Gene Therapy and Viral Vectors 2
1/323
Earn XP
Description and Tags
ends lecture 10 slide 6
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
324 Terms
Herpes Simplex Virus: Classification and Core Properties
Family: Herpesviridae (α-herpesvirus subgroup)
Genome type: Linear double-stranded DNA (dsDNA)
Genome size: ~150–200 kb (HSV-1 ≈ 152 kb)
One of the largest genomes among human viruses used in gene therapy
Herpes Simplex Virus: Diagram
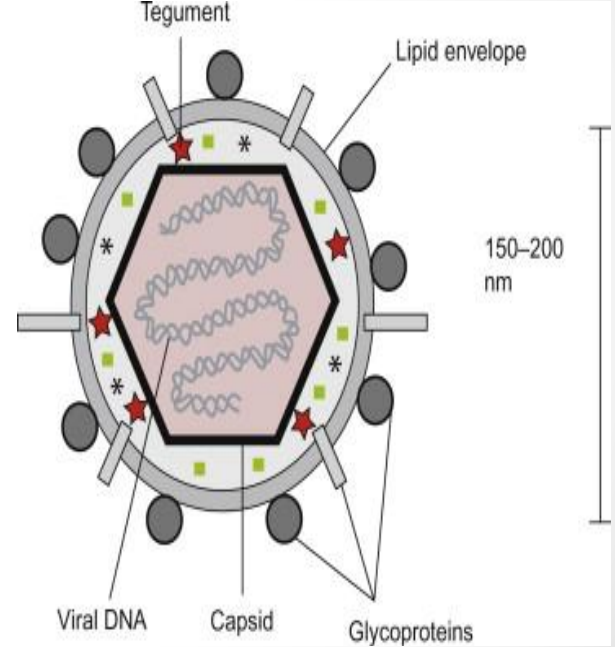
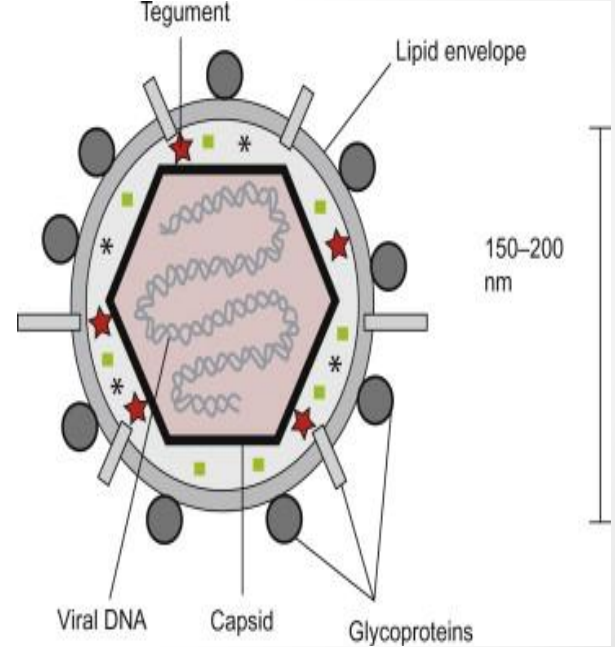
Structural Components of Herpes Simplex Virus
Core
capsid (Icosahedral protein shell)
Tegument
Envelope (lipid bilayer + glycoproteins)
Structural Components of Herpes Simplex Virus: Core
Contains linear dsDNA genome (~152 kb)
DNA is tightly packed under high internal pressure inside capsid
Delivered directly into nucleus through nuclear pore complex
Functional implication
Rapid genome release → immediate access to host transcription machinery
No need for cytoplasmic replication steps
Structural Components of Herpes Simplex Virus: Capsid (Icosahedral protein shell)
Surrounds and protects viral DNA
Structure: icosahedral symmetry (T=16)
Depth
Composed mainly of:
Major capsid protein VP5
Highly stable:
Protects genome during:
Extracellular transmission
Intracellular transport
Mechanistic role
Travels along microtubules (dynein-mediated) to nucleus
Docking at nuclear pore → DNA injection
Structural Components of Herpes Simplex Virus: Tegument
Protein layer between capsid and envelope
Contains regulatory viral proteins
Major tegument proteins:
VP16 → activates immediate-early (α) gene transcription
VHS (virion host shutoff protein) → degrades host mRNA
Functional consequences
Virus controls host cell immediately upon entry:
Shuts down host protein synthesis
Redirects machinery toward viral gene expression
Structural Components of Herpes Simplex Virus: Envelope (lipid bilayer + glycoproteins)
Derived from host membranes
Embedded with ~10 viral glycoproteins
Key glycoproteins
gB, gC → initial attachment (heparan sulfate binding)
gD → receptor engagement (HVEM, nectin)
Depth
Entry mechanism:
Membrane fusion, not endocytosis (in many cells)
This allows:
Direct release of capsid into cytoplasm
Functional implication
Determines:
Cell tropism (which cells can be infected)
functions of tegument in HSV
Initiates viral gene expression
VP16 activates Immediate Early (α) genes → rapid transcription onset
Shuts off host protein synthesis
VHS protein degrades host mRNA → shifts translation to viral proteins
Enables capsid transport to nucleus
Interacts with dynein/microtubules → efficient delivery to nuclear pore
Suppresses host immune response
Interferes with interferon signaling → delays detection
Supports virion assembly
Acts as a bridge between capsid and envelope during maturation
Synthesis: Tegument proteins allow HSV to immediately control host processes, ensuring rapid gene expression, efficient genome delivery, and evasion of early immune responses.
HSV glycoprotein function
Initial attachment to host cells
gB and gC bind heparan sulfate on cell surface → concentrates virus on membrane
Receptor recognition and entry specificity
gD binds entry receptors (e.g., HVEM, nectin) → determines cell tropism
Membrane fusion and viral entry
gD activation triggers gB + gH/gL fusion machinery
Leads to fusion of viral envelope with host membrane → capsid released into cytoplasm
