Electron Transport Chain
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
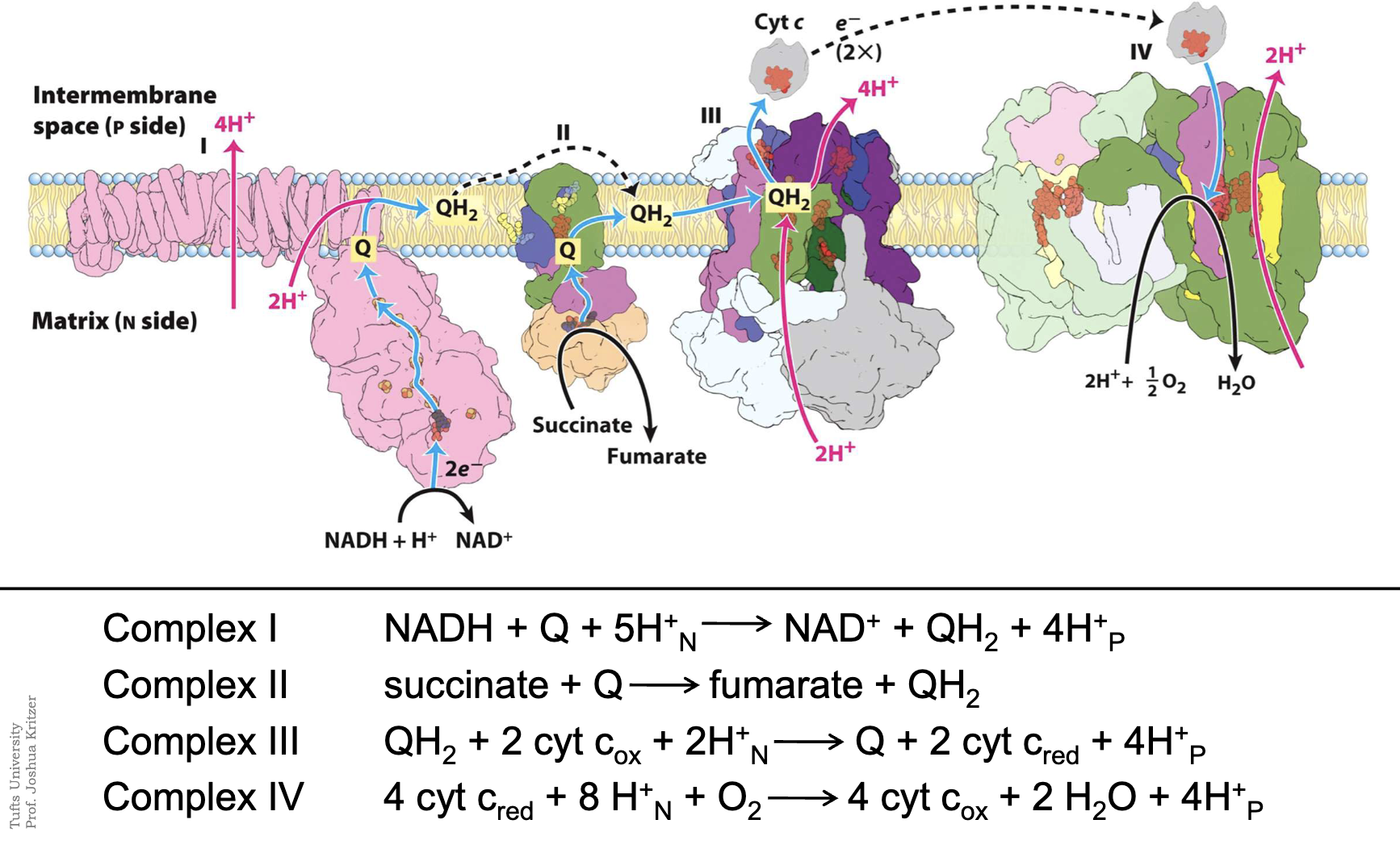
How is ATP production powered?
