Metallurgy Midterm Reference Sheet
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
What are the five main strengthening mechanisms for metals?
Solution strengthening (solid solution strengthenining)
cold work strengthening (work hardening)
grain refinement
precipitation strengthening (hardening)
transformation strengthening (hardeningn)
You run a tensile test and have an original gage length of 2 inches. After the test your gage length is 2.40. Calculate your % elongation.
Equation:
%Elongation = (final length - original length) x 100
_____________________
Original length
Answer: 20%
Explain how solution strengthening of metals work
impedes the movement of dislocation by distorting the crystal structure
substitutional solutes and interstitial solutes distort the crysal structure in different ways
simplest of all strengthening mechanisms
occurs during manufacture of the alloy - no further processing or heat treatment is required
welding has the least effect on solution strengthening
one of the most weldable alloys
Which would require a higher preheat temperature before welding a steel with a carbon equivalent of .5 or steel with a carbon equivalent of .3. Why?
Carbon Equivalent of .5 would need a higher preheat temperature depending on the thickness of the metal
carbon equivalent:
CE is under .40 you can weld without any precautions (room temp)
CE is between .4 and .5 you need to think about preheat, especially in thick sections
CE is between .5 and .6 you need to preheat most thichnesses and you should think about post weld heat treatment.
CE above .6 requires preheat and post weld heat treatment for all thicknesses
What does the acronym “BONCH” stand for and why is it important in metallurgy?
Boron, Oxygen, nitrogen, carbon, hydrogen
*IMPORTANCE-
These five elements matter in metallurgy because they are tiny atoms that easily mix with hot metals and strongly change how metals behave when cooled, bent, or used.
What is dislocation? Why it it important to metallurgy?
crystals that have a defect
important because dislocation reduces the aount of force required to plastically deform a metal
the easier it is to move dislocation the softer and more ductile the metal
you can make metal stronger by impeding movement of dislocation
every strengthening mechanism works by impeding the movements of dislocation
When talking about solubility in metals what is the saturation point?
the maximum amount of solute (such as carbon or alloy elements) that a metal solvent can hold in a solid solution at a specific temperature
What is the relationship between solubility and temperature?
-Higher temp. increases solubility
*if you heat the water that has crystals in the bottom, the crystals will dissolve and you can addmore
*if you cool it back down the salt crystals will form and drop to the bottom again
-solid metals act in the same way
In general, what is the relationship between strength and ductility in metals?
Ductility - ability of a material to stretch without breaking (how much a material can plastically deform)
Strength- material resistance to deformation
Describe how welding affects solution strengthen metals
Welding has the least effect on solution strengthening than any other strengthening mechanisms
any strength loss will be due to change in grain structure or segregation of solute atoms
stress concentration from the weld bead may also reduce strength
strength loss would be typically be less than 10%
Of the steel chemistries below, which would you expect to have the highest hardenability? Why?
A. 1% manganese, .3% carbon, .2% nickel, .5%chromium, .7% molybdenum
B. 1% manganese, .3% carbon, .4% nickel, .8% chromium, .9% molybdenum
C. 1% manganese, .3% carbon, .3% nickel, .7%chromium, .7% molybdenum
D. 1% manganese, .3% carbon, .3% nickel, .8% chromium, .6% molybdenum
B will have the highest hardenability.
All four steels have the same carbon and manganese, so hardenability differences come from nickel, chromium, and especially molybdenum, which is the strongest hardenability raiser among common alloying elements.
Explain what is happening metallurgically when you austenitize and quench 4140 steel?
When you austenitize and quench 4140, you are changing its internal structure from a softer, mixed microstructure to very hard martensite by heating and then “freezing” the atoms in place with a fast cool.
What is “pig iron”?
Another word for cast iron that is produced in a blast furnace
fluid when molten
cast iron melts at a relatively low temp.
shrinks very little as it cools
excellent material for making castings
Which alloying element has the greatest effect on the strength of steel and why?
Carbon because it raises tensile strength, hardness and resistance to wear and abrasion. Lowers ductility, toughness
in the 6010 sample of the hydrogen cracking lab, why didn’t the weld metal turn to martensite even though you did no preheat?
6010 is a high hydrogen electrode that has a cellulose coating that forms hydrogen when welding and it cools quickly in the HAZ so the microstructure is mostly ferrite and pearlite
In the lab that you ran on 5052 aluminum welded with ER5356 filler, why was the weld bead harder than the base material?
weld filler are designed to provide a chemistry in the weld pool that has minimizes crack sensitivity
filler metal is harder because the filler metal is richer in magnesium than the base metal causing a solution strengthening in the weld metal
What is the difference between an atom and a molecule?