Cell-to-cell spread
Glycoproteins mediate fusion between infected and adjacent cells
Enables spread without exposure to extracellular immune defenses
HSV Genome Structure Diagram
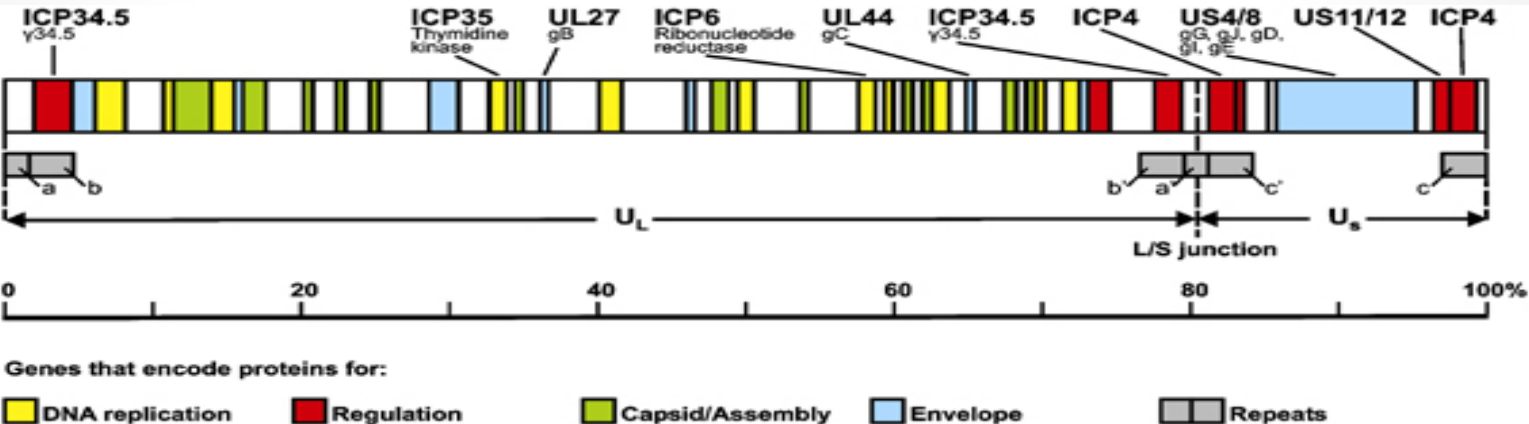
HSV Genome size and composition
HSV-1 genome: ~152 kb dsDNA
Encodes: ~84 viral genes
Depth
This is a high coding capacity genome, allowing:
Structural proteins
Enzymes
Regulatory proteins
Compared to smaller viruses, HSV encodes more of its own replication machinery, reducing dependence on host
HSV: Essential vs non-essential genes
~50% genes are:
Essential → required for viral replication
Remaining genes:
Non-essential (“accessory”)
Depth
Essential genes include:
DNA polymerase
Helicase-primase complex
Non-essential genes mainly:
Modify host environment rather than replication itself
HSV: functions of non-essential genes
These genes are important for:
Immune evasion
Replication in non-dividing cells
Shutdown of host protein synthesis
Depth
Immune evasion:
Inhibits interferon signaling
Non-dividing cell replication:
Critical for infection of neurons
Host shutoff:
Ensures viral dominance over cellular machinery
HSV: Gene deletion and vector design
Non-essential genes can be:
Deleted
Replaced with exogenous (therapeutic) DNA
Depth (this is the key concept)
Common deletions:
ICP34.5 → reduces neurovirulence
ICP4 → blocks replication (creates replication-deficient vector)
Result
Space created for:
Large therapeutic inserts (~30–50 kb)
HSV Epidemiology: Classification within α-herpesviruses
HSV belongs to alphaherpesviruses (α-HVs)
Human α-HVs include:
HSV-1
HSV-2
Varicella Zoster Virus
Depth
α-herpesviruses are characterized by:
Rapid replication (lytic phase)
Ability to establish latent infection in sensory neurons
HSV Epidemiology: Transmission and disease types
HSV-1
Transmitted via oral contact
Causes oral herpes (cold sores)
HSV-2
Sexually transmitted
Causes genital herpes
Depth
Both viruses:
Infect epithelial cells initially
Then establish latency in neurons
HSV-1 can also cause genital infections (increasingly common)
HSV Epidemiology: Global prevalence
HSV-1:
~3.7 billion people (<50 years) → ~67% infected
HSV-2:
~491 million (15–49 years) → ~13% infected
Depth (important implication)
Extremely high prevalence → many individuals have:
pre-existing immunity
HSV: Clinical presentation
Most infections:
Asymptomatic
When symptomatic:
Painful blisters or ulcers
Depth
Virus replication causes:
Cell lysis → tissue damage → lesions
Immune response contributes to:
Inflammation and pain
HSV: Associated diseases
HSV infections linked to:
Oral/genital herpes
Varicella Zoster Virus causes:
Chickenpox
Depth
Important distinction:
HSV → recurrent localized lesions
VZV → systemic infection (chickenpox) + later reactivation (shingles)
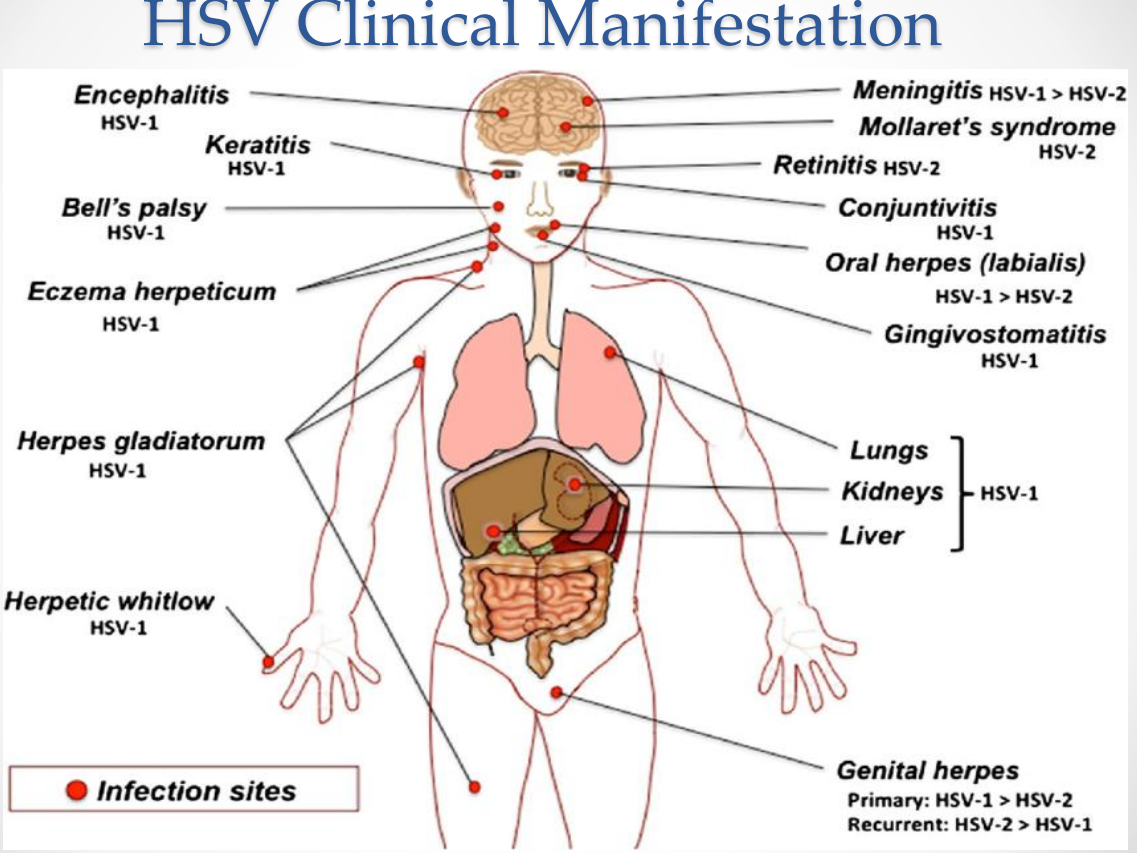
HSV Clinical Manifestations:
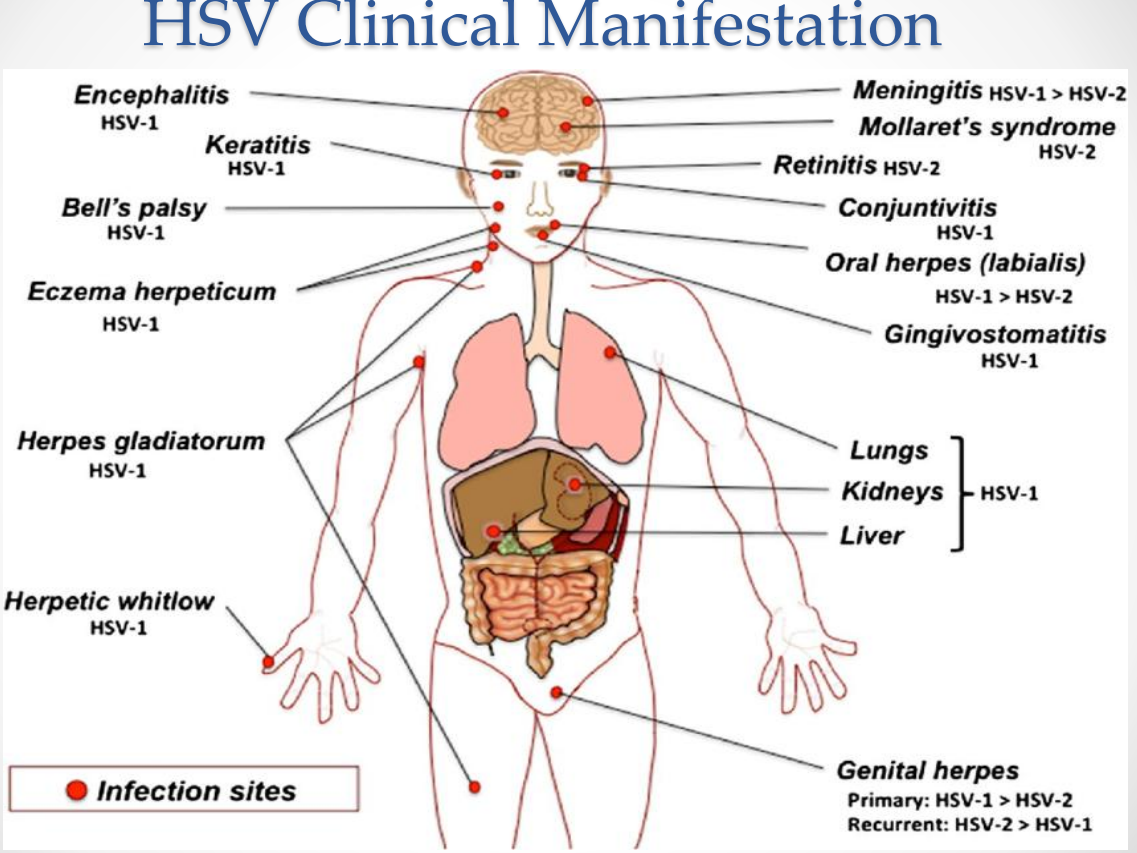
HSV Clinical Manifestations: Neurotropic manifestations
Encephalitis (HSV-1)
Meningitis (HSV-1 > HSV-2)
Mollaret’s meningitis (HSV-2)
Bell’s palsy (HSV-1)
Depth
HSV has strong neurotropism → infects sensory neurons
Travels via:
retrograde axonal transport → CNS
HSV encephalitis:
Typically affects temporal lobe
Due to viral replication + immune-mediated damage
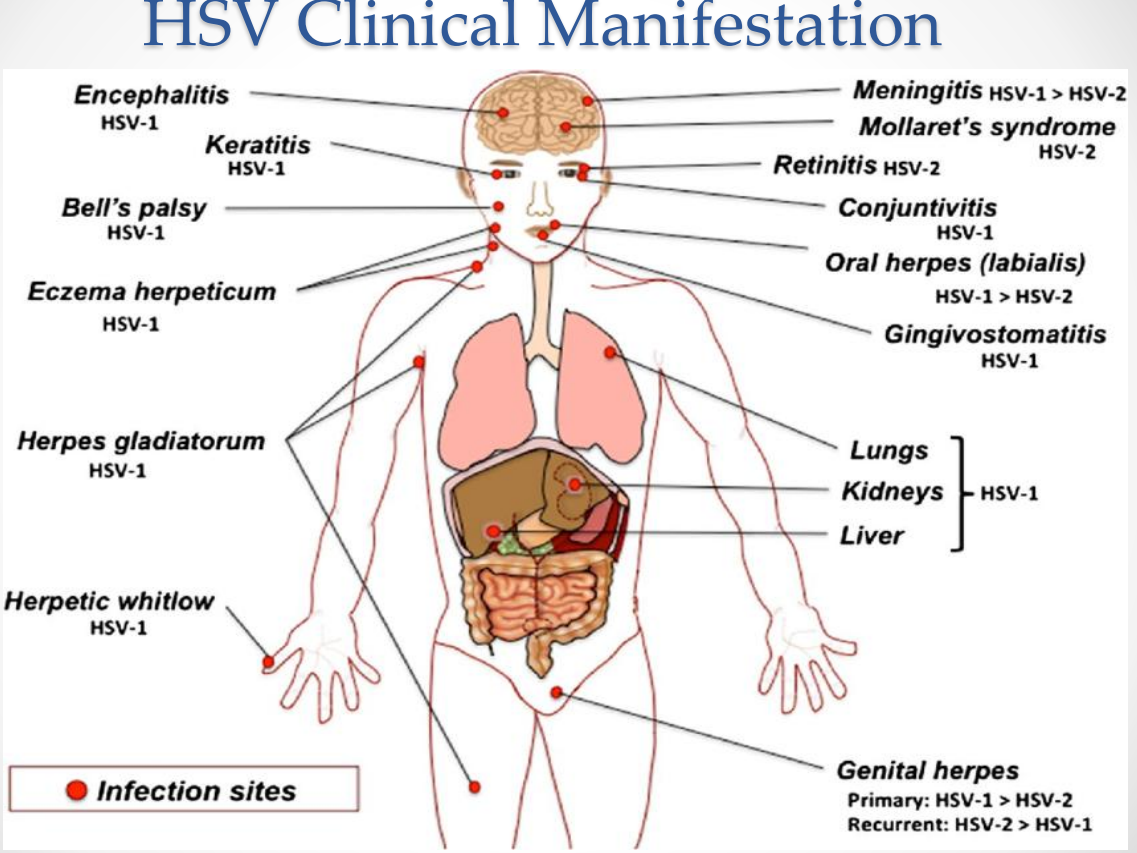
HSV Clinical Manifestations: Ocular infections
Keratitis (HSV-1)
Conjunctivitis (HSV-1)
