 organic chem 1
1/172
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
173 Terms
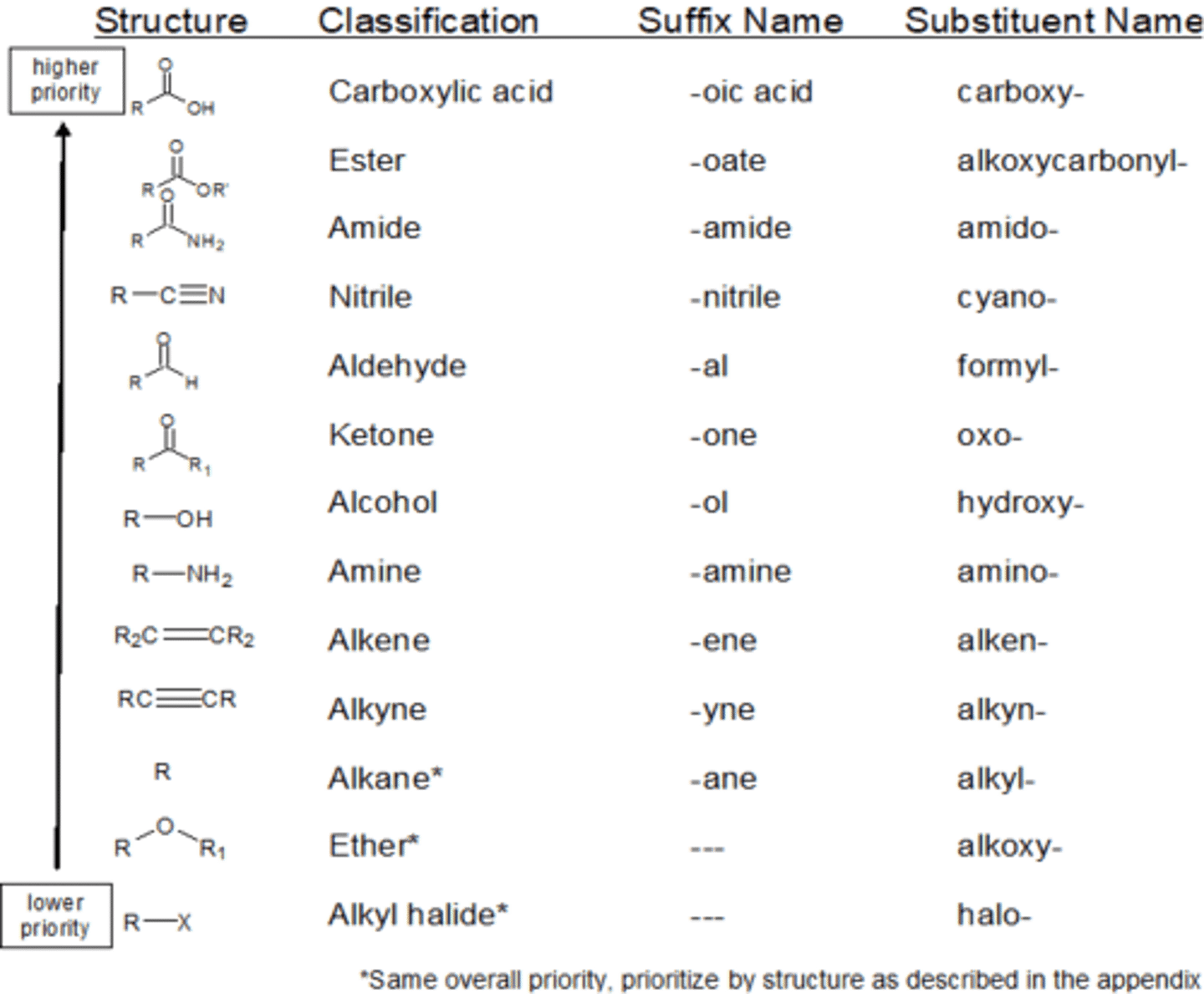
definition of functional group
a group of atoms responsible for the characteristic reactions of a particular compound
describe 2 characteristics of a homologous series
- same general formula
- same functional group
- trend in physical properties
steps for naming an organic compound
1. identify the longest unbranched carbon chain to give the stem name
2. name any substituent groups, if more than one, alphabetically
3. identify position of substituent group
rules of naming an organic compound
- count from the end that gives the lowest number
- if two of the same group are on the same carbon, repeat number and add di- suffix
- if more than one of the same group is on different carbon, number and add di-, tri-, tetra-
which of the following has the longest bond length
A) Cl-Cl
B) H-Cl
C) O=O
D) N≡N
A
a triple bond is the shortest bond, followed by a double bond
H-Cl exhibits polar bonding due to Cl being very electronegative
define stereoisomers
compounds with the same structural formula but a different arrangement of atoms in space
explain what must happen for geometric isomerism to be able to occur
- there must be restricted movement somewhere in the molecule
- often in a double bond or cyclic compound
how does a C=C bond result in the potential for geometric isomerism?
- the π bond formed by the sideways overlap of p orbitals fixes the carbon atoms in position
- resulting in no free rotation
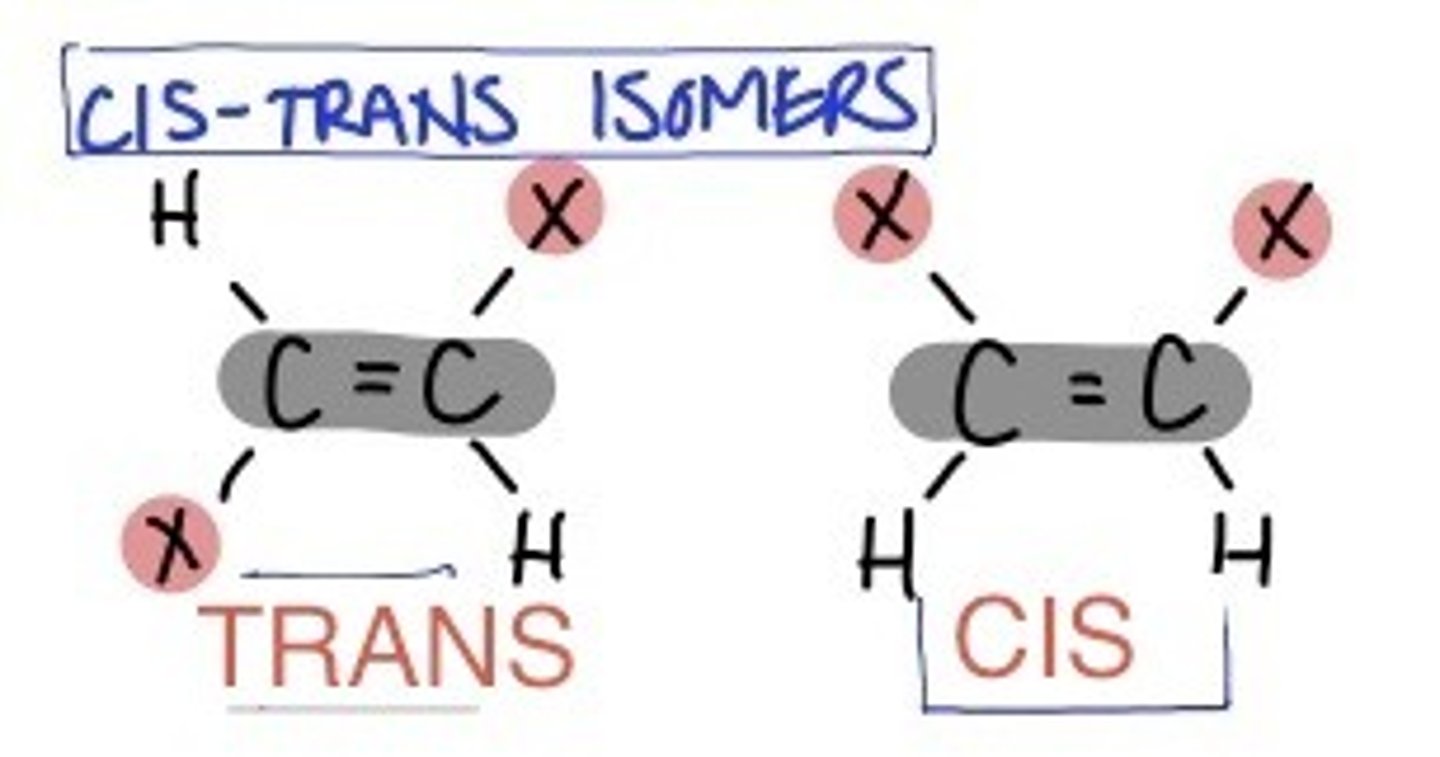
rules for naming geometric isomers
- if there is only 2 different groups out of the 4 around the C=C bond, then the cis/trans method can be used
- if there are 3-4 different species around the C=C bond, then the E/Z method must be used
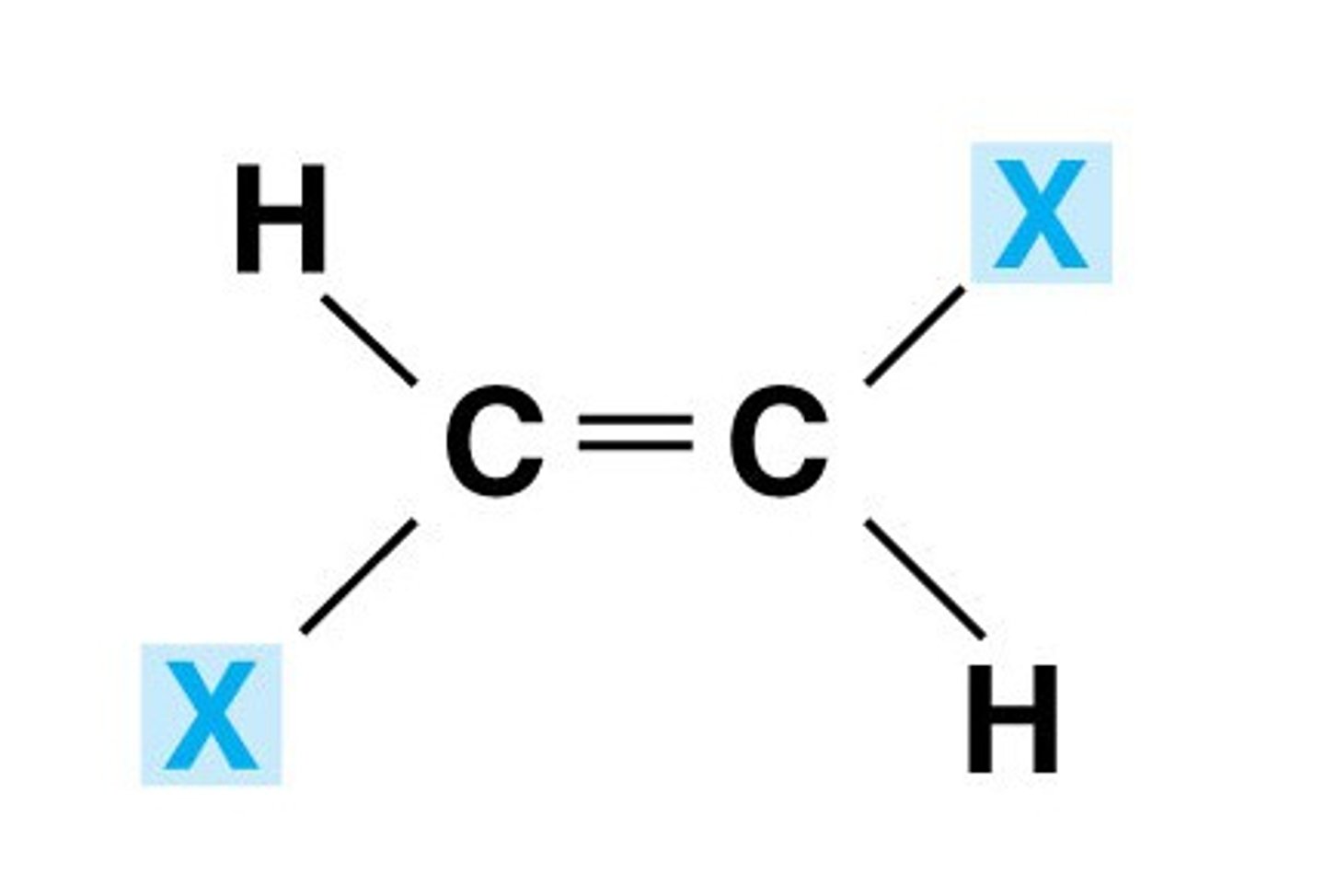
trans isomer
- priority groups are on OPPOSITE sides of the carbon chain (transport across the city, ie cross the double bond)
- line of symmetry is horizontal
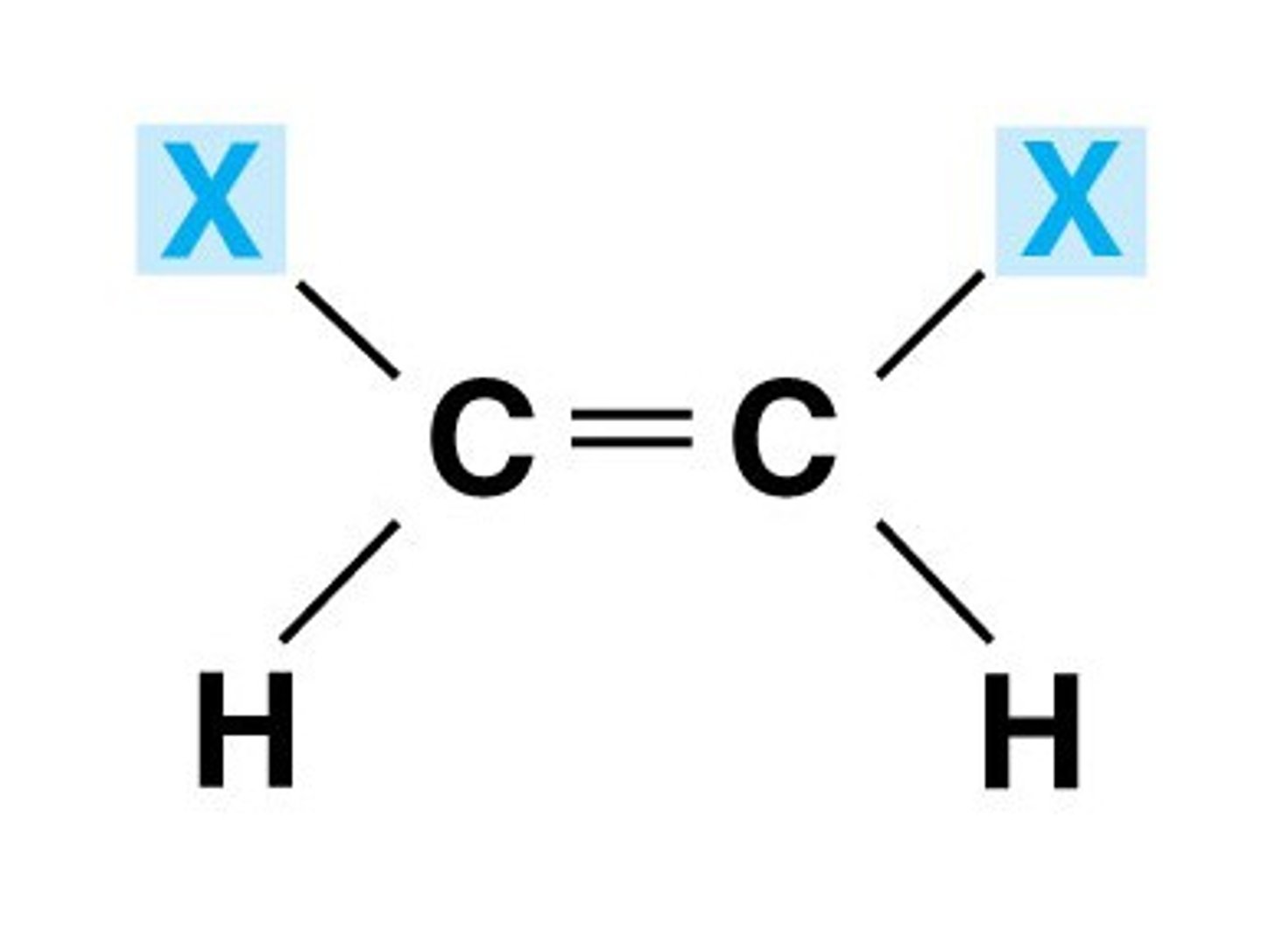
cis isomer
- priority groups are on the same side of the carbon chain (a bucket is formed)
- line of symmetry is horizontal
E (entgegen) isomer
- priority groups are on opposite side of carbon chain
- vertically compare atomic numbers to determine group with highest priority
- line of symmetry is horizontal
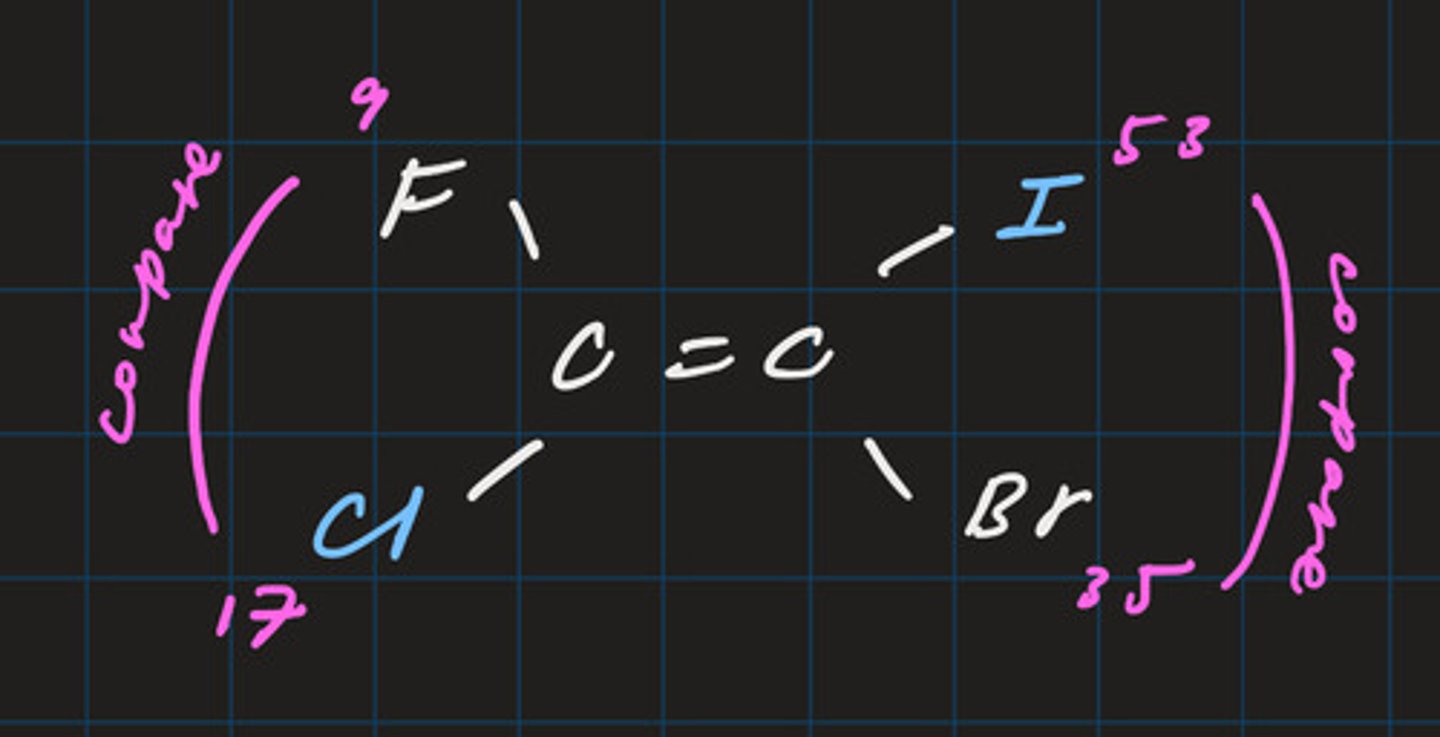
Z (zusammen) isomer
- priority groups are on the same side of carbon chain