Atoms:
All matter is made of atoms
smallest unit that can be considered specific material
atoms are made up of three basic subatomic particles (protons, neutrons, electrons)
Molecules:
atoms combines with one another forms molecules
can be very simple such as a water molecule
can also be complex containing several atoms
can be extremely complex containing millions or billions of atoms of different kinds such as in human DNA molecule (204 molecule)
What crystal structure does iron have at 1900 degree F?
Cubic Face centered (FCC) (because FCC is soft metals, ductile, strecthable, heat iron, it changes structure)
What are the three types of crystal structures (lattice)?
cubic body centered (bcc) - unit cell that is a cube with an atom at each corner and one in the center
Cubic face centered (FCC) - is a cube with an atom at each corner and on e in the centerre and one in the center of each side of the cube,
Close packed Hexagonal (CPH)- unit cell is a hexagonal with a reference atom that is surrounded by 12 atoms that are an equal distance from the reference atom.
Explain the relationship between hardenability and weldability in steel.
hardenability goes up, weldability usually goes down
high hardenability makes welding difficult
as weld cools shrinkage increase
if the material cannot give to relieve the shrinkage stresses, the material will crack
if hydrogen level is high and martensite is high, delayed cracking is likely
formation of high levels of martensite during welding must be avoid
What crystal structure does iron have at room temperature?
Cubic body centered (BCC)
Explain how transformation strengthening strengthens metal.
how most steel acheive their strength
possible due to to the fact that iron undegoes a phase transformation (allotropic transformation) upon heating and cooling
What is the difference between substitutional and interstitial solutes?
Substitutional solutes - replaces Solent atoms in the atomic structure
distorts the crystal structure by the difference in size between the solute atom and the base material atom
Interstitial Solutes - fits in the space (interstitial space) between solvent atoms in the atomic structure
distorts the crystal structure by getting between atoms and pushing them apart
pushing on small atoms in between other atoms to create distortion
What is carbon equivalent and how does it affect preheat temperature?
Carbon equivalent- factor that indicates the total relationship of the percentage of carbon, silicone, and phosphorus in the chemical compostion
How it affects preheat temp.:
CE is under .40 you can weld without any precautions, (room temp)
CE is between .4 and .5 you need to think about using preheat, especially in thick sections
CE is between .5 and .6 you nedd to preheat most thicknesses and you should think about pst weld treatment
CE is above .6 requires preheat and post weld heat treatment for all thicknesses
Describe how grain boundaries are formed during weld metal solidification.
During the process of neucleation - Liquid metal solidifies small crystal (dendrites) begin to grow in the liquid
as the liquid beings to solidify the dentrites grow larger and eventually grow together to form grain boundaries
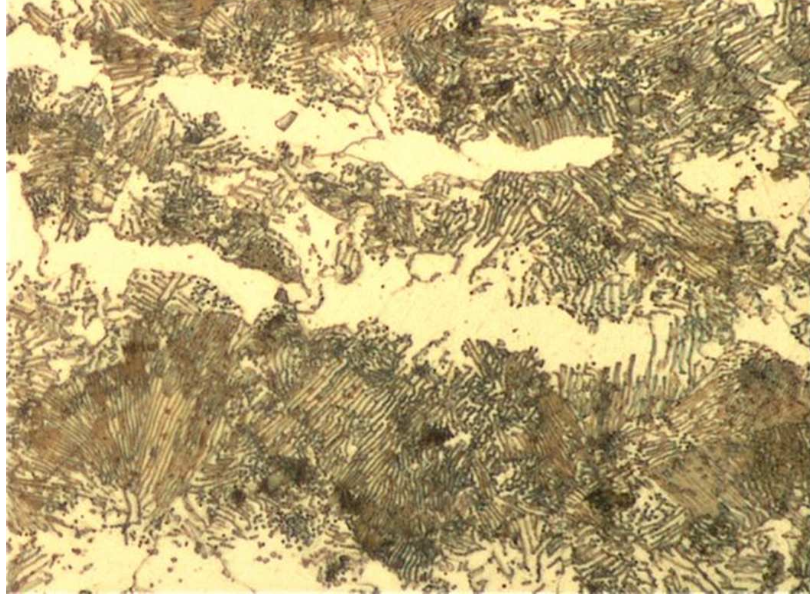
Explain this microstructure
mircrostructure is a mixture of ferrite and pearlite
Which of the following forms would have a grain structure most similar to a weld?
