IB Chemistry HL: How much? The amount of chemical change?
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
What are symbol and word equations?
A symbol equation is a shorthand way of describing a chemical reaction using chemical symbols to show the number and type of each atom in the reactants and products
A word equation is a longer way of describing a chemical reaction using only words to show the reactants and products
How do you balance equations and what should you remember?
When balancing equations remember:
- Not to change any of the formulae
- To put the numbers used to balance the equation in front of the formulae
- To balance firstly the carbon, then the hydrogen and finally the oxygen in combustion reactions of organic compounds
When balancing equations follow the following the steps:
- Write the formulae of the reactants and products
- Count the numbers of atoms in each reactant and product
- Balance the atoms one at a time until all the atoms are balanced
- Use appropriate state symbols in the equation
How can you calculate the number of moles?
It is important to be clear about the type of particle you are referring to when dealing with moles
- E.g. 1 mole of CaF2 contains one mole of CaF2 formula units, but one mole of Ca2+ and two moles of F- ions

How do you calculate the reacting masses?
The masses of reactants are useful to determine how much of the reactants exactly react with each other to prevent waste
To calculate the reacting masses, the chemical equation is required
- This equation shows the ratio of moles of all the reactants and products, also called the stoichiometry, of the reaction
To find the mass of products formed in a reaction the following pieces of information are needed:
- The mass of the reactants
- The molar mass of the reactants
- The balanced equation
What is Avogadro's Law?
Avogadro’s law (also called Avogadro’s hypothesis) enables the mole ratio of reacting gases to be determined from volumes of the gases
Avogadro deduced that equal volumes of gases must contain the same number of molecules
- At standard temperature and pressure(STP) one mole of any gas has a volume of 22.7 dm3
The units are normally written as dm3 mol-1(since it is 'per mole')
The conditions of STP are
- a temperature of 0o C (273 K)
- a pressure of 100 kPa
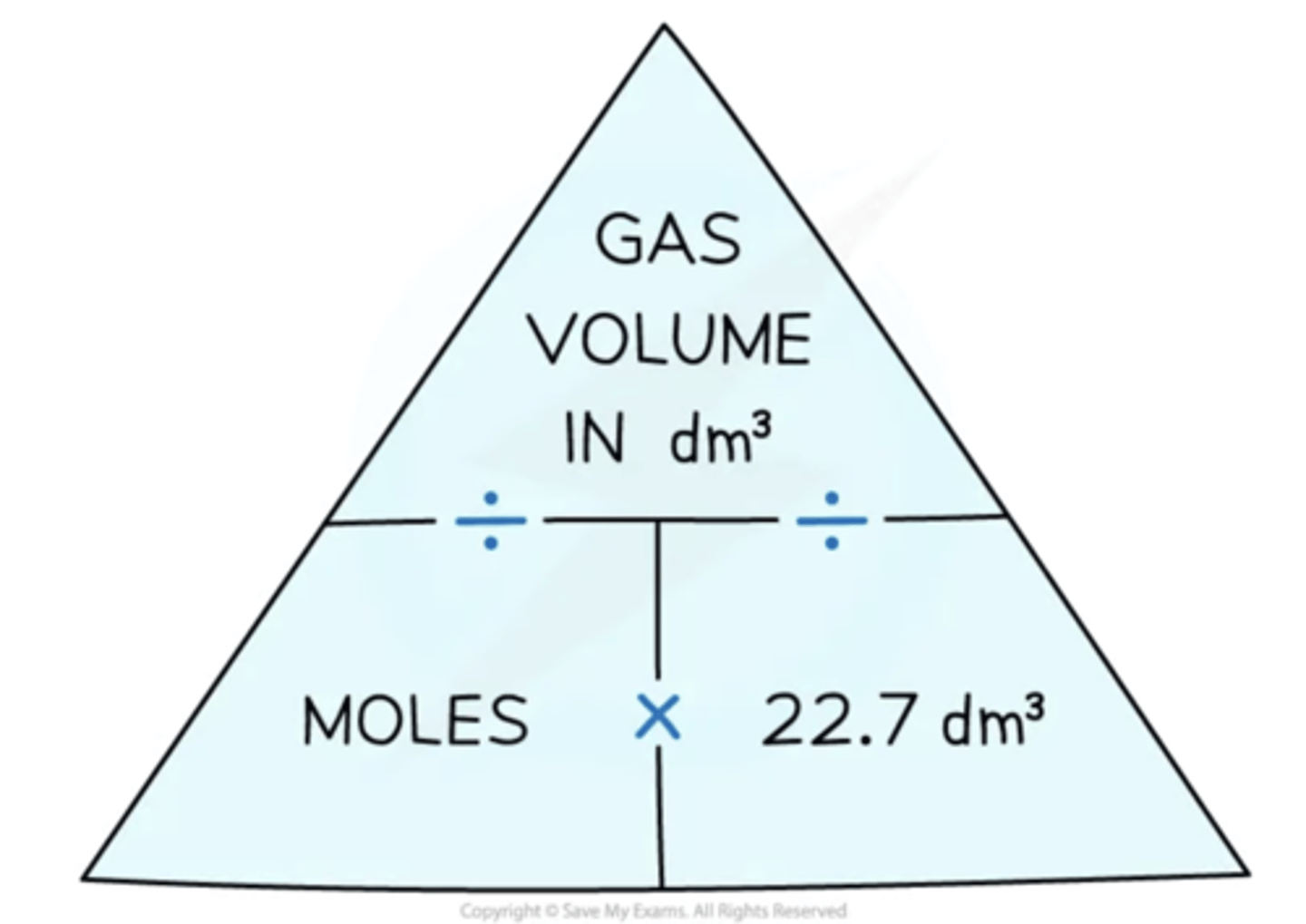
What can the molar gas volume be used to find?
The molar gas volume of 22.7 dm3 mol-1 can be used to find:
- The volume of a given number of moles of gas:
volume of gas (dm3) = amount of gas (mol) x 22.7 dm3 mol-1
- The number of moles of a given volume of gas
What is a titration?
Volumetric analysis is a process that uses the volume and concentration of one chemical reactant (a standard solution) to determine the concentration of another unknown solution
- The technique most commonly used is a titration
The volumes are measured using two precise pieces of equipment, a volumetric or graduated pipette and a burette
The steps in a titration are:
1. Measuring a known volume (usually 20 or 25 cm3) of one of the solutions with a volumetric or graduated pipette and placing it into a conical flask