Proton gradient → proton motive force across membrane (electrochemical gradient)
ATP Synthase: use nrg stored in gradient to make ATP
Complex I (NADH Dehydrogenase)
pumps 4 H into intermembrane space
NADH → NAD+ (donates 2 e-) (cap: 2 e-, 1 H+)
which is accepted by FMN (flavin mononucleotide) → FMNH2
e- move through Fe S clusters (1 e- carrier cycling b/w Fe 2+ / Fe 3+) → NADH -→ FMN —> CoQ red to CoQH2
Coenzyme Q
lipid soluable (bc long "fatty" isoprenoid tail) + mobile e- carrier
interior of membrane + diffuses freely
From Complex I: Receives 2 e- from NADH (via FMN + Fe-S clusters)
From Complex II: Receives 2 e- from Succinate (via FAD + Fe-S clusters).
Once Q is”full” (QH2): to Complex III: delivers e- to the Cyt-B (ox to Q)
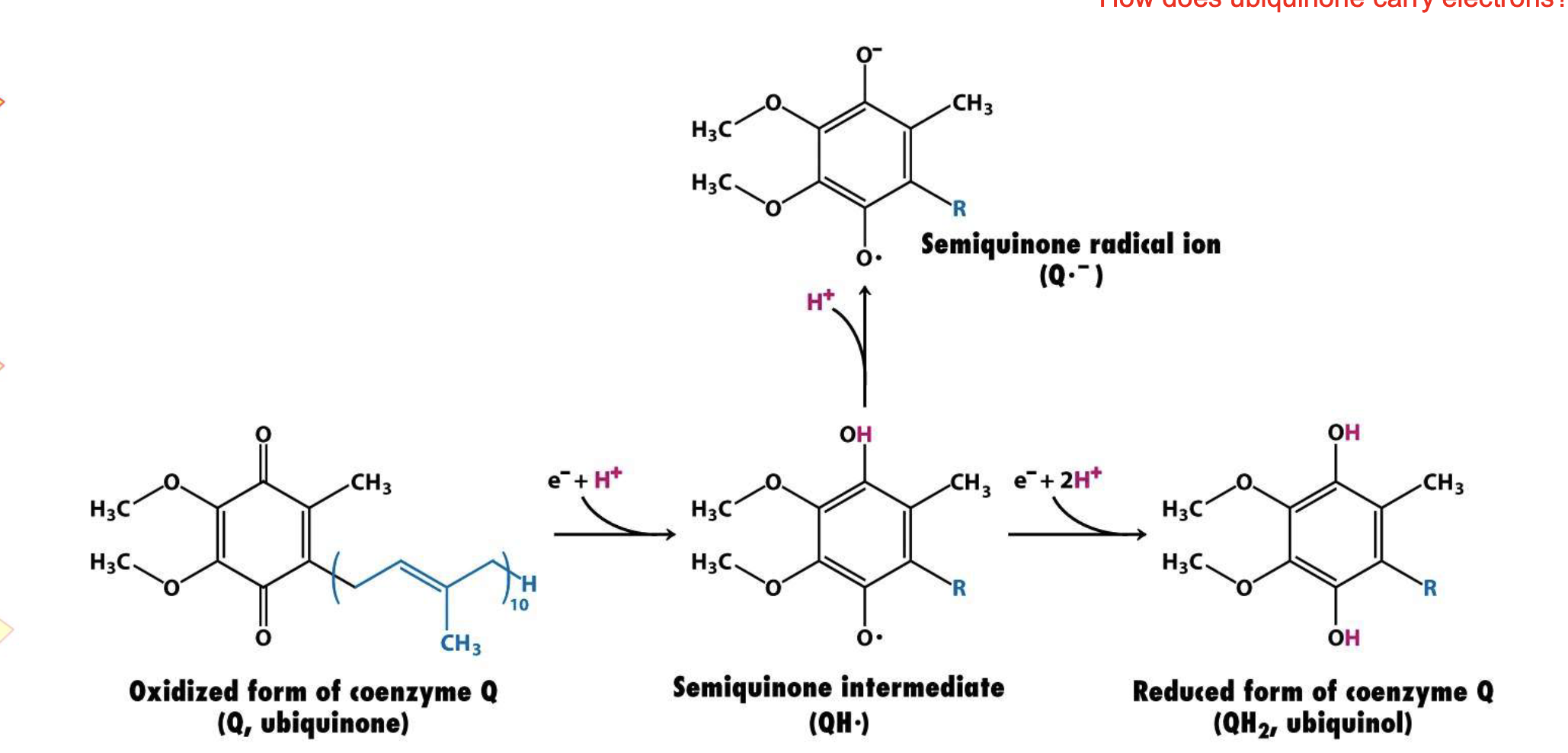
Ubiquione (Q) Redox States
Q (Ubiquinone): fully oxidized, "empty."