Forging
Rolled Plate
Extrusion
Casting
Casting
What is the relationship between grain size and a metal’s toughness?
As grain size gets smaller- the metal gets tougher
As grain size gets smaller, the metal gets tougher
Describe how precipitation hardening strengthen metal
The precipitates distort the crystal structure and impede the movement of dislocation
precipitation hardening has 2 steps:
solution treating/ quenching
aging
You run a tensile test and get a maximum load of 4560 lbs. Your width and thickness are ½ inch wide .125 inch thick. Calculate the tensile strength
First find cross section area: A=TxW
Secon calculate UTS: UTS= (breaking load) divded by cross sectin area
answer: 72,960 psi
What grain direction in a cold rolled plate has the most strength?
A. longitudinal
b. long transverse
c. short transverse
d. martensite
longitudinal
What is the main function of preheating a steel alloy prior to welding?
lower mositure on the joint at the beginging of welding reduces the possibility of porosity and hyrdogen induced cracking
preheat reduces residual stresses and distortion
preheat is used to slow the cooling rate of welds in steels with high hardenability
Which grain direction in cold rolled plate has the least strength?
a. longitudinal
b. long transverse
c. short transverse
d. Martensite
short transverse
Explain why a metal with small grain size is stronger than a metal with large grain size.
The warpage of the crystal structure at grain boundaries blocks the movement of dislocation which makes metals stronger and tougher
Explain the difference between elastic and plastic deformation
Elastic: is not permanent
the part will return to it’s original shape after the load is removed
stong brittle metals deform more elastically
knife balde
springs
titanium
Plastic: is permanent
the part will not return to its original shape after the load is removed
plastic defromation is called “worl"k”
weak soft metals deform more plastically
copper plumbing
lead
gold
carbon steel
Explain the equation below in respect to resistance welding
Q= I2RT
how much heat is generated in resistance welding, and why current, resistance, and time are so critical.
The process relies on Joule heating: when high current passes through the contact between sheets, their resistance concentrates heat at that interface and forms the weld nugget
Which metal would have a greater ductility. One with 20% cold work or one with 50% cold work?
One with 20% cold work
What effect does annealing have on cold worked metal?
annealing removes the cold work strengthening process; plastic deformation can be continued again
Describe how welding affects the strength of precipitation hardened metals.
the heat from welding affects differeent allys in different ways
in aluminum and magnesium alloys welding heat overages the base metal in the heat affected zone very rapidly
the higher the heat input the greater the degree of overaginging
strength loss is typically around 30% to 50%
Name two ways to obtain fine grains in metal.
chemically- grain refining additions during manufacture of the metal
heat treatment- heating to just above the recrystallization temperature in materials that undergo a phase transformation or have cold work
cold work- cold rolling during processing of the metal into shapes
What is a metal ore?
Ores are naturally occurring rocks that contain metals or metal compounds in sufficient amounts to make it worthwhile extracting them.
most metals like steel, aluminum, and titanium are ectracted from ore
How does steel differ from cast iron?
Composition?
Properties?
Cast iron: produces in blast furnace, very fluid when molten, melts at low temp., little shrinkage when cooled
poor weldability, used for non structural items
Composition: 4% to 6% carbon which makes it very brittle
Properties: high compressive strength, excellent material for making castings
Steel: pure iron has excellent weldability
Compostion: manganese and carbon are added to make carbon steels
chromium, molybdenum, nickel, vanadium, managanese and carbon are added to make high strength steels
chromium and nickel are added to make stainless steels
alloying elements are added to achieve carbon steels
Properties: high tensile strength, durability and ductility
How does cold working strengthen metal?
as dislocation get pinned, plastic deformation becomes more difficult
the more you plastically deform, the more dislocation you create and the stronger the material gets
work hardened materials can be much stronger than annealed materials
What happens metallurgically when a precipitation hardened metal overages?
if you age long enough, the precipitates will grow so large they become incoherent
when this happens they no longer distort the crystal structure and the alloy loses strength
Name three “forms” of a metal.
the form of a metal refers to how it is made it’s shape
there are many metal forms:
sheet and plate, shapes (S and W beams angle, channel, etc. ), foil, wire, castings and forgings, extrusions
most metals forms start as a large ingot of material
the ingot is basically a giant casting
What effect does welding have on cold strengthened metal?
the heat from weld effectively eliminates all work hardening and grain refining in the weld and heat affected zone
work hardened and grain refined metals are softened by welding
failure will occur in the heat affected zone
the metal exhibits full soft properties in the weld and heat affected zone
the strength will gradually ramp up to full base metal properties
the higher the heat input the greater the distance it takes to get to full properties
Explain the metallurgical difference between a hot rolled and a cold rolled metal.
