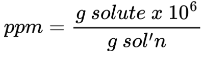LESSON 1: Solutions and their Properties
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Solutions
homogeneous mixtures ; are mixtures of two or more substances evenly distributed throughout a single phase
Solubility
usually increases with temperature ; how much can be dissolved ; is the maximum amount of a solute that can dissolve in a given amount of solvent at a specific temperature
Solute
is the susbstance dissolved in a solution and is usually present in smaller amount
Solvent
is the dissolving medium
Unsaturated Solution
contains less solute than the solvent’s capacity to dissolve
Saturated Solution
contains the maximum amount of solute that the solvent can dissolve at a certain temperature
Supersaturated Solution
contains more dissolved solute than is present in a saturated solution
Concentration
is a measure of the amount of solute in a given amount of solvent or solution
Mole Fraction
equal to the number of moles of that component divided by the total number of moles of all the components present
Mole Fraction Formula
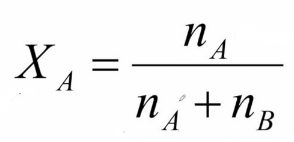
Molality
is the number of moles of solute per kilogram of solvent
Molality Formula
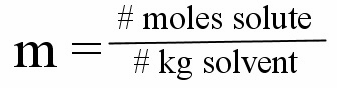
Molarity
is the number of moles of solute per liter of solution
Molarity Formula
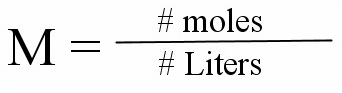
Temperature
a measure of the average kinetic energy of the molecules ; if it increases it raises the kinetic energy of the solution, allowing solvent to more effectively break apart the intermolecular forces of attractions between solute particles
Pressure
has an appreciable effect on the solubility of gases in gas-liquid solutions, but it has no effect on the solubility of solid salts in water ; according to Henry's Law, increasing the pressure of a gas above a liquid increases its solubility, while decreasing the pressure decreases its solubility
Soluble
something that can dissolve
Insoluble
something that cannot be dissolved
Suspension
is a heterogeneous mixture containing large solid particles dispersed in a liquid or gas, which do not dissolve and will settle out over time due to gravity
Nature of Solute and Solvent
most ionic solutes are soluble in water ; refer to solubility rules ; soluble compounds with group IA, NH4+. NO3 ; for covalent compounds: “Like dissolves like”
Mole Fraction Formula
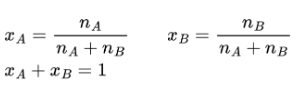
Percent by Mass Formula
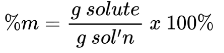
Percent by Volume Formula
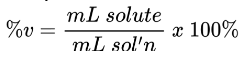
Percent by Mass to Volume Formula
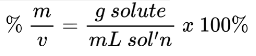
Parts per Million Formula