- vertically compare atomic numbers to determine group with highest priority
- line of symmetry is horizontal
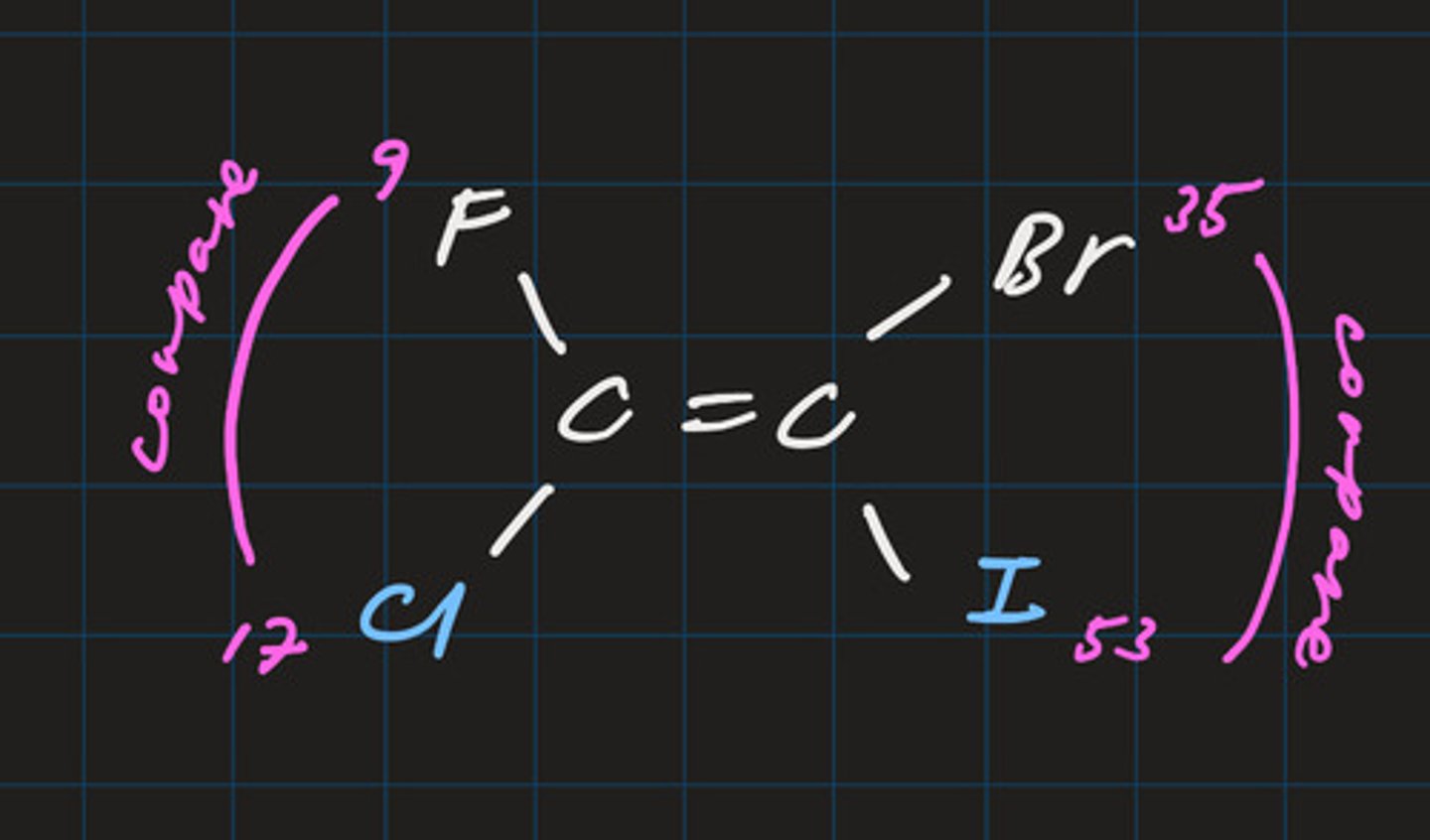
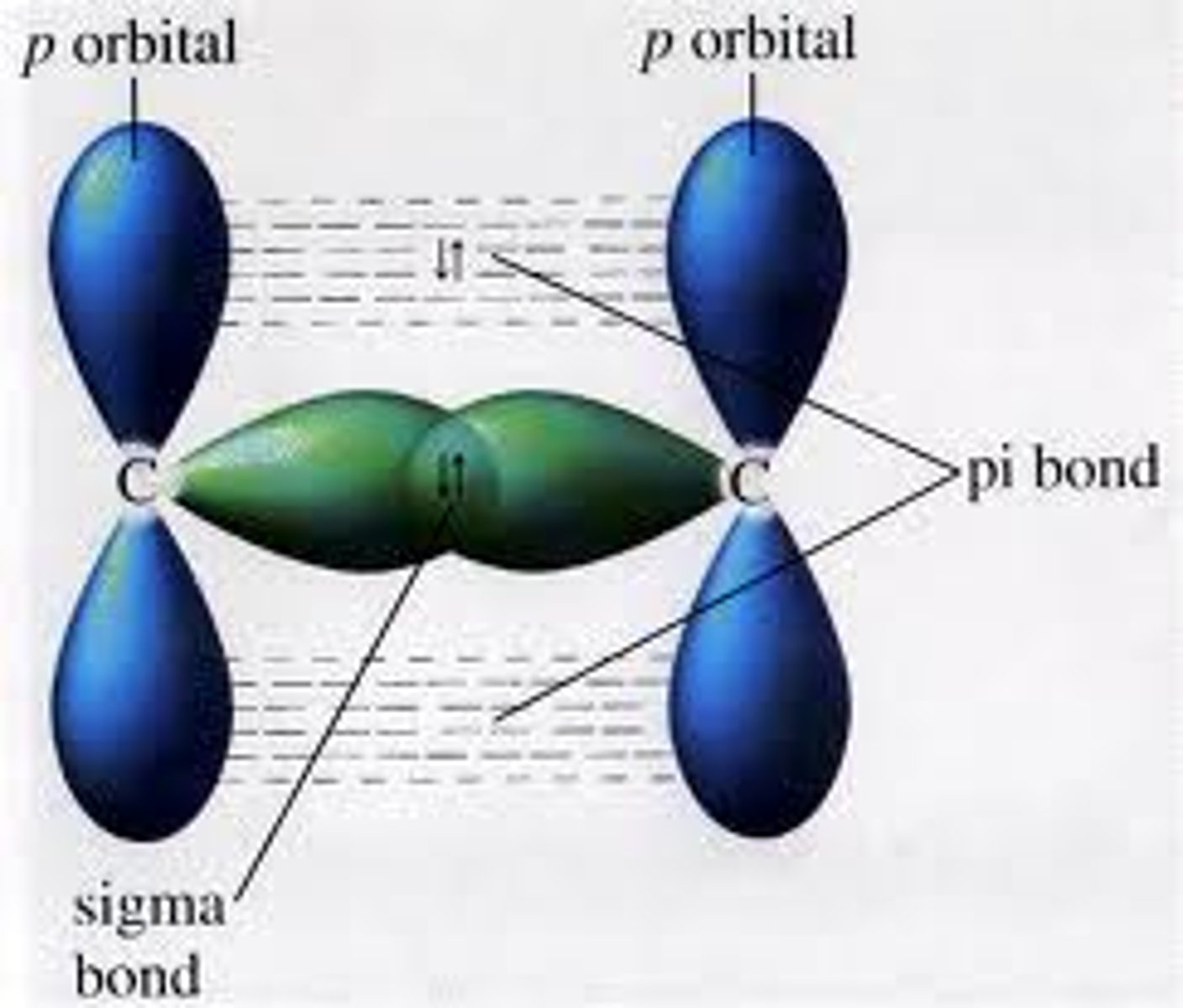
describe in detail the structure of the C=C bond in alkenes and hence explain why alkenes are more reactive than alkanes
- alkenes contain a π bond and a σ bond
- head on overlap of σ bond's p orbitals
- sideways overlap of π bond's p orbitals
- hence the C=C bond is an area of high electron density
- more susceptible to electrophilic attacks => more reactive
amount of sigma and pi bonds in a triple bond
1 sigma, 2 pi

why is a sigma bond stronger than a pi bond?
- sigma bond is the head-on overlap of orbitals
- electron density to be concentrated in the plane of the atoms
- pi bonds are formed due to sideways overlap of p orbitals
- electron density is above and below the molecule
explain the difference between cracking and reforming
- cracking: breaking the carbon chain to give a shorter chain hydrocarbon
- reforming: converting a straight chain to a more branched chain (changes chemical and physical properties)
define 'cracking'
- use of high temperature
- to break large molecules
- producing alkenes and shorter chained alkanes
define 'reforming'
- the processing of straight chain hydrocarbons into branched chain alkanes and cyclic hydrocarbons
- for efficient combustion as it prevents knocking (where alkanes explode when compressed which is more likely in straight chains)
write an equation for reforming heptane into cycloheptane, showing the skeletal formulae of the organic molecules
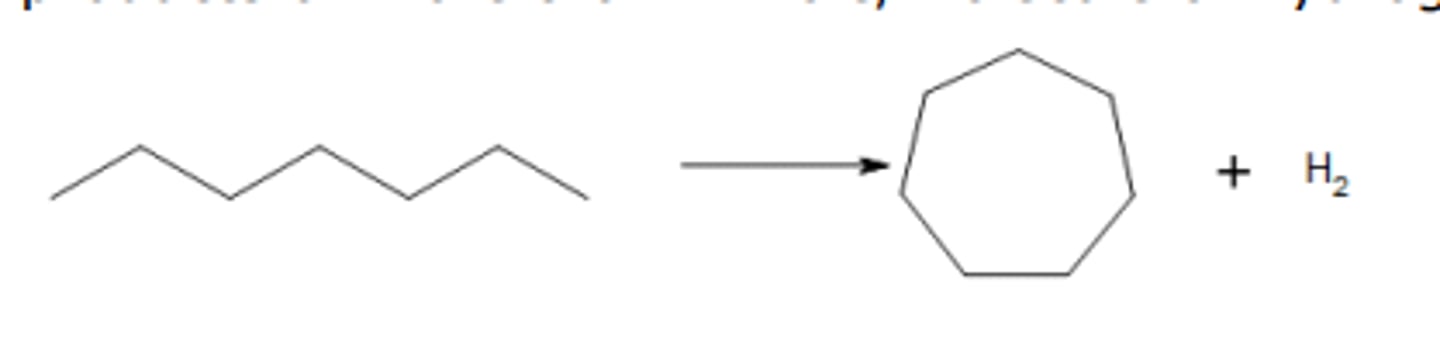
what happens to the proportions of branched chain alkanes and cyclic hydrocarbons during the reforming process?
both increases
conditions for cracking
- 600-700°C
- silica or alumina catalyst
explain two major reasons for cracking hydrocarbons
- shorter chain alkanes AND alkenes formed
- alkenes are useful for making polymers
- shorter chains are more in demand / can be used as fuel
describe how fractions in crude oil are separated during fractional distillation
- vapour condenses at different levels and temperatures
- the higher the bp the lower the level at which it condenses
suggest one difference between fractional distillation of air and of crude oil
oil must be heated until it is a gas
give a reason why oxygen should be excluded during steam cracking
prevent combustion
explain what is meant by the term fuel
substance that releases energy when burnt