Retinitis (HSV-2)
Depth
Keratitis is:
One of the leading causes of infectious blindness
Mechanism:
Viral replication damages corneal epithelium
Recurrent infections worsen damage due to immune scarring
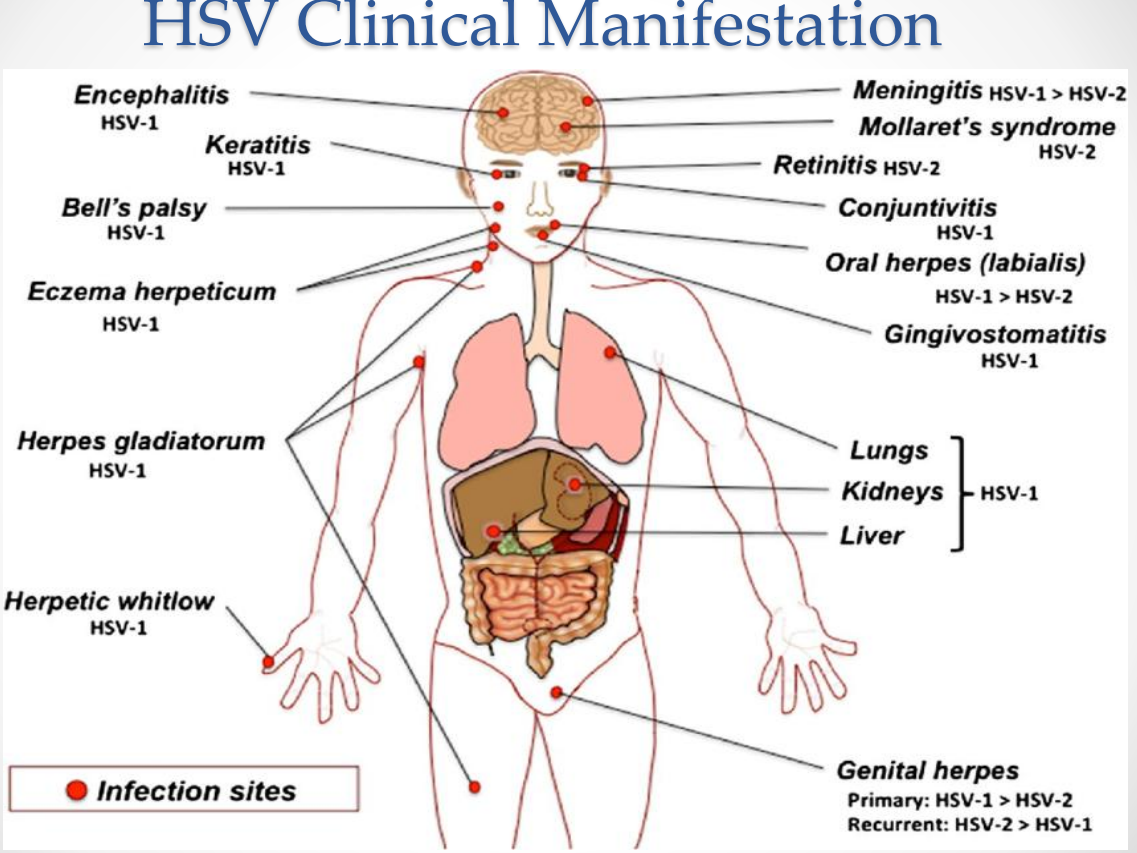
HSV Clinical Manifestations: Oral and facial infections
Oral herpes (labialis) — HSV-1 > HSV-2
Gingivostomatitis — HSV-1
Depth
Primary infection:
Often gingivostomatitis (severe, widespread lesions)
Reactivation:
Localized cold sores
Occurs due to:
Reactivation from trigeminal ganglion latency
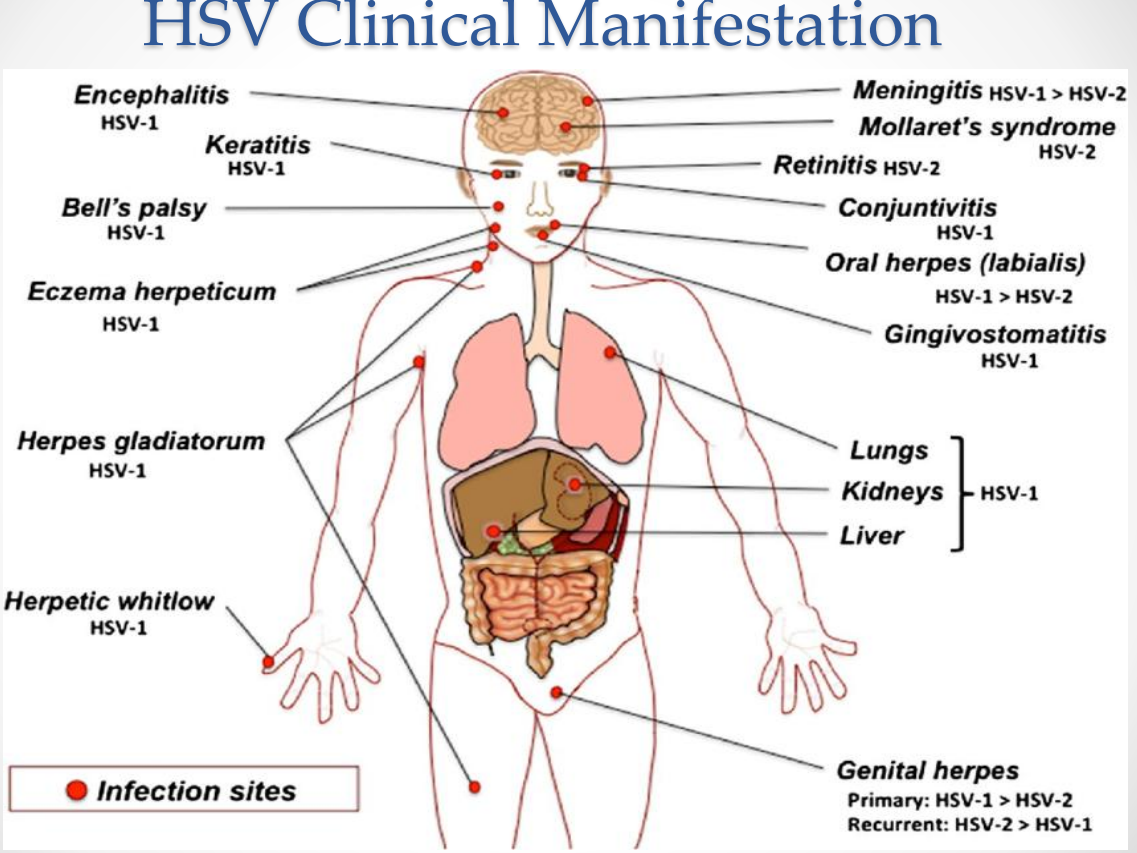
HSV Clinical Manifestations: Skin infections
Eczema herpeticum (HSV-1)
Herpes gladiatorum (HSV-1)
Herpetic whitlow (HSV-1)
Depth
Occur when virus enters through:
broken skin barrier
Gladiatorum:
Seen in contact sports (skin-to-skin transmission)
Whitlow:
Infection of fingers (common in healthcare workers)
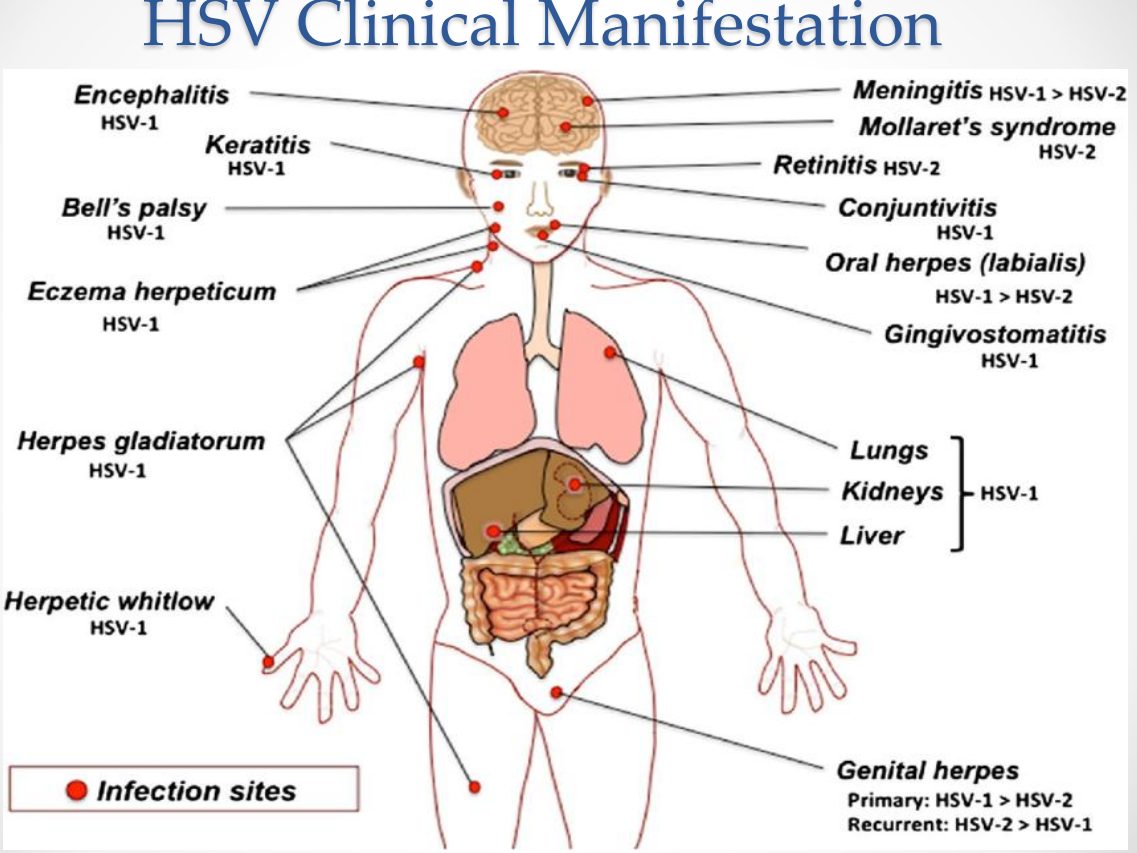
HSV Clinical Manifestations: Genital infections
Genital herpes
Primary: HSV-1 > HSV-2
Recurrent: HSV-2 > HSV-1
Depth (important distinction)
HSV-2:
Better adapted to genital tract latency
Causes more frequent reactivation
HSV-1:
Increasing cause of primary genital infections
But less recurrent
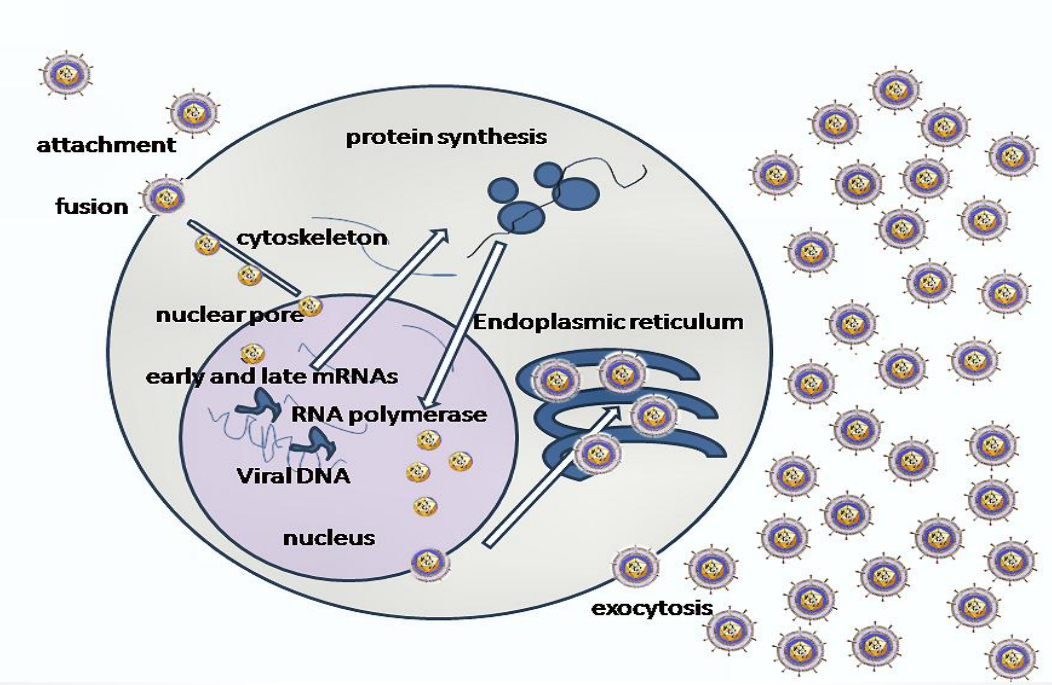
Life Cycle of Herpes Simplex Virus: Attachment and Entry
Viral glycoproteins:
gB, gC → bind heparan sulfate
gD → binds entry receptors (HVEM, nectin)
Triggers:
gB + gH/gL–mediated membrane fusion
Outcome
Viral envelope fuses with host membrane
Capsid + tegument released into cytoplasm
Life Cycle of Herpes Simplex Virus: Capsid Transport to Nucleus
Capsid moves via:
microtubules (dynein-mediated retrograde transport)
Outcome
Capsid docks at:
nuclear pore complex
Viral DNA is:
injected into nucleus
Life Cycle of Herpes Simplex Virus: Immediate Host Takeover (Tegument action)
VP16 → activates α (immediate early) genes
VHS → degrades host mRNA
Outcome
Rapid shift from:
host → viral gene expression
Life Cycle of Herpes Simplex Virus: Transcriptional Cascade
Three phases:
α (Immediate Early)
Regulatory proteins
β (Early)
DNA replication enzymes
γ (Late)
Structural proteins
Outcome
Controlled, sequential gene expression
Life Cycle of Herpes Simplex Virus: Viral DNA Replication
Genome:
Linear → circularizes in nucleus
Replication mechanism:
Starts as theta replication
Switches to rolling circle replication
Outcome
Formation of:
concatemeric DNA (long repeats)
Life Cycle of Herpes Simplex Virus: Assembly
Capsid assembly:
Occurs in nucleus
Viral DNA:
Packaged into capsid
Tegument addition:
Occurs during:
cytoplasmic transit
Life Cycle of Herpes Simplex Virus: Envelopment and Release
Virus acquires envelope by:
budding through nuclear membrane
Outcome
Mature virions transported via:
vesicles → released by exocytosis
HSV: Lytic vs Latent Pathways
Lytic cycle
Active replication
Cell lysis → virus release
Latent cycle
Viral DNA persists as:
episome in sensory neurons
Only:
LAT expressed
Reactivation
Triggered by stress, immunosuppression
Virus travels:
anterograde → epithelial cells → re-infection
HSV lifecycle overview + diagram
Attachment and Entry
Capsid Transport to Nucleus
Immediate Host Takeover
Transcriptional Cascade
Viral DNA Replication
Assembly
Envelopment and Release
All α-HVs initially infect _______ cells (primary site of infection), and later spread to infect ________
epithelial
sensory neurons.