Cold Rolling: sometimes the steel is descaled and cold rolled
cold rolling allows greater precision in dimension and provides a smooth surface finish
cold rolling also results in higher strength due to cold working
Hot Rolling: vast majority of ingots are heated and rolled to a more useful size or shape known as a billet
the heating makes the metal soft and ductile and prevents work hardening
What is the difference in grain structure between forged material and a cast material?
Forging: forging results in shapes that ranges from simple to complex
results in desirable grain flow patterns which are useful for their strength in different directions
a good example of cold forging is thread rolling (30% stronger than machined threads due to the good grain flow around the thread root
Cast: generally have large grains which varies in size from one area of casting to another
involves pouring hot metal into a mold and letting it solidify
complex shapes can be made economially by casting
casting creates shapes that would be difficult or impossible to produce any other way
You run a tensile test on a sample that have been welded and some that have not been welded. The strengths were:
Welded - 132, 462 psi
Unwelded - 189,278 psi
Calculate the joint efficiency
%Joint Efficiency - welded UTS x 100 / unwelded UTS
answer: 69.9827 psi
69.9827 psi
Which metal would lose the largest percentage of strength when welded? Why?
1/8 hard 304 stainless steel
17-4 PH, annealed
7050 aluminum, precipitation strengthen
7050 aluminum, precipitation strengthen
gets most of its strength from a carefully controlled precipitation‑hardened temper; welding locally heats it into the solution/overaging range and destroys the optimized precipitate structure in the HAZ, causing a very large drop in strength relative to its original high value.
What function does the quench step perform in precipitation hardening?
from the solution temperature the material is quenched
quench rate is different for each alloy
7050- aluminum- water
17-4 PH Stainless - air
quench rate must be fast enough to not allow the alloying elements to precipitate during cooling
if quench rate is fast enough, a super saturated solid solution is created
this saturation where the alloying elements are frozen in solution
they want to precipitate out but the rapid quench did not allow them to
What is the relationship between diffusion rate and temperature?
Diffusion rate - the higher the temperature the faster and farther these atoms ove
the diffusion rate doubles with every 20 F increase in temp
In the lab on precipitation hardening what happened to the strength of the 6061 aluminum when heated? Why?
metal becomes weaker because of aging the alloy at higher temperatures and the time spent overaging
What happens metallurgically during aging of a precipitation hardening material?
first the metal is heated up to a temperature where all the alloy elements are dissolved in a solid solution
the metal is then quenched fast so that the dissolved alloy elements do not have enough time to precipitate out-forming a super saturated solid solution.
Second: involves heating the metal up to a lower temp. to allow the alloy elements to precipitate out in a controlled way. Fine precipitates warp the cruystal structure of the material and impede the movement of dislocation. when dislocation movement is impeded, the metal becomes stronger
What 3 functions of flux in SMAW?
Shielding the molten weld pool
The flux melts and produces gases that surround the arc and molten metal, protecting it from atmospheric contamination (oxygen, nitrogen, moisture).
This reduces porosity and other gas-related defects in the weld.
Forming slag to protect and shape the weld
Molten flux solidifies on top of the weld as slag, shielding the hot metal during cooling and slowing the cooling rate.
This helps refine the weld metal structure and shapes the bead profile; the slag is chipped off afterward.
Stabilizing and improving the arc and weld metal
Flux ingredients help start and stabilize the arc, making it smoother and easier to control.
They can also add alloying and deoxidizing elements to the weld metal, improving strength, toughness, and cleanliness.
What is the major difference between a low hydrogen and a high hydrogen coating on a SMAW electrode?
Describe how welding affects the properties in the HAZ of transformation strengthened high strength steels.

Which of these microstructure would you want in the HAZ of your weld?
austenite - first image
Why do you store 7018 SMAW electrodes in an electrode oven?
7018 is a low hydrogen electrode that will capture moisture is left out.
to keep the electrode dry, improperly stored or damp low hydrogen coatings can produce large amounts of hydrogen
Which of the 5 main strengthening mechanisms is least affected by welding?