Q- (Semiquinone): radical intermed. "half-full" (1 e-) + unstable—If it leaks e- to Oxygen, creates Superoxide (ROS)
QH2 (Ubiquinol): fully reduced. "Full" (2 e- + 2 H)
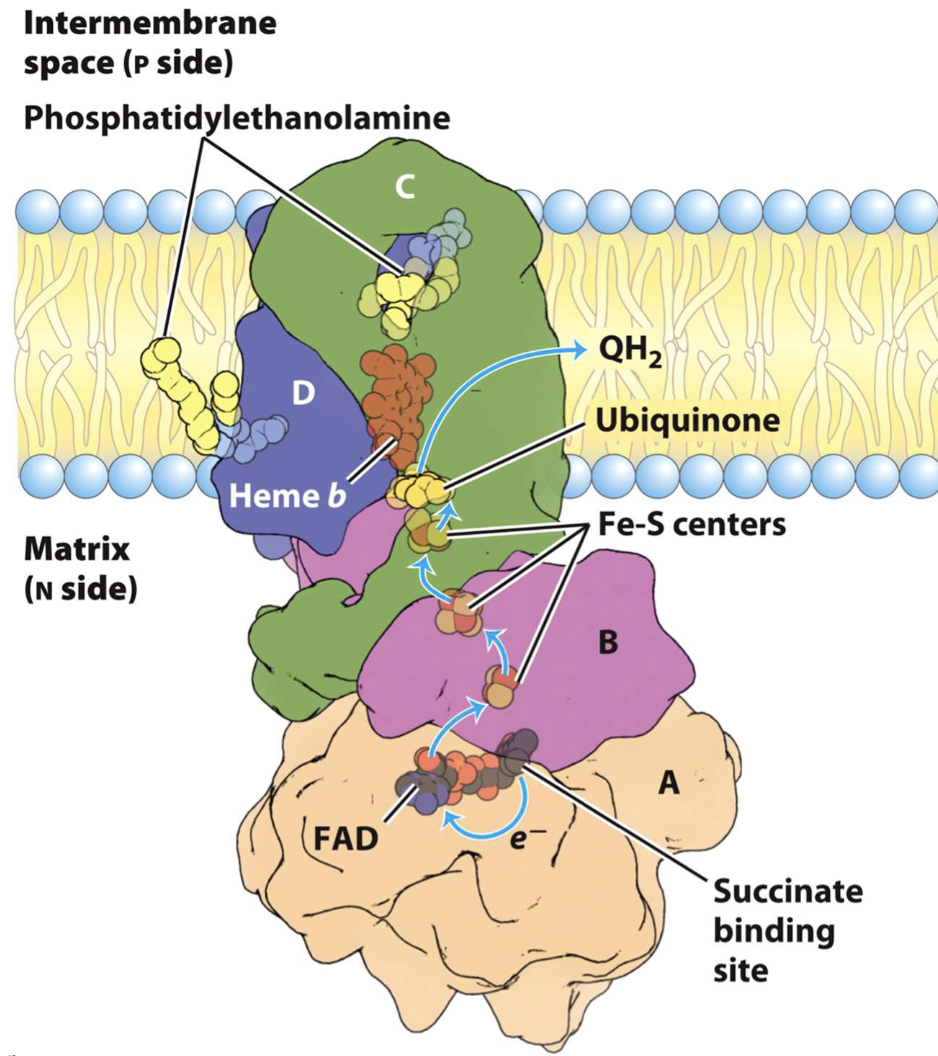
Complex II-Succinate dehydrogenase
Succinate → FAD: Succinate is oxidized to Fumarate + FAD is reduced to FADH2.