HSV as a Latent Neurotropic Virus: Acute Infection
Infection begins in peripheral epithelial cells
What’s happening mechanistically
Virus undergoes lytic replication:
Produces virions
Causes epithelial cell death
Leads to:
Cold sores
Viral shedding (transmission stage)
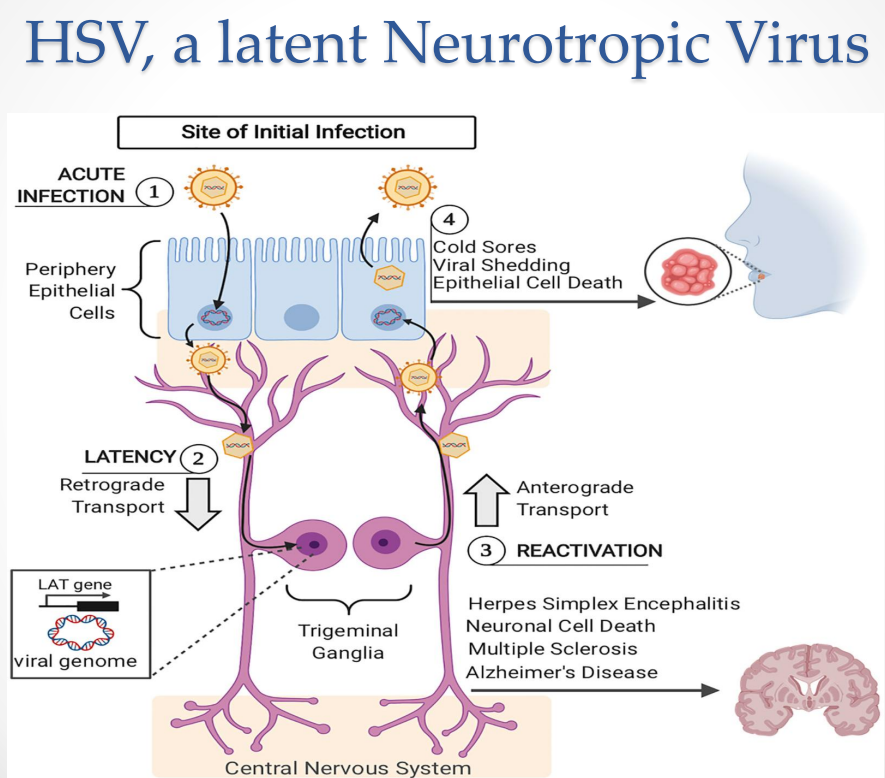
HSV as a Latent Neurotropic Virus: Establishment of Latency
Virus enters sensory neurons
Moves via:
Retrograde axonal transport → trigeminal ganglia
Inside neuron
Viral DNA:
Circularizes → episomal form
Transcription is largely silenced except:
LAT (Latency-Associated Transcript)
Functional role of LAT
Suppresses:
Viral lytic gene expression
Prevents:
Apoptosis of infected neuron
→ Ensures long-term persistence
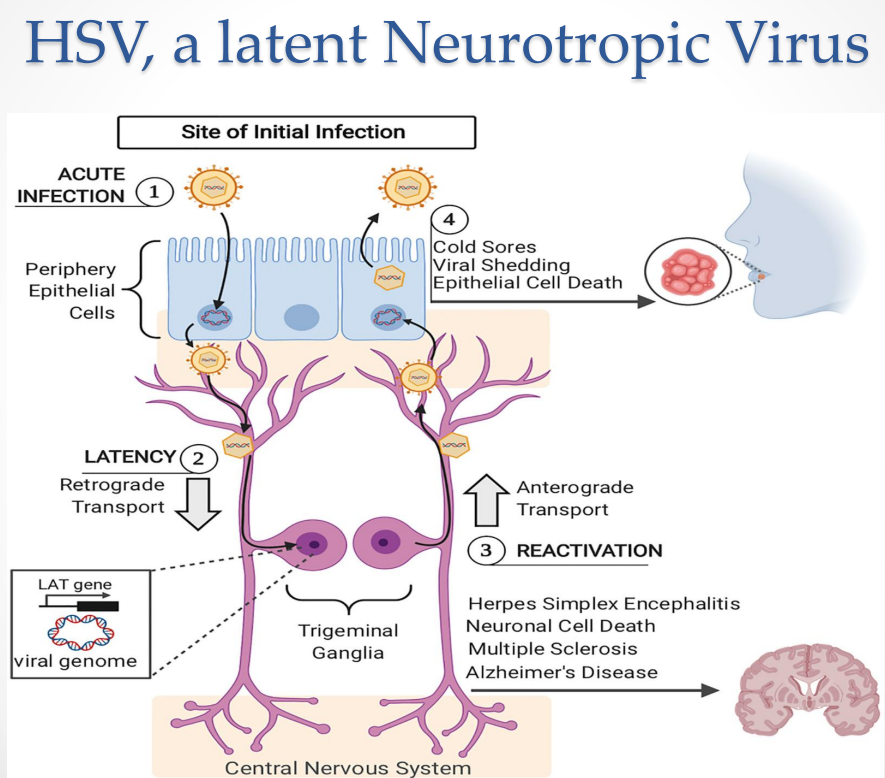
HSV as a Latent Neurotropic Virus: Reactivation
Triggered by:
Stress, UV light, immunosuppression
Mechanism
Viral genome reactivates:
Lytic genes expressed again
Virus travels via:
Anterograde transport → back to epithelial cells
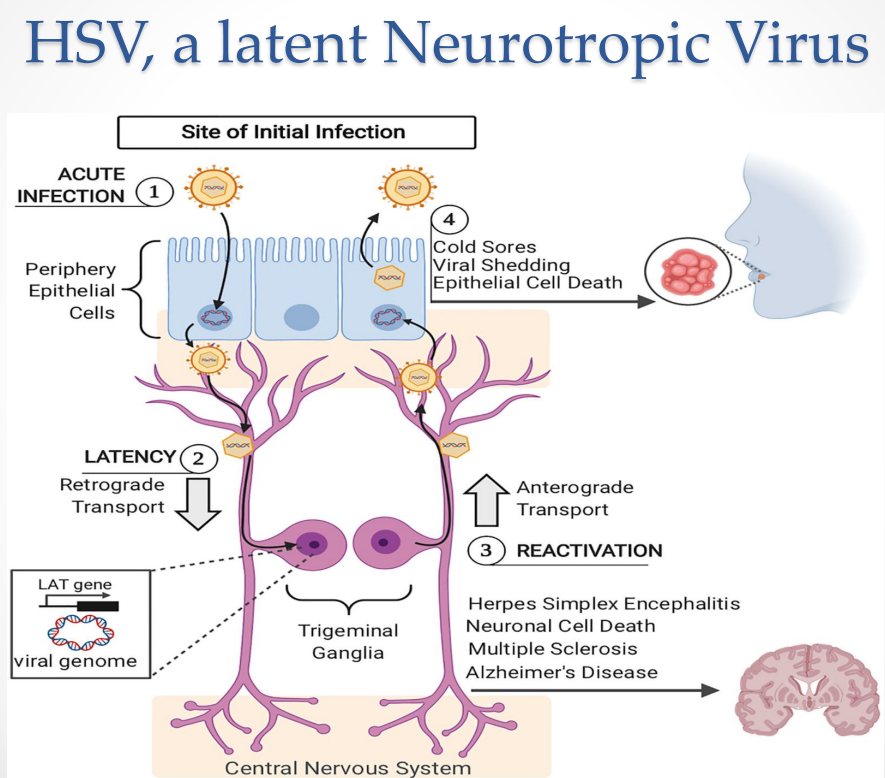
HSV as a Latent Neurotropic Virus: Secondary Infection / Recurrence
Virus reaches epithelial cells again
Outcomes (from slide)
Cold sores
Viral shedding
Epithelial cell death
Key insight
Recurrence occurs at:
same anatomical site
Due to fixed neuronal reservoir
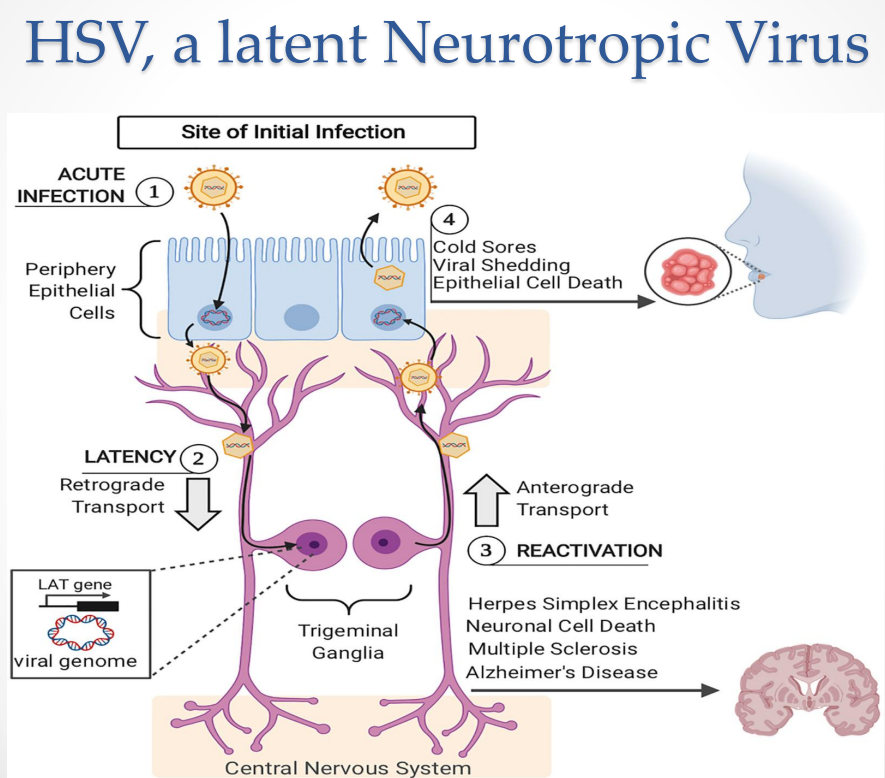
HSV as a Latent Neurotropic Virus: CNS involvement
Possible outcomes:
Herpes simplex encephalitis
Neuronal cell death
Associations with:
Multiple sclerosis
Alzheimer’s disease
Depth
Occurs when virus spreads beyond peripheral neurons into:
central nervous system
HSV-1 encephalitis:
Often targets temporal lobe
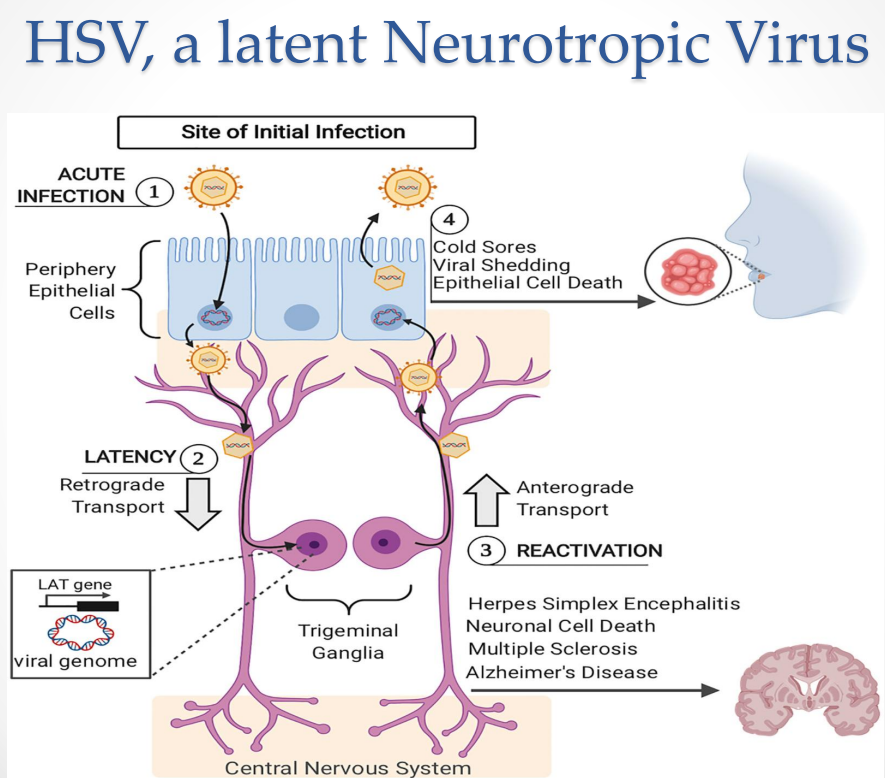
HSV as a Latent Neurotropic Virus Diagram
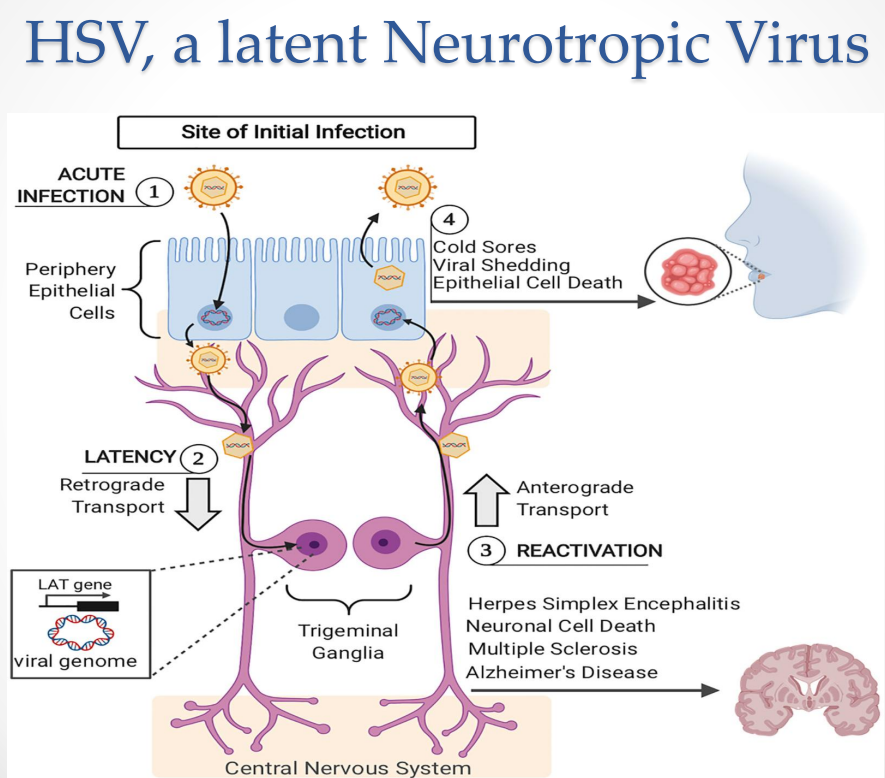
Challenges of HSV as a Gene Therapy Vector
Immunogenicity
Packaging Constraints
Random Integration
Cytotoxicity / Lytic Nature
Challenges of HSV as a Gene Therapy Vector: Immunogenicity
HSV particles trigger a strong immune response
Mechanistic depth
Viral proteins (especially envelope glycoproteins + tegument proteins) are recognized by:
Innate immunity (TLRs, interferon response)
Adaptive immunity (neutralizing antibodies, T cells)
Consequences
Rapid vector clearance
Reduced transgene expression duration
Difficulty with repeat dosing
Challenges of HSV as a Gene Therapy Vector: Packaging Constraints
Despite large genome, there are limits to how much DNA can be inserted
Mechanistic depth
Capsid has a physical size limit → cannot exceed stable genome length
Overloading genome:
Disrupts capsid assembly
Reduces viral stability
Practical implication
Although HSV can carry ~30–50 kb inserts:
Insert size must be balanced with essential genome elements
Challenges of HSV as a Gene Therapy Vector: Random Integration
HSV DNA is mainly episomal but can rarely integrate into host genome
Mechanistic depth
Integration may occur via:
host DNA repair pathways (non-homologous recombination)
Risks
Insertional mutagenesis:
Disruption of host genes
Potential activation of oncogenes
Challenges of HSV as a Gene Therapy Vector: Cytotoxic
HSV naturally undergoes lytic replication
Depth:
Causes host cell death
Problem for:
Non-cancer gene therapy
Requires:
attenuation (e.g., ICP34.5 deletion)
Benefits of Herpes Simplex Virus as a Vector
Broad Cell Tropism
Natural Cytolytic Activity
Large Genome → Gene Insertion Capacity
Engineering Flexibility
Synergy with Other Therapies
Episomal Persistence
Benefits of Herpes Simplex Virus as a Vector: Broad Cell Tropism
HSV can infect a wide variety of cell types
Depth
Due to multiple entry receptors (heparan sulfate, HVEM, nectins)
Infects:
Dividing cells (tumors)
Non-dividing cells (neurons)
Why this is impressive
Many vectors (e.g., retroviruses) cannot infect non-dividing cells
→ HSV is versatile across tissues
Benefits of Herpes Simplex Virus as a Vector: Natural Cytolytic Activity
HSV replication leads to cell lysis
Depth
Viral replication:
Disrupts cellular machinery
Causes membrane breakdown
Application
Direct killing of:
Cancer cells (oncolysis)
Benefits of Herpes Simplex Virus as a Vector: Large Genome → Gene Insertion Capacity
Contains many non-essential genes
Depth
These can be:
Deleted
Replaced with therapeutic genes
Outcome
Can insert:
Large or multiple genes (~30–50 kb)
Supports:
Complex therapies (e.g., gene + regulatory elements)
Benefits of Herpes Simplex Virus as a Vector: Engineering Flexibility
HSV can be re-engineered
From slide
Can express:
Cytotoxic genes
Immune-stimulating genes
Depth
Examples:
Prodrug-activating enzymes
Cytokines (e.g., GM-CSF)
Enables:
targeted tumor destruction + immune activation
Benefits of Herpes Simplex Virus as a Vector: Synergy with Other Therapies
Works well with:
Radiation therapy
Chemotherapy
Depth
Viral infection can:
Increase tumor sensitivity to radiation
Enhance immune-mediated tumor clearance
Benefits of Herpes Simplex Virus as a Vector: Episomal Persistence
HSV genome remains episomal (non-integrating)
Why it matters
Reduces:
Insertional mutagenesis risk
Supports:
Safer gene delivery
Major Types of Cancer
-Carcinoma is a cancer that begins in the skin or in tissues that line or cover internal organs.
• Sarcoma is a cancer that begins in bone, cartilage, fat, muscle, blood vessels, or other connective or supportive tissue.
• Leukemia is a cancer that starts in blood-forming tissue, such as the bone marrow, and causes large numbers of abnormal blood cells to be produced and enter the blood.
• Lymphoma and multiple myeloma are cancers that begin in the cells of the immune system.
• Central nervous system cancers are cancers that begin in the tissues of the brain and spinal cord.
“Drivers” of Cancer: proto-oncogenes
1. Proto-oncogenes → Oncogenes (from slide)
Normal role: promote cell growth and division
When altered → become oncogenes
Mechanism
Gain-of-function mutations:
Overexpression
Constitutive activation
Examples (from slide)
HER2, Ras, Myc
Functional outcome
Cells:
Proliferate without external growth signals
Avoid normal growth limits
“Drivers” of Cancer: tumor suppressor genes
Normal role: inhibit cell division / control cell cycle
Mechanism
Loss-of-function mutations:
Remove growth inhibition
Disable cell cycle checkpoints
Examples (from slide)
p53, p10
Functional outcome
Cells divide:
Uncontrollably
Even when damaged
“Drivers” of Cancer: DNA repair genes
Normal role: repair damaged DNA
Mechanism
Mutation → defective repair system
Leads to:
Accumulation of mutations
Examples (from slide)
BRCA1, BRCA2
Functional outcome
Genomic instability:
Accelerates cancer progression
Stages of Cancer
1. Purpose of Staging (from slide)
Determines:
Location of cancer
Extent of spread
Impact on other body parts
Depth
Staging is not just descriptive — it directly guides:
Treatment selection
Prognosis estimation
2. Role in Treatment Planning (from slide)
Helps decide:
Surgery (localized tumors)
Chemotherapy (systemic disease)
Radiation therapy (targeted control)
Depth
Early-stage:
Often treated with localized therapies
Advanced-stage:
Requires systemic approaches
3. Predicting Recurrence (from slide)
Indicates:
Likelihood cancer will return after treatment
Depth
Higher stage → higher chance of:
Residual disease
Metastasis
4. Predicting Survival / Recovery (from slide)
Used to estimate:
Patient prognosis
Depth
Lower stage:
Better survival rates
Higher stage:
Poorer outcomes due to spread
5. Standardized Communication (from slide)
Provides a common language for:
Doctors
Researchers
Depth
Ensures:
Consistency across hospitals and studies
Enables:
Accurate comparison of patient outcomes
TNM: — Primary Tumor Size & Extent
Answers:
How large is the tumor?
Where is it located?
Depth
T staging reflects:
Tumor size (mm/cm)
Degree of local invasion into surrounding tissue
Higher T → greater:
Local tissue damage
Surgical difficulty
TNM: Node — Lymph Node Involvement
Answers:
Has cancer spread to lymph nodes?
