AQA A level Chem 3.3 Halogenoalkanes
1/17
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
State the benefit to life on Earth of ozone in the upper atmosphere. (1)
Absorbs harmful UV radiation (1)
Suggest one reason why the use of CFCs was not restricted until several years after Rowland and Molina published their research. (1)
One of these reasons:
• lack of evidence that ozone was being depleted
• lack of alternatives to CFCs
• commercial interest to continue to use CFCs
• hard to obtain international agreement (1)
CFC-11 is a greenhouse gas that can contribute to global warming.
State and explain how CFC-11 is able to contribute to global warming (2)
absorbs infrared radiation (1)
molecule has polar bonds (1)
Which compound is not formed by reacting 3-bromo-3-methylhexane with warm, ethanolic potassium hydroxide?
A 2-ethylpent-1-ene
B 3-methylhex-1-ene
C 3-methylhex-2-ene
D 3-methylhex-3-ene (1)
B (1)
The question below refers to the reaction of 1-bromopropane with a solution of potassium cyanide in aqueous ethanol.
What is the organic product of this reaction?
A propylamine
B butylamine
C propanenitrile
D butanenitrile
D (1)

The question below refers to the reaction of 1-bromopropane with a solution of
potassium cyanide in aqueous ethanol.
The reactions of 1-bromopropane and 1-chloropropane with potassium cyanide
in aqueous ethanol occur at different rates under the same conditions.
Which row correctly shows the compound that has a faster rate of reaction and the correct reason for this?(1)
A (1)
Which species could act as a nucleophile?
A BH3
B NH4+
C PH3
D SiH4 (1)
C (1)
When 2.0 cm3 of 1-bromo-2-methylpropane (Mr = 136.9) were reacted with
an excess of sodium hydroxide, 895 mg of 2-methylpropan-1-ol (Mr = 74.0)
were obtained.
The density of 1-bromo-2-methylpropane is 1.26 g cm−3
Calculate the percentage yield for this reaction. (3)
Amount 1-bromo-2-methylpropane (= (2 × 1.26) / 136.9 = 2.52/136.9) = 0.0184 mol (1)
mass of 2-methylpropan-1-ol expected (= 0.0184 × 74.0) = 1.36 g (1)
100 × (0.895/1.36) = 65.7%
A student compares the rates of hydrolysis of 1-chlorobutane, 1-bromobutane and 1-iodobutane.
The suggested method is:
• add equal volumes of the three halogenoalkanes to separate test
tubes
• add equal volumes of aqueous silver nitrate to each test tube
• record the time taken for a precipitate to appear in each test tube.
State and explain the order in which precipitates appear. (2)
Silver iodide then silver bromide then silver chloride (1)
bond strength C−I < C−Br < C−Cl (1)
When 2-bromobutane reacts with ethanolic potassium hydroxide, two structurally isomeric alkenes are produced, one of which shows stereoisomerism.
Explain why two structurally isomeric alkenes are formed (2)
H is lost from different carbon atoms (1)
H removes from C1 and C3 to give two isomers (1)
Reaction of 2-bromobutane with potassium hydroxide can produce two types of product depending on the solvent used. In aqueous solution, the formation of an alcohol, E, is more likely but in ethanolic solution the formation of alkenes is more likely.
Name alcohol E and draw its structural formula. By reference to the structure of the halogenoalkane, explain why the initial step in the mechanism of the reaction producing the alcohol occurs.(5)
Alcohol =butan-2-ol (1)
Appropriate structure for CH3CH(OH)CH2CH3 (1)
C-Br bond is polar (1)
Lone pair on OH-(1)
Attacks the CS+(1)
The boiling point of iodomethane (CH3I) is higher than that of fluoromethane (CH3F) even though electronegativity of iodine is less than of fluorine.
Explain why iodomethane has the higher boiling point by considering the forces that act between CH3I molecules and comparing these forces with the forces between CH3F molecules (3)
Iodine is a bigger molecule than fluorine so the van der Waals forces between CH3I molecules are stronger than CH3F (1)
The dipole-dipole forces between CH3F is greater than CH3I (1)
As van der Waals forces are the strongest, CH3I takes more energy to break and therefore has a higher boiling point (1)
Which statement is not correct about ozone?
A It absorbs harmful ultraviolet radiation in the upper atmosphere.
B It decomposes to form oxygen.
C Its decomposition is catalysed by chlorine molecules.
D Ozone holes are regions of the upper atmosphere where there
is a reduced concentration of ozone (1)
C (1)
Which one of the following statements explains best why fluoroalkanes are the least reactive haloalkanes?
A - Fluorine is much more electronegative than carbon
B - The F- ion is the most stable halide ion
C - The C-F bond is the most polar carbon-halogen bond
D - The C-F bond is the strongest carbon-halogen bond (1)
D (1)
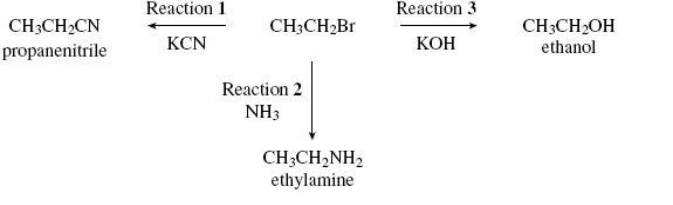
Nucleophiles react with bromoethane in substitution reactions. This type of reaction is illustrated in the following scheme.
Explain why an excess of ammonia is needed in Reaction 2 to produce a high yield of ethylamine. (1)
The idea of removing a proton from the intermediate alkylammonium ion (1)
State what the • represents in Cl• (1)
An unpaired electron (1)
Write equations for the following steps in the mechanism for the reaction of fluorine with fluoromethane (CH3F) to form difluoromethane (CH2F2).
Initial step
First propagation step
Second propagation step
A termination step leading to the formation of 1,2-diflouroethane (4)
Initiation : F2 → 2F• (1)
First propagation : F• + CH3F → •CH2F + HF (1)
Second propagation : F2 + •CH2F → CH2F2 + F• (1)
Termination : 2• CH2F → CH2FCH2F (1)
Nitrogen monoxide (NO) catalyses the decomposition of ozone into oxygen.
Use the overall equation to deduce Step 3 in the following mechanism that shows how nitrogen monoxide catalyses this decomposition (1)
Overall Equation 2O3 → 3O2
Step 1 O3 → O + O2
Step 2 NO+ O3 → NO2 + O2
Step 3 (1)
O + NO2 → NO + O2 (1)