Solution Strengthening
Explain the difference in heat treat cycle to get the pearlitic transformation versus the martensitic transformation.
To get pearlite (slower cooling)
Heat steel to the austenitizing temperature (above the critical temperature so it becomes austenite).
Then cool it slowly – for example, in the furnace or in still air, so the temperature drops gradually through the eutectoid range.
Because cooling is slow, carbon has time to diffuse and separate into ferrite + cementite layers, forming pearlite.
Result: moderate hardness, good ductility and toughness – useful for parts that need strength but also some bend.
To get martensite (quenching)
Again, heat to the austenitizing temperature so the structure is austenite.
Now quench it – cool it very quickly in water, oil, or another quench medium.
The rapid cooling is so fast that carbon cannot diffuse, so the austenite transforms by a diffusionless shear into martensite, a very hard, supersaturated phase.
Result: very high hardness and strength, but low ductility (brittle) unless you temper it afterward.
Explain the difference in strength and ductility between untempered martensite and tempered martensite.
tempered martensite- in this brittle quenches state, steel is virtually useless
to get rid of brittleness of the as-quenched martensite, steel is heated up again to a temperature below the austenzitizng range
this slightly softens the martensite but gets rid of the brittleness
the result is a very strong tough steel
Untempered Martensite
highest strength/hardness but very brittle
very high strength and hardness (often the maximum the steel can reach after quench), but with large internal stresses and a highly strained, supersaturated structure.
Has very low ductility and toughness; it tends to crack suddenly with little plastic deformation, which is why as‑quenched martensite is rarely used directly in service.
When talking about heat treating steel, what is the critical cooling rate?
Critical cooling rate is below the rate that will form martensite
Name 3 things you can do to avoid hydrogen cracking?
eliminate hydrogen (using low hydrogen welding processes)
Eiminate tensile strength ( minimize restaint to reduce residual stress)
Eliminate Susceptible microstructure aka. martensite ( preheat slows the cooling rate to prevent martensite formation during cooling
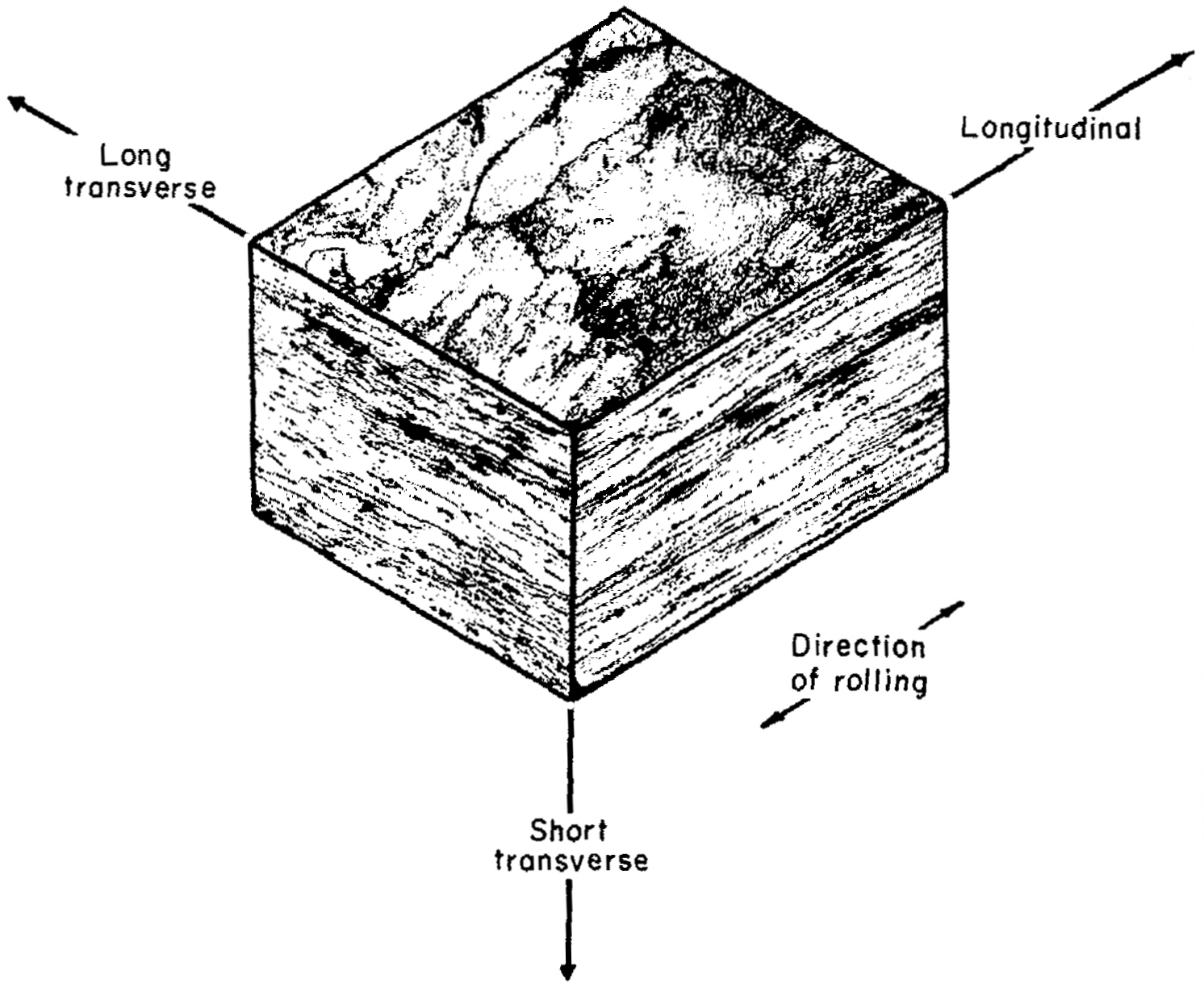
Identify the longitidinal, short transverse, long transverse direction.
Image
What is the relationship between carbon content and weldability of a steel?
What is the “condition” or “temper” of a material?
Low‑carbon steels (about 0.25–0.30% C and below) are the easiest to weld; they stay relatively ductile in the HAZ and have a low risk of cracking under normal procedures.