FADH2 → Fe-S clusters: 3 different Fe-S clusters act as a wire to move e- toward the membrane.
Fe-S clusters → Heme b: A heme group acts as an intermed (control e- flow to prevent ROS)
Heme b → Q: Q picks up the 2 e- and 2 H from matrix to become QH2
NO PROTON PUMP (skips proton pumping of Complex I, e- entering via Complex II (from FADH2) result in less ATP (1.5 ATP) vs. NADH (2.5 ATP)
Smallest complex
FAD in Complex II is covalently bound, FADH2 enters at lower nrg level
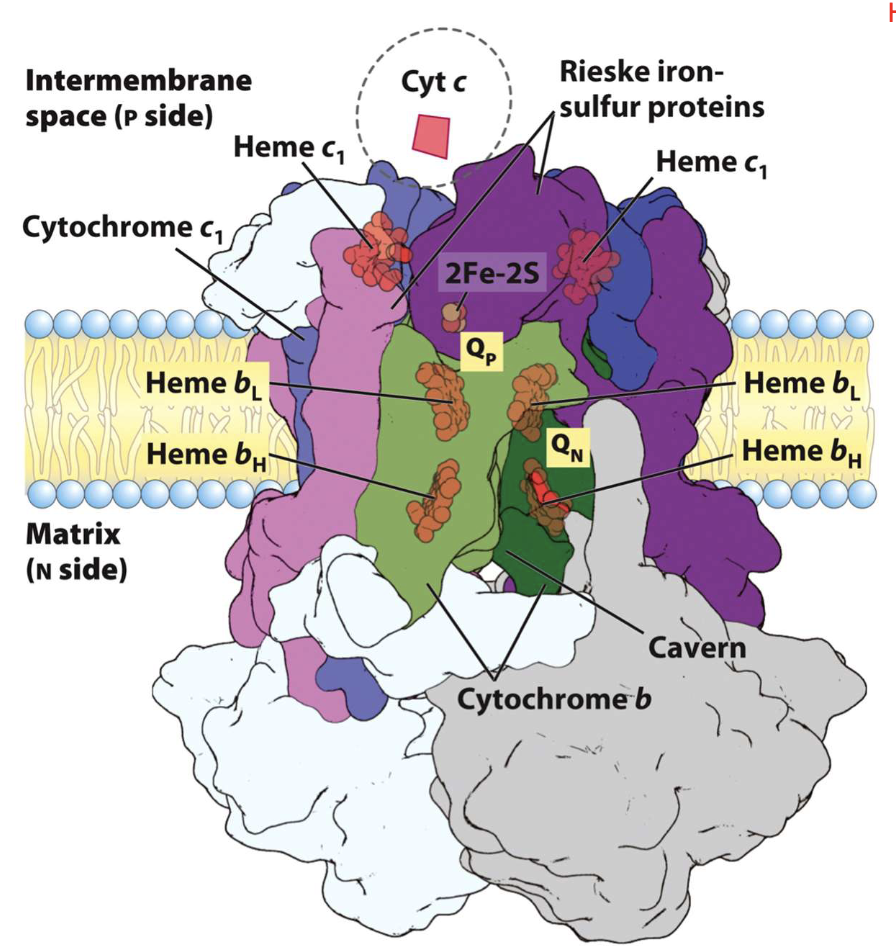
Complex III
use Fe-S cluster transfer e- from CoQH2 to heme forming cyt c
dimer in inner membrane: each monomer has a cyt b subunit lining an inner "cavern."