If yes: how many and where?
Depth
Lymphatic spread is often the first route of metastasis
More nodes involved → higher likelihood of:
Systemic dissemination
Regional lymph nodes act as:
checkpoint for cancer spread
TNM: Metastasis (M) — Distant Spread
Answers:
Has cancer spread to other parts of the body?
If yes: where and how much?
Depth
Indicates spread via:
blood (hematogenous)
or advanced lymphatic spread
Presence of metastasis (M1):
Automatically indicates advanced-stage cancer
TNM Stage grouping: Stage Grouping + diagram
Stage 0:
Abnormal cells, no invasion
Stage I:
Early, localized spread
Stage II:
Larger tumor ± limited lymph node involvement
Stage III:
Extensive regional spread (more lymph nodes, larger tumor)
Stage IV:
Distant metastasis

Treatment of cancer
Surgery
• Chemotherapy
• Radiation Therapy
• Immunotherapy (Oncolytic viral therapy)
• Stem Cell Transplant
• Hyperthermia
• Gene Therapy
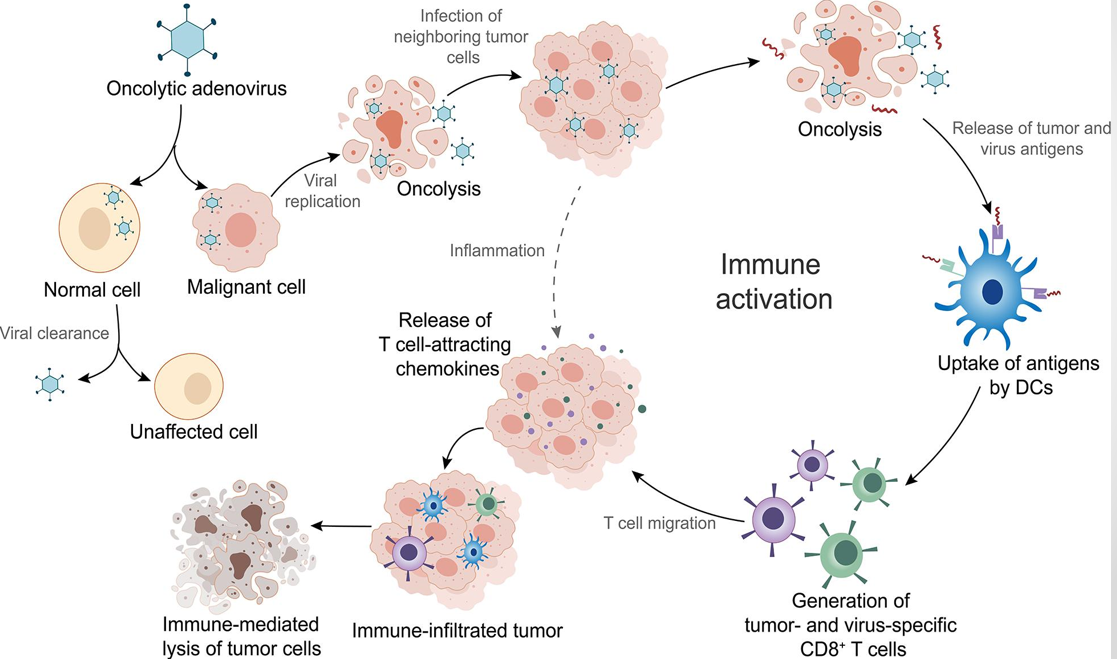
Oncolytic HSV Mechanism: Infection of Tumor Cells
Oncolytic HSV infects tumor cells
Depth
Tumor cells often have:
Defective antiviral responses (e.g., interferon pathway)
This makes them:
More permissive to viral entry and replication
Oncolytic HSV Mechanism: Viral Replication in Tumor Cells
Virus replicates efficiently inside tumor cells
Depth
Cancer cells:
Have high metabolic activity
Provide resources for rapid viral genome replication
Engineered HSV strains:
Preferentially replicate in abnormal cells
Oncolytic HSV Mechanism: Tumor Cell Lysis
Infected tumor cell ruptures (lysis)
Depth
Lysis results from:
Accumulation of viral particles
Breakdown of cellular integrity
Outcome
Cell death + release of:
new virions
Oncolytic HSV Mechanism: Viral Spread
Released virions infect neighboring tumor cells
Depth
Creates a self-amplifying cycle:
Infection → replication → lysis → spread
Allows:
Progressive destruction of tumor mass
Oncolytic HSV Mechanism: Effect on Healthy Cells
Virus:
Does not replicate efficiently in healthy cells
Depth
Normal cells:
Have intact antiviral defenses
Activate interferon pathways → inhibit viral replication
Outcome
Healthy cells:
Survive infection or clear virus
Oncolytic HSV Mechanism diagram HF10 project
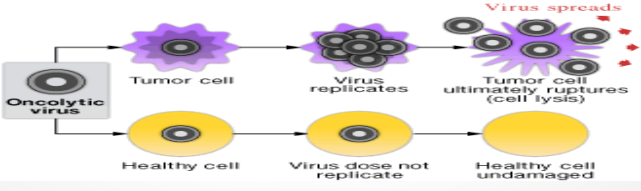
Oncolytic virus
An oncolytic virus is a virus that preferentially infects and kills cancer cells. As the infected cancer cells are destroyed by oncolysis, they release new infectious virus particles or virions to help destroy the remaining tumour.
Oncolytic virotherapy
Clinical Trial Phases
1. Phase I — Safety (from slide)
Purpose: Check for safety
Sample: 10–20 healthy volunteers
Depth
Determines:
Maximum tolerated dose (MTD)
Dose-limiting toxicities
Unexpected side effects:
Common at this stage
2. Phase II — Efficacy (from slide)
Purpose: Check for efficacy
Sample: ~200 patients
Depth
Evaluates:
Does the treatment actually work?
Many treatments fail here because:
Effectiveness is lower than expected
3. Phase III — Large-scale confirmation (from slide)
Purpose: Confirm findings in large population
Sample: >1000 people
Depth
Compares:
New treatment vs standard therapy/placebo
Detects:
Rare side effects (due to large sample size)
4. Phase IV — Post-marketing surveillance (from slide)
Purpose: Long-term safety in real-world population
Sample: General patient population
Depth
Conducted after:
Drug approval
Identifies:
Rare or delayed adverse effects
Effects in:
Previously untested groups
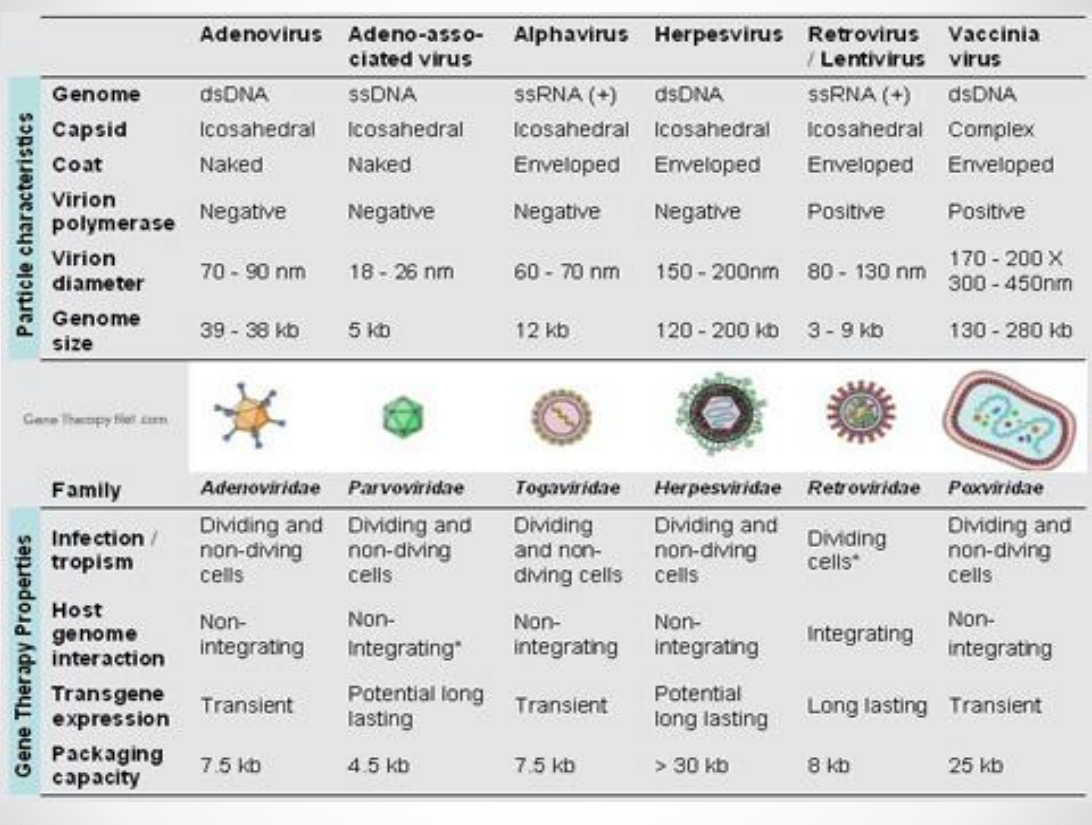
Table of properties for viruses
Delivery methods
• Micro-injection
• Electroporation
• Gene gun
• Tattooing
• Laser
• Ultrasound
Electroporation
What it is:
Application of short electrical pulses to create temporary pores in the cell membrane.
Technical details:
Electric field disrupts lipid bilayer → transient permeability
DNA enters through these pores
Key features:
Works on many cells at once
Efficiency depends on voltage, pulse duration
Exam insight:
Widely used for bacteria, mammalian cells, and in vivo gene delivery
Gene Gun (Biolistic method)
What it is:
DNA-coated metal particles (gold/tungsten) are shot into cells at high velocity.
Technical details:
Physical penetration delivers DNA directly into cytoplasm/nucleus
Often used for plant cells (cell wall barrier)
Key features:
No need for vectors
Can target tissues directly
Limitation:
Cell damage + shallow penetration
Tattooing
What it is:
Uses rapid needle punctures (like a tattoo machine) to deliver DNA into skin.
Technical details:
Creates micro-injuries → enhances DNA uptake
Often used in DNA vaccines
Key features:
Simple, low-cost
Works well for skin immune responses
Laser
What it is:
Laser creates temporary holes in cell membranes.
Technical details:
Highly controlled, localized membrane disruption
DNA diffuses into cell after pore formation
Key features:
Precise targeting
Requires specialized equipment
Ultrasound
What it is:
Uses sound waves (sonoporation) to increase membrane permeability.
Technical details:
Often combined with microbubbles
Cavitation effect → membrane disruption
Key features:
Non-invasive
Can target deep tissues
Microinjection diagram
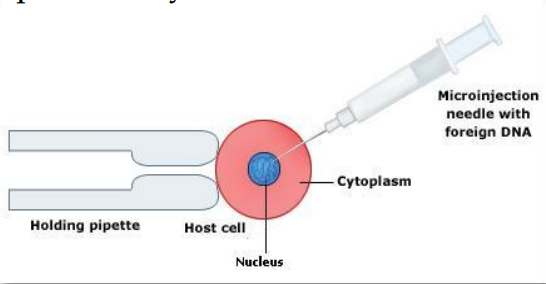
What microinjection actually does (mechanism)
A glass micropipette (~0.5–1 µm tip) physically penetrates the cell membrane
DNA is directly deposited into:
Cytoplasm or
Nucleus (more effective for expression)
Key point:
No reliance on endocytosis → no degradation in endosomes/lysosomes
Microinjection Target cells
Slide mentions:
Eggs
Oocytes
Embryos
Plant protoplasts
Why these?
