CHEM 1112 -Test #3
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
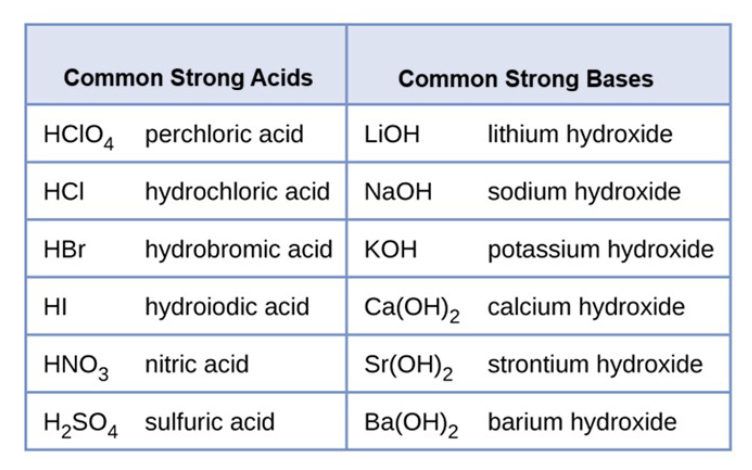
Acid vs. Base
Acid: A proton (H+) donor: is a substance that produced H+ ions in aqueous solution
Base: A proton Acceptor: is a substance that produces OH- ions in aqueous solutions
Lewis Acid vs. Lewis Base
Lewis Acid: electron pair acceptor
Lewis Base: electron pair donor
Conjugate Base vs. Conjugate Acid
Conjugate Base: the base formed when an acid donates a single proton
Conjugate Acid: the acid formed when the accpetance of single proton by a base
What is the Auto IONIZATION of Water?
The Kw (Equil. constant of Water) ONLY AT 25*C is = 1.0×10-14 and the equation is:
Kw = [H+]x[OH-]
Where [H+] = [OH-] = 1.0×10-7
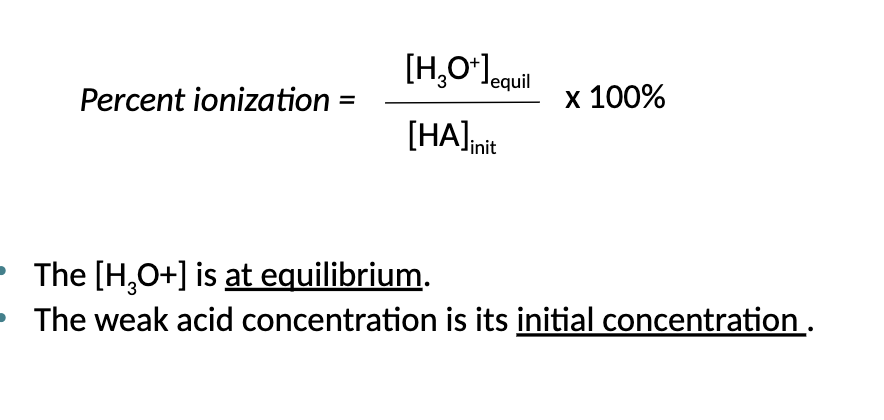
What is the Ka Represent?