The "cavern" allows 2 Q’s to bind simultaneously at distinct sites:
Qo Site: (P-side/Intermembrane space):QH2 is oxidized (gives up e-)
Qi Site: (N-side/Matrix):Q or Q- is reduced (accepts e-)
3 hemes (bH, bL + c1).
All protoporphyrin IX (same structures)
diff reduction potentials (Eo) bc of unique microenvironments, allowing a specific "downhill" path for e-
Rieske 2Fe-2S Cluster: Fe-S protein essential for the "upstairs" path to Cyt c.
Coordinated to His instead of the Cys
Result: higher Eo (higher affinity for e-), allowing it to pull e- from QH2 effectively
Cyt c: Last destination for e- in complex
- mobile, water-soluble protein that stays on the P-side (intermembrane space)
PUMPS 4 PROTONS
Complex IV: cytochrome c oxidase
Role: uses cytochromes + Cu2+ to transfer e- in form of H- from cyt c → O2 → H2O
contains: 2 heme groups (a + a3), 3 copper atoms (CuA/CuA + CuB)
Specific E- Path:
Cytochrome c binding: Occurs at the intermembrane space (P-side).
Di-copper center (CuA): The first internal recipient.
Heme a: Intermediate carrier
Heme a3: final + leading to the oxygen-binding site
Pump 4 protons physically moved to the intermembrane space.
4 protons removed from the matrix to form water.
Net Result: 8 H cleared from the matrix (N-side)
Lethal Inhibitors:
Cyanide (CN-) & Azide (N3-): Bind Fe3+ in heme a3, stopping e- transfer to O2.
Carbon Monoxide (CO): Competes with O2 for binding at Fe2+ in Heme a3
Complex IV mechanism
O2 + 4cyt c(red) + 8H (matrix) + → 2H2O + 4cyt c(ox) +4 H+ (intermem space)
First 2 e- arrive (from 2 CytCred)
2 reduced cyt c each donate 1 e⁻ → total 2 e⁻
e- move to:
1 e⁻ → heme a₃ (Fe)
1 e⁻ → CuB
✅ Fe–Cu center is now reduced and ready to bind O₂
O₂ binds b/w Fe (heme a₃) + CuB accepts 2 e- → forms a peroxide bridge (O₂²⁻)
Next 2 e- arrive (2 more CytCred): 2 e- further reduce the bound oxygen intermed (dangerous intermediate (like O₂•⁻ / peroxide) is fully held in place at the Fe–Cu center (never released)
2 protons from the matrix (H⁺) are added: Breaks the O–O bond + forms Hemea3–OH and Cub –OH
Obtain 2 more protons from matrix + ox. Hemea3 + Cub into original state + release 2 H2O
Complex III transfer: THE Q CYCLE
QH2 + 2cyt c(oxidized) + 2H (n-matrix)+→Q + 2 cyt c(reduced)+ 4H + (p: ntermembranespace)
QH2 carries 2 e-, but Cyt c can only accept 1
FIRST HALF: First QH₂:
Releases 2 H⁺ into intermembrane space
1 e⁻ → cyt c
1 e⁻ → Q → forms semiquinone (Q•⁻) (held in the “cavern”)
Second QH₂:
Releases 2 more H⁺ into intermembrane space
1 e⁻ → another cyt c
1 e⁻ → reduces Q•⁻ → QH₂ (fully reduced) using 2 H⁺ from matrix
Overall: Converts 2-e- carrier → 1-e transfers
Produces 2 cyt c (each carrying 1 e⁻)
Proton movement:
4 H⁺ released into intermembrane space
2 H⁺ taken up from matrix
drives ATP synthesis
OVERALL EQUATIONS FOR ETC
2 overall paths for e- in ETC:
Complexes I,III,IV 2 NADH + O2 + 22H+(n) → 2 NAD+ + 2H2O + 20H+ (p side)
Complexes II,III,IV 2 succinate + O2 + 12H+(n) → 2 fumarate + 2H2O + 12H+ (p side)