TDL2 - Work & Gas Expansion
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
1st TD law
ΔU=Q−W
So ΔU = heat supplied to system minus the work (energy it spends) it performs on the surroundings
closed, open, isolated sys
Can exchange energy but not matter (Eg - stoppered flask) Energy crosses the boundary (heats/cools) but the contents inside cannot leave so matter stays fixed. |
Can exchange both energy and matter with its surroundings (Eg - open flask, cells)
Matter can leave, e.g. evaporation. Matter can be added e.g. - add water |
Can exchange neither heat or matter (Eg - sealed flask that is thermally, mechanically, and electrically insulated from its surroundings) Universe is an example too. This is an idealisation, isolated systems aren't really a thing in practice |
diathermic vs adiabatic walls
A diathermic wall allows thermal contact, meaning two systems separated by it will eventually reach thermal equilibrium (same temperature).
An adiabatic wall prevents heat transfer, so systems cant equalise temperature via heat flow unless energy is transferred in another way (e.g., work).
define work in terms of gas expansion
transfer of energy that achieves or utilises uniform motion of atoms in the surroundings.
W can be classified as mechanical, when work is done all atoms are moving in the same direction, the force is constant - gas pushes air molecules away in the same direction
What is heat in terms of gas expansion
Heat is the transfer of energy because of ΔT between the system and its surroundings.
Mode of E transfer that causes/utilises random atomic or molecular motion in the surroundings.
When a gas expands and pushes back an external pressure, the infinitesimal work done (one tiny segment) is:
dw = −Pex dV
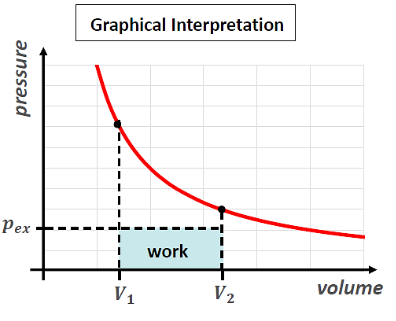
total work from intial (V1) to final volume (V2) gas expansion constant AND non-constant pex

draw a graph of (adiabatic) gas expansion against pex
This is irreversible work
what does isothermal gas expansion mean, how does the WD compare to adiabatic
T is constant during all parts of expansion. Also no change in ΔU
rev = gas is expanding so slowly that Pex = Pgas at all times
ΔU = 0 so q = -W
w = -nRT ln(V2/V1)
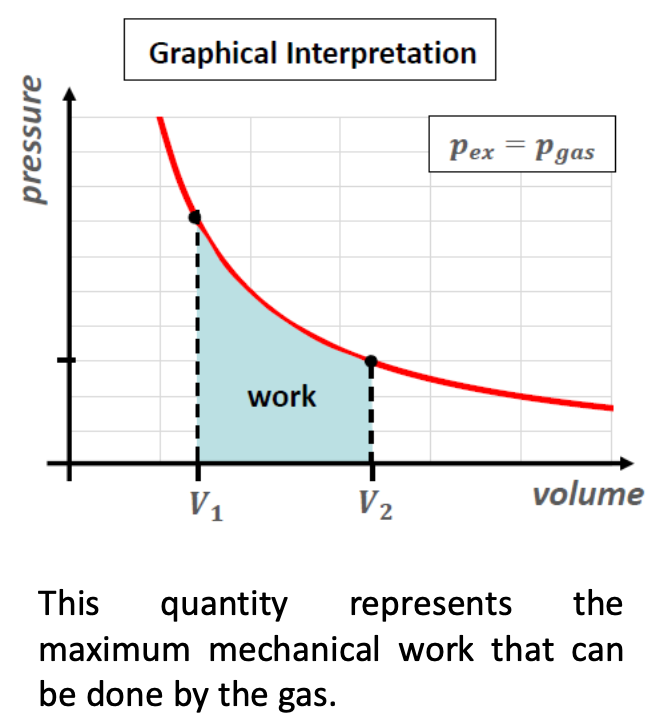
When does a system do max expansion work? Why?
WDsys = pex ΔV
WDsys is max when the sys is always infinetisemally close to mech eq. This happens in reversible expansion.

graph of rev isothermal gas expansion