Ka Represents the equilibrium constant for the dissociate of an acid (it indicates strength of Acid)
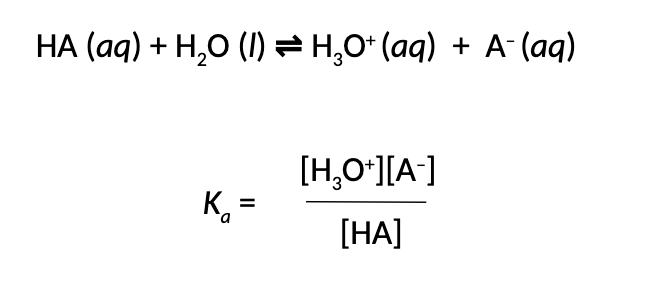
What is the formula for the Percent Ionization of a weak Acid
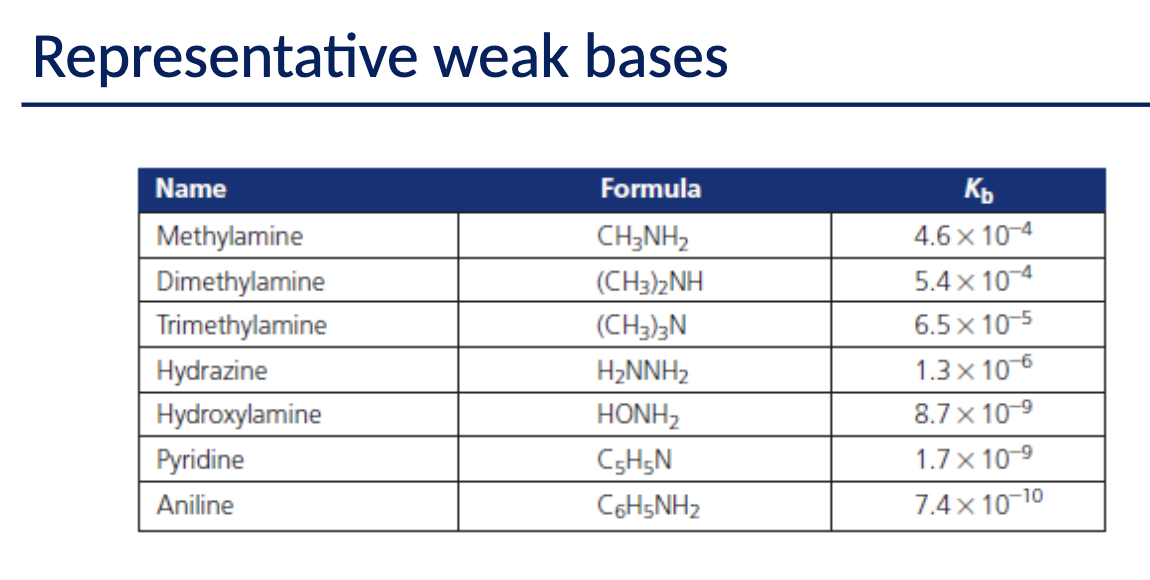
What are the types of Common Acids and Common Bases?
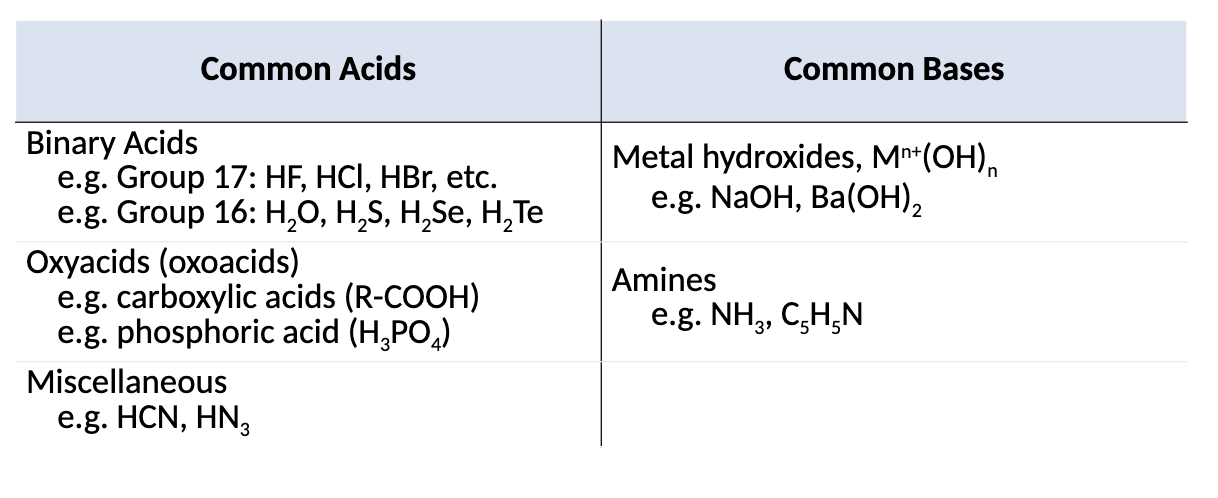
What is are Oxoacids?