As carbon rises into medium and high‑carbon ranges, the steel becomes stronger and harder but also more brittle, so the weld and HAZ are much more likely to form hard martensite and crack (especially with hydrogen), requiring preheat, controlled heat input, and often post‑weld heat treatment.
A common rule‑of‑thumb summary: more carbon = higher strength and hardness, but lower ductility and worse weldability (higher cracking risk).
What is the relationship between amount of alloying elements in a steel and it’s hardenability?
Hardenability = how deep into the part you can form martensite when you quench it, not just how hard the surface is.
When you add alloying elements like chromium, manganese, molybdenum, nickel, etc., they slow down the “soft” transformations (like pearlite and bainite), so martensite can form even when the steel cools more slowly and deeper inside a thick section.
So, as the total alloy content goes up, the steel can harden through a thicker cross‑section with the same quench → higher hardenability.
Describe what happens metallurgically during annealing of a highly cold worked metal.
Before annealing: cold‑worked state
Cold working (like rolling, bending, drawing at room temperature) squeezes and stretches the metal’s grains and stuffs them full of defects called dislocations.
This makes the metal stronger and harder but less ductile (it can’t stretch much before it cracks).
During annealing: three main stages
Recovery
You heat the metal, but not enough to melt it.
Atoms start to move just enough to rearrange some dislocations, so internal stresses drop, and the metal gets a bit softer and more ductile, but the grain shape still looks elongated under a microscope.
Recrystallization
At a higher temperature (above the recrystallization temperature), brand‑new, strain‑free grains start to form inside the cold‑worked structure.
These new grains grow and replace the old, deformed grains, so the metal’s strength drops and ductility jumps back up toward its original, “as‑new” condition.
Grain growth (if you keep heating/holding)
If you hold it hot for longer, those new grains grow larger.
This extra growth usually makes the metal even softer and more ductile, but with lower strength.
What is the effect of welding on grain size in the HAZ?
n the HAZ, welding usually makes the metal grains bigger, especially close to the weld.
When you weld, the metal right next to the weld gets heated very hot, almost to where it would melt.
At those high temperatures, the tiny crystals (grains) in the steel start to grow and combine into bigger grains.
Bigger grains make the metal weaker in toughness (it’s easier for cracks to start and spread), even if the strength might still be high.
Farther away from the weld, where it doesn’t get as hot, the grains don’t grow as much and can even stay fairly fine.
Grain Boundaries
are the interface between grains
at grain boundaries the structure is disrupted
most important part of the microstructure
in almost every situation fine grain structures is better than coarse grain structure