molecules containing 6 carbon atoms are found in both the petrol fraction and the higher boiling kerosene fraction
explain why isomers of hexane have lower bp and so are found in the petrol fraction
- isomers in petrol fraction have branched chains
- branched chains have LOWER SA as they do not pack so closely together
- intermolecular forces are weaker / so bp is lower
state 2 problems that result from the incomplete combustion of alkane fuels
- CO is toxic
- less energy produced
- soot deposits
evaluate the use of alternative fuels such as biodiesel and alcohol derived from renewable sources
- biodiesel obtained from refining renewable fats and oils
- bioethanol from the fermentation of sugar from maize crops
- carbon neutral process as the CO2 produced are absorbed by plants as they grow
- reduces landfill and toxic gas
- however petrol cars may need to be modified to use fuels at high ethanol concentrations
ethanol is a fuel and can be made by either the fermentation of carbohydrates or the hydration of ethene. how is the ethanol formed by the fermentation of carbohydrates classified?
a biofuel and renewable
3 multiple choice options
some buildings are made from limestone, which is mainly calcium carbonate. gases in the atmosphere such as sulfur dioxide, SO2, and nitrogen dioxide, NO2, can be responsible for damaging these buildings.
describe how these gases come to be present in the atmosphere and explain how they can damage a limestone building
- sulfur / nitrogen oxides form when fossil fuels are burnt
- react with water to form to form sulfuric acid (acid rain)
- which reacts with limestone to form soluble compounds (neutralisation reaction)
the reaction between decane and nitrogen monoxide produces carbon dioxide, water and nitrogen as the only products.
give a possible reason why the reaction may not proceed according to the equation.
- oxygen is present so decane may react with oxygen
- NO may reaction with CO
explain how oxides of sulfur and nitrogen can be formed from the combustion of fossil fuels
- sulfur compounds in fuel will react with O₂ in the air
- N2 in air will react with O₂ in air under high temperature
explain why this compound is likely to have a higher bp than hydrocarbons of similar molecular mass and shape
- the OH groups can form hydrogen bonds
- so more energy is needed to break the intermolecular bonds
- hydrocarbons only have LDF
alkenes have the same general formula as cycloalkanes.
explain why this general formula is different from that of alkanes. include the general formula in your answer.