acids that contain an inner atom bonded to a variable number of oxygen and acidic OH groups
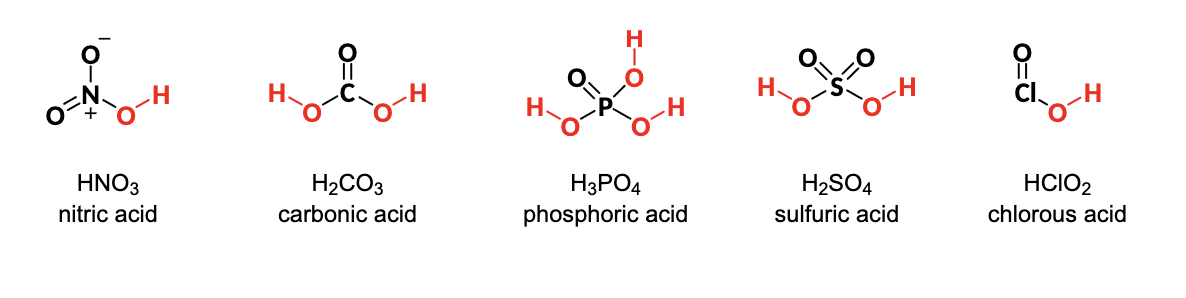
Any acids with a _____ in their molecular formula is a weak Acid
CO2H = usually weak acids
What can you tell about an acid if their Ka < 1?
Weak Acid
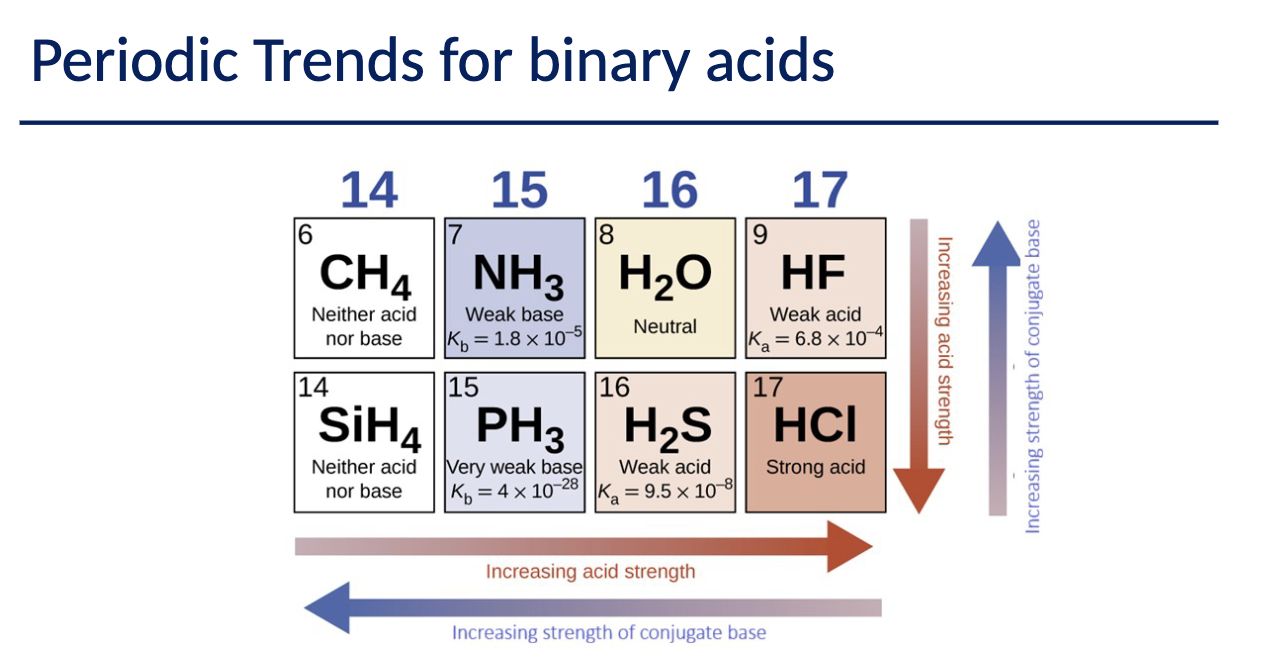

HIO3 as its the most stable, contain more electronegative Ions (3O), has more resonance structures in respect with Oxygen.