Large size → easier to inject
Visible nucleus → precise targeting
Protoplasts lack cell wall → easier penetration
Microinjection Equipment
Slide mentions:
Specialised microscope
Manipulator
Phase-contrast microscope
Expanded breakdown:
Micromanipulator → controls needle movement in micrometers
Holding pipette → stabilizes the cell using suction
Injection needle → delivers DNA
Phase-contrast microscope → allows visualization of transparent cells
Computerized control (from slide):
Improves:
Accuracy
Reproducibility
Speed
Video systems help monitor injection in real time
Role of dye in microinjection
Dye is co-injected with DNA
Helps identify:
Whether injection was successful
Which cells received DNA
Exam angle:
Acts as a visual marker, not functional in gene expression
microinjections limitations
Very low throughput (one cell at a time)
Requires high technical skill
Risk of cell damage or lysis
Expensive equipment
Microinjection: ideal cell characteristics
Large size
Non-adherent
Pronounced nucleus
Why these matter (technical reasoning)
Large cells → easier needle insertion, lower chance of rupture
Non-adherent cells → easier to manipulate and position under microscope
Pronounced nucleus → allows accurate nuclear injection, which increases gene expression efficiency
Microinjection problematic cell types
Contractile cells (e.g., muscle)
Monolayer adherent cells
Why they are difficult
Contractile cells:
Rapid shape change during injection
Calcium influx triggers contraction → needle displacement → cell damage
Adherent cells (monolayer):
Attached to surface → harder to position and stabilize
Needle insertion angle becomes difficult
Increased mechanical stress during injection
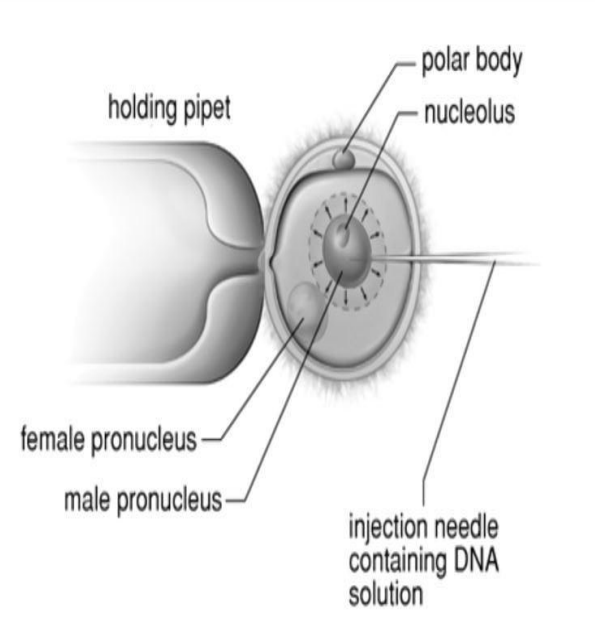
DNA Injection (Pronuclear stage)
Following fertilization, male and female pronuclei remain separate for a few hours before fusion
This allows microinjection of desired genes into the larger male pronucleus
Technical integration:
Male pronucleus = larger + more visible → easier targeting
Injection at this stage ensures DNA is present before first mitotic division
Increases chance of genome-wide distribution of transgene
DNA Injection (Site of injection)
Injection is performed into the male pronucleus
Technical integration:
Nuclear injection avoids cytoplasmic degradation
Promotes random integration into host genome
Higher efficiency than cytoplasmic delivery
DNA Injection (Embryo survival and transfer)
Eggs that survive injection are transferred into oviducts of a pseudopregnant female mouse
Technical integration:
Pseudopregnancy = hormonally prepared uterus (via mating with vasectomized male)
Provides:
Proper implantation environment
Normal embryonic development conditions
