2.28 - 2.32 Acids, alkalis and titrations
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
How to use phenolphthalein to distinguish between acidic and alkaline solutions
Alkaline - pink
Acidic - colourless
How to use methyl orange to distinguish between acidic and alkaline solutions
Alkaline - yellow
Acidic - red
How to use litmus solution to distinguish between acidic and alkaline solutions
Alkaline - blue
Acidic - red
How to use litmus paper to distinguish between acidic and alkaline solutions
Blue litmus paper goes red in acidic and stays blue in alkaline
Red litmus paper goes blue in alkaline and stays red in acidic
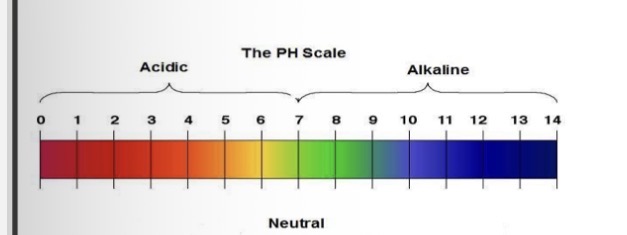
How to read pH scale
pH 7 is neutral
< pH 7 is acidic (0 is strongest acid)
> pH 7 is alkaline (14 is strongest alkali)
How to use the universal indicator to measure the pH of an aqueous solution
Add drops of solution to universal indicator paper and observe what colour it goes
What does acids in aqueous solutions produce
H+ ions (hydrogen ions)
What do alkalis in an aqueous solution produce
OH- ions (hydroxide ions)
What is a neutralisation reaction
Reaction between an acid and base
What is the ionic equation for any alkali- acid neutralisation reaction
H+ (aq) + OH- (aq) → H2O (l)