What is the reaction of an Acid and Base called?
Acid-base neutrali
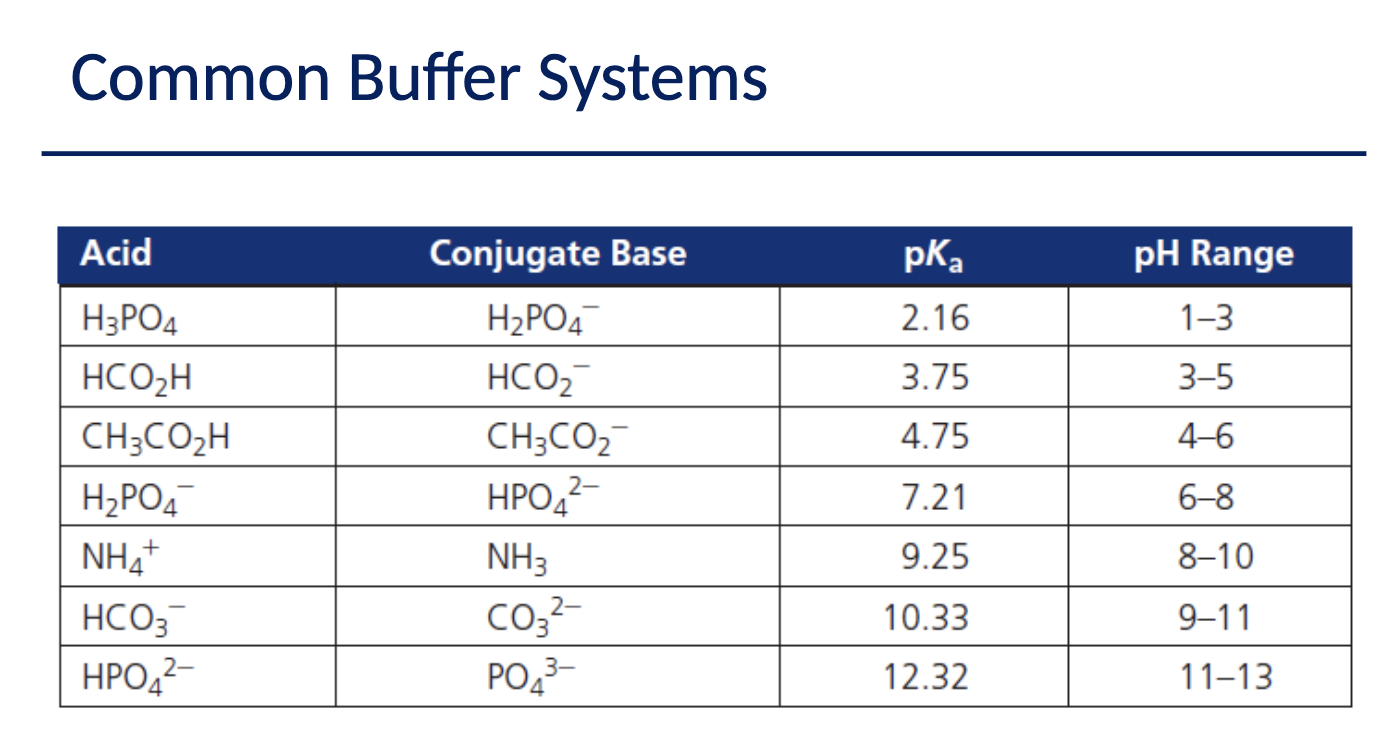
What do Buffers do?
resist changes in pH upon the addition of acid or base
Oxidized vs. Reduced
Oxidized molecules lose electrons (thus they are the reducing agent)
Reduced molecules gain electrons (thus they are the oxidizing agent)