DNA Injection (Founder mouse generation)
Leads to generation of a Founder mouse, from which permanent transgenic lines can be established
Technical integration:
Founder = organism with integrated transgene
Used for breeding → stable inheritance (germline transmission)
Not all offspring are transgenic due to:
Random integration
Possible mosaicism
DNA Injection (Detection of transgene)
Presence identified by:
PCR analysis
Southern blot hybridization
Technical integration:
PCR → fast detection of gene presence
Southern blot → confirms:
Integration into genome
Copy number
Insertion pattern
DNA Injection steps
Fertilization → pronuclei visible
Inject DNA into male pronucleus
Select surviving embryos
Transfer to pseudopregnant female
Birth of offspring
Screen using PCR/Southern blot
Identify founder → establish transgenic line
DNA injection diagram
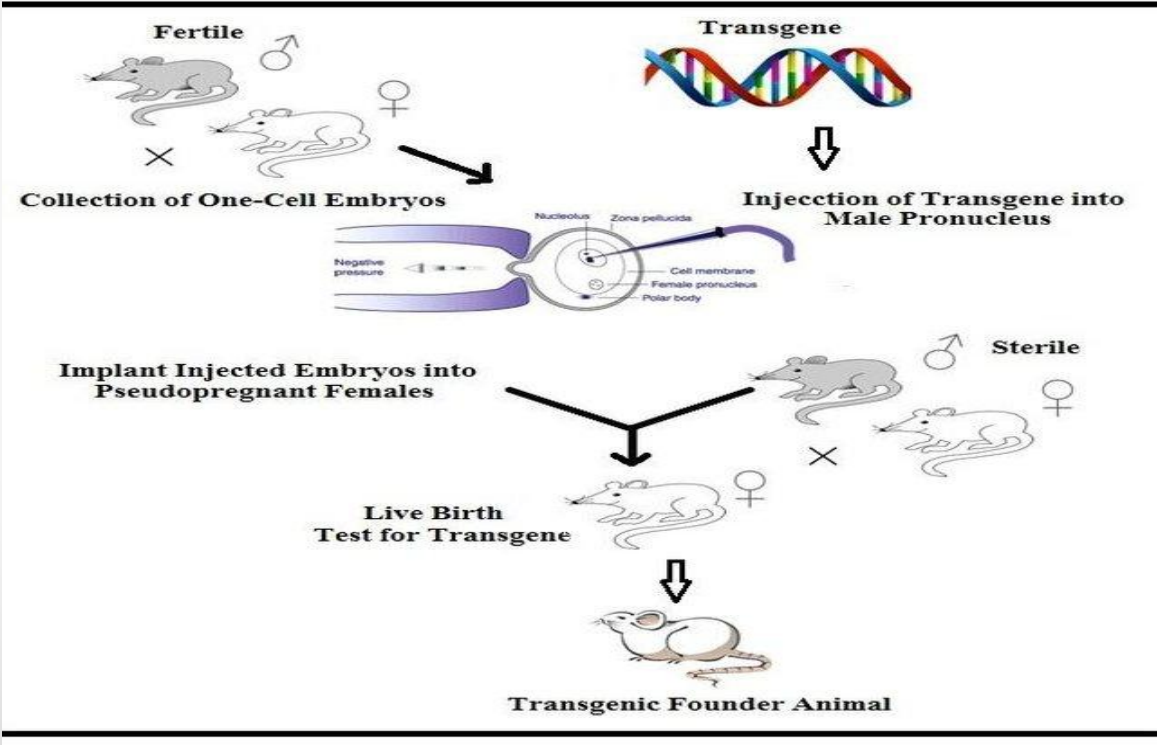
Applications: Transgenic Animals (Fertile mating → one-cell embryos)
Fertile male × female → zygote formation
Collection of one-cell embryos (early stage)
Technical integration:
One-cell stage = ideal for genetic modification
Ensures any inserted DNA can propagate to all cells (germline inclusion)
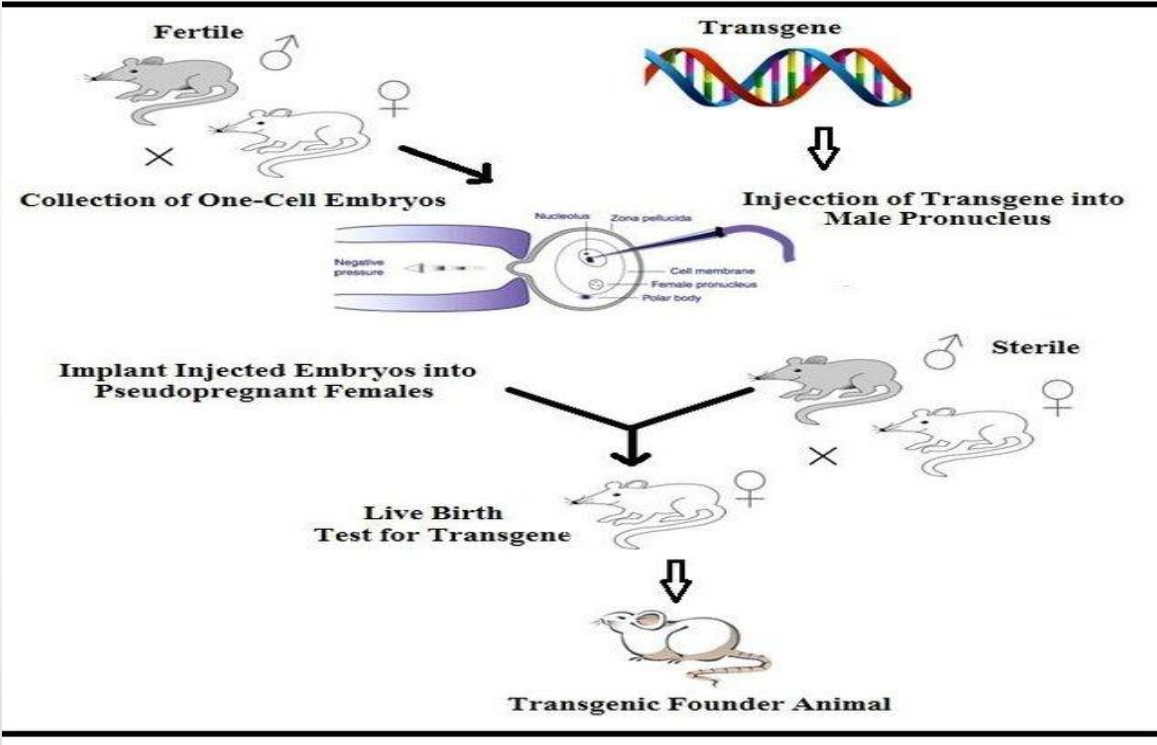
Applications: Transgenic Animals (Injection of transgene into male pronucleus)
Transgene is injected into male pronucleus
Technical integration:
Male pronucleus:
Larger → easier targeting
More transcriptionally active early on
DNA integrates randomly into genome
Occurs before first division → increases stable inheritance
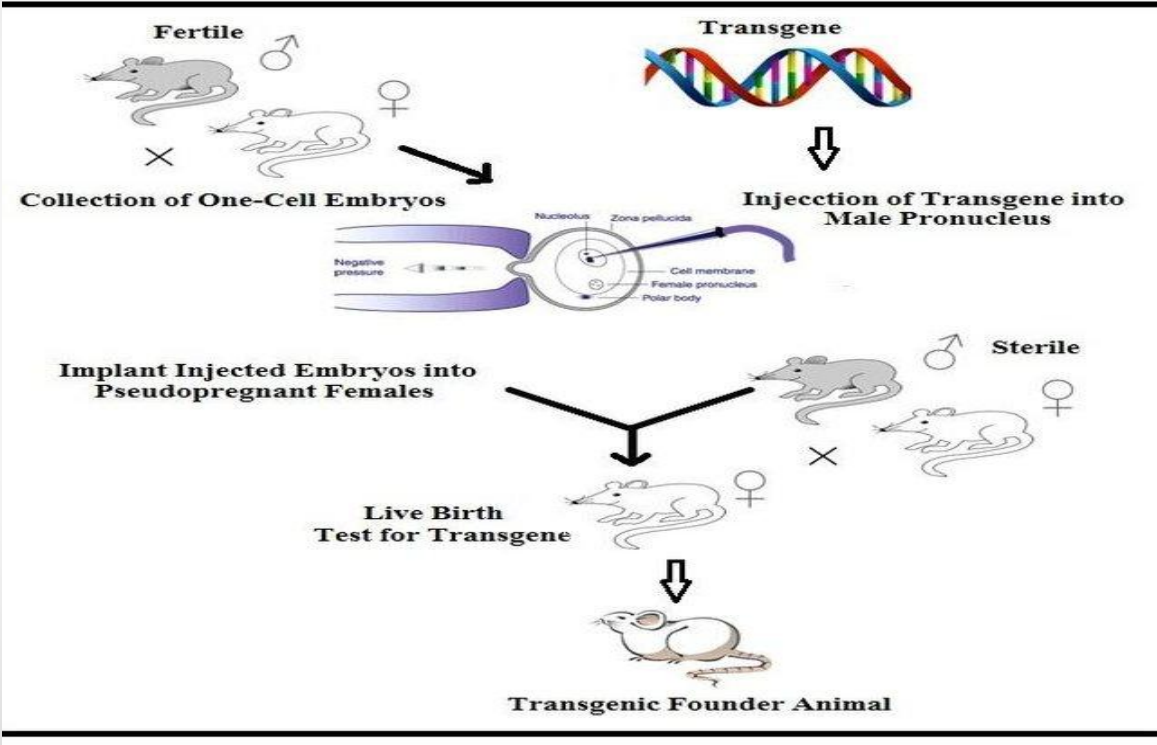
Applications: Transgenic Animals (Embryo implantation)
Injected embryos are implanted into pseudopregnant females
Technical integration:
Pseudopregnant female = hormonally primed uterus
Required because:
Embryos cannot develop ex vivo
Implantation site: oviduct/uterus
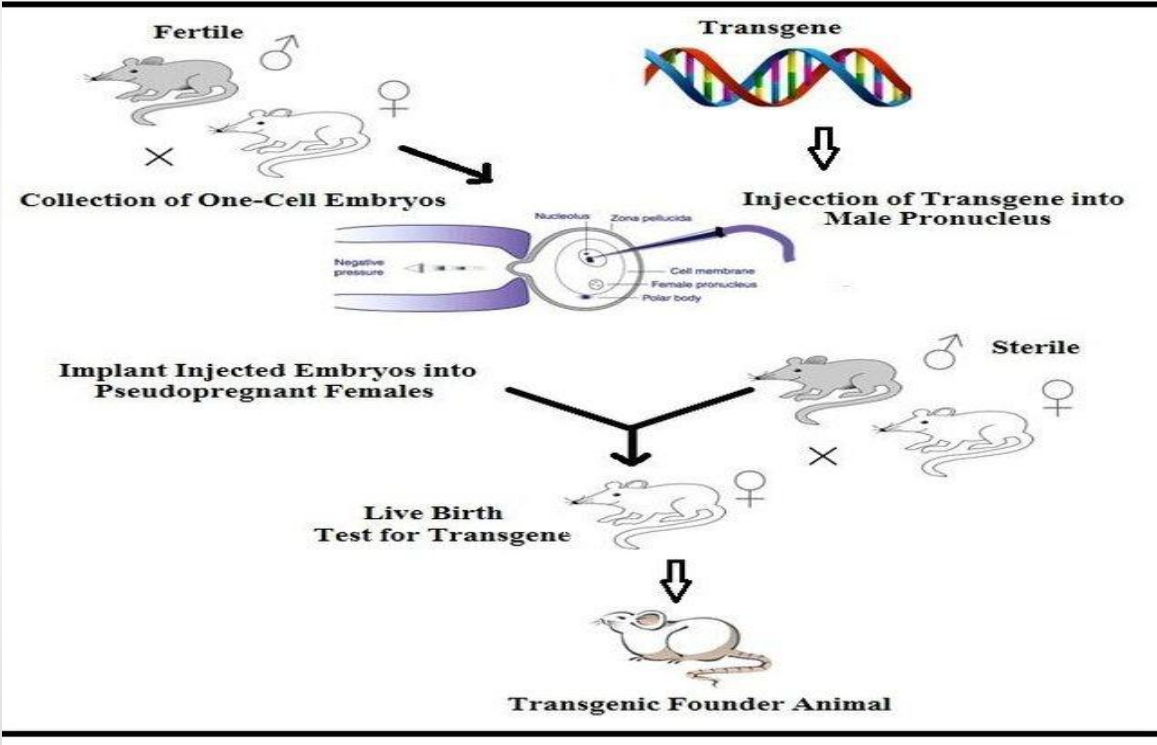
Applications: Transgenic Animals (Use of sterile male)
Female is mated with sterile (vasectomized) male
Technical integration:
Triggers:
Hormonal changes (progesterone increase)
Uterine receptivity
Ensures no competing fertilization
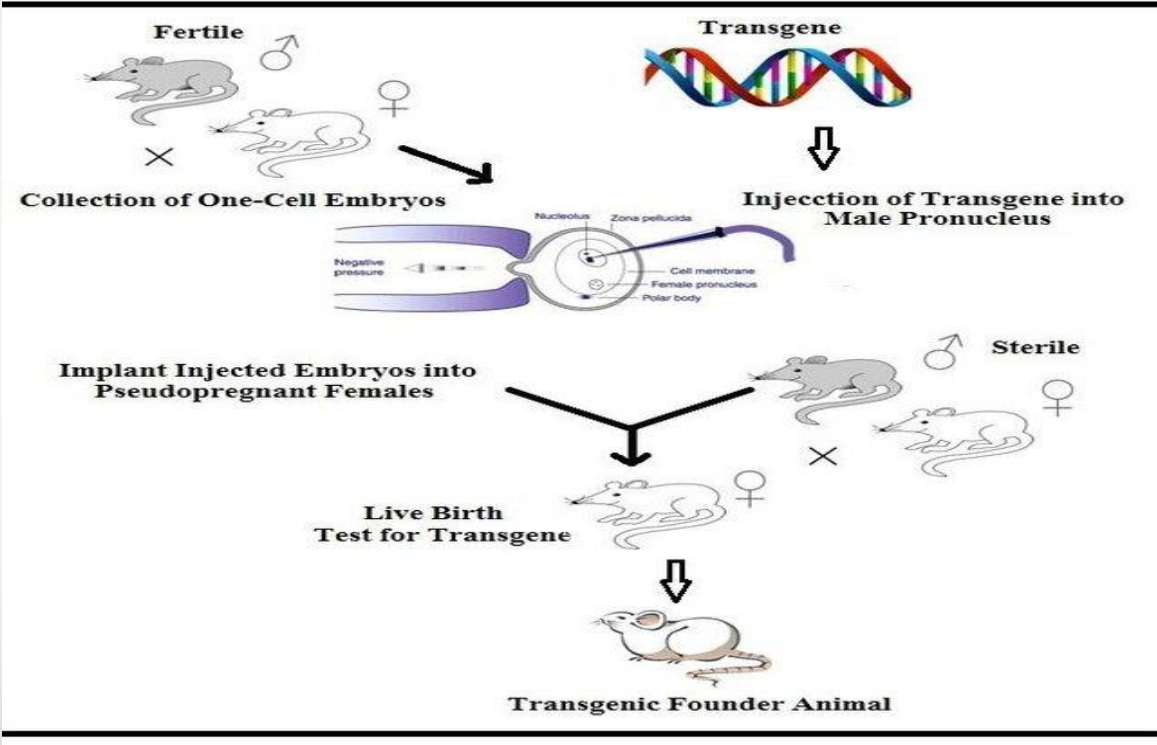
Applications: Transgenic Animals (Live birth + testing)
Offspring are born → tested for transgene
Technical integration:
Not all offspring are transgenic
Screening methods:
PCR → presence of gene
Southern blot → integration pattern
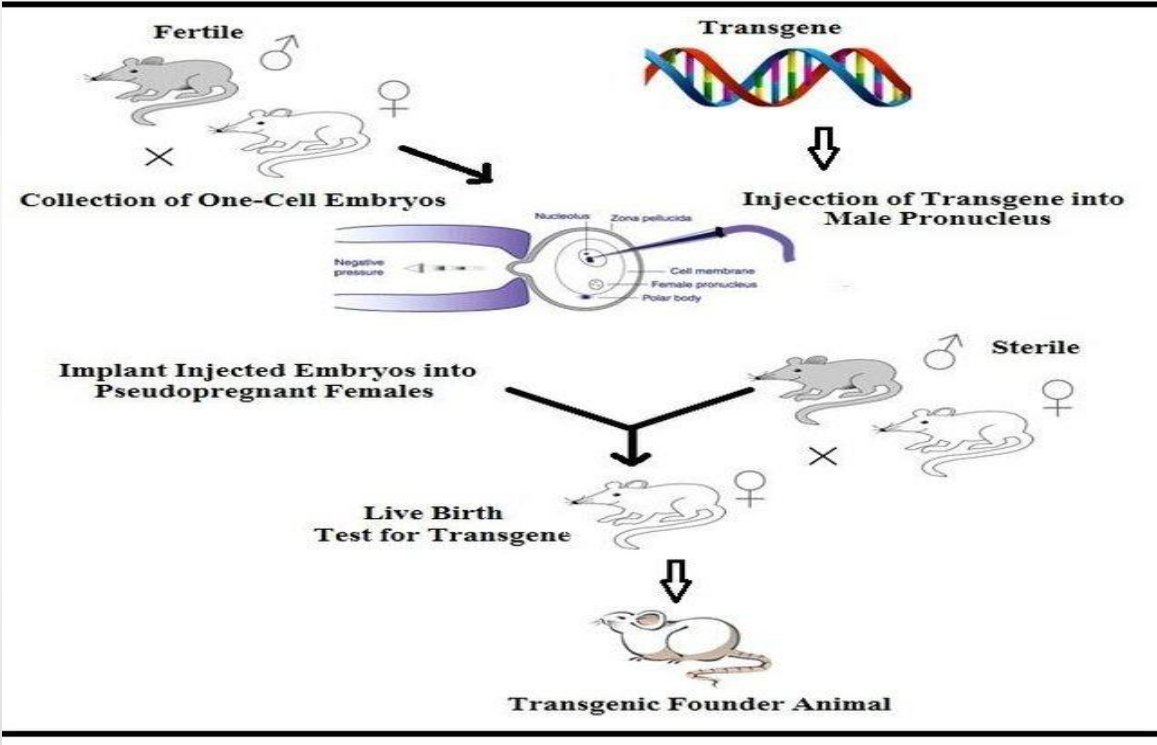
Applications: Transgenic Animals (Transgenic founder animal)
Positive offspring = Transgenic founder
Technical integration:
Founder carries gene in germline
Can be bred → stable transgenic lineage
Important for:
Functional gene studies
Disease models
Drug testing