AP Chemistry
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Filtration
a physical separation technique used to isolate an insoluble solid (precipitate) from a liquid or solution.
Distillation
a physical separation technique that purifies liquid mixtures based on boiling point
Paper chromatography
separates chemical species in a solution based on differences in their intermolecular interactions (polarities) with a mobile phase and a stationary phase
Column Chromatography
a separation and purification technique that splits a mixture of components based on their differing affinities (intermolecular forces) for a stationary phase and a mobile phase, more appropriate for separating the components in a mixture on a larger scale.
Anything that DECREASES the amount of light passing through during a spectrophotometer experiment cause an _______ in absorbance
increase
The absorption of photons of infrared radiation is associated with
transitions in molecular vibrational levels
Absorption of photons in the visible range will cause
transition of electrons between electron energy levels.
By Kinetic Molecular Theory, gas particles are always moving at ____ speeds
a variety of different
By coloumb’s law, ionic compounds with bigger charges have
higher melting points
By coloumb’s law, ionic compounds with the same charge but have smaller ions have
higher melting points
Isotopes differ in
neutrons
Generally, smaller atoms will have ___ and therefore ____ bond lengths
shorter, stronger
by VSEPR Theory, 2 groups means its shape is
Linear
by VSEPR Theory, 3 groups, NO LONE PAIRS means its shape is
Trigonal planar
by VSEPR Theory, 3 groups with ONE LONE PAIR means its shape is
bent
by VSEPR Theory, 4 groups with NO LONE PAIRS means its shape is
tetrahedral
by VSEPR Theory, 4 groups with ONE LONE PAIR means its shape is
Trigonal Bipyrimidal
by VSEPR Theory, 4 groups with TWO LONE PAIRS means its shape is
Bent
Stronger intermolecular forces results in
Higher melting and boiling point, lower vapor pressure, higher viscosity
Properties of ionic solids
brittle, conduct electricity in molten state only, held together by ionic bonds
Properties of covalent networks
Nonmetals and metalloids, rigid and hard, high melting points, held together by covalent bonds
Properties of molecular solids
Made of individual molecules, low melting point, held together by IMFs
Metallic solids
conduct electricity and heat, held together by metallic bonds
When will gasses have lower pressure than expected
at low temperatures or with gasses with stronger IMFs
When will gasses have higher pressure than expected
Extremely small volumes
At high pressures, what will volume do?
volume will be bigger than expected
How do you experimentally determine the order of a reaction
Change reaction concentration and observe how rate responds
Infrared radiation is used to study
bonds within molecules
Solid to liquid is called
melting/fusion
liquid to gas is called
vaporization
solid to gas is called
sublimation
gas to solid is called
deposition
When manipulating Keq, when you multiply a reaction by a coefficient you must
raise it to the power of that coefficient
When manipulating Keq, when you add two reactions you must
multiply their Keqs
Stronger acids(or bases) have ____ conjugate bases(or acids).
weaker
Stronger species (acids/bases) are
less stable and more reactive
Strong acids
H2SO4, HI, HBr, HNO3, HCL, HClO4
What are the strong bases
Group 1 and 2 hydroxides (LiOH, NaOH, etc)
What is the heat of vaporization
the amount of energy needed to break all intermolecular forces
Vapor pressure
the pressure exerted by the gas above a liquid at equilibrium, indicates how easily a liquid's molecules escape into the gas phase
Volatility
a measure of how easily a substance evaporates at a specific temperature
Properties when a substance has stronger IMFs
higher boiling point, heat of vaporization, surface tension, and viscosity, lower vapor pressure and volatility
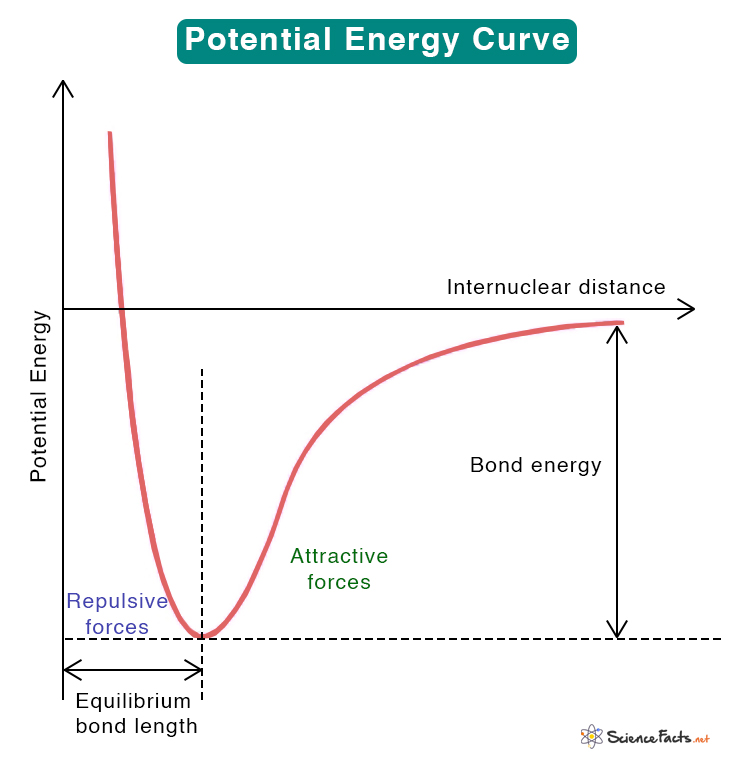
A molecule with a SHORTER bond length will result in what changes to the graph
shift to the left and lower peak (shorter=greater energy)
Gasses stray from ideal gas law due to
low temperatures, stronger IMFs, large particles, small containers, high pressures
In paper chromatography, the stationary phase is the _____ and the mobile phase is the ____
paper (what the solution separates into), solvent (the solvent that passes through the stationary phase)
The pH of the conjugate base of a strong acid is
neutral
The PH of the conjugate acid of a weak base is
acidic
The more electronegative an atom is (acids/base) + trend on periodic table
the more acidic; across a row= more acidic
The bigger an atom ______, + periodic trend
the weaker the acid, down a column= weaker acids
Acids with resonance are ____ than acids without resonance
stronger
Strong acid + strong base will result in a pH ____
neutral
Strong acid + weak base will result in a pH ____
less than 7 (acidic)
Weak acid and strong base will result in a pH
greater than 7 (basic)
A buffer is more effective when
the concentration of the conjugate pair are higher
The autoionization of water is an ______ process, so warming up water ______ the pH
endothermic, decreases