- alkanes have a double bond (unsaturated) while cycloalkanes do not (saturated)
- cycloalkanes have carbons in a ring-like structure
- alkene have a general formula of CnH2n, alkanes have a general formula of CnH₂n+2
state one use of ethene
to make polymers / ethanol
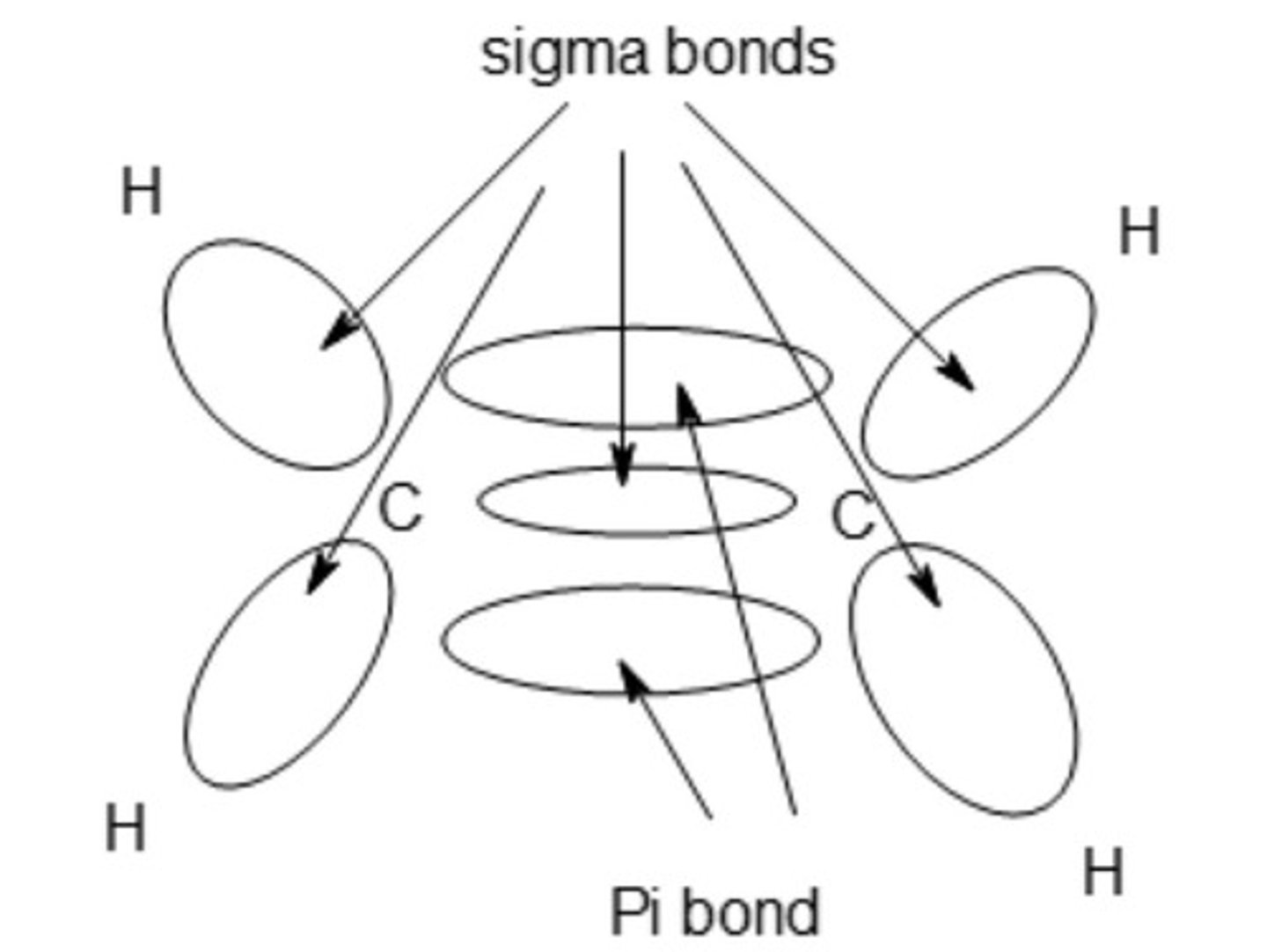
draw a diagram of an ethene molecule showing the shape of all the bonding electron clouds.
include labels for all the bond types within the molecule.
MP1 - sigma bond drawn and identified
MP2 - diagram identifying pi bond and sigma bond between C atoms
MP3 - C-H bonds drawn at least one identified as sigma bond
explain why most polymers are so difficult to break down
- C-C and C-X bonds are very strong so they resist chemical and biological attacks
- polymers are not naturally occurring in nature so no enzymes have evolved to break them down
★ some plastic products can be cleaned and used again
give two other uses of waste plastic
- recycling (uses a huge amount of energy)
- incineration to release energy
- as a feedstock for cracking
what are some problems regarding addition polymers
- non biodegradable due to inert carbon bonds
- incineration causes toxic gas to be produced
★ give a reason why some polymers cannot be recycled
- polymer degrades during heating (for recycling)
- heavy pigmentation / colour of plastic to be recycled
cyclohexene form addition polymers
draw a section of the polymer, showing 2 repeat units
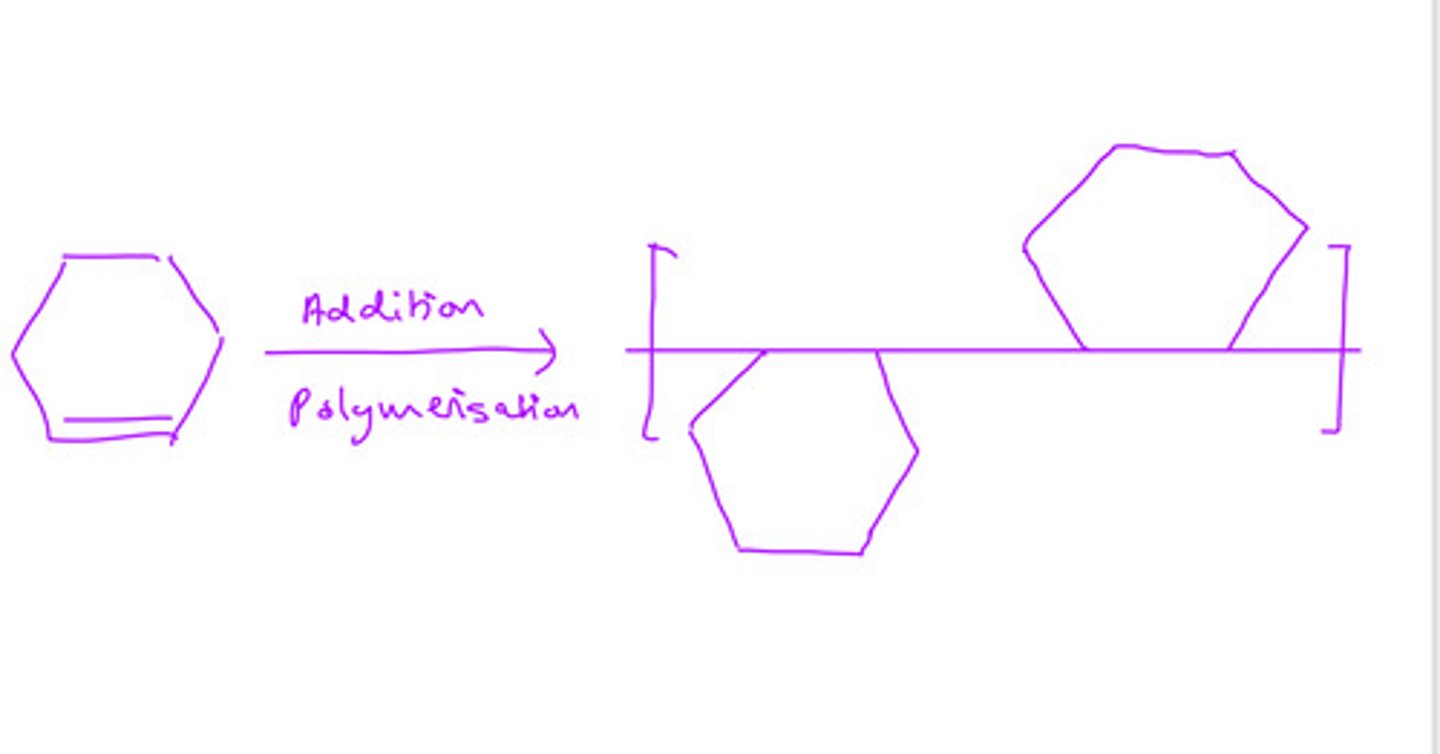
some polymers are disposed of by incineration.
ignoring any economic considerations, explain why incineration is not a suitable method for the disposal of poly(chloroethene).
- incineration produces HCl
- which is corrosive and causes acid rain
describe how the toxic gas hydrogen chloride produced during the incineration of poly(chloroethene) could be removed instead.
pass through alkaline solution (eg NaOH)
scientists are developing sources of energy as alternatives to fuels produced form crude oil, which of the following is incorrect?
A) crude oil is being used up faster than it is being formed
B) burning hydrocarbons affect global carbon dioxide levels
C) hydrocarbons from crude oil are a source of essential chemicals other than fuels
D) carbon dioxide produced by burning hydrocarbons is toxic to plants
D
★ incinerators used to dispose of polymer waste have a system to remove waste gases
suggest why these systems contain solid calcium hydroxide and why it is finely powdered
- calcium hydroxide is basic
- so it will neutralise HCl / SO2 / NOx in waste gases
- fine powder increases SA
- to ensure fast reaction
state a safety precaution that chemists should take when using HCl
use a fume cupboard
observation during combustion of propane and what it's caused by
black smoke
carbon remnants
define homolytic fission
- when a single bond breaks and one electron goes to each atom to form radicals
- eg free radical substitution
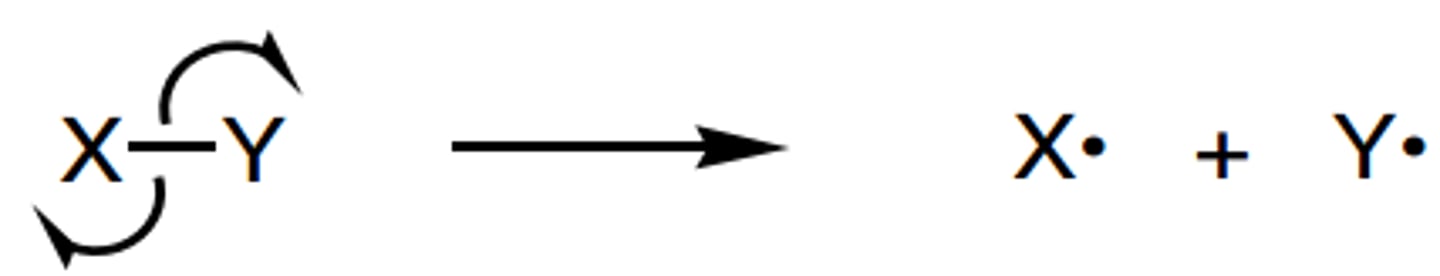
define heterolytic fission
- when a single bond breaks and both electrons go to one atom to form a cation and an anion
- in most mechanisms, bonds (eg C-X) break heterolytically
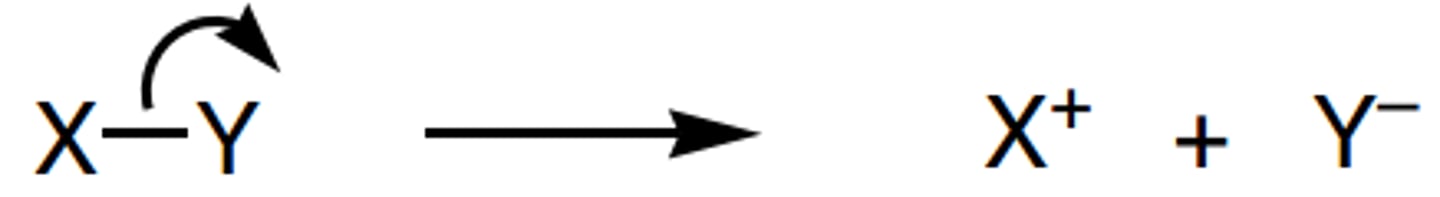
the heterolytic bond fission of a sigma bond in an alkane would produce:
ions
explain what is meant by the term free radical
- a species with an unpaired electron
- represented in mechanisms by a single dot
- formed by homolytic fission of a covalent bond
what do fish hook arrows represent?
shows the movement of ONE electron only
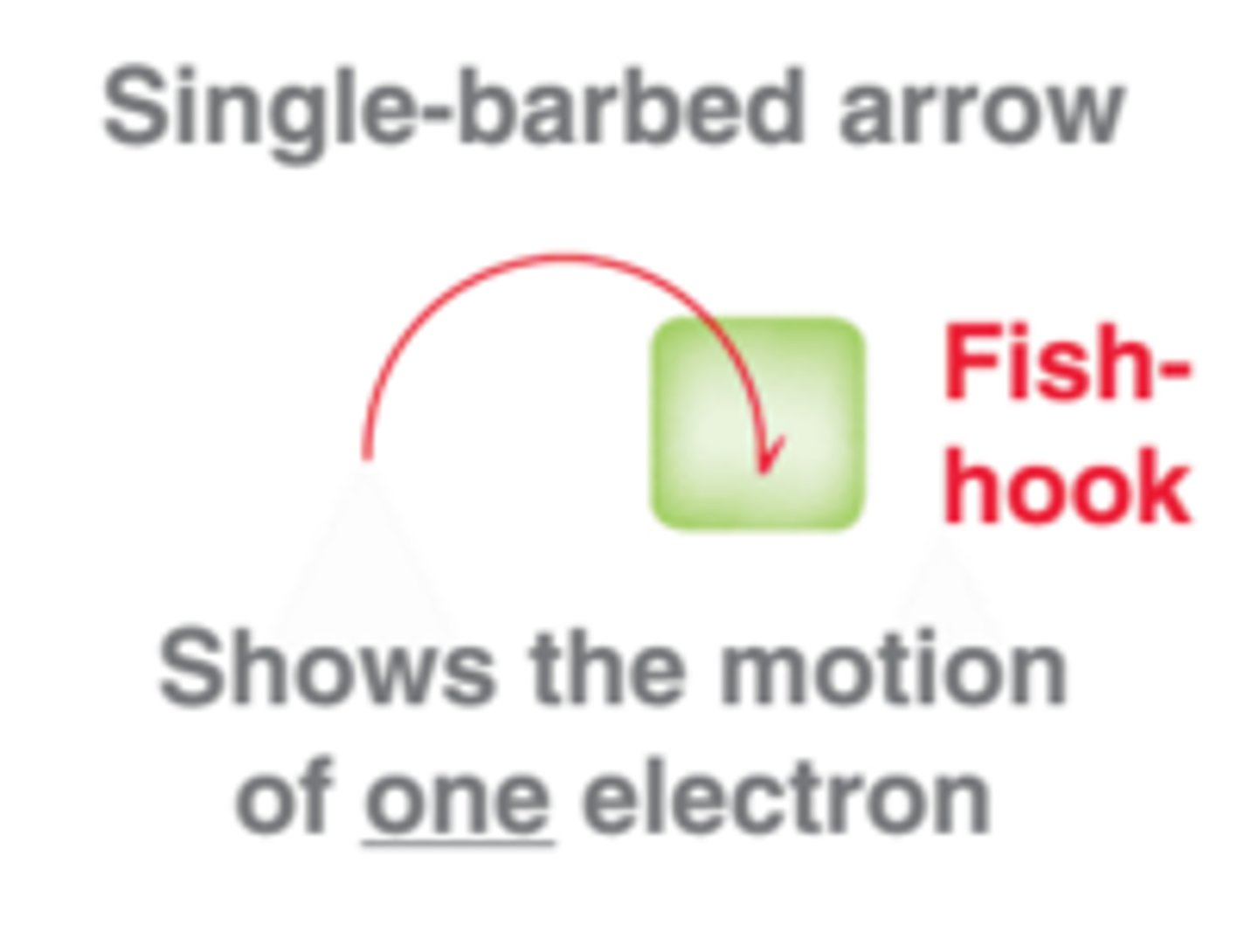
what is the meaning of curly arrows in a reaction mechanism?
- movement of a pair of electrons
- from an electron pair OR a double bond
write the equation for the initiation step of chlorine
Cl₂ → 2Cl•
(UV required)
write two equations for the propagation step of propane and chlorine
(stable compound + radical → stable compound + radical)
C₃H₈ + Cl• → C₃H₇• + HCl
C₃H₇• + Cl2 → C₃H₇Cl + Cl•
this is a CHAIN reaction
write two equations for the termination step of propane and chlorine
(radical + radical → stable compound)
C₃H₇• + Cl• → C₃H₇Cl
C₃H₇• + C3H7• → C₆H₁₄
write the overall equation for the free radical substitution of propane and chlorine
C₃H₈ + Cl₂ → HCl + C₃H₇Cl
- ignore all radicals
- stable products formed during propagation
limitations of free radical substitution
- mixture of products (low yield)
- structural isomers can form, eg 1-chloropropane, 2-chloropropane
- further substitution reactions can occur
if the reaction with propane is carried out with an excess of chlorine, how are the products of the reaction affected?
further substitution reactions can occur
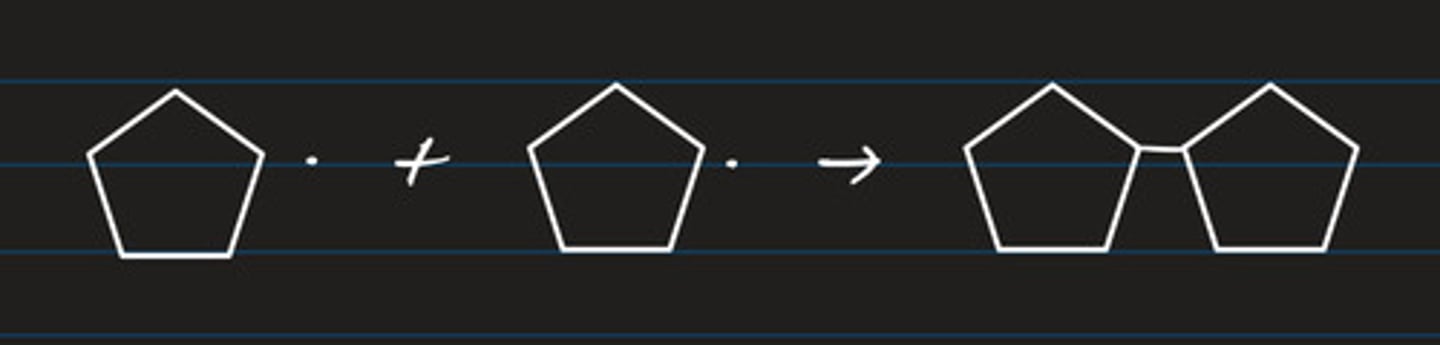
write two equations for the propagation step of cyclobutane and chlorine
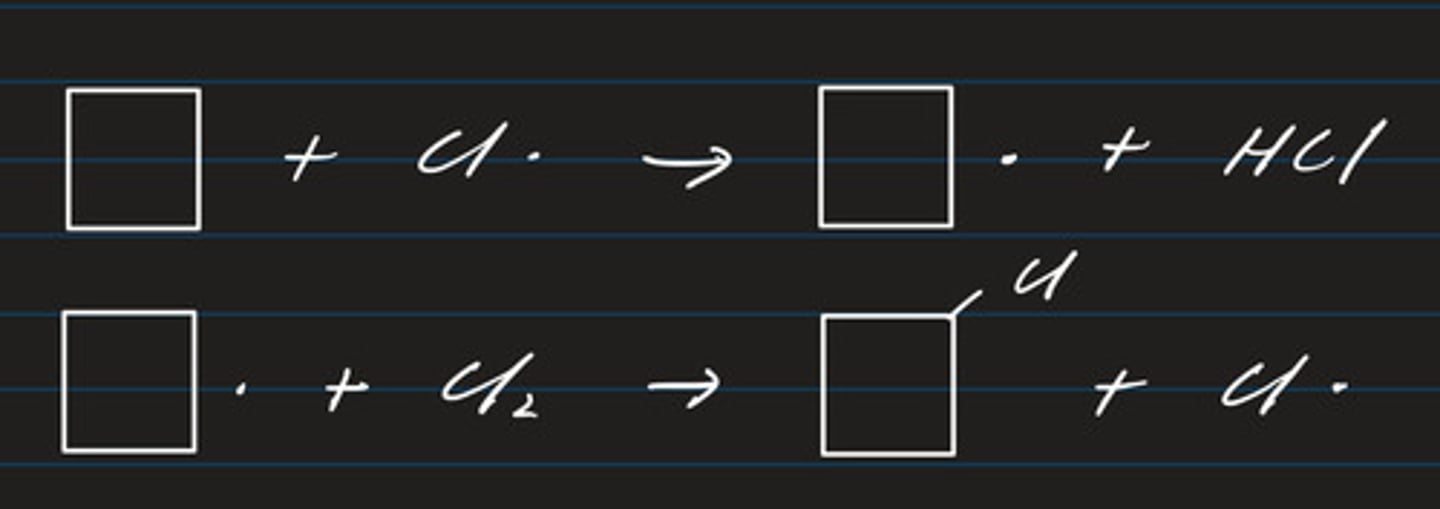
write a termination equation step for cyclopentane and chlorine
a student is asked to devise a laboratory synthesis of 1,2-dichloroethane. the student suggests reacting ethane with chlorine in the presence of ultraviolet radiation.
give two reasons why this is not a good method to prepare 1,2-dichloroethane. (2)
- you cannot control how many chlorine atoms will substitute
- mixture of unwanted products will form
- need to separate the 1,2-dichloroethane from any other products made
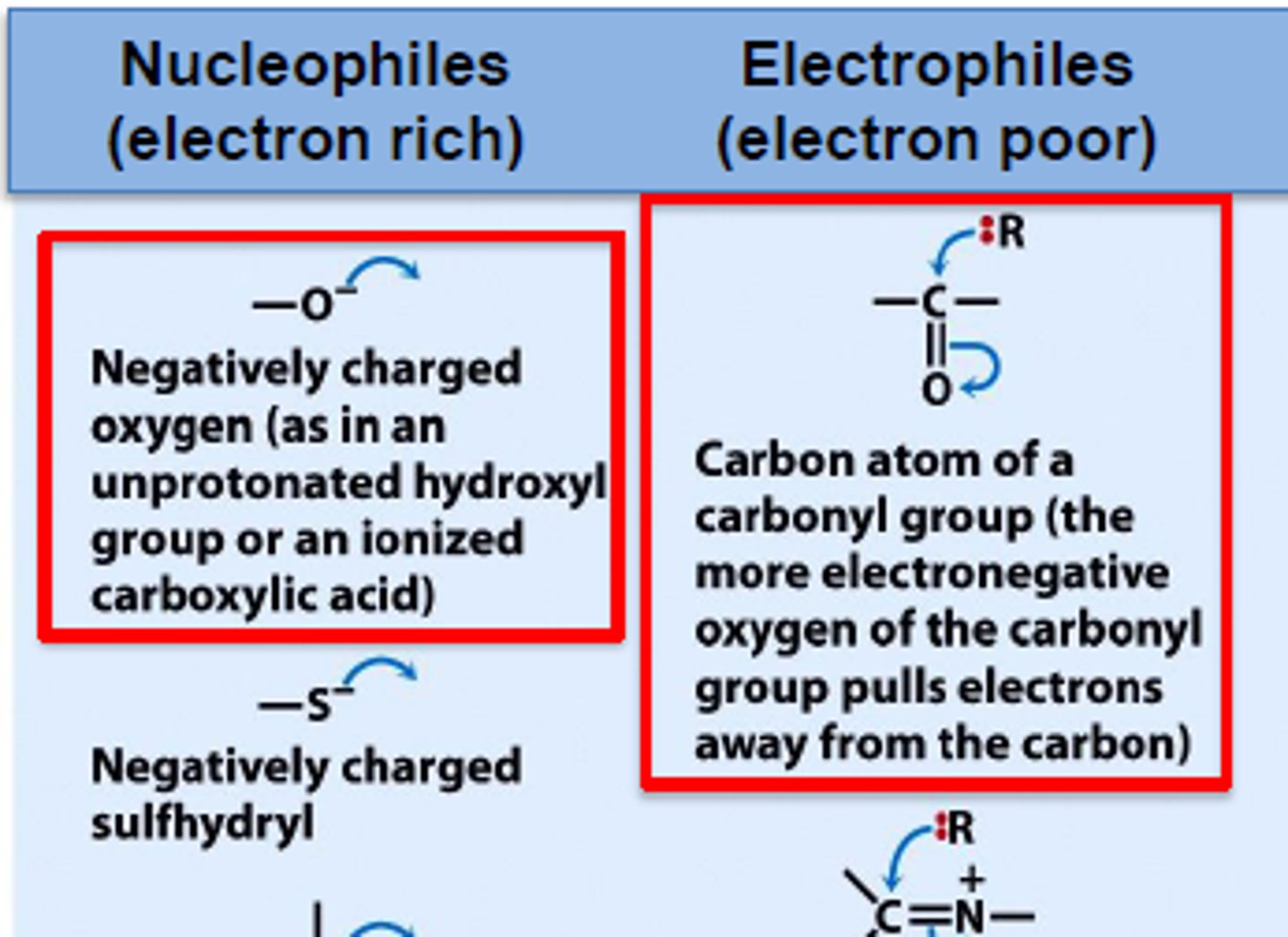
define electrophile
- an electron pair acceptor
- attracted to the electron rich C=C double bond
what are the 4 most electronegative elements? (important for assigning δ+ and δ- signs)
F, O, N, Cl
what process do alkenes undergo to form alkanes
electrophilic addition w/ hydrogen (aka hydrogenation)
conditions and catalyst required for alkene → alkane
- add hydrogen gas (H₂)
- nickel catalyst
- 150°C
draw the mechanism for the electrophilic addition of Br₂ to C₂H₄
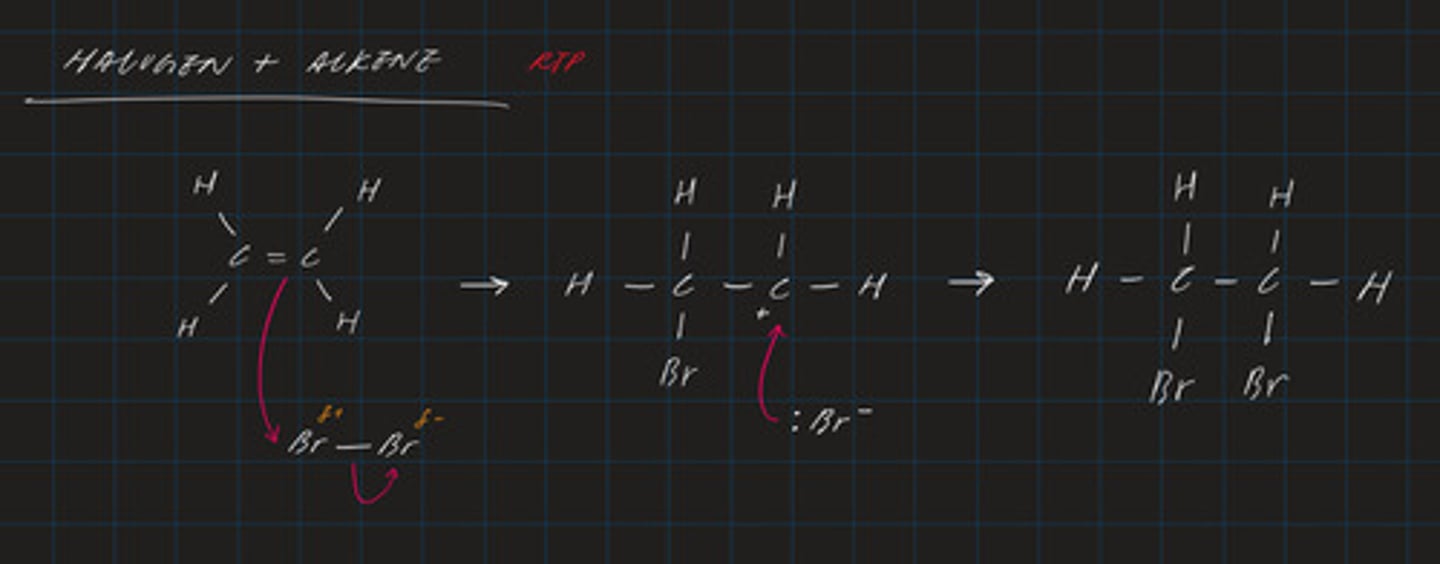
draw the mechanism for the electrophilic addition of HBr to C₂H₄
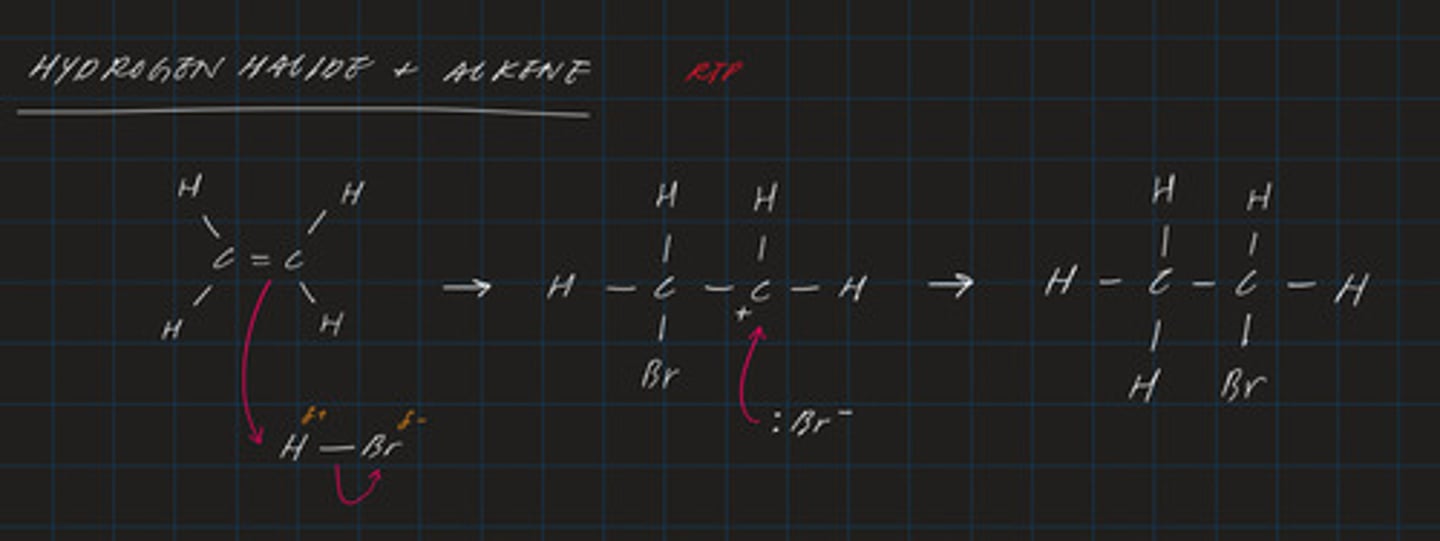
what reaction does alkene undergo to form alcohol?
electrophilic addition reaction w/ steam (H2O)
(2 products can be formed if asymmetrical alkene like propane)
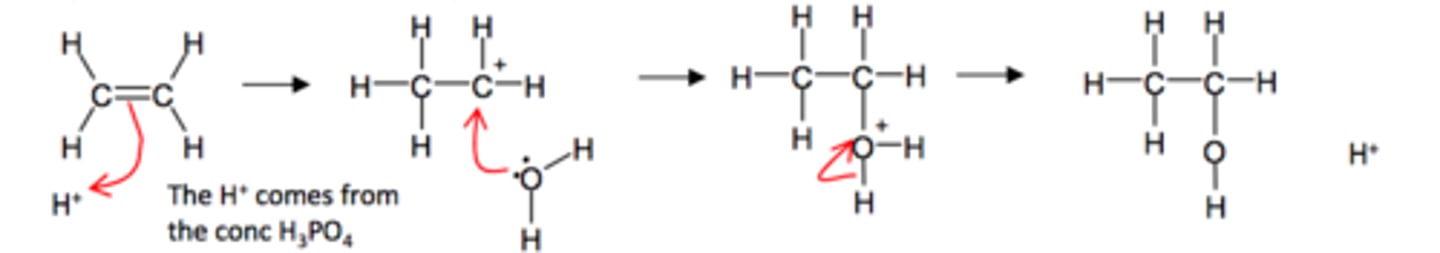
conditions and catalyst required for alkene → alcohol
- phosphoric acid (catalyst provides H+)
- steam
- 300°C
- 60-70 atm
what is catalytic hydrogenation used for in the industry?
- manufacture margerine
- from unsaturated vegetable oil
LiAlH4 is a source of hydride ions, H−.
give a possible reason why LiAlH4 cannot be used to reduce alkenes.
- H- is a nucleophile (electron rich)
- so the hydride ion will not attack the C=C
- instead it would be repelled by regions of high electron density
state the colour change during test for c=c bond with bromine water
orange to colourless (di-bromoalkane formed)
what is formed as the MAJOR product when ethene reacts with bromine water?
- bromine water is aqueous, meaning it contain both Br₂ & H₂O
- so the major product formed is 2-bromoethanol instead of 1,2-dibromoethane
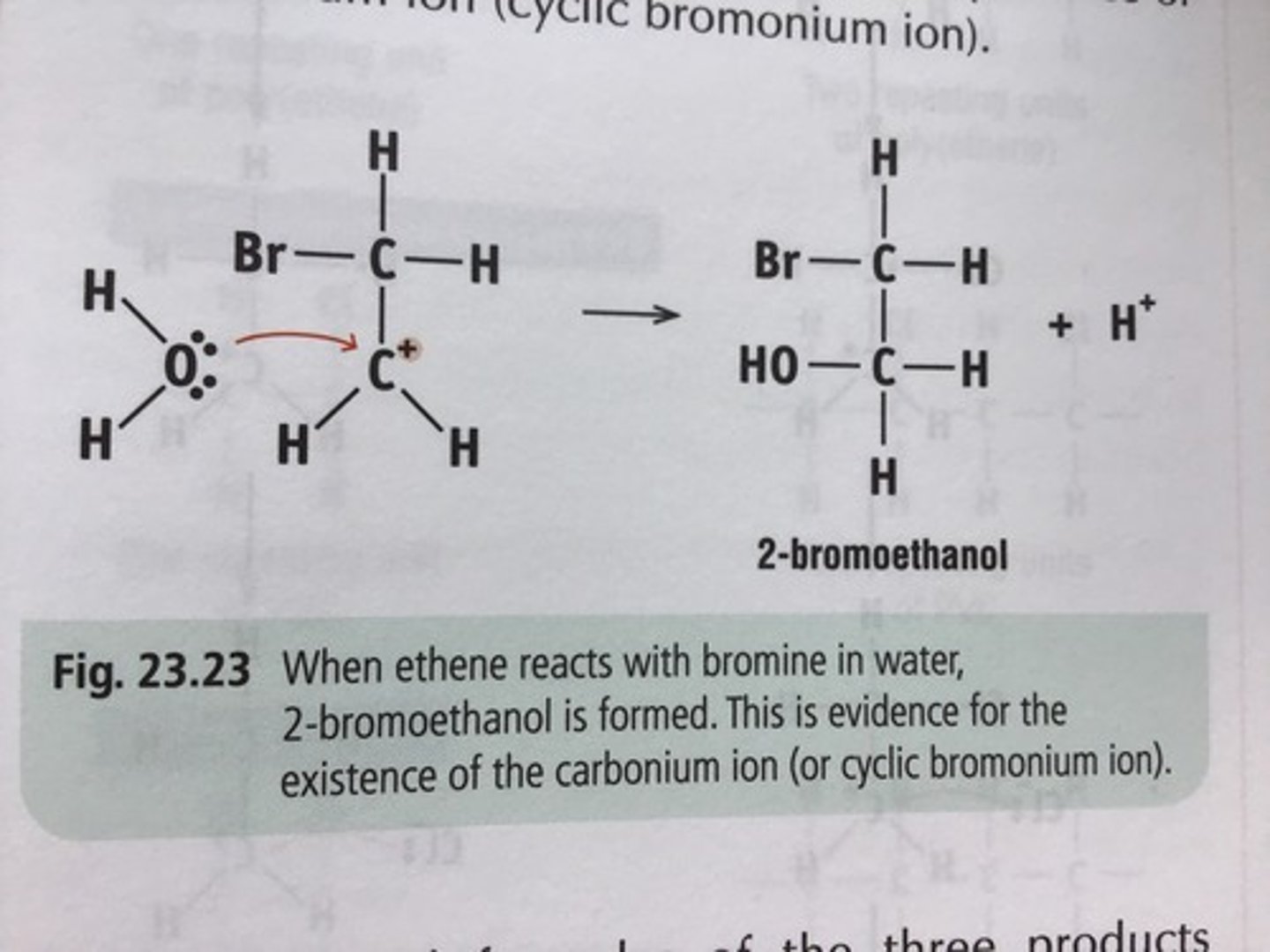
state the colour change during test for C=C bond with potassium manganate (VII)
purple to colourless
describe what happens to the alkene when it is shaken with acidified potassium manganate
- the C=C bond is oxidised to form a diol (2 -OH groups)
- the O from OH is provided by MnO₄- and H₂O
- the H from OH provided by H₂O
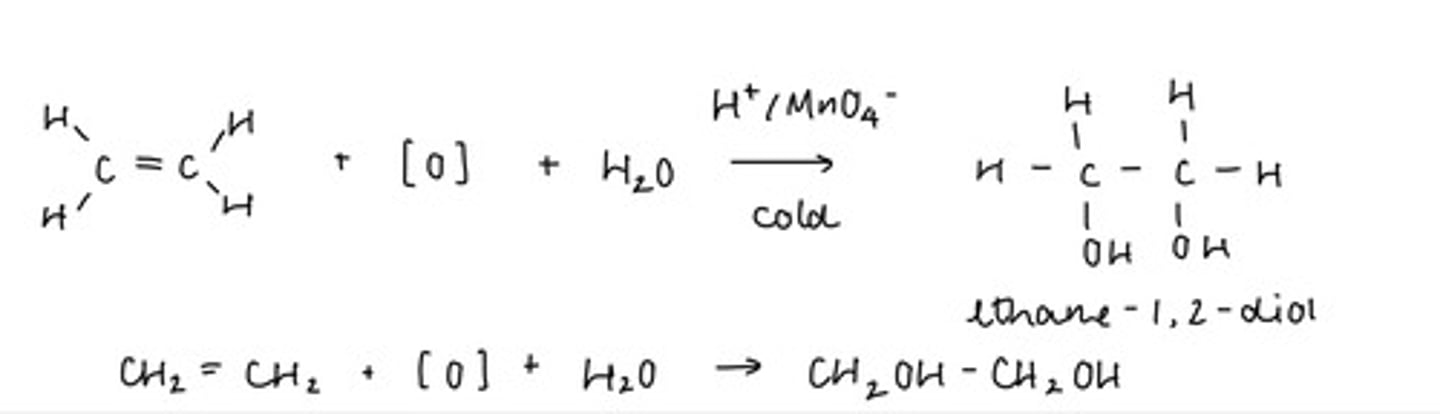
what is formed when ethene reacts with potassium manganate?
- potassium manganate is a strong oxidising agent
- forms a diol with ethene → ethane-1,2-diol

ethene reacts with acidified potassium manganate(VII). which of the statements below are correct?
1) the organic product can show geometric isomerism
2) the type of reaction that takes place is oxidation
3) the bond angles around each carbon atom are greater in the product than in ethene
4) the acidified potassium manganate(VII) decolourises in the reaction
only 2 and 4
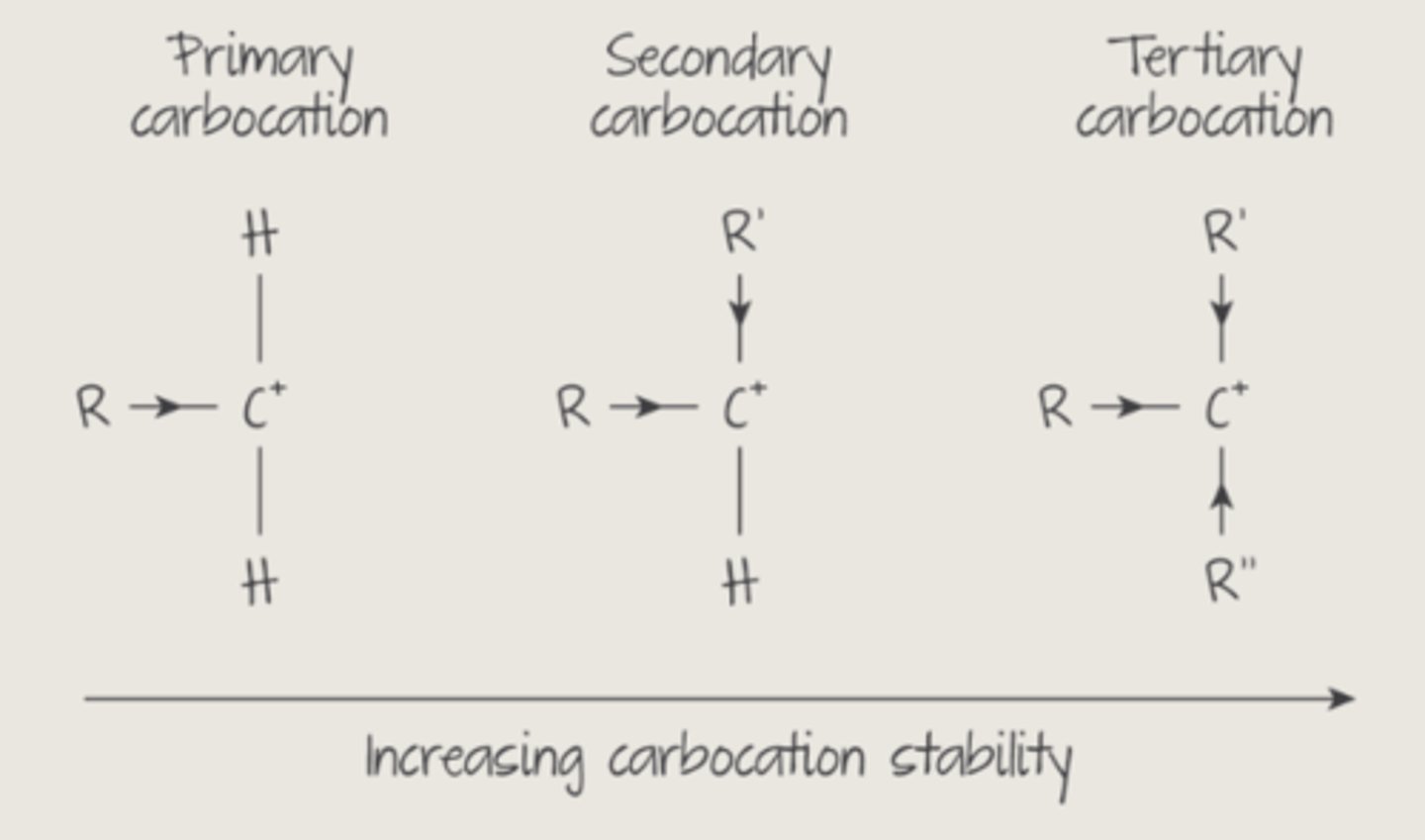
what are 1°, 2° and 3° carbocations and which is most stable? illustrate with reference to the positive inductive effect
most stable 3° > 2° > 1° least stable
alkyl groups tend to push e- away, which means the alkyl group becomes slightly positive and the C they're attached to becomes slightly negative (positive inductive effect)
differentiating major / minor product
- the H+ always joins to the C which is bonded to the most H atoms (major product)
- only on asymmetrical alkenes
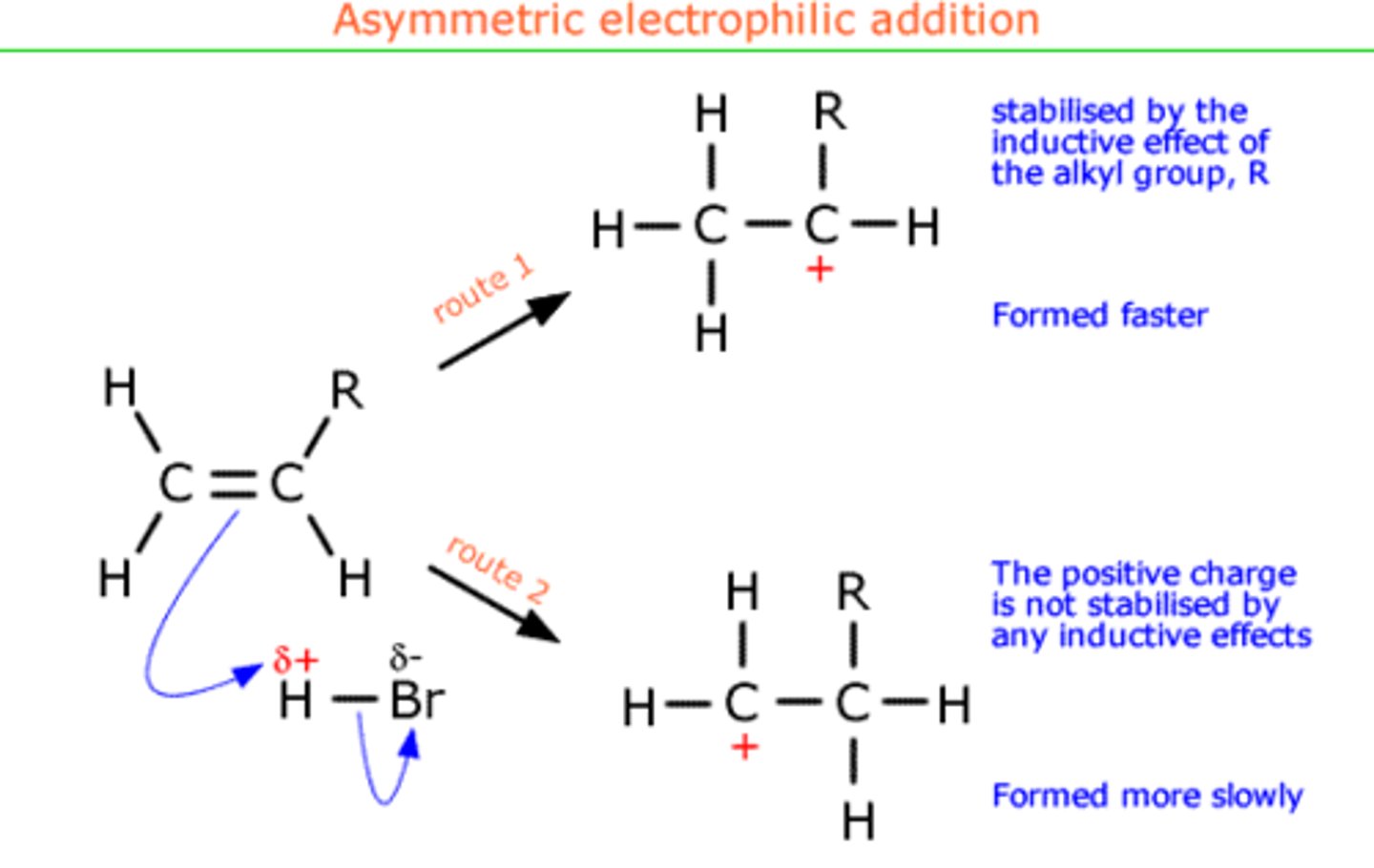
explain why 1,2-dibromopropane is the major product instead of 1,3-dibromopropane
- 1,2-dibromopropane formed from secondary carbocation
- secondary carbocation is more stable
define nucleophile
- an electron pair donor (electron rich)
- species with a lone pair
-seek out areas with high positive charge density (eg electron deficient carbon)
- eg OH⁻, CN⁻, NH₃, H₂O
explain why are halogenoalkanes are open to attack by nucleophiles
- halogens have a greater negativity than carbon
- a dipole is induced in the C-X bond, causing it to become polar
- carbon is thus open to nucleophilic attack
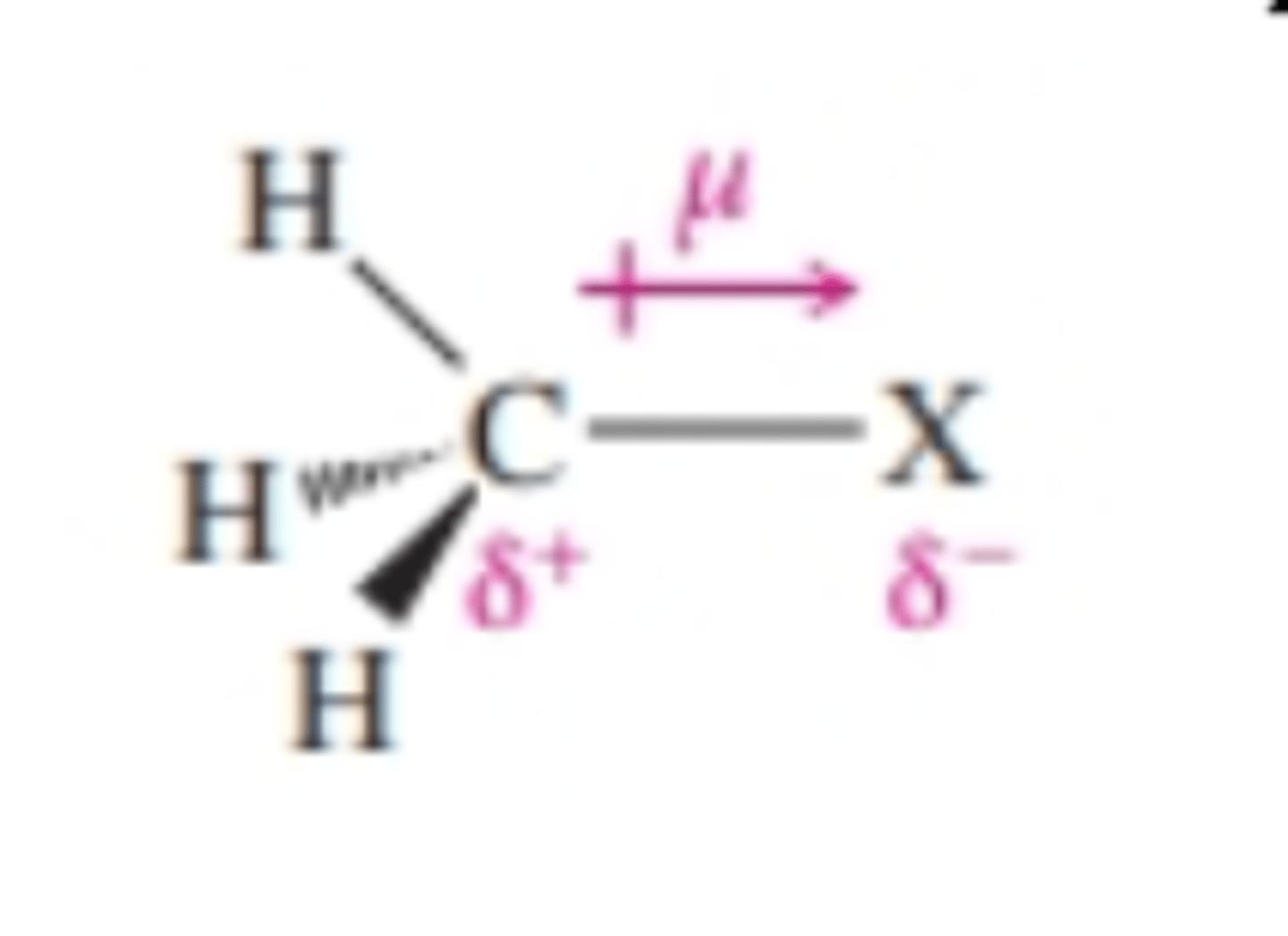
state whether halogenoalkanes are soluble in water
- insoluble as C-H bonds are non-polar
- not compensated for enough by the polarity of the C-X bond polarity
what is the effect of a charged rod on a stream of liquid 1-bromobutane
stream is diverted because halogenoalkanes are somewhat polar due to the C-X bond
describe the trend in bp in relation to the reactivity of halogenoalkanes
- all have higher bp than their alkene counterparts because they have more electrons
- stronger LDF
- polarity of C-X bond also creates permanent dipole-dipole interactions
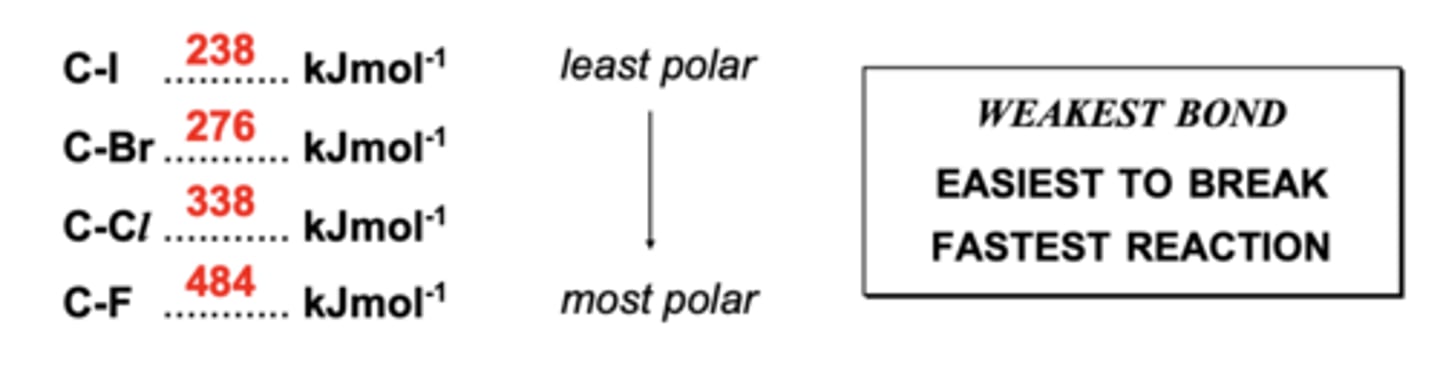
describe the trend in reactivity of chloro-, bromo- and iodoalkanes
- iodoalkanes are hydrolysed the quickest
- this is because C-I has the lowest bond enthalpy, so the bond breaks first
- the larger the halogen, the longer the C-X bond, hence the lower the bond enthalpy
- size of halogens increase down the group
devise an experiment to compare the rates of hydrolysis of 2-chlorobutane, 2-bromobutane and 2-iodobutane.
state the trend in the rates of reaction and justify your answer.
- ETHANOL: use of ethanol as a cosolvent
- FAIR TEST: use of equal amounts / volume in each test tube
- SILVER NITRATE: add silver nitrate solution to each test tube
- TIME: find the time taken for precipitate to form
- RATE: expected trend is 2-iodobutane > 2-bromobutane > 2-chlorobutane
- BOND ENTHALPY: bond enthalpy in C-I is the weakest / bond strength decreases down the group
explain why ethanol was added to each test tube
to increase solubility of halogenoalkane
give a reason why the test tubes were left in the water bath for a long period of time before the halogenoalkanes were added
to allow the solution to equilibrate / reach the require temperature
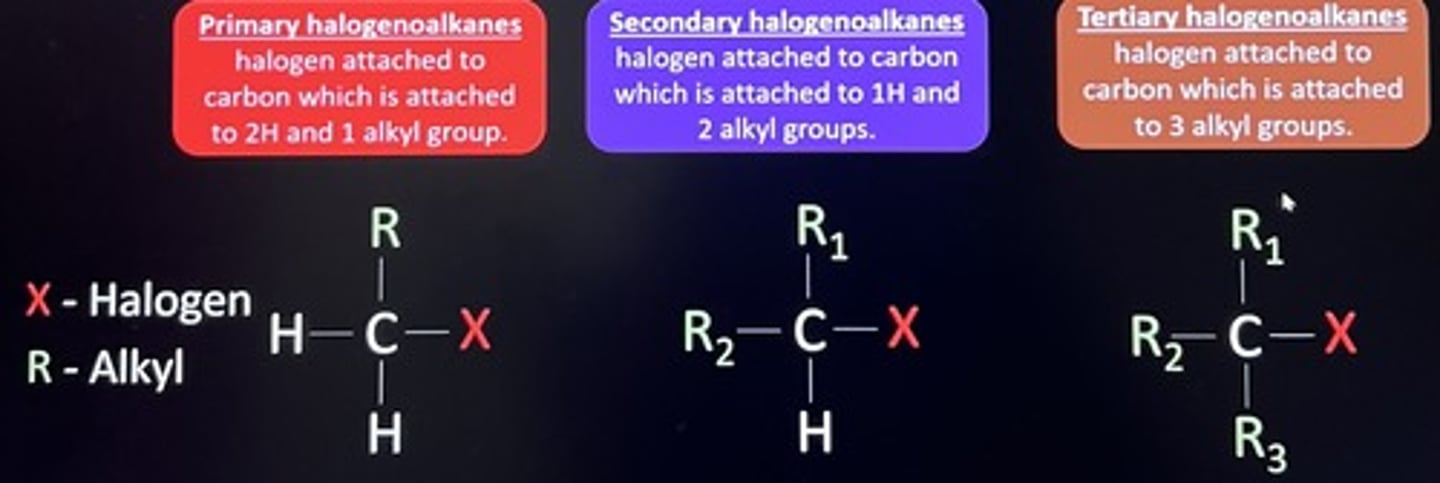
★ describe the order of rate of reaction between primary, secondary and tertiary halogenoalkanes
least stable (fastest rate)
3° > 2° > 1°
most stable (slowest rate)
the reactions of ammonia and of hydroxide ions with halogenoalkanes are similar.
compare the rate of reaction of ammonia with 1‑chlorobutane and with 2‑bromo‑2‑methylpropane. justify your answer.
- rate of reaction is slower for 1-chlorobutane
- C-Cl has higher bond enthalpy than C-Br
- 1-chlorobutane is a primary halogenoalkane while 2-bromo-2-methylpropane is a tertiary halogenoalkane
describe an experiment to compare the rate of substitution reaction between primary, secondary and tertiary halogenoalkanes
your plan should include:
- chemicals you will use
- outline of experiment
- observation or measurements you will make
- use equal amounts of halogenoalkanes
- equal amounts of silver nitrate solution
- use isomeric primary, secondary and tertiary bromoalkanes
- time how long it takes for each precipitate to form
- shorter the time = faster the rate
- tertiary > secondary > primary
further test to determine halogenoalkane with ammonia
- AgCl is soluble in both dilute and concentrated ammonia
- AgBr is soluble only in concentrated ammonia
- AgI is insoluble
a silver halide dissolved in concentrated ammonia to form a colourless solution.
explain why this result does not prove conclusively that the silver halide was silver bromide and give a further test to confirm that the silver halide is silver bromide
- both silver chloride and silver bromide dissolve in concentrated ammonia
- however if the solid doesn't dissolve in dilute ammonia then it is silver bromide
describe the test for halogenoalkane
1. warm in NaOH and ethanol
2. add nitric acid
3. add silver nitrate solution
→ WHITE (AgCl), CREAM for (AgBr), YELLOW for (AgI)
write the ionic equation, including state symbols, for the reaction involving the silver nitrate in the test tube with 1-chlorobutane
Ag⁺ (aq) + Cl⁻ (aq) → AgCl (s)
★ precipitate form as a result of reactions between aqueous silver ions and aqueous halide ions.
explain why halide ions are present in the mixture containing a halogenoalkane which has only covalent bonds.
- halogenoalkane is hydrolysed by water
- the C-X bond breaks heterolytically, producing ions