2. The other solution is placed in the burette
3. A few drops of the indicator are added
4. The tap on the burette is carefully opened and the solution added, portion by portion, to the conical flask until the indicator just changes colour
5. Multiple trials are carried out until concordant results are obtained
How can you calculate the concentration?
The four steps involved in problem solving are:
1. write the balanced equation for the reaction
2. determine the mass/ moles/ concentration/ volume of the of the substance(s) you know about
3. use the balanced equation to deduce the mole ratios of the substances present
4. calculate the mass/ moles/ concentration/ volume of the of the unknown substance(s)
What is a back titration?
A back titration is a common technique used to find the concentration or amount of an unknown substance indirectly
The principle is to carry out a reaction with the unknown substance and an excess of a further reactant such as an acid or an alkali
The excess reactant, after reaction, is then analysed by titration and the mole ratios are used to deduce the moles or concentration of the original substance being analysed
What are limiting and excess reactants?
Sometimes, there is an excess of one or more of the reactants (excess reactant)
- The reactant which is not in excess is called the limiting reactant
To determine which reactant is limiting:
- The number of moles of the reactants should be calculated
- The ratio of the reactants shown in the equation should be taken into account
What is percentage yield? How do you calculate it?
In a lot of reactions, not all reactants react to form products which can be due to several factors:
- Other reactions take place simultaneously
- The reaction does not go to completion
- Products are lost during separation and purification
The percentage yield shows how much of a particular product you get from the reactants compared to the maximum theoretical amount that you can get
The actual yield is the number of moles or mass of product obtained experimentally
The theoretical yield is the number of moles or mass obtained by a reacting mass calculation

What is atom economy? How do you calculate it?
The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product
- The rest of the atoms or mass is wasted
It is found directly from the balanced equation by calculating the Mr of the desired product
In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product
- Whenever there is only one product, the atom economy will always be 100%
The atom economy could also be calculated using mass, instead or Mr

What is the significance of efficient processes?
Efficient processes have high atom economies and are important to sustainable development
- They use fewer resources
- Create less waste
As well as atom economy and percentage yield there are other factors that can be used to gauge the efficiency of a chemical process
- Rate
- Quantities of reagents such as catalysts and solvents
- Energy uses
